Abstract
Background
The etiology of burning mouth syndrome (BMS) patients remains unclear and systemic conditions concurrently with BMS are viewed with greater scrutiny. The present study aimed to reveal whether gastroesophageal reflux disease (GERD) is the possible comorbidity of BMS.
Methods
In our study, a real-world design was employed, enrolling BMS patients from an oral medicine clinic and capturing data in a non-interventional, cross-sectional setting. Endoscopy and the Gastroesophageal Reflux Disease Questionnaire (GERDQ) were used to assess the potential GERD patients. Demographics, clinical symptoms and signs were compared between both the GERD and non-GERD groups, and GERDQ ≥8 and <8 groups. Meanwhile, GERDQ items were assessed to determine predictive value.
Results
In total, 124 BMS patients with GERDQ scores were enrolled, of whom 84 patients were screened by endoscopy. GERD were confirmed by endoscopy in 30.95% (26/84) of patients with BMS, whereas 69.05% (58/84) showed no definite evidence of GERD. When GERDQ with a cutoff score of 8 was utilized for screening all 124 BMS patients, 25.8% (32/124) had GERDQ scores ≥8. When considering the endoscopic diagnosis as the golden standard due to the low availability of 24-h pH monitoring, the specificity and sensitivity were 79.2% and 46.2% respectively. Clinically, altered taste (p = 0.022) and thickened tongue coating (p = 0.001) were significantly more common in the GERDQ ≥8 group, whereas no significant difference was revealed between endoscopy approved GERD and non-GERD groups.
Conclusions
GERD may represent a potential systemic comorbidity in BMS patients and GERDQ might serve as the screening tool assisting healthcare professionals. Altered taste and thickened tongue coating might be suggestive for potential GERD symptoms in BMS patients. Further research is desired to elucidate the mechanisms linking such conditions.
This is a visual representation of the abstract.
Keywords
Introduction
Burning mouth syndrome (BMS) was defined by the International Association for the Study of Pain (IASP) as a chronic intraoral burning sensation that has no identifiable cause either local or systemic condition or disease (1). In practice, BMS is diagnosed with excluding local and systemic causes, including medication induced, anemia, vitamin deficiencies, Sjögren's syndrome and endocrine disease (hypothyroidism, diabetes mellitus) (2,3). Clinically, there is keen interest in both the academic and clinical communities to explore such comorbidities for uncovering the common pathogenesis of BMS.
Among the possible comorbidities, gastroesophageal reflux disease (GERD) is one of the common systemic conditions of BMS patients according to a scoping review (4). Our previous clinical study found that gastrointestinal disorders ranked first among the combined systemic conditions and acid reflux was the most common combined symptom in patients with burning mouth complaints (5). To date, gastrointestinal disorders such as GERD have not been excluded from the diagnostic criteria of International Headache Society (IHS) and International Classification of Orofacial Pain (ICOP) of BMS. Hence, further studies are still required to determine the relationship between BMS and GERD.
GERD is a condition in which the reflux of gastric contents into the esophagus results in symptoms and/or complications, including heartburn, oesophageal chest pain and regurgitation (6). Some epidemiological characteristics of GERD are similar to those of BMS because it is more likely to be diagnosed in advanced age and to co-occur with other chronic conditions including depression, anxiety and some neurological disorders (7,8). Passage of gastric contents of gastric acid and pepsin into the mouth was suggested to be linked with oropharyngeal mucosal damage and the burning mouth sensation (9,10). Exploring the relationship between GERD and BMS contributes to promote a more nuanced understanding of the multifactorial etiology of BMS, enable the establishment of an appropriate diagnosis and facilitate timely referral or therapeutic intervention.
To date, evidence about the relationship between BMS and GERD remains inadequate. A symptom-based GERD questionnaire (GERDQ) for screening applied in this study is validated with respect to ease of use in settings with limited access to endoscopy and ambulatory pH-metry. The aim of this study was to explore the possible link between BMS and GERD by identifying BMS patients accompanied with GERD and related symptoms in the clinic, assessing the predictive value of GERDQ, and providing appropriate referrals for these patients.
Methods
Study design
This cross-sectional, real-world study was conducted at Peking University School and Hospital of Stomatology. The cohort was derived from consecutive patients and variables were captured from electronic health records in non-interventional settings. Prior to the commencement of the study, ethical approval was obtained from the Ethics Committee of Peking University Health Science Center (PKUSSIRB-202059167). Informed consent was acquired from all patients in this study.
Study populations
Patients diagnosed with BMS based on the criteria of the International Headache Society (IHS) in 2016 and the International Classification of Orofacial Pain (ICOP) in 2020 from January 2023 to December 2023 were included in the study. The inclusion criteria were listed: (i) superficial oral burning sensation in the oral mucosa; (ii) symptoms recurring for more than two hours a day over a period of at least three months; and (iii) absence of identifiable causative lesions upon clinical examination. Exclusion criteria included: (i) presence of local lesions such as candidiasis, lichen planus or xerostomia and (ii) combined systemic diseases including medication-induced, anaemia, deficiencies of vitamin B12 or folic acid, Sjögren's syndrome and diabetes (2,3).
Clinical data collection
Demographic characteristics, clinical symptoms and signs were recorded for eligible patients. Clinical symptoms encompassed the location of symptoms, duration, xerostomia, altered taste, and severity measured by the visual analog scale (VAS). Clinical signs included findings such as erythema of anterior pillar, coated tongue and erythema of foliate papillae.
Questionnaire assessment
The GERDQ is recommended by the guidelines of the Lyon Consensus in 2018 and could be used as a preliminary diagnostic tool of GERD in BMS patients in clinical settings (11). The GERDQ consists of six questions inquiring about the frequency of heartburn, regurgitation, upper stomach pain, nausea, sleep disturbance and the use of additional medication to alleviate heartburn or regurgitation over the past week (12). The total score ranges from 0 to 18, with a cumulative score of 8 indicating a likelihood of having GERD (12). It is suggested that GERDQ with a cutoff value of 8 or more had a modest diagnostic value for GERD (13).
Statistical analysis
The demographic and clinical characteristics of BMS patients were analyzed using the descriptive statistics method. Numerical variables were described as the mean ± SD and range, and categorical variables were reported as frequency and percentage. Data normality was assessed by the Shapiro–Wilk test. An independent samples t-test or the Mann–Whitney U-test was used to compare continuous variables between two groups. Categorical data were analyzed using the chi-squared test or Fisher's exact test. p < 0.05 was considered statistically significant. Data analysis and processing were performed using SPSS, version 26.0 (IBM Corp.).
Results
Demographic characteristics
In total, 124 BMS patients were enrolled, with 105 women (84.7%) and 19 men (15.3%). The mean ± SD age of the patients was 55.14 ± 7.95 years. Combined systemic diseases were screened, hypertension without the use of angiotensin-converting enzyme inhibitors was found in 13.8% of patients, hyperlipidemia was found in 7.3% of patients, thyroid disease was found in 8.1% of patients, and GERD was diagnosed by endoscopy in 21.0% of patients. GERDQ scores were taken from all the BMS patients, with 92 patients having GERDQ score <8 and 32 patients having GERDQ score ≥8 (Figure 1). There was no significant difference in sex and age between two groups (p = 0.232 and 0.721, respectively), with a dominant majority of female patients in both groups (Table 1).

Flow chart of burning mouth syndrome (BMS) participants distribution. GERD = gastroesophageal reflux disease; GERDQ = Gastroesophageal Reflux Disease Questionnaire.
Demographic characteristics of patients with burning mouth syndrome (BMS) (n = 124).

GERD, gastroesophageal reflux disease; GERDQ, Gastroesophageal Reflux Disease Questionnaire.
*p < 0.05.
**p < 0.001.
Among the 84 BMS patients undergoing endoscopy, the diagnosis of GERD was confirmed in 26 (31.0%) patients (Table 2) and GERDQ score ≥8 was significantly frequent in this group of patients (p = 0.017).
Demographic characteristics and scores for the Gastroesophageal Reflux Disease Questionnaire (GERDQ) in burning mouth syndrome (BMS) with or without gastroesophageal reflux disease (GERD) diagnosed by endoscopy (n = 84).

Smoking (p = 0.047) and drinking (p = 0.014) were significantly more prevalent in the endoscopy-diagnosed GERD group, whereas systemic conditions including hyperlipidemia, thyroid disorders, hypertension and psychological problems showed no significant differences across GERD/non-GERD subgroups (all p > 0.05).
Evaluation of the questionnaire
The mean ± SD scores of GERDQ in patients with BMS are shown in Table 3. The overall score was 6.86 ± 1.51. The mean ± SD scores of heartburn, regurgitation and sleep difficulty were significantly higher in BMS patients with GERDQ scores ≥8 compared to those with scores <8 (p < 0.001). The mean score of nausea also showed a significant difference between groups (p = 0.043). In addition, no significant differences were observed in the mean score of upper stomach pain and additional medication between groups (p = 0.449 and 0.102, respectively).
The mean scores for the Gastroesophageal Reflux Disease Questionnaire (GERDQ) in patients with burning mouth syndrome (BMS) (n = 124).
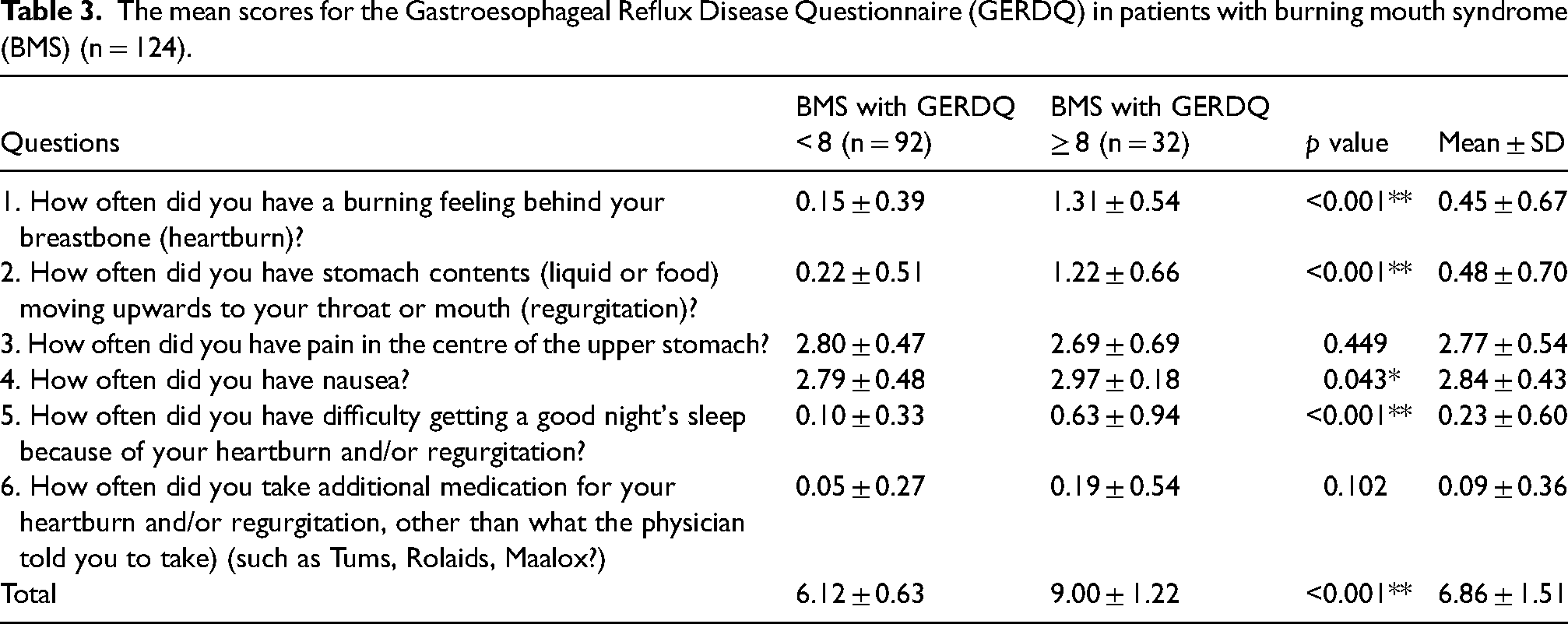
Particularly, compared with BMS patients for whom GERD was excluded by endoscopy, the GERDQ scores of the endoscopy-confirmed group were higher (7.15 ± 1.83 vs. 6.74 ± 1.29) without any significant difference (p = 0.279). Among 40 BMS patients who had not yet undergone endoscopy, 25.0% had a GERDQ score ≥8, warranting further GERD evaluation.
Clinical symptoms of BMS patients
The clinical symptoms of BMS patients are presented in Table 4. Altered taste was significantly more common in the BMS with GERDQ ≥8 group (p = 0.022), with bitter taste reported in the majority (28.1%). The prevalence of xerostomia was higher in the BMS with GERDQ ≥8 group (53.1%) compared to the GERDQ <8 group (38.0%), although the difference was not statistically significant (p = 0.216). There was no significant difference in VAS between the two groups (p = 0.363), and low VAS were more frequently noted in the BMS with GERDQ ≥8 group. The history of symptoms did not differ significantly between the two groups (p = 0.796), with the majority of patients experiencing symptoms for more than one year. Symptom locations varied, and the entire tongue was the most commonly affected area in both groups.
Clinical symptoms of patients with burning mouth syndrome (BMS) (n = 124).

GERDQ, Gastroesophageal Reflux Disease Questionnaire; VAS, visual analog scale.
The clinical symptoms of 84 BMS patients with and without GERD diagnosed by endoscopy are summarized in the supplementary material (Table S1). The prevalence of xerostomia and altered taste was higher in the BMS with GERD confirmed by endoscopy group, with no statistically significant difference between the two groups (p = 0.234 and 0.745, respectively). Regarding the location of symptoms, the tongue was the most commonly affected area in both groups (69.0% vs. 69.2%, p = 0.981). VAS scores did not differ significantly between the two groups, with the majority of patients reporting low VAS scores (0–5) in both groups.
Clinical signs of BMS patients
The clinical signs mainly focused on the coated tongue, anterior pillar and foliate papillae (Table 5). Notably, a coated tongue was significantly more common in the BMS with GERDQ ≥8 group (p = 0.001). In the group of BMS with GERDQ ≥8, 28.1% of the subjects had thickened tongue coating, whereas only 6.5% of the subjects in the GERDQ <8 group had thickened tongue coating. The erythema of the anterior pillar and the foliate papillae were more common in the BMS with GERDQ ≥8 group, although there was no significant difference between the two groups (p = 0.102 and 0.592, respectively).
Clinical signs of patients with burning mouth syndrome (BMS) (n = 124).

GERDQ, Gastroesophageal Reflux Disease Questionnaire.
In the 84 BMS patients with endoscopy, the prevalence of edema ± erythema of foliate papillae was higher in the BMS with GERD diagnosed by endoscopy group (11.5%) compared to the BMS without GERD diagnosed by endoscopy group (5.2%), with no significant difference between the groups (p = 0.556). Erythema of the anterior pillar and coated tongue also showed no significant difference between the groups (p = 1.000 and 0.885, respectively) (see supplementary material, Table S1).
The predictive value of GERDQ
Considering the limited availability of 24-h pH monitoring, the GERDQ was assessed using endoscopic and clinical diagnoses as the gold standard. The results showed that GERDQ achieved a predictive accuracy of 69.0%, with a negative predictive value (NPV) of 76.7% and a positive predictive value (PPV) of 50.0%. The specificity of GERDQ, representing its ability to correctly identify patients without GERD, was 79.2%, whereas its sensitivity, reflecting its capacity to detect true GERD cases, was 46.2% (Table 2).
Discussion
The present study aimed to further explore whether GERD is a potential comorbidity of BMS. BMS is a common chronic orofacial pain condition affecting 1.73–7.72% of the population worldwide (14), which impacts the quality of life and places a recognizable burden on the patient and healthcare system (15). Our study suggested that GERD may represent a potential systemic comorbidity in BMS patients, and altered taste and thickened tongue coating might be suggestive for potential GERD symptoms in BMS patients. The GERDQ might serve as the screening tool assisting healthcare professionals, with limited access to endoscopy and ambulatory pH-metry. Four GERDQ items (heartburn, regurgitation, sleep disturbance and nausea) demonstrated higher discriminatory power between the GERDQ ≥8 and <8 groups. Further consulting in the gastroenterology department should be recommended when it is suspected.
According to current guidelines, the diagnosis of GERD has no single gold standard and is based on a combination of symptoms, endoscopy, reflux monitoring and response to therapy (6). For the detection of reflux episodes, pH-impedance monitoring is considered to be the gold standard with limitations as an expensive, not widely available and time-consuming tool (11). Practically, upper endoscopy is widely used for evaluating esophageal mucosa, particularly in detecting erosive esophagitis and Barrett's esophagus (6,16). Nevertheless, 60–70% of GERD patients have non-erosive reflux disease, which is characterized by reflux symptoms but without mucosal breaks on endoscopy (17). On the other hand, for dental practitioners, a simple, non-invasive diagnostic tool is desirable to screen GERD in clinic. As recommended by the Lyon Consensus in 2018 and the Chinese guideline for diagnosis and treatment of gastroesophageal reflux disease in 2023 (11,18), the utilization of GERDQ assessments emerges as a non-intrusive and patient-friendly alternative, which might potentially be a valuable screening tool based on reflux symptoms, with specificity of 71% and sensitivity of 65% (12). In summary, the comprehensive use of GERDQ and endoscopy could assist the screening and diagnosis in different subgroups of patients, although no strong correlation between them was observed in our study.
Our study identified altered taste and thickened tongue coating as significantly more prevalent among BMS patients with GERDQ ≥8. Notably, bitter taste was reported in majority, consistent with previous studies suggesting dysgeusia, particularly bitter taste, as a common feature of BMS (19,20). GERD-related taste alterations, including bitter and acidic sensations, may arise from gastric acid reflux into the oral cavity (21–23). In addition, thickened tongue coating, another significant finding, has been proposed as an extraesophageal manifestation of GERD (24). Changes in metabolic patterns and microecological indexes of tongue coating are associated with various gastrointestinal diseases (25). Exfoliated cells of the tongue coating in GERD patients were tested in some studies, and showed different genotypes from controls (25,26). Therefore, it is suggested that the potential GERD symptoms should be highly suspected in these BMS patients with altered taste and thickened tongue coating in clinic.
The mechanisms linking GERD and BMS remain unclear. Proposed hypotheses include acid and pepsin-induced injury to the oral mucosa (9,10) and a shared neurological pathway (4). Evidence indicates that BMS patients are predisposed to somatic symptoms, including gastrointestinal disturbances (27), supporting the hypothesis that central sensitization may underlie BMS pathophysiology. Central and peripheral sensitization of nociceptive receptors may amplify sensory nerve activation, contributing to both esophageal symptoms such as heartburn and pain-related comorbidities, ultimately exacerbating clinical symptoms and impairing quality of life (28,29). A key candidate of the potential mechanistic link between BMS and GERD is the transient receptor potential vanilloid 1 (TRPV1) receptor. Elevated TRPV1 expression in the epithelial fibers of BMS patients correlates with persistent pain (30). Studies have demonstrated increased mRNA and protein levels of TRPV1 in GERD patients, inducing substantial calcium ion influx, which activates downstream signaling pathways and transmits nociceptive signals to higher brain centers, potentially contributing to hyperalgesia (31–33).
In sum, GERD may be one of the potential systemic comorbidities or common symptoms of BMS patients. It is imperative to elevate the consciousness of healthcare providers regarding the relationship between BMS and GERD. In the clinic, it is necessary for dental practitioners to be vigilant about the potential comorbidity of GERD in BMS with appropriate use of questionnaires, and prompt referral should be made when it is suspected. GERDQ, with a specificity of 79.2% and a sensitivity of 46.2%, is more effective for excluding non-GERD patients than confirming GERD. It serves as an effective initial screening tool in primary care or resource-limited settings to reduce unnecessary invasive diagnostics. However, for patients with persistent GERD symptoms, additional methods such as endoscopy or 24-h pH monitoring are essential for accurate diagnosis.
We acknowledge the limitations in this cross-sectional, real-world study. First, heterogeneity of the study population may have introduced biases. Second, due to the limited access of 24-h pH monitoring, we mainly used endoscopy as the diagnostic method for GERD in this study. Third, because chronic pain is associated with race, the patients included in our study inevitably had regional limitations (5). Additionally, the sample size in our study may not fully represent the broader population. Future studies should aim to address these limitations through larger, multiple regions and more homogeneous cohorts to provide more definitive conclusions.
Conclusions
In conclusion, our cross-sectional, real-world study suggests the importance of considering GERD in the diagnosis and management of BMS, and GERD may represent a potential systemic comorbidity in BMS patients. GERDQ might serve as the screening tool assisting healthcare professionals. Altered taste and thickened tongue coating might be suggestive for potential GERD symptoms in BMS patients. More studies with appropriate study designs are necessary to further explore the mechanism of the link between them.
Clinical implications
There is a notable high prevalence of GERD in BMS patients, highlighting a potential comorbid relationship between the two conditions.
GERDQ might serve as valuable screening tool assisting healthcare professionals, especially in resource-limited settings.
Significantly more common in BMS patients with GERDQ ≥8, altered taste and thickened tongue coating are considered key clinical indicators suggestive for potential GERD symptoms in this population.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251336139 - Supplemental material for Is gastroesophageal reflux disease a comorbidity of burning mouth syndrome? A cross-sectional, real-world study
Supplemental material, sj-docx-1-cep-10.1177_03331024251336139 for Is gastroesophageal reflux disease a comorbidity of burning mouth syndrome? A cross-sectional, real-world study by Linman Li, Shuangshuang Wu, Luling Wang, Xinming Zhang, Yajuan Cui and Zhimin Yan in Cephalalgia
Footnotes
Acknowledgments
This work is supported by National Natural Science Foundation of China (82170967).
Author contributions
LL was responsible for study conceptualization, methodology, formal analysis, investigations and writing the original draft. SW was responsible for data curation, methodology and investigations. LW was responsible for resources, supervision and validation. XZ was responsible for supervision and validation. YC was responsible for supervision and validation. ZY was responsible for funding acquisition, project administration, resources, supervision, and reviewing and editing.
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the authors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
Ethical approval was obtained from the Ethics Committee of Peking University Health Science Center (PKUSSIRB-202059167). Informed consent was acquired from all patients in this study.
Funding
This work was supported by National Natural Science Foundation of China (82170967).
Supplemental material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
