Abstract
Background
Evidence of an association between trigeminal neuralgia (TN) and subsequent cancer risk remains sparse. The present study aimed to examine the association between TN and cancer risk in the Danish population.
Methods
A nationwide population-based cohort study using hospital diagnoses collected routinely and prospectively from Danish population-based registries in 1994–2022.
Results
We identified 7846 individuals with a first-time diagnosis of TN. Within one year of diagnosis, we observed 193 cancer cases (standardized incidence rate (SIR) = 2.45, 95% confidence interval (CI) = 2.11–2.82). Absolute risk (AR) for all cancers within one year of TN diagnosis was 2.5% (95% CI = 2.2–2.9). Cancers of the head, neck and nervous system were most strongly associated with TN (AR 0.9% (95% CI = 0.7–1.1); SIR = 13.5 (95% CI = 10.5–17.0)) and the risk was persistently elevated one year after TN diagnosis. We observed 827 cancer diagnoses beyond one year after TN diagnosis, where smoking related cancers were associated with elevated cancer risk (SIR 1.13 = (95% CI = 0.98–1.29)).
Conclusions
TN was associated with an elevated risk of cancers of the head, neck and nervous system including site-specific cancers in the area. Our results suggest the potential importance of smoking related tumors in TN, either as a symptom, cause or shared risk factor.
This is a visual representation of the abstract.
Introduction
Trigeminal neuralgia (TN) is a condition characterized by recurrent episodes of unilateral, non-radiating facial pain in the distribution of one or more divisions of the trigeminal nerve (1). The pain is typically severe with an electric shock-like, shooting, stabbing or sharp quality, lasting only from a fraction of a second to two minutes (2). Pain is typically precipitated by otherwise innocuous sensory stimuli within the affected trigeminal distribution, including tooth brushing, chewing and yawning (3,4). Although TN can occur at any age, the average age at onset is 55 years and ranges from 53–57 years for both sexes. The incidence increases with age, and women are more frequently affected than men (5,6). Identified risk factors for TN include hypertension (7), arteriosclerotic changes (8), migraine headache (9) and diabetes (10).
Prior estimates of TN incidence vary substantially from 4.3 to 27 diagnoses per 100,000 annually (11–13), and prevalence estimates from prior studies range from 0.03% to 0.3% (1), depending on the population studied. Because TN is relatively rare, delays in diagnosis and misdiagnosis are common.
TNs are classified as classical, secondary and idiopathic (14–17). Classical TN arises from intracranial vascular compression by the superior cerebellar artery and is associated with morphological changes of trigeminal root at the site of the conflict. Idiopathic TN, in which no apparent underlying cause can be identified, accounts for an estimated 10% of cases. Secondary TN accounts for approximately 15% of cases and occurs in the setting of other disease processes, including multiple sclerosis and tumors of the cerebellopontine angle. Magnetic resonance imaging is generally recommended in individuals diagnosed with primary TN to visualize a possible neurovascular etiology and to rule out other secondary causes.
Although meningiomas and vestibular schwannomas are generally recognized as the tumors that most frequently underlie secondary TN, evidence regarding TN and subsequent cancer risk remains sparse. It is also plausible that TN and certain cancers may co-occur because of common underlying causes such as cigarette smoking.
To date, there have been no large population-based studies of TN and cancer incidence to date and existing studies have been generally limited by reliance on patient populations derived from a single hospital department or region or restricted to a limited age range (18–25). Furthermore, prior research describing distant and metastatic cancers in relation to TN remains sparse. Comprehending the cancer risk in patients with TN could yield valuable insights for diagnosis and long-term care for TN patients.
In this nationwide, population-based cohort study, we examined the relationship between hospital diagnoses of TN and cancer in the general Danish population. We estimated the association between TN and all cancers combined, cancer groups and site-specific cancer diagnoses. We included cancers outside of the head, neck and nervous system because cancers originating in remote locations may produce TN symptoms through mass effects from distant metastases or paraneoplastic processes.
Methods
Setting, design and population
We conducted a nationwide cohort study using routinely and prospectively collected data from Danish population-based registries in the period 1994 to 2022 (26). The Danish population is covered by a tax-funded health care system. All provided health care services are recorded and linked within the registries via an individually assigned personal identification number given at birth or immigration (27). The cohort was established by linking data from the Danish Civil Registration System (27), the Danish National Patient Registry (DNPR) (28) covering all Danish hospitals and the Danish Cancer Registry (DCR) (29). The DNPR contains records of hospital admissions since 1977, and contacts to emergency departments and outpatient clinics since 1995. Inpatient, outpatient and emergency department discharge diagnoses were used to identify patients of all ages diagnosed with TN coded using code G50.0 from the International Classification of Diseases, 10th Revision (ICD-10) (30). Further subclassification into classical, secondary or idiopathic TN was limited by our use of ICD-10 codes to identify TN diagnoses. Individuals with TN prior to 1 January 1994 were identified using diagnostic code 351 from the ICD-8 and were excluded (28). We included all patients with a first-time discharge diagnosis of TN recorded in the DNPR between 1994 to 2022. Patients with TN- or cancer diagnosis (except non-melanoma skin cancer (NMSC)) at any time prior to TN diagnosis were excluded from the analysis. NMSC-cancers are recorded incompletely in the DCR and DNPR and were therefore excluded from the analysis.
In Denmark, a diagnosis of TN follows the recommendations of the European Academy of Neurology guideline on TN. These recommendations include detailed history, neurologic examination and neuroimaging with magnetic resonance imaging to ensure a correct diagnosis and to distinguish among classical, primary and secondary TN (31).
Cancer diagnoses
Cancers were identified and classified according to the DCR, which has maintained records on all incident cases of primary cancer in Denmark since 1943, and has had mandatory reporting since 1987 (29). The DCR is between 95% and 98% complete and valid (29). We included all primary cancers recorded since 1 January 1994. Cancers were classified into the following groups according to their etiology: (i) hormone-related cancers; (ii) gastrointestinal cancers; (iii) smoking-related cancers; (iv) cancers of the head, neck and nervous system (including benign cerebral tumors); (5) hematologic cancers; (6) immune-related cancers; and (7) all other cancers (32–34) (see supplementary material, Table S1). Patients with NMSC were excluded from the analysis because these cancers are recorded incompletely in the DCR and DNPR.
Covariate
Covariates included in our analysis derived from the DNPR and CRS were sex (male or female), age group (0–17, 18–29, 30–39, 40–49, 50–59, 60–69 or ≥70 years) and calendar year of TN diagnosis (1994–1999, 2000–2004, 2005–2009, 2010–2014, 2015–2019 or 2020–2022). We identified comorbid diagnoses including stroke, cerebral infections, cranial trauma, benign cerebral tumors, ontological interventions, anxiety and depression, which could potentially be associated with TN (3,31).
Chronic obstructive pulmonary disease (COPD) was used as a proxy for smoking. Cerebral infections, stroke and cranial trauma were identified only within the 12 months prior to TN diagnosis. We additionally calculated the Charlson's comorbidity index (CCI) score, which has been extensively used and validated for the prediction of short-term mortality and has been adapted for use with hospital discharge data (35,36). The CCI score is derived by summing the assigned weights of all comorbid conditions presented by a patient, categorized into low (CCI = 0), moderate (CCI = 1–2) or high (CCI ≥ 3) scores. We used a modified CCI score excluding any tumor, leukemia, lymphoma and metastatic solid tumor groups from the calculation. Covariates, including those defined in the CCI score, are listed in the supplementary material (Table S2).
Statistical analysis
Our analysis included all individuals with a new diagnosis of TN between 1 January 1994 and 31 December 2022. Follow-up extended from the date of TN diagnosis until cancer diagnosis, death, emigration or administrative censoring at the end of the study period, whichever occurred first. We included all individuals with an ICD-10 code of TN diagnosis in the study period and no post-hoc power calculations were performed.
The population's characteristics were described with respect to frequency (N), percentage (%) of the total, the median calculated as the middle value of the data and the interquartile range (IQR) reported as the quartiles of the data. We calculated standardized incidence ratios (SIRs) for the associations of TN with overall cancer, cancer groups and site-specific cancer diagnoses. Age-, sex- and calendar year-standardized SIRs were calculated as the ratio of observed to expected numbers of cancers among patients with TN. The expected number of cancers was calculated based on the entire Danish population in the study period. This analysis was stratified by follow-up time, and SIRs were calculated separately for cancers diagnosed within one year or beyond one year after TN diagnosis. Stratification by follow-up time was chosen to account for diagnostic delays after TN diagnosis. We calculated the absolute risk (AR) of cancer among individuals diagnosed with TN within the first year after TN diagnosis as the cumulative incidence, by treating death as a competing event.
The 95% confidence intervals (CIs) were computed according to the assumption that the observed number of site-specific cancer cases followed a Poisson distribution. Exact limits were used when the observed number of cases was fewer than ten; otherwise, Byar's approximation was used. All analyses were conducted in SAS, version 9.4 (SAS Institute, Cary, NC, USA).
Secondary analyses
Compared to the general population, patients diagnosed with TN in an inpatient setting may be more likely to have comorbid illnesses registered that may either directly increase their risk of cancer or lead to increased rates of diagnosis through increased surveillance. Therefore, we stratified the calculation of SIRs for cancer overall according to the encounter type of first-time TN diagnosis (inpatient, outpatient or emergency department diagnosis). Additional secondary analyses were conducted within subgroups of individuals with TN, defined by sex, year of diagnosis, age at diagnosis, smoking using COPD as proxy and concurrent comorbidities, as evaluated with the CCI.
Patient involvement
Patients were not involved in determining the research question or the outcome measures, nor in developing plans for study design or implementation. No patients were asked to advise on the interpretation or writing up of results. No plans are in place to disseminate the results to study participants or the relevant patient community.
Results
We identified 7846 individuals with a first diagnosis of TN in Denmark between 1994 and 2022 (65% women) (Table 1). Of these, 20.5% were diagnosed in an inpatient setting, 72.5% were diagnosed in an outpatient clinic and 7% were diagnosed in an emergency department. The median age at diagnosis was 56.8 years (IQR = 44.5–69.9 years). The median length of follow-up was 7 years (IQR = 3.3–13.0 years). Most patients with TN had a low CCI score (68.8%) and 5% had a high CCI score.
Characteristics for all patients with a hospital diagnosis of trigeminal neuralgia 1994–2022.

Charlson comorbidity index, excluding any tumor, leukemia, lymphoma and metastatic solid tumor.
All cancers
We observed 1020 cancer diagnoses in the entire follow-up period among patients with TN, 193 of which occurred within the first year of TN diagnosis (Table 2). The cancers most frequently diagnosed within the first year of TN diagnosis were cancers of the head, neck and nervous system (n = 71), smoking-related cancers (n = 36) and hormone-related cancers (n = 28). The SIR for the overall study period was 1.19 (95% CI = 1.12–1.27). The AR for any cancer within the first year TN diagnosis was 2.5% (95% CI = 2.2–2.9) and the corresponding SIR was 2.45 (95% CI = 2.11–2.82).
Absolute risks (ARs) and standardized incidence ratios (SIRs) with 95% confidence intervals (CIs) for all cancer groups and site-specific cancers, by duration of follow-up after trigeminal neuralgia (TN) diagnosis.
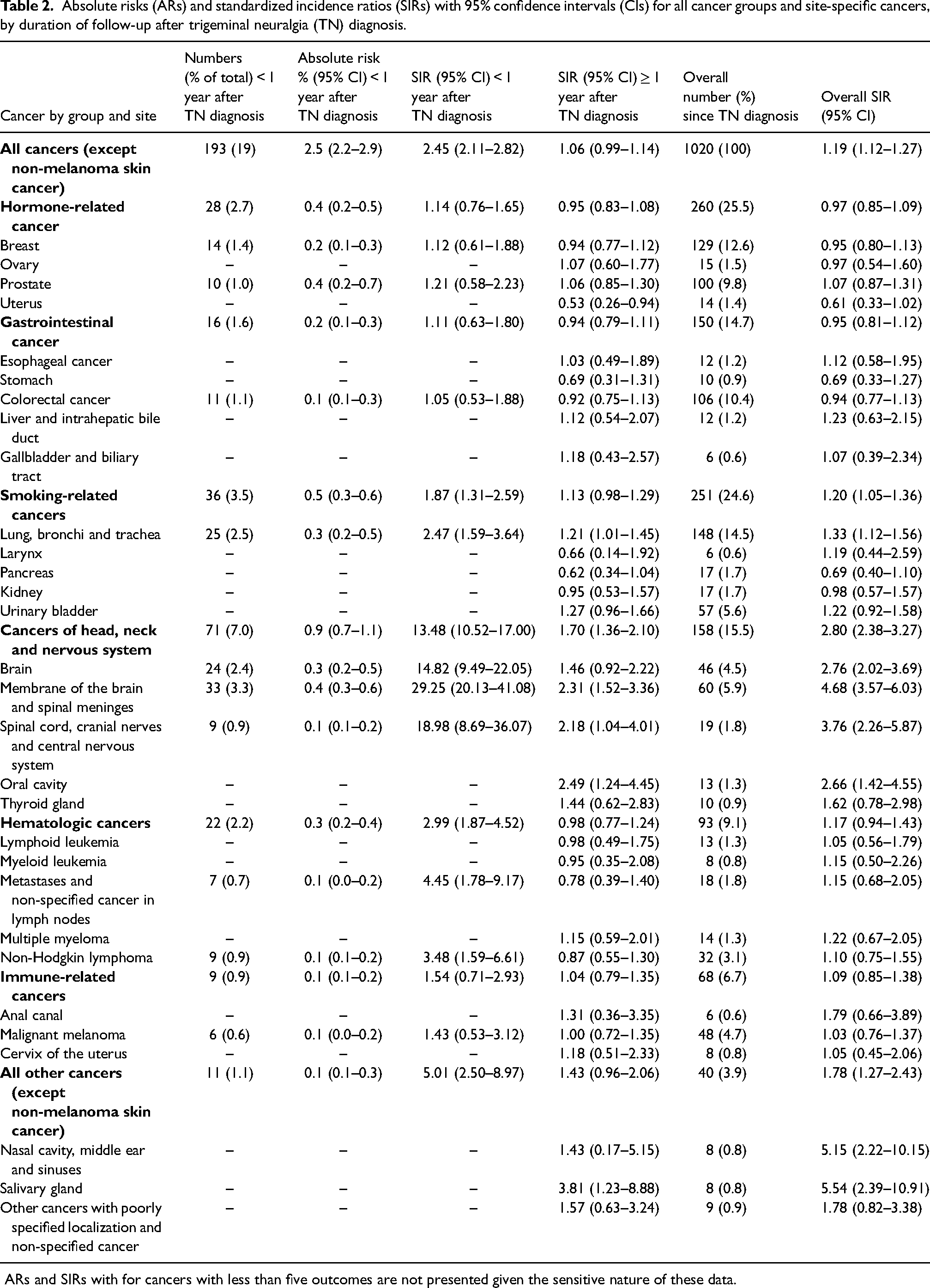
ARs and SIRs with for cancers with less than five outcomes are not presented given the sensitive nature of these data.
Cancer groups
For the study period overall, we observed elevated risks for cancers of the head, neck and nervous system (SIR = 2.80 (95% CI = 2.38–3.27)), smoking-related cancers (SIR = 1.20 (95% CI = 1.05–1.36)) and hematologic cancers (SIR = 1.17 (95% CI = 0.94–1.43)) (Table 2).
Within the first year of TN diagnosis, we observed elevated risk for cancers of the head, neck and nervous system (AR = 0.9% (95% CI = 0.7–1.1); SIR = 13.5 (95% CI = 10.5–17.0)), hematologic cancers (AR = 0.3% (95% CI = 0.2–0.4); SIR = 2.99 (95% CI = 1.87–4.52)) and smoking-related cancers (AR = 0.5% (95% CI = 0.3–0.6); SIR = 1.8 (95% CI = 1.31–2.59)).
Site-specific cancer diagnoses
Finally, our analysis considered site-specific cancer diagnoses. Several site-specific cancers demonstrated markedly increased SIR within the first year of TN diagnosis, including cancers of the brain (SIR = 14.82 (95% CI = 9.49–22.05)), membrane of the brain and spinal meninges (SIR = 29.25 (95% CI = 20.13–41.08)), spinal cord, cranial nerves and central nervous system (SIR = 18.98, (95% CI = 8.69–36.07)). Also, cancer sites in close relation to the head and neck had increased SIRs within the first year of TN diagnosis, including cancer of the lung, bronchia and trachea (SIR = 2.47 (95% CI = 1.59–3.64)), non-Hodgkin lymphoma (SIR = 3.48 (95% CI = 1.59–6.61)) and metastases and non-specified cancer in lymph nodes (SIR = 4.45 (95% CI = 1.78–9.17)) (Table 2).
Beyond one year of follow-up, sites of more distant relation to the cranial nerves had increased SIR, including lung, bronchi and trachea, oral cavity, meninges, spinal cord, cranial nerves and nervous system, and salivary gland. SIRs for liver and gall bladder, prostate, esophagus, urinary bladder, thyroid, anal canal and cervix- and ovary are based on a few observations, resulting in imprecise estimates with wide confidence intervals.
Secondary analyses
SIRs stratified by TN diagnosis (inpatient, outpatient and emergency department diagnosis) were similar across contact type for all follow-up periods. SIRs were similar according to gender. Within the first year after TN diagnosis, SIRs were increased for patients younger than 50 years. Beyond one year of follow-up, the SIR were similar across age groups. Stratified by COPD, the risk of cancer following TN diagnosis was increased for patients with concomitant COPD compared to patients without COPD. The risk was most prominent within the first year of TN diagnosis but persisted beyond one year of follow-up (see supplementary material, Table S3). The SIRs were similar across CCI score groups for all follow-up periods. Stratified analyses by patient characteristics are shown in the supplementary material (Table S3).
Discussion
The present study has examined cancer risk associated with TN diagnoses in Denmark over a 29-year period. We found that TN was associated with persistently increased risks of cancers of the head, neck and nervous system and cancers of the lung, bronchi and trachea. Importantly, we found that, in analyses restricted to cancer diagnoses made at least one year after the initial TN diagnosis, the elevated overall cancer rates persisted.
the findings from the present study are generally consistent with prior research that suggests TN is associated with increased rates of cancer of the head, neck and central nervous system. For example, prior systematic reviews and retrospective cohort studies have consistently demonstrated an association between TN and cancers of the central nervous system, causing direct pressure on the trigeminal nerve (15,16,19,21,23–25,37). The estimated point prevalence of brain cancers in the referred studies varied between 0.4% and 10.8% (primary or secondary tumor) for primary tumors of the cerebellopontine angle, meningiomas and posterior fossa tumors. The association found in previous studies may reflect brain cancer as the cause of TN, making TN a marker of cancer. TN could also be associated with other cancers through shared risk factors, where our study indicates that such a shared risk factor could be smoking. Notably, these prior studies did not explicitly consider or report the time elapsed from TN diagnosis to cancer diagnosis, nor did they consider cancers outside the central nervous system. Our analysis further extends the literature on TN and cancer by providing comprehensive consideration of cancers at sites other than the head and neck.
Our results demonstrate increased rates of cancer diagnoses within the first year of TN diagnosis. These findings may reflect increased rates of detection because of the more careful surveillance of patients diagnosed with TN. The persistent overall association between TN and cancers beyond the first year of TN diagnosis that we observed in the preent study could be a result of several underlying mechanisms. First, this finding could reflect compression of key structures by slow growing tumors. Space-occupying lesions in the central nervous system are assumed to produce the symptoms of TN, for example through dural stretch, thereby leading to diagnosis of TN before diagnosis of the underlying malignancy. Second, it is possible that the persistent association between TN and cancer is explained by underlying paraneoplastic processes. Although rare, this phenomenon could be reflected in the increased SIR of cancers of the membrane of the brain and spinal meninges beyond one year after TN diagnosis. A few descriptions of paraneoplastic syndromes in relation to TN has been described in the literature (38,39). In this context, diagnostic delay has been frequently reported for TN. In a Danish, prospective study of 158 patients with TN, the diagnostic delay was one year, although this delay may be longer in other settings 5 as a result of unpredictable periods of complete remissions, or because people may initially seek dental care if the mandibular branch is involved (1,5,40).
In Denmark, diagnosis and treatment of TN follows the recommendations of the European Academy of Neurology guidelines. These include recommended neuroimaging with magnetic resonance imaging to ensure correct diagnosis and distinguish among classical, primary and secondary TN (31). The persistent association between TN and cancers diagnosed more than one year after the initial TN diagnosis could also reflect diagnostic delays, perhaps reflecting some combination of delays in specialty care, delays in neuroimaging or trials of medications for presumed idiopathic TN.
Finally, the findings from the present study further suggest that TN and cancer may co-occur because of shared, underlying risk factors. For example, we found that the SIR for cancers of the lung, ronchi, and trachea was increased for cancer diagnoses made within one year and at least one year after the initial TN diagnosis. Smoking is an established risk factor for these cancers, and prior research has suggested that smoking modulates trigeminal pain processing (41). Hypertension (7) and atherosclerotic changes (8), both of which are associated with smoking, are also known risk factors for TN. We further observed an association between TN and cancers of the oral cavity, which may be attributed to the common underlying cause of smoking (42). Because data on smoking are incompletely captured in Danish registry data, we were unable to specifically examine smoking as a potential common cause of certain cancers and TN. Future research that examines the role of smoking and other potential common causes in the observed association between TN and various cancer types is needed.
Limitations
Although the present study leverages routinely collected registry data with high quality and complete long-term follow up the high validity of the DCR (29), generalizability to study populations outside of Denmark may be limited. Selection bias is minimized because the DNPR captures the entire Danish population (28). Outcome misclassification is also unlikely, given prior studies that demonstrate the high validity of the DCR (29). In our analysis, we were unable to differentiate between classical, secondary or idiopathic TN because we identified diagnoses of TN and subsequent cancer risk as a result of constraints related to using ICD-10 codes. Furthermore, TN is a clinical diagnosis and may be difficult to distinguish from other trigeminal and autonomic cephalalgias, such as trigeminal neuropathy, with which it shares some clinical features (43,44). Although uncertainty regarding the diagnosis of TN may be a source of exposure misclassification, we have no reason to believe assume that this uncertainty would be differential with respect to eventual cancer diagnosis; therefore, the associations would probably have been attenuated toward the null.
Conclusions
TN was associated with persistently elevated risk of cancers of the head, neck and nervous system, including cancers of the oral- and nasal cavity and salivary gland. After one year following TN diagnosis, the risk of cancer of the lung, bronchi, trachea and urinary bladder was also elevated, implying common symptoms risk factors, symptoms or causes of disease.
Clinical implications
Trigeminal neuralgia should prompt examination of underlying cancer. In particular, cancers of the head, neck and nervous system, including cancers of the oral- and nasal cavity, may be suspected when patients present with trigeminal neuralgia
Supplemental Material
sj-docx-1-cep-10.1177_03331024241292637 - Supplemental material for Trigeminal neuralgia and risk of cancer: A population-based cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024241292637 for Trigeminal neuralgia and risk of cancer: A population-based cohort study by Malene Risager Lykke, Holly C. Elser, Cecilia Hvitfeldt Fuglsang, Dóra Körmendiné Farkas and Henrik Toft Sørensen in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Center for Population Medicine and the Lundbeck Foundation, Grant R433-2023-1140.
Ethical statement
According to Danish law, registry-based research does not require ethical approval and informed consent but requires only permission from the Danish Data Protection Board. This study was recorded at Danish Data Protection Agency made by Aarhus University (record number 2016-051-000001/812.).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
