Abstract
Background/Hypothesis
Levcromakalim has previously been shown to induce attacks of migraine with aura in certain individuals. In this study, we tested the migraine-inducing effect of levcromakalim in a cohort of participants with migraine aura without headache.
Methods
In a double-blind, randomized, placebo-controlled cross-over study, eight adult participants with migraine with aura received intravenous infusions of levcromakalim and saline. Headache, aura and associated symptoms were evaluated for 24 h following administration of the study drug. The primary endpoint was occurrence of migraine-like attacks with or without aura in the 24-h observation period.
Results
Five participants (62.5%) developed migraine of any type following levcromakalim compared with three participants (37.5%) following placebo. No participants developed aura following levcromakalim.
Conclusion/Interpretation
Our findings suggest that the aura-inducing effect of levcromakalim is likely not based on direct induction of cortical spreading depression but may involve activation of the trigeminovascular system. This hypothesis should be further explored in future studies.
ClinicalTrials.gov identifier
NCT04905654
Introduction
Levcromakalim, an adenosine triphosphate-sensitive potassium (KATP) channel opener, triggers migraine-like headache in individuals with a history of migraine and non-migraine headache in healthy volunteers (1–4). The responsible mechanism might involve activation of the trigeminovascular system (TVS) causing cephalic vasodilation and activation of perivascular trigeminal pain fibers (5). However, the differential impact of TVS activation on individuals with or without a migraine history remains unknown. Attacks of migraine with aura (MA) without headache are unlikely to involve activation of the TVS.
In some individuals with a history of MA with headache, levcromakalim triggers usual attacks of migraine with aura accompanied by headache. Similarly, other substances such as calcitonin gene-related peptide (CGRP), sildenafil, and nitroglycerin, have shown a modest aura-inducing effect in individuals with a history of aura with headache (6,7). The recognized pathophysiological underpinning of migraine aura is cortical spreading depression (CSD). Levcromakalim might be able to directly trigger CSD by acting in the central nervous system, thereby giving rise to aura symptoms (5). Alternatively, levcromakalim-induced TVS activation may subsequently lead to CSD initiation. The interplay between CSD and the TVS remains an important research area, and further insights may guide development of treatments of MA in the future. Investigations of patients not expected to undergo activation of the TVS during migraine aura may be instrumental in this regard.
The present explorative study investigates the migraine-inducing effects of levcromakalim specifically in participants with a history of MA without headache, shedding further light on the complex interplay between KATP channels and migraine pathophysiology.
Methods
Design
The study was designed as a double-blinded, randomized, placebo-controlled cross-over study. Each participant underwent three visits, which included a preliminary screening session and two experimental days.
Participants
The full study methodology has been previously published (5). Briefly, individuals with at least six episodes of migraine aura in the past year were recruited, with at least 80% of these episodes being without any accompanying headache. Headache diagnoses were made according to the International Classification of Headache Disorders 3rd edition (8). Headache was defined as any painful or pressing sensation in the head. Participants were recruited via the out-patient clinic at the Danish Headache Center, advertising in public forums and on social media, through invitation via the Danish Blood Donor Study (9), and by referral from private practicing neurologists.
The Ethics Committee of the Capital Region, Denmark, approved the study (H-20049785). The study conformed to the Declaration of Helsinki of 1964 with later revisions. The study was registered at ClinicalTrials.gov (NCT04905654). All participants provided a written informed consent prior to any study-related activities.
Procedures
Participants were assigned to receive intravenous levcromakalim (20 ml of 50 µg/ml over 20 min) or placebo (20 ml of 0.9 mg/ml saline over 20 min) in random order in a double-blind cross-over study design (5). The study drug and placebo were prepared, blinded and randomized by the Capital Region Central Pharmacy. Headache characteristics, associated symptoms and aura symptoms were recorded every 10 min during the in-hospital phase (0–120 min) and every hour during the out-of-hospital phase (2–24 h). We measured blood pressure and heart rate every 10 min during the in-hospital phase.
Outcomes and measures
Our pre-defined primary endpoints were the differences in the incidence of migraine-like attacks of any type, migraine attacks with aura, or any headache within the 24-h period following the start of the infusion. For this substudy we were particularly interested in the incidence of migraine aura following infusion of levcromakalim and placebo, respectively. Migraine aura was considered present if symptoms fulfilled the International Classification of Headache Disorders, third edition (ICHD-3) criteria B and C for 1.2 migraine with aura. Migraine headache was considered present if ICHD-3 criteria C and D for 1.1 migraine without aura were fulfilled (moderate/severe headache defined as a pain intensity of 4 or above on a numeric rating scale (NRS) from 0–10), or if participants reported that headache mimicked usual migraine and used acute medication. Presence of any migraine was defined as either set of criteria being fulfilled.
Data and statistical analysis
We previously estimated that we needed to include 27 participants with MA without headache to demonstrate a difference in incidence of migraine attacks with aura without headache between levcromakalim and placebo infusion. The power calculation was based on a projected 50% induction rate of aura after levcromakalim and 10% after placebo (5).
Continuous variables are presented as median (range) or mean (standard deviation [SD]).
Data availability
Upon reasonable request, the corresponding author will provide the necessary data and materials to interested researchers for the purpose of academic scrutiny, reproducibility, and further scientific investigation.
Results
Participants
We invited approximately 400 individuals with a history of MA via phone or personal mail, and an additional approximately 150 individuals contacted us through our advertisements. Of these, we recruited nine of a planned 27 participants, and further recruitment was deemed unfeasible due to the low inclusion rate. Eight participants completed both experimental days. The mean age was 50.5 (SD 7.6) years, and the mean weight was 78.7 (SD 13.7) kg. All participants reported a history of visual aura, two reported sensory aura, and three reported speech and/or language aura. Three participants reported exclusively aura without headache in spontaneous aura episodes, and the remaining five participants reported headache in 1–20% of aura episodes. Two participants had a history of migraine without aura in addition to a history of migraine with aura.
Incidence of migraine attacks with and without aura, and any headache
Two participants developed aura following placebo (after 2 h 24 min and 22 h, respectively) compared to none following levcromakalim.
Five participants (62.5%) developed migraine of any type following levcromakalim compared with three participants (37.5%) following placebo (Table 1). Two participants developed migraine without aura both experimental days. The median peak headache intensity was higher following levcromakalim than following placebo (Figure 1).

Headache intensity scores. Individual and median headache scores on the active (levcromakalim) and placebo days on a numeric rating scale from 0–10. Abbreviations: NRS: Numeric rating scale.
Headache characteristics during spontaneous attacks of migraine with aura and on each experimental day.
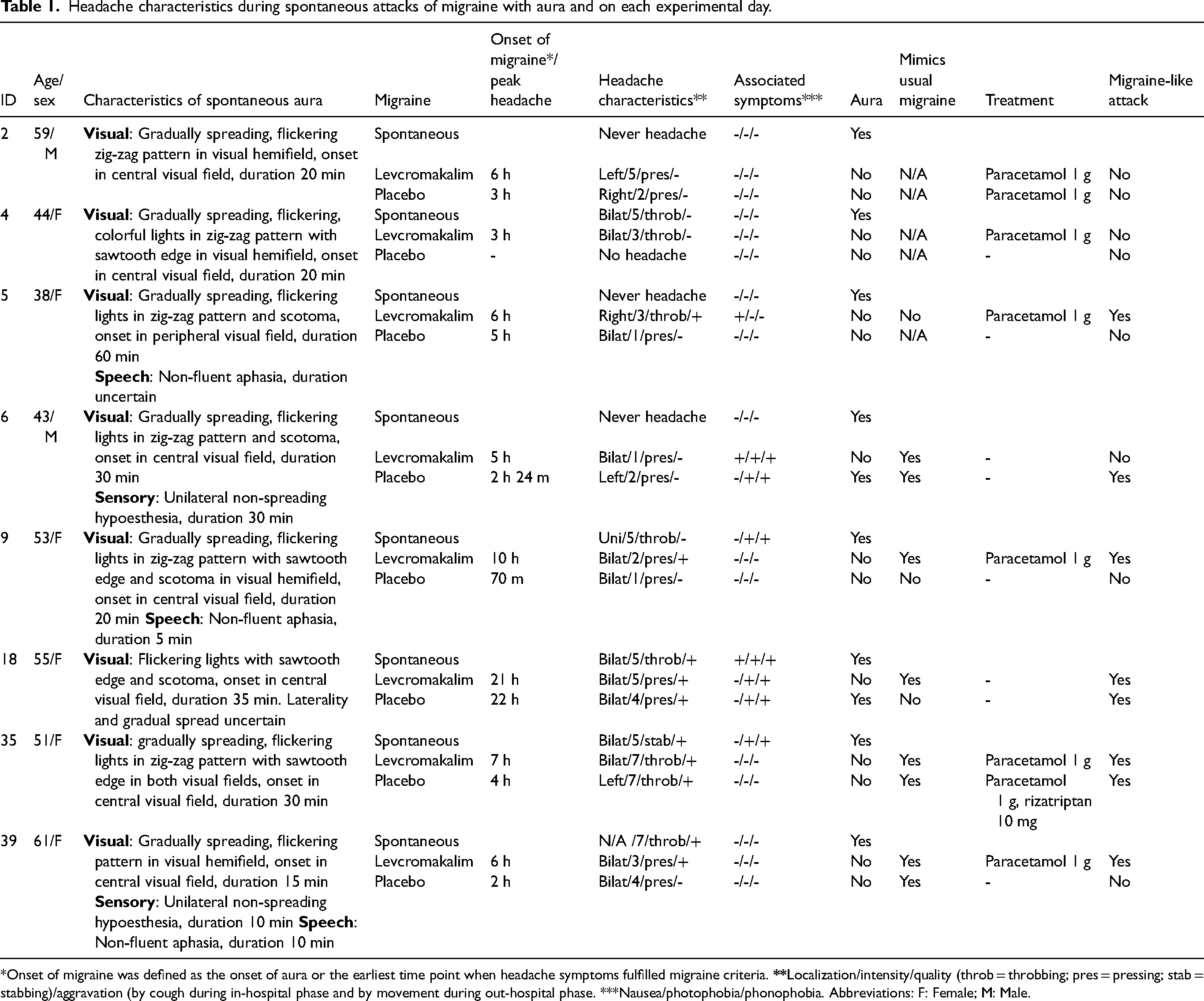
*Onset of migraine was defined as the onset of aura or the earliest time point when headache symptoms fulfilled migraine criteria.
Discussion
Our study focused on a distinctive cohort of individuals with a history of migraine aura without headache, representing the first investigation of migraine induction in this specific population. Median peak headache intensity post-levcromakalim infusion was higher than that following placebo, indicating TVS activation in our cohort. None of the participants reported migraine aura symptoms after levcromakalim. If levcromakalim directly induced CSD, we would our participants to develop migraine aura following levcromakalim. Since migraine aura episodes in these individuals likely depend on CSD initiation as a primary event and not TVS activation, this suggests that levcromakalim's aura-inducing effect likely does not rely on directly inducing CSD. Considering its pharmacological properties and limited animal data (10), there is a possibility that levcromakalim may cross the blood-brain-barrier in humans. However, our findings, alongside recent rat studies (11), argue against the notion that levcromakalim triggers CSD via direct neuronal or glial action, as KATP channel opening typically leads to cell membrane hyperpolarization, which would inhibit rather than provoke CSD. Our findings, while constrained by a small participant pool and a notable nocebo migraine-induction rate, align with previous studies demonstrating aura-induction following infusion of CGRP, which is not expected to cross the blood-brain-barrier (7,12). Further, anti-CGRP antibodies, which also act peripherally, have shown efficacy in reducing both migraine with and without aura frequency (13). If TVS-activation occurs secondarily to CSD, inhibitory modulation by anti-CGRP antibodies might not affect CSD, i.e., anti-CGRP antibodies would be expected to reduce migraine headache frequency while not affecting the frequency of migraine aura. Based on present and previous findings (2,5), we propose that levcromakalim likely triggers migraine aura through its action on the TVS. While distinguishing between induced and spontaneous migraine attacks may be challenging, the simplistic sequential hypothesis of aura-headache causality—where CSD precipitates headache—might not fully capture the complex aura-headache interplay (5).
Although only two participants had a history of migraine without aura, migraine-like headache without aura manifested in five participants following levcromakalim infusion and one following placebo infusion. These findings may indicate a predisposition to migraine headache among our participants. Two participants developed migraine aura following placebo; one shortly after the in-hospital phase and the other on the following day. As investigators and participants were all blinded, we speculate that these aura episodes may have been spontaneous aura episodes.
Limitations
Recruitment challenges prevented us from reaching our target of 27 participants, potentially significantly limiting the power and generalizability of our findings. However, given previous studies (2,5), we anticipated at least some participants out of the eight to develop aura following levcromakalim, rendering it unlikely that tripling number of participants would alter our outcomes.
Conclusions
In this explorative study involving participants with migraine aura without headache, levcromakalim did not induce migraine aura. Future studies should aim to explore how migraine aura may be triggered through the activation of the TVS. This could involve further studies of pharmacological triggers of the TVS not expected to cross the blood-brain-barrier.
Clinical implications
Levcromakalim did not induce migraine aura in a small group of participants with migraine aura without headache
Levcromakalim likely induces migraine aura via effects on the trigeminovascular system rather than direct induction of cortical spreading depression.
Further studies should evaluate the potential for activation for trigeminovascular system to trigger migraine aura.
Footnotes
Acknowledgements
We are grateful to Thomas Folkmann Hansen and Ole Birger Pedersen of the Danish Blood Donor Study for access to the Danish Migraine Population Cohort. The authors wish to thank all the study participants and the research staff at the Danish Headache Center. The authors additionally give thanks to doctors Jakob Møller Hansen, Lili Kokoti, Peter Thede Schmidt-Hansen, Janu Thuraiaiyah and Zixuan Alice Zhuang.
Author contributions
M.A, M.M.A-K, and A.H conceived and initiated the study. M.M.A-K, A.V.T., and S.R.O. participated in recruitment. A.V.T. and M.M.A-K. participated in data acquisition. A.V.T. analyzed the data and drafted the first version of the manuscript. All authors revised the manuscript for intellectual content, approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.V.T., M.M.A-K., and S.R.O. declare no competing interests. A.H. reports receiving personal fees from AbbVie, Eli Lilly, Lundbeck, Novartis, Teva and Pfizer. A.H. also serves as an associate editor of Headache. M.A reports receiving personal fees from AbbVie, Amgen, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva Pharmaceuticals outside of the submitted work. MA also serves as an associate editor of The Journal of Headache and Pain, and an associate editor of Brain.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a research grant from the Lundbeck Foundation (grant number R310-2018-3711).
