Abstract
Background
Data from some population-based studies have indicated an increased risk of atrial fibrillation (AF) among patients with migraine, particularly among individuals with migraine with aura. The present study aimed to assess the association between primary headache disorders and AF.
Methods
In a population-based 9-year follow-up design, we evaluated the questionnaire-based headache diagnosis, migraine and tension-type headache (TTH) included, collected in the Trøndelag Health Study (HUNT3) conducted in 2006–2008, and the subsequent risk of AF in the period until December 2015. The population at risk consisted of 39,340 individuals ≥20 years without AF at HUNT3 baseline who answered headache questionnaire during HUNT3. The prospective association was evaluated by multivariable Cox proportional hazard models with 95% confidence intervals (CIs).
Results
Among the 39,340 participants, 1524 (3.8%) developed AF during the 9-year follow up, whereof 91% of these were ≥55 years. In the multivariable analyses, adjusting for known confounders, we did not find any association between migraine or TTH and risk of AF. The adjusted hazard ratios (HRs) were respectively 0.84 (95% CI = 0.64–1.11) for migraine, 1.16 (95% CI = 0.86–1.27) for TTH and 1.04 (95% CI = 0.86–1.27) for unclassified headache. However, in sensitivity analyses of individuals aged ≥55 years, a lower risk of AF was found for migraine (HR = 0.53; 95% CI = 0.39–0.73).
Conclusions
In this large population-based study, no increased risk of AF was found among individuals with migraine or TTH at baseline. Indeed, among individuals aged ≥55 years, migraine was associated with a lower risk for AF.
Keywords
Introduction
Migraine, particularly migraine with aura (MA) has been suggested as a risk factor for cardiovascular disease and stroke (1,2). Among patients with MA, a recent meta-analysis that included prospective cohort studies noted an increased risk of stroke (hazard ratio 1.67; 95% confidence interval (CI) = 1.26–2.22) and increased cardiovascular mortality (hazard ratio (HR) = 1.27; 95% CI = 1.14–1.42) (3). Several mechanisms have been proposed for the relationship between migraine and ischemic stroke, including common cardio-cerebrovascular risk factors, cortical spreading depression, blood coagulation, platelet aggregation, paradoxical embolism, genetic predisposition and vascular dysfunction (4,5). Arial fibrillation (AF) is a major risk factor for stroke (6) and there is a possible link between migraine and AF. However, existing studies have demonstrated conflicting results (4,7–12). Two population-based large-scale follow-up studies from Denmark and the USA, as well as a Korean retrospective study, have found increased risk of AF among patients with MA (4,7,12). On the other hand, the association between migraine without aura (MO) and AF has been less evident (4,7). A few cross-sectional studies of selected groups including artificial intelligence-enabled ECG AF prediction study reported a higher prevalence of AF in migraine patients (8,9,13), whereas others did not (10,11). For example, among young stroke patients, the risk of AF in MA increased by five-fold compared with MO (8). Because systemic embolism is highly prevalent in people with AF (14), it is important to clarify the relationship between migraine (in particular MA) and AF. Studies reporting a positive association between MA and AF have hypothesized that AF might result from autonomic dysfunction during migraine attacks and is most evident in individuals with visual aura (7,15) and, alternatively, that emboli caused by AF may trigger cortical spreading depression (16). However, a recent systematic review highlighted that current knowledge for the association between headache disorders and AF does not allow firm conclusions relevant for clinical practice (17). So far, a large-scale follow-up study evaluating the relationship between primary headache disorders including migraine and tension-type headache (TTH) and AF in an unselected population is lacking. The main aim of this prospective, population-based, 9-years follow-up study was therefore to evaluate the risk associated with primary headache disorders for development of AF. Based on the previous studies, we hypothesized that individuals with migraine, particularly MA would have an increased risk of AF.
Methods
Study design
This was a large-scale population-based 9-year follow-up study evaluating the impact of questionnaire-based headache diagnosis collected in the third wave of the Trøndelag Health Study (HUNT3) performed between 2006 and 2008 (18) and the subsequent risk of confirmed AF until December 2015.
Study population
In HUNT3, all residents of Nord-Trøndelag County aged ≥20 years were invited to answer a large number of health-related questions in two sets of questionnaires (Q1 and Q2), Q2 including 14 questions about headaches. Participants were also invited to undergo a clinical examination measuring height, weight and blood pressure. The diagnosis of AF was verified by experts evaluating medical records from the two hospitals in Nord-Trøndelag County in the period between 2006 and December 2015. These AF data were then linked to HUNT3 data using the Norwegian personal identification number.
Included in the present study were individuals aged ≥20 years who answered headache questionnaire during HUNT3. Included were also those who self-reported previous myocardial infarction and stroke in HUNT3.
Excluded from the study were HUNT3 participants without response on headache question, and AF diagnoses before 2006 that included either paroxysmal or persistent AF. Furthermore, individuals with AF within 7 days after cardiac surgery, AF with no information on ECG or an uncertain diagnosis were excluded. Excluded were also those with AF as a result of other conditions/illness including acute phase of myocardial infarction or other serious conditions with major hemodynamic instability, including septic shock during or shortly after major non-cardiac surgery. However, recurrence of AF during the follow-up period and not related to other conditions was included as incident AF.
Among 93,860 individuals invited to HUNT3, 50,806 (54%) answered Q1, of whom 39,696 (42%) answered the self-administered headache questionnaire in Q2 (Figure 1). One specialist in cardiology and one specialist in internal medicine evaluated the medical records of the two hospitals in Nord-Trøndelag County (19). AF was confirmed in a total of 1988 individuals during the study period (2006–2015). Of the total 39,696 participants, we excluded individuals with AF before HUNT3 (n = 256), AF episodes within 7 days after cardiac surgery (n = 54), AF as a result of other conditions/illness including acute myocardial phase of infarction (n = 41) and diagnosis of AF with no information on ECG or an uncertain diagnosis (n = 5). Thus, in total, 39,340 subjects (including 1524 incident AF) who had headache data at baseline and follow-up AF status were included in the analysis (Figure 1). Among these, myocardial infarction was self-reported by 1320 (3.3%) individuals and stroke by 1083 (2.7%) individuals.

Flow diagram of the study population in HUNT3 and confirmed incident atrial fibrillation (AF) during follow-up until 2015.
Headache diagnoses
The self-administered headache questions were designed to determine whether participants suffered from headache and, if so, to assess the frequency, quality, intensity, location, associated symptoms and duration of headaches (18). Participants who answered “yes” to the screening question “Have you suffered from headache in the past 12 months?” answered 13 additional questions. Headache sufferers were classified into three groups (migraine, tension-type headache and unclassified headache) according to slightly modified criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) and were mutually exclusive (20). In the slightly modified criteria, individuals fulfill the migraine criteria even if the attack lasted less than 4 h, because the question “How long does the headache usually last?” did not ask about untreated attacks (21).
The validity of these questionnaire-based headache diagnoses in HUNT3 has been reported elsewhere (21). For headache, sensitivity was 88% and specificity was 86% (kappa value was 0.70); for migraine, sensitivity was 51% and specificity was 95% (kappa value was 0.50); and, for migraine with aura (MA), sensitivity was 50% and specificity was 95% (kappa statistic was 0.44). Considering migraine and tension-type headache (TTH) ≥ 1 day/month, these values changed to sensitivity, specificity and kappa statistics of 58%, 91% and 0.54 for migraine, and 96%, 69% and 0.44 for TTH.
Atrial fibrillation
The diagnoses of AF were retrieved from discharge records that were coded with ICD-10 code 148 at the two governmental hospitals in Nord-Trøndelag County. AF diagnoses were then verified by two physicians based on electrocardiographic criteria as recommended by the European Society of Cardiology (22). Incident AF included both paroxysmal and persistent AF diagnosed after 2006. The validation process of AF is presented previously (19). For AF, the sensitivity was 73.7% and specificity was 99.7%, positive predictive value was 88.5% and negative predictive value was 99.2% (19).
Confounders
In the present study, potential confounders were selected based on evidence from epidemiological studies that identified factors associated with both AF and migraine, including age, body mass index (BMI), smoking status, alcohol consumption and high blood pressure (23–29). Measurements for weight and height were performed by trained nurses at the HUNT baseline examination. BMI was calculated and categorized as <25 kg/m2, 25–29.9 kg/m2 or ≥30 kg/m2. Blood pressure measurement was determined as an average of the second and third readings and used as a continuous variable in the regression model. Self-reported data collected at baseline included smoking status (never, former, daily or occasionally), alcohol consumption (abstainers, light drinkers, moderate drinkers or heavy drinkers) and physical activity. Responses to physical activity were categorized as no physical exercise, ≤ 1 time per week, 2–3 times per week and almost everyday. Alcohol consumption was classified as abstainers (no alcohol during last year or never drank alcohol), light drinkers (<1 time per week), moderate drinkers (1–3 times per week) and heavy drinkers (4–7 times per week). Self-reported common chronic disorders including history of stroke and myocardial infarction has also been included.
Statistical analysis
Descriptive data at baseline in HUNT3 are presented with numbers and percentages for categorical variables, and with the mean ± SD for continuous variables. We used Cox proportional HRs with 95% CIs to assess the associations between headache types at baseline in HUNT3 and the subsequent risk of AF. Time was defined as years from admission or inclusion to diagnosis of AF. We censored participants without AF or those who did not meet the AF definition until the end of follow-up in December 2015. Risk for AF was calculated by headache diagnosis, using headache-free individuals as the reference category. The results are presented in three different models based on the number of potential confounders included. In model 1, we adjusted for age (continuous variable) and sex; in model 2, smoking status and BMI (categorical variable) were added. Other covariates such as high blood pressure (continuous variable), physical activity, alcohol consumption and comorbidities (self-reported stroke or myocardial infarction), use of over-the-counter (OTC) medications were added in model 3. Supplementary sensitivity analysis was performed using median age of 55 years as the cutoff. Statistical analyses were performed using STATA, version 17.0 (StataCorp LP, College Station, TX, USA).
Ethical approval
The Norwegian Data Inspectorate, the Norwegian Health Authority and the Regional Committee for Ethics in Medical Research had approved the HUNT3 study. In addition, the Regional Committee on Ethics in Medical and the Nord-Trøndelag Health Research approved the present study (ID 478096). All participants have provided their written informed consent.
Results
The baseline characteristics of the study population are presented in Table 1. The total population of 39,340 individuals had a mean age of 53 years and 56% of these were female. Mean BMI and total cholesterol were identical in all headache groups. Individuals with MA had a higher proportion of daily smokers (24%). Individuals who answered the headache questionnaire (n = 39,696) were somewhat older and less likely to smoke than the 11,110 HUNT3 participants who did not fill out the headache questionnaire (see supplementary material, Table S1).
Baseline characteristics of population at risk in HUNT3 with valid data of atrial fibrillation until 2015.

TTH = tension type headache; MA = migraine with aura; MO = migraine without aura; BMI = body mass index; HADS = Hospital Anxiety and Depression Scale; aCholesterol measured in mmol/L. bAlcohol abstainers during last year. cSelf-reported.
Headache disorders and atrial fibrillation risk
Of the 39,340 subjects without AF at baseline in HUNT3, 1524 (3.8%) developed AF over the 9-year follow-up period. The majority of incident AF occurred in the age group ≥55 years (91%) and AF was more common among men (59%) than women (41%). Baseline characteristics among the 1524 persons who developed AF are presented in the supplementary material (Table S2), showing that persons with MA were more likely to smoke and had higher HADS score in age group <55 years compared to other headaches groups. In the multivariable analyses using Cox proportional hazard model, no association was found between primary headache disorders including migraine and TTH at baseline, and the risk of AF (Table 2). In model 2, adjusted for age, gender, smoking status and BMI, the hazard ratios were 1.16 (95% CI = 0.86–1.27) for TTH, 0.84 (95% CI = 0.64–1.11) for migraine and 1.04 (95% CI = 0.86–1.27) for unclassified headache. Additional adjustment for alcohol consumption, systolic and diastolic blood pressure, physical activity, use of OTC medications and self-reported history of stroke or myocardial infarction did not change the results (Table 2). Furthermore, supplementary analyses evaluating the impact on sex and headache frequency were carried out, but no differences in the results were detected (see supplementary material, Tables S3 and S4). In supplementary sensitivity analyses using age of 55 years as cutoff, no association was found for headache types at baseline and risk of AF in individuals below 55 years. Conversely, in individuals aged ≥55 years, migraine and its subtypes were observed as a protective factor for AF (Table 3). Compared to headache-free participants, the hazard ratios were 0.53 (95% CI = 0.39–0.73) for migraine, 0.58 (95% CI = 0.38–0.87) for MA and 0.48 (95% CI = 0.30–0.78) for MO.
Hazard ratio (HR) with 95% confidence interval (CI) of atrial fibrillation during study period (2006–2015).
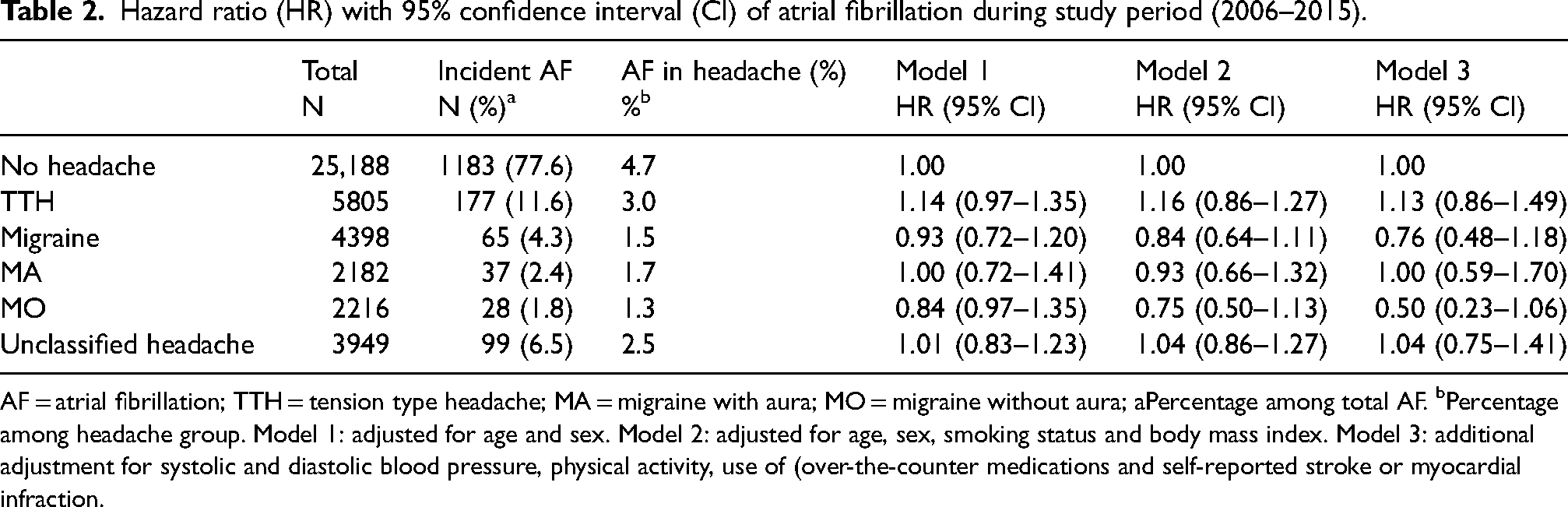
AF = atrial fibrillation; TTH = tension type headache; MA = migraine with aura; MO = migraine without aura; aPercentage among total AF. bPercentage among headache group. Model 1: adjusted for age and sex. Model 2: adjusted for age, sex, smoking status and body mass index. Model 3: additional adjustment for systolic and diastolic blood pressure, physical activity, use of (over-the-counter medications and self-reported stroke or myocardial infraction.
Hazard ratio (HR) with 95% confidence interval (CI) of atrial during study period (2006–2015) by sex and age.

TTH = tension type headache; MA = migraine with aura; MO = migraine without aura. Model 1: adjusted for age and sex. Model 2: adjusted for age, sex, smoking status and body mass index.
Discussion
In this large-scale population-based 9-year follow-up study, no association was found between types of headache at baseline and the subsequent risk of AF. In the supplementary analyses categorizing the population by age, individuals with migraine aged ≥55 years had lower risk of AF compared to headache-free participants.
Comparison with previous studies
In contrast to our results, a 20-year follow-up study among 11,939 individuals aged 45 to 65 years from the Atherosclerosis Risk in Community (ARIC) noted an increased incidence of AF in patients with migraine with visual aura (HR = 1.30; 95% CI = 1.03–1.62) (7). Furthermore, in a nationwide Danish 19-year follow-up study of 51,032 migraine patients and 510,320 matched controls, the risk of AF or atrial flutter was higher in individuals with MA (HR = 1.31; 95% CI = 1.11–1.55) (4). However, the analysis by years since migraine diagnosis categories showed that the risk was significantly higher only in the first year after MA diagnosis (HR = 2.73; 95% CI = 1.73–4.32) (4), whereas no such association was found in the subsequent years of diagnosis. The HR was 1.22 (95% CI = 0.91–1.63) for >1–5 years since MA diagnosis and 1.21 (95% CI = 0.97–1.51) for >5–19 years since diagnosis (4). Several methodological differences such as follow-up time, participation rate, statistical power and case definition could at least partly explain the difference in results. The follow-up duration in the present study was approximately 10 years shorter than the previous population-based studies (4,7), which might impact the possibility of finding a positive result between migraine and risk of AF. Furthermore, in contrast to the low participation rate of 42% in the present study, which impacted the generalization of the results, the other studies had high participation rates. Regarding statistical power, the present study had higher number of participants (n = 39,340) than the US study (7), but much lower number than the Danish study (4). Furthermore, there are also differences in the case definition. The present study used hospital validated medical record-based diagnoses for AF and validated self-administered headache diagnoses accepting headache lasting less than 4 h for migraine diagnoses. In the US study, the diagnosis of AF was hospital-based, and headache diagnosis was based on interviews conducted by trained interviewers using somewhat stricter migraine criteria fulfilling headaches for at least 4 h (7). Thus, a possibility of including more severe migraine patients only with high frequency of migraine episodes could have shown a positive association. In the Danish study, AF diagnosis was obtained from the patient registry and hospital-based migraine diagnosis (4). The authors of this study acknowledged that the validity of such registry for migraine diagnosis remains uncertain (4). Furthermore, several outcomes were measured in the Danish study, including myocardial infarction, stroke, peripheral artery disease, venous thromboembolism, heart failure and AF (4). Thus, the outcome that occurred first decided the diagnosis. By contrast, in the present study AF was the only outcome measured, and we excluded AF as a result of myocardial infarction, cardiac surgery or other serious conditions.
A Korean nationwide retrospective study demonstrated that, regardless of aura status, the severity of migraine significantly increased the risk of AF (12). The diagnosis of migraine required at least two clinic visits during a year, and severe migraine was defined as a migraine requiring medical attention or active therapeutic intervention or chronic migraine that persists for more than three months. This study is prone to selection bias and not directly comparable to the present study because only people using the National Health Insurance Service were included. Furthermore, the proportion of MA was lower (0.12%) than in previous population-based studies (2.1% to 4.1%), and the MA:MO ratio of 1:21 was in the lower end compared to the ratio of 1:2–4 observed in other studies including HUNT (30,31). Another recent retrospective cross-sectional study from the USA predicting artificial intelligence-enabled ECG AF observed increased risk of AF in patients with MA (13). These findings are not supported by the results from our unselected study population because no association between migraine frequency or migraine subtypes and AF was detected.
The majority of AF diagnoses were observed in individuals aged ≥55 years (91%), in men (59%) and in non-headache groups (77.6%). As expected, there was a lower incidence of migraine associated with increasing age. The higher AF incidence among non-headache group can be described by the differences observed in the baseline characteristics. Compared to patients with migraine, individuals in non-headache group were older and had a higher prevalence of comorbidities, including higher mean systolic blood pressure and a higher self-reported history of stroke and myocardial infarction.
Interestingly, participants aged ≥55 years with migraine and its subtypes had a lower risk for incident AF, which is inconsistent with the previous follow-up study (7). It should be noted that the mean age of participants in Danish study was 10 years lower compared to the present study (35 vs. 45 years) (4). The reason for the finding of lower risk of AF among older migraine patients is unclear. However, because multiple comparisons were performed, we cannot rule out the possibility of these negative statistical findings by change alone. A much more speculative explanation is the possible link between the use of migraine preventive medication and a potential cardioprotective effect that lowers the risk of AF. With advancing age, there is a natural rise in the proportion of patients with hypertension and other cardiovascular diseases. However, on the other hand, the need of preventive medication decreases in older migraine patients. A recent US study detected that the risk of ischemic stroke among older people with migraine differed by migraine medication used (32). Unfortunately, comprehensive information about prescription of medication was not available in the present study. Information on the use of OTC medications for treatment of headache was available, but adjustment for the use of OTC medications did not affect the main results.
Interpretation
In this unselected general population, no increased risk of AF was found for individual with migraine with or without aura. In line with our results, genetic studies have also failed to show common genetic factors for migraine and AF (33,34). Data by AF type are not available; thus, the possibility that migraine is only linked with paroxysmal AF cannot be ruled out. Of relevance for such a theory, electrocardiographic changes such as sinus bradycardia, non-specific ST-T changes and right bundle branch block have been identified during migraine attacks (15). Furthermore, QTc interval and P-wave dispersion can be increased during migraine attacks compared to attack-free periods (15). Case reports and case series have also indicated increased incidence of AF during migraine attacks, but such data are limited by selection bias and low numbers (15,35). The impact of use of prescribed medication on the association between migraine and AF could not be evaluated in the present study. Further investigations of the underlying mechanisms linking use of migraine medication and AF are therefore warranted.
Strengths and limitations
The major strengths of the present study are the population-based 9-year prospective follow-up design and the expert validation of AF diagnoses in hospital medical records including ECG. Furthermore, the questionnaire-based headache diagnosis has been validated by expert review. In the present study, we used headache-free individuals as the reference group. By contrast, previous population-based studies (4,7) have compared migraine patients with migraine-free individuals, potentially including bilateral headaches or TTH in the reference group.
The limitations of the present study include the fact that the headache diagnoses were based on self-administered questionnaire with moderate sensitivity for migraine diagnosis (21). Thus, diagnostic accuracy of questionnaire-based diagnoses of headache may not be optimal, and the misclassification cannot be ruled out. Furthermore, we lack information on migraine medication including prescription of preventive medication and attack treatment, which may impact the risk of AF (36–38). In addition, only hospital-based AF diagnoses were included. Thus, we may have missed individuals with AF treated just outside the two hospitals in the country. A Swedish study reported that 22% of patients with AF were seen only in primary care centers (39). On the other hand, the consequence of a lower sensitivity of the diagnosis in focus is less threatening to the validity of an epidemiological study compared to a lower specificity (19,40). The present study also lacks follow-up data to assess the endpoint. The information of myocardial infarction and stroke used in the adjusted analyses were self-reported, which may limit their validity. In addition, we could not ignore the influence of unmeasured or residual confounding factors, including lack of information about diabetes and details about previous heart disease, which could influence the results. Furthermore, only 42% of the invited population in HUNT3 has information on headache and AF diagnoses. Thus, generalization of the results to the entire population must be made with caution. However, a non-participant study performed in HUNT3 did not find a significant difference in headache prevalence between participants and non-participants (42.1% vs. 44.7% in women and 27.8% vs. 27.3% in men). Likewise, the migraine prevalence was also almost similar (10.9% vs. 11.8% in women and 5.0% vs 5.3% in men) (41).
Conclusions
In this large-scale, population-based follow-up study, we found no association between TTH, MA, MO or unclassified headache and the subsequent risk of AF. In future follow-up studies evaluating the risk of AF in migraine patients, comprehensive evaluation of prescribed migraine medication is recommended to further clarify potential underlying protective mechanisms.
Neither migraine with or without aura, nor tension-type headache was associated with increased risk of atrial fibrillation. In this population-based study, individuals with migraine aged ≥55 years had lower risk of atrial fibrillation compared to headache-free participants. The study findings do not support screening of migraine patients for atrial fibrillation.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241254517 - Supplemental material for Migraine and risk of atrial fibrillation: A 9-year follow-up based on the Trøndelag Health Study
Supplemental material, sj-docx-1-cep-10.1177_03331024241254517 for Migraine and risk of atrial fibrillation: A 9-year follow-up based on the Trøndelag Health Study by Samita Giri, Erling Tronvik, Håvard Dalen, Hanne Ellekjær, Jan P Loennechen, Alexander Olsen and Knut Hagen in Cephalalgia
Footnotes
Acknowledgements
The Trøndelag Health Study (HUNT) is a collaboration between HUNT Research Centre (Faculty of Medicine and Health Sciences, The Norwegian University of Science and Technology NTNU), Trøndelag County Council, Central Norway Regional Health Authority and the Norwegian Institute of Public Health.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The Regional Committee on Ethics in Medical and the Nord-Trøndelag Health Research has approved the present study (ID 478096). All participants have provided written their informed consent.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
