Abstract
Background
Idiopathic intracranial hypertension is a secondary headache disorder potentially causing visual loss. Neurofilament light chain is a candidate, prognostic biomarker, but further studies of neuronal biomarkers are needed. Our objective was to investigate neurofilament light chain in cerebrospinal fluid (cNfL) and plasma (pNfL), amyloid-beta 42 (Aβ-42), total-tau and phosphorylated-tau in cerebrospinal fluid in new-onset idiopathic intracranial hypertension.
Methods
Prospective case-control study including new-onset idiopathic intracranial hypertension and age, sex and BMI matched controls. Biomarkers were compared between patients and controls and related to papilledema, visual fields and opening pressure.
Results
We included 37 patients and 35 controls. Patients had higher age-adjusted cNfL (1.4 vs. 0.6 pg/mL, p-adjusted < 0.001), pNfL (0.5 vs. 0.3 pg/mL, p-adjusted < 0.001) and total-tau/Aβ-42 (0.12 vs. 0.11, p-adjusted = 0.039). Significant, positive linear correlations were found between cNfL, pNfL, total-tau/Aβ-42 and opening pressure. Patients with severe papilledema had elevated cNfL compared to mild-moderate papilledema (median cNfL: 4.3 pg/mL (3.7) versus 1.0 pg/mL (1.4), p-adjusted = 0.009). cNFL was inversely associated with perimetric mean deviation (r = −0.47, p-adjusted < 0.001).
Conclusions
cNfL, pNfL and total-tau/Aβ-42 were elevated in new-onset idiopathic intracranial hypertension. cNfL was associated with severity of papilledema and visual field defects at diagnosis. This indicates early axonal damage. Neurofilament light chain is a candidate biomarker for disease severity.
Introduction
Idiopathic intracranial hypertension (IIH) is strongly associated with female sex, childbearing age, and obesity, and is characterized by chronically elevated intracranial pressure (ICP) (1,2). The burden of disease is increasing in parallel with pandemic obesity (3,4). Blindness is a feared complication occurring in 1–2% and 50% suffer persistent visual field defects (5–7). Early identification of patients with a poor prognosis can prevent visual loss, but no prognostic biomarkers exist (8–10).
Recently, elevated cerebrospinal fluid (CSF) neurofilament light chain (NfL) in patients with IIH was shown to be associated with higher opening pressure (OP), moderate-severe papilledema and optic nerve damage, suggesting NfL as a prognostic biomarker for irreversible optic nerve damage (9,11,12). NfL is a cytoskeletal protein specific for neurons, and elevated levels reflect neuroaxonal damage to large myelinated axons, e.g. the optic nerves, and can be measured in CSF and blood (13). Elevated NfL is found in Alzheimer’s disease (AD), multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), traumatic brain injury and atypical Parkinson’s disease (13,14).
Additionally, neuropsychological complaints are common in IIH, and reduced processing speed and reaction time has been shown (15–17), begging the question whether the elevated NfL levels exclusively reflects optic nerve damage, or whether the chronic ICP-elevation also causes cortical damage. This may be answered by investigating a broader profile of biomarkers related to neuronal damage. Previously, neuro-specific enolase has been shown to be normal in IIH (8), but other neuronal biomarkers have not been investigated. AD biomarkers amyloid-beta 42 (Aβ-42), total tau-protein (t-tau) and phosphorylated tau-protein (p-tau) could provide novel insight into whether tau pathology or altered Aβ-42 clearance is present in IIH. T-tau is a micro-tubule protein particularly abundant in thin, unmyelinated cortical axons. It stabilizes neuronal axons and is associated not only with AD, but also with acute cortical damage e.g. after stroke or traumatic brain injury (18–20). Elevated p-tau (tangle formation) is specific for AD (20). Increased t-tau and NfL in IIH might suggest extensive axonal damage cortically and subcortically. Conversely, isolated NfL-elevation suggests neuronal damage in large, myelinated axons, e.g., the optic nerves, without cortical affliction. Aβ-42 is a waste product linked to glymphatic dysfunction (21). As CSF dysregulation is a hallmark of IIH, it has been proposed that glymphatic dysfunction is implicated in the pathogenesis, possibly resulting in amyloid-beta accumulation (22). Studies find that other conditions besides AD can affect Aβ-42, e.g. traumatic brain injury studies have found decreased Aβ-42 and increased tau (23). Altered Aβ-42 clearance has also been associated with normal pressure hydrocephalus (24,25).
Our aim was to investigate NfL, Aβ-42, t-tau, p-tau in CSF and NfL in plasma from patients with new-onset IIH compared to healthy age, sex, and BMI matched controls. We hypothesized that CSF dysregulation and ICP elevation in IIH could result in altered Aβ-42 clearance, increased NfL (cNfL) and t-tau compared to controls. Important secondary outcomes were associations between biomarkers at baseline and clinical findings at baseline and ocular remission.
Methods
Study population
This was a prospective case-control study consecutively including patients with clinically suspected, new-onset IIH (26) at two tertiary centres (Danish Headache Center (DHC), Rigshospitalet-Glostrup and Department of Neurology, Odense University Hospital (OUH)) from January 2018-April 2021. Healthy controls were recruited from July 2020-May 2021 on social media platforms. Inclusion criteria for patients were diagnosis with new-onset, definite IIH according to the diagnostic criteria (26) and ≥18 years old. Inclusion criteria for healthy controls were age, sex and BMI matched to the patient cohort and headache frequency ≤1 day/week. Healthy controls were recruited from the community and samples were taken exclusively for research purposes. Exclusion criteria for all participants were: pregnancy, breastfeeding, prior IIH, previous treatment for CSF disorders, recent CSF withdrawal (14 days), known neurodegenerative disorders, recent or severe head trauma, secondary causes of elevated ICP (e.g. anaemia), significant comorbidities, conditions contraindicating lumbar puncture (LP) (e.g. coagulopathies), signs/symptoms of ICP-dysregulation (controls). Participants with previous, minor concussions were not excluded. There is no overlap between the study population presented and our previous study on NfL in IIH (9).
Baseline investigations
Screened patients had a standardized diagnostic workup to confirm or exclude IIH (history, neurological and neuro-ophthalmological examination, routine blood collection, LP including OP and CSF status, cerebral MRI and venography). Healthy controls were rigorously screened for neurological or neuro-ophthalmological disease, frequent headache, any non-primary headache disorders or other signs of ICP-dysregulation. Their work-up included fundus examination using scanning laser ophthalmoscopy (Compass, Padova, Italy) (27), history, neurological examination, routine blood collection and LP including OP and routine CSF status. LP was standardized as previously described elsewhere (28).
Neuro-ophthalmological findings were assessed by a neuro-ophthalmologist (SMH, EAW, LDM). Papilledema was graded according to Frisén (29). Visual field exams were performed by automated perimetry with a Humphrey Field Analyzer (24-2, Zeiss systems) at OUH or an OCTOPUS 900 perimeter (30-2, dynamic, Haag-Streit) at DHC. A perimetric mean deviation score (MD) was calculated, and the patient excluded if the neuro-ophthalmologist found significant signs of poor cooperation. Optical coherence tomography (OCT) was obtained by Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany) to image the posterior segment of the eye. The macula and optic nerve head were imaged with Heidelberg Spectralis eye tracking and automated real-time averaging features. Scans were repeated if necessary to obtain a good quality (signal to noise ratio >25 dB). The optic nerve head was imaged in high resolution while the macular scan was in high-speed mode, 20° × 20°, with fovea in the center (5.9 mm). High resolution scans of the optic nerve head were recorded with ≥30 frames. Macula was imaged with ≥9 frames. Circle scan 12.0° (3.5 mm) was used for retinal nerve fiber layer (RNFL) measurement. Ganglion cell layer (GCL) and RNFL were measured using Heidelberg Eye Explorer software. The semi-automatic segmentation method for GCL measurement was used (30).
Follow-up (ocular remission)
Patients were followed with neuro-ophthalmological examination until ocular remission (remission of papilledema). Bilateral optic nerve atrophy was a combined assessment of OCT findings, visual field defects and fundus images by the neuro-ophthalmologist. OP and biomarkers were not repeated.
Sample preparation and analysis
CSF samples were collected and stored in polypropylene tubes (31). CSF was centrifuged at 400G for 10 minutes and CSF-supernatant was frozen within 3 hours (−80°C). Samples were analysed at the Department of Clinical Biochemistry, Aarhus University Hospital, a routine laboratory accredited by the Danish Accreditation Fund (DANAK) according to the DS/EN ISO/IEC 15189 standard. CSF Aβ-42, t-tau and p-tau were analyzed with Electrochemiluminescence immunoassay (ECLIA) using Cobas 8000 (Roche Diagnostics GmbH, Penzberg, Germany). cNfL and pNfL were measured on a single-molecule array (Simoa) HD-1 analyzer (Quanterix Corp, Massachusetts, USA). The measuring range was 0.2–45000 pg/mL for cNfL, 0.2–1800 pg/mL for pNfL, 200–1700 pg/mL for Aβ-42, 80–13000 pg/mL for t-tau and 8–120 pg/mL for p-tau. When results exceeded this, they were evaluated as the highest or lowest detectable value. A subset of cNfL samples (n = 10) were analyzed at Rigshospitalet-Glostrup (Neuroimmunology Research Laboratory) due to logistic reasons (same assay).
Statistics
Sample size was estimated to 32 participants in each group based on previous studies of Aβ-42 in normal pressure hydrocephalus (NPH) (24) and traumatic brain injury (23) with an 85% power and alpha 0.05 to detect a 30% reduction in Aβ-42.
Frequencies and percentages described categorical variables. For continuous variables, means and standard deviations or medians and interquartile range (IQR) were used depending on normality of the data. For comparison of BMI, age and OP (controls vs patients) the Welch t-test was used. Differences between neuronal biomarkers (controls vs patients) were analyzed using the Wilcoxon rank sum test as data did not follow a normal distribution (Table 1). For categorical variables Fishers exact test was used (papilledema, atrophy). For correlation analyses between neuronal markers (cNfL, pNfL, Aβ-42, t-tau, p-tau, t-tau/Aβ-42, p-tau/Aβ-42) and OP, RNFL, GCL and visual fields Spearman correlation was used, and a correlation matrix was created (Figure 1). Simple linear regression described the correlations between cNfL, pNfL, Aβ-42, t-tau, p-tau, t-tau/Aβ-42, p-tau/Aβ-42 and OP, RNFL, GCL and visual fields. Regression analyses were carried out with and without log transformation to best meet the conditions of a linear model and diagnostic regression plots were examined. To account for the age-dependent variations observed in healthy individuals (32,33) a normalization procedure was implemented for cNfL and pNfL. cNfL values were divided by the mean cNfL value for individuals of the corresponding age as determined by Vågberg et al. (32). Similarly, pNfL values were divided by the mean pNfL value for individuals of the corresponding age using the upper limit of the interval reported by Hviid et al. (33). A significance level of p < 0.05 was used for all analyses. All results related to biomarkers were corrected for multiple testing using the false discovery rate (FDR) method. The upper limit of a standard issue manometer in Denmark is 50 cm CSF, five patients had an OP exceeding this and were evaluated to the highest measurable value. Papilledema, OCT measurements and visual fields were evaluated as ‘worst eye’. Patients with missing data (legends Tables 1–2 and Figure 2) were excluded from the given analysis. Statistical analyses were performed in R Studio (version 2022.07.1 + 554).
Demographics and clinical findings.
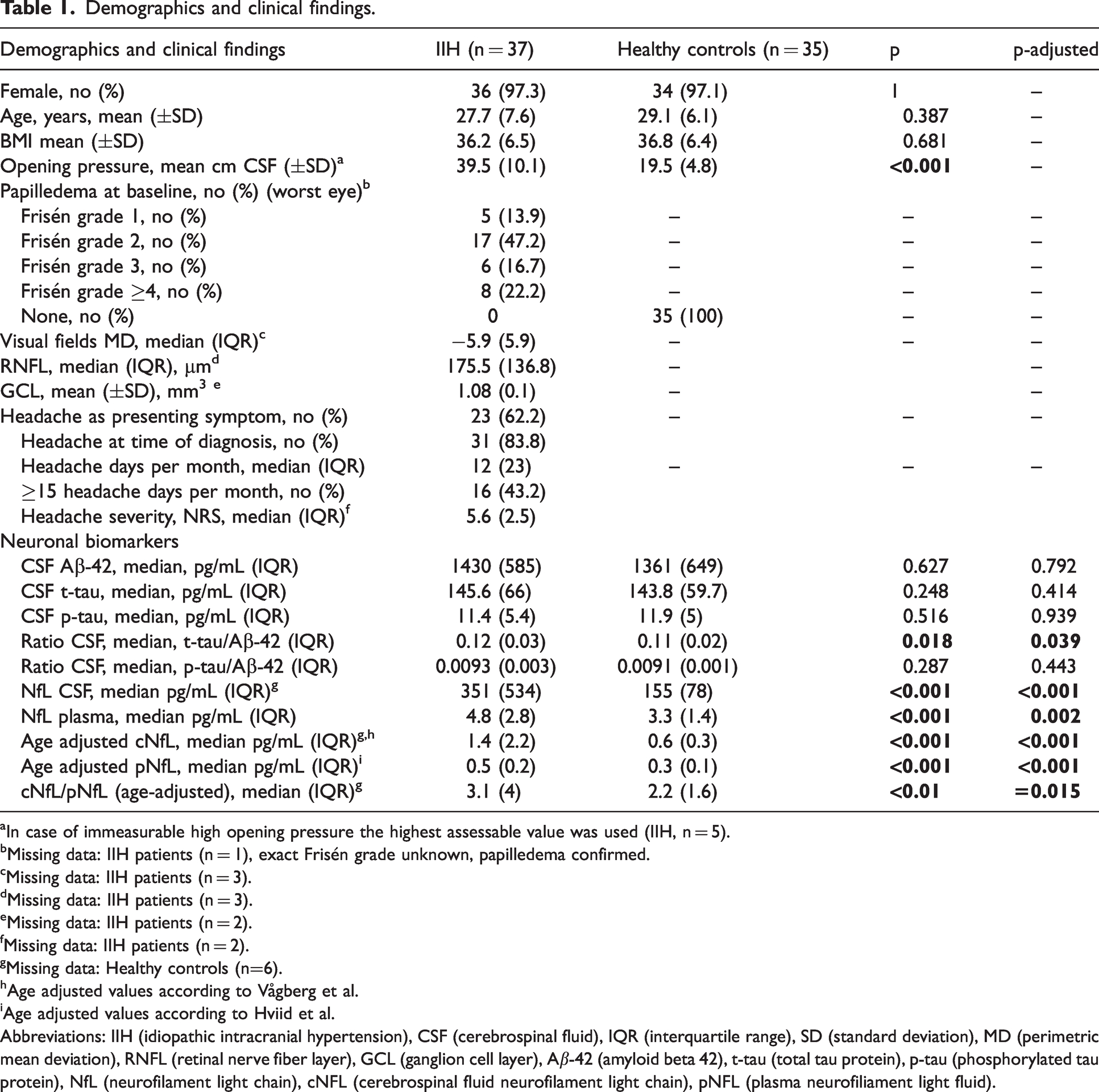
In case of immeasurable high opening pressure the highest assessable value was used (IIH, n = 5).
Missing data: IIH patients (n = 1), exact Frisén grade unknown, papilledema confirmed.
Missing data: IIH patients (n = 3).
Missing data: IIH patients (n = 3).
Missing data: IIH patients (n = 2).
Missing data: IIH patients (n = 2).
Missing data: Healthy controls (n=6).
Age adjusted values according to Vågberg et al.
Age adjusted values according to Hviid et al.
Abbreviations: IIH (idiopathic intracranial hypertension), CSF (cerebrospinal fluid), IQR (interquartile range), SD (standard deviation), MD (perimetric mean deviation), RNFL (retinal nerve fiber layer), GCL (ganglion cell layer), Aβ-42 (amyloid beta 42), t-tau (total tau protein), p-tau (phosphorylated tau protein), NfL (neurofilament light chain), cNFL (cerebrospinal fluid neurofilament light chain), pNFL (plasma neurofiliament light fluid).

Spearman Correlation matrix. Abbreviations: IIH (idiopathic intracranial hypertension), CSF (cerebrospinal fluid), NfL (neurofilament light chain), Aβ-42 (amyloid beta 42), t-tau (total tau protein), RNFL (retinal nerve fiber layer), MD (mean deviation), GCL (ganglion cell layer).
Clinical findings (ocular remission).
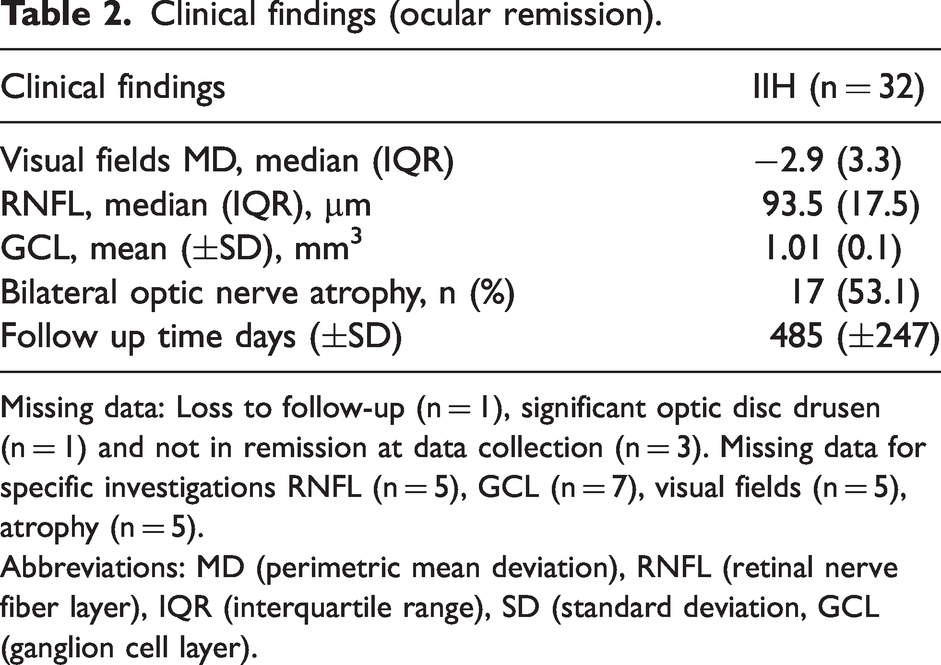
Missing data: Loss to follow-up (n = 1), significant optic disc drusen (n = 1) and not in remission at data collection (n = 3). Missing data for specific investigations RNFL (n = 5), GCL (n = 7), visual fields (n = 5), atrophy (n = 5).
Abbreviations: MD (perimetric mean deviation), RNFL (retinal nerve fiber layer), IQR (interquartile range), SD (standard deviation, GCL (ganglion cell layer).

Inclusion of IIH patients and healthy controls. Abbreviations: IIH (idiopathic intracranial hypertension), IIH-WOP (Idiopathic intracranial hypertension without papilledema).
Standard protocol approvals, registrations, and patient consents
The study was approved by the local ethical committee (Region of Southern Denmark, project ID: S-20170058) and followed the Helsinki declaration and Danish data regulations. Participants provided written informed consent.
Data availability
De-identified data can be shared with researchers who provide a methodologically sound proposal. It is a requirement by Danish law that a data processing agreement is signed. Raw neuro-ophthalmological data are shared locally for technical reasons. Data are available two years after publication. Proposals can be directed to the corresponding author.
Results
We evaluated 186 patients with suspected, new-onset IIH, 37 were eligible, 149 were excluded. We included 35 healthy controls. Inclusion is shown in Figure 2 along with reasons for exclusion and alternative diagnoses in excluded patients (legend, Figure 2).
Demographics and clinical characteristics are given in Table 1. Patients and controls were matched for sex (97.3% female versus 97.1% female, p = 1), age (27.7 years versus 29.1 years, p = 0.387) and BMI (36.2 versus 36.8, p = 0.681). Patients had higher OP than controls (39.5 cm CSF versus 19.5 cm CSF, p < 0.001). At baseline 5 (13.9%) patients presented with Frisén grade 1 papilledema, 17 (47.2%) with Frisén grade 2, 6 (16.7%) with Frisén grade 3 and 8 (22.2%) patients with Frisén grade ≥4. In patients baseline median MD and RNFL were −5.9 (IQR: 5.9) dB and 175.5 (IQR: 136.8) μm, respectively, and mean GCL was 1.08 (SD: ±0.1) mm3. In 62.1% (n = 23) of IIH patients a new or changed headache was the presenting symptom, and 83.8% (n = 31) reported having headache at diagnosis. The median number of headache days was 12 (IQR: 23) days/month, and 43.2% (n = 16) had headache at least 15 days/month. Median headache severity (NRS, Numerical Rating Scale 0–10) was 5.6 (IQR: 2.5).
Neuronal biomarkers at baseline
IIH patients had higher age-adjusted cNfL and pNfL compared to controls (median cNfL: 1.4 (IQR: 2.2) versus 0.6 (IQR: 0.3) pg/mL, p-adjusted < 0.001 and pNfL: 0.5 (IQR:0.2) versus 0.3 (IQR: 0.1) pg/mL, p-adjusted < 0.001) and higher t-tau/Aβ-42 ratio (0.12 (IQR)0.03 versus 0.11 (IQR: 0.02), p-adjusted = 0.039). The ratio cNfL/pNfL (age-adjusted) was significantly higher in patients with IIH compared to controls (3.1 (IQR: 4) versus 2.2 (IQR: 1.6), p-adjusted = 0.015). There was no significant difference in CSF concentration of Aβ-42 (1430 versus 1361 pg/mL, p-adjusted =0.792), t-tau (145.6 versus 143.5 pg/mL, p-adjusted =0.414), p-tau (11.4 versus 11.9 pg/mL, p-adjusted = 0.939) or p-tau/Aβ-42 ratio (0.0093 versus 0.0091, p-adjusted = 0.443) between patients and healthy controls (Table 1).
Age adjusted cNfL and pNfL were significantly correlated to CSF OP (cNfL: Spearman rho = 0.44 p = 0.001, pNfL: Spearman rho = 0.38, p = 0.002) (Figure 1 and 3). We found a positive linear correlation between age-adjusted cNfL (log) and OP (r = 0.475, p-adjusted < 0.001) and age-adjusted pNfL (log) and OP (r = 0.38, p-adjusted = 0.003). Furthermore, we found a positive linear correlation between (log)t-tau/Aβ-42 ratio and OP (r = 0.36, p-adjusted = 0.009). Before correction for multiple testing log(t-tau) was positively, weakly, correlated with OP (r = 0.28, p = 0.018), however this was not significant after correction (p-adjusted = 0.092) (Figure 3). There was no correlation between Aβ-42, p-tau, p-tau/Aβ-42 and OP (data not shown).

Biomarkers and CSF opening pressure. NfL CSF: Age adjusted values according to Vågberg et al. NfL plasma: Age adjusted values according to Hviid et al. Abbreviations: IIH (idiopathic intracranial hypertension), CSF (cerebrospinal fluid), NfL (neurofilament light chain), Aβ-42 (amyloid beta 42), t-tau (total tau protein).
Patients with severe papilledema (Frisén grade ≥4) had higher age-adjusted cNfL compared to patients with mild-moderate papilledema (median age-adjusted cNfL: 4.3 pg/mL (IQR: 3.7) versus 1.0 pg/mL (IQR: 1.4), p-adjusted = 0.009, Figure 4). Likewise, there was a positive linear correlation between RNFL and age-adjusted cNfL (Figure 5, r = 0.48, p-adjusted = 0.004). Baseline MD was inversely correlated with age-adjusted cNfL (Figure 6, r = −0.47, p-adjusted <0.001). We found no associations between remaining biomarkers and ophthalmological data (data not shown).

Baseline papilledema (Frisén grade) and NfL in patients with IIH. NfL CSF: Age adjusted values according to Vågberg et al.
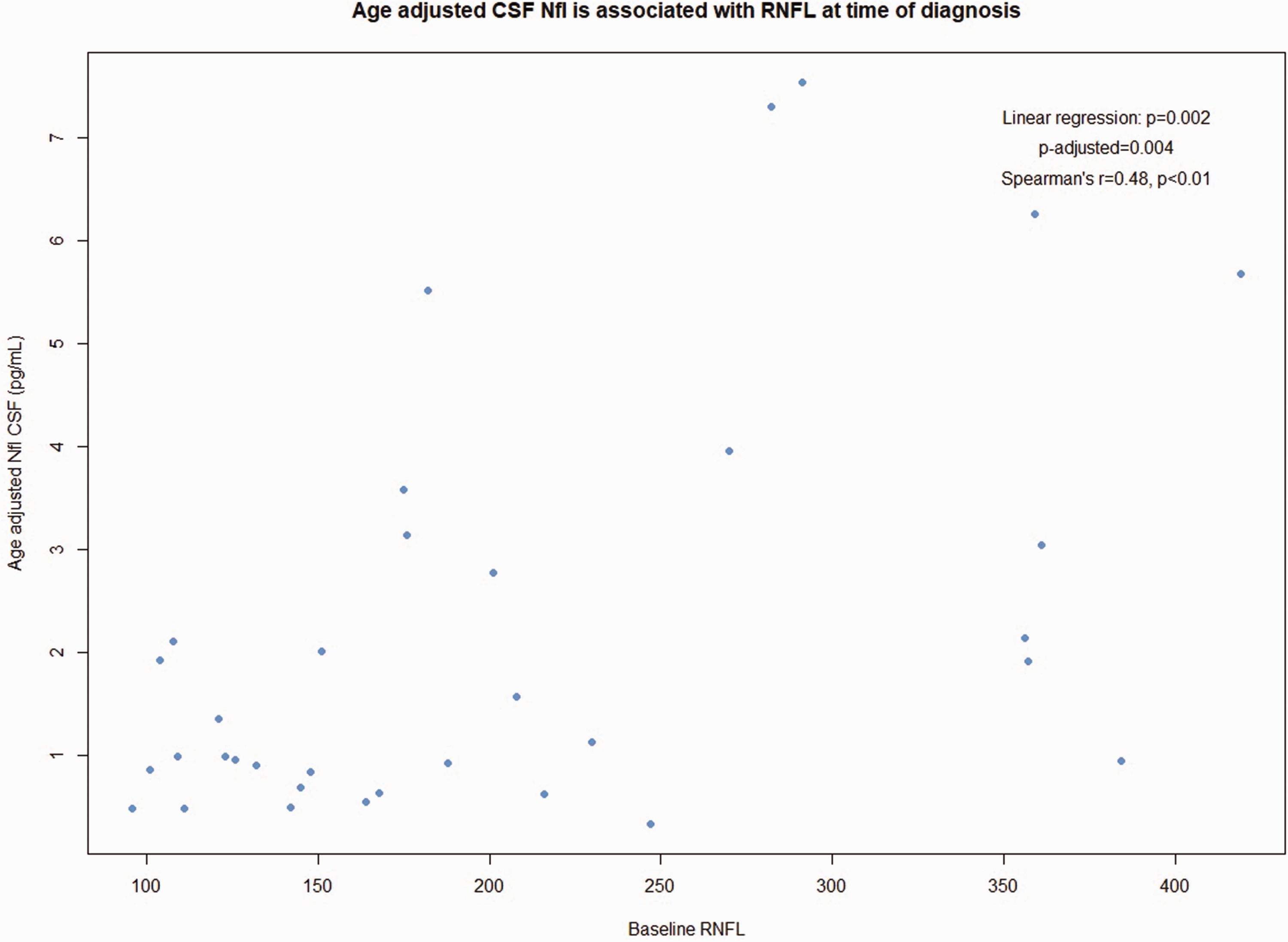
Baseline papilledema (OCT-RNFL) and CSF NfL. Abbreviations: CSF (cerebrospinal fluid), NfL (neurofilament light chain), OCT (optical coherence tomography), RNFL (retinal nerve fiber layer).

Baseline visual fields (PMD) and CSF NfL. Abbreviations: CSF (cerebrospinal fluid), NfL (neurofilament light chain), PMD (perimetric mean derivation).
Ocular remission
Follow-up data was available for 32 patients (mean follow-up time 485 (SD: 247) days). Median perimetric MD and RNFL were −2.9 (IQR: 3.3) dB and 93.5 µm (IQR: 17.5), respectively. Mean GCL was 1.01 (SD: 0.1) mm3. Bilateral optic nerve atrophy was diagnosed in 53.1% (n = 17, Table 2). We found no associations between age-adjusted cNfL or pNfL at diagnosis and GCL, RNFL, MD or bilateral optic nerve atrophy at remission (data not shown).
Discussion
Establishing novel biomarkers in IIH is an important research priority (34), nevertheless, only few have been investigated (8–10). We present a prospective case-control study investigating NfL, Aβ-42, t-tau and p-tau in IIH. Our aim was to investigate the type and character of neuronal damage and altered CSF dynamics in new-onset IIH. We demonstrate that cNfL and pNfL are higher in new-onset IIH compared to controls, indicating that axonal damage is present at diagnosis. Progression of optic nerve damage, and subsequent visual loss, is a feared complication of IIH, and the elevated NfL levels early in the course of the disease highlight that IIH is far from a benign disorder.
Importantly, NfL was associated with severity of papilledema at diagnosis (OCT and Frisén score). cNfL was also associated with functional optic nerve impairment at diagnosis, as the visual fields were poorer in patients with cNfL elevation. Furthermore, cNfL and pNfL levels correlated positively with OP in accordance with previous studies (9). This indicates that a disease state with high OP, severe papilledema and visual field defects at diagnosis is associated with more extensive axonal damage. We correlated the biomarkers to outcome at ocular remission (bilateral atrophy, GCL, RNFL and MD), but found no associations to baseline cNfL or pNfL. Moreover, cNfL and pNfL levels in the present study were lower than in our previous study of cNfL in IIH (9). In both studies, the same assay and the same method was used for age-adjusting the findings. However, in our first study, we analyzed CSF and serum samples, which had been collected at different time-points of the participants’ course of disease (9). In the current manuscript, we only analyzed CSF and serum samples which had been collected at diagnosis, explaining the lower NfL levels in CSF and serum compared to our first study. In both of our studies, NfL concentrations in CSF correlated with severity of papilledema. However, whereas NfL concentrations in CSF correlated with persistent bilateral visual field defects and bilateral optic nerve atrophy at remission in our first study, we did not find this prognostic impact in the current study, although we used the same outcomes (bilateral optic nerve atrophy) in both studies.
CSF and blood samples used in the first study were from patients referred to our hospitals due to insufficient or delayed treatment receiving a lumbar puncture to lower intracranial pressure while optimizing medical treatment. Thus, these patients' higher and prognostic NfL levels in CSF reflect a significant damage of their optic nerves due to delayed and insufficient treatment, whereas the lower NfL levels in CSF and blood samples found in the current study reflect that the samples were collected early, at diagnosis, and the patients got immediate and sufficient treatment preventing clinically, objectifiable long-term sequelae.
Thus, low NfL levels correlate with disease severity and seem to reflect minor, clinically not yet objectifiable optic nerve damage whereas higher NfL levels correlate with both disease severity and ophthalmological sequelae as they seem to reflect clinically objectifiable optic nerve damage. In agreement with previous studies, we show that there is a highly significant linear correlation between pNfL and cNfL (r = 0.54, p < 0.001)(9).
We found no significant difference in Aβ-42, t-tau, p-tau or p-tau/Aβ-42 ratio between patients with IIH and controls. Hyperphosphorylation pathology (elevated p-tau) was not present in IIH. Consequently, our study finds no direct evidence of altered AB-42 dynamics, tau pathology or major cortical damage. Nevertheless, as was shown in a previous study, and replicated here, patients with IIH have an increased cNfL/serum NfL ratio compared to controls, which is associated with OP (12). It has been suggested that this difference is caused by reduced outflow of CSF in IIH, and consequently, reduced permeability of NfL from the CNS. Such changes in CSF dynamics have been proposed to be part of IIH pathophysiology, and it has been speculated that this could be related to glymphatic congestion in IIH (12). Interestingly, in our patients with IIH there was also a significantly higher t-tau/Aβ-42 ratio than controls and this too correlated with OP. Elevated t-tau/Aβ-42 and p-tau/Aβ-42 ratios reflects Aβ-42 accumulation and neuronal damage associated with elevated tau-proteins in AD (35–37). This finding may reflect a subtle change in the biomarker levels, which is associated with ICP, even though we found no overall difference in Aβ-42, t-tau, and p-tau. In AD elevated t-tau/Aβ-42 and p-tau/Aβ-42 ratios have been associated with prediction of future dementia in cognitively normal individuals (35,36), however, the t-tau/Aβ-42 ratio in patients with IIH was much lower than what is seen in AD, and within the normal range (35,37). The increased t-tau/Aβ-42 ratio is considered a sign of Aβ-42 deposition, causing neuronal damage in AD, but, in our group of younger subjects the increased ratio – if anything – more likely reflects mild neuronal injury in combination with altered Aβ-42 clearance. It has been hypothesized that impairment of Aβ-42 clearance in various neurological diseases might be caused by glymphatic dysfunction (38). A recent imaging study investigating the glymphatic system in IIH found increased enhancement and reduced clearance of the MRI tracer Gadobutrol in the frontal and temporal regions of patients with IIH compared to age and sex matched controls (39). This suggests an impaired glymphatic system in IIH, and interestingly, these areas match the areas typically affected by amyloid and tau deposition in AD (39). The elevated t-tau/Aβ-42 ratio and cNfL/pNfL-ratio in the present study justifies future studies of possible glymphathic congestion and reduced clearance of Aβ-42 in IIH.
There was no association between t-tau and neuro-ophthalmological findings reflecting that t-tau is not sensitive enough to assess disease severity or optic nerve damage.
Strengths and limitations
The significant strengths were the prospective design, inclusion of healthy controls and measurement of several neurodegenerative biomarkers. This study was the first to investigate NfL, Aβ-42, t-tau, p-tau and ratios t-tau/Aβ-42 and p-tau/Aβ-42 in a prospective study of patients with new onset, treatment-naïve IIH. Patients were included at diagnosis after a systematic work up at two specialized centers and followed until ocular remission. Healthy controls were matched for sex, age, and BMI, and thoroughly screened for neurological disease. As NfL is influenced by age and BMI (40) potential bias was minimized. These significant strengths allow us to characterize the entire neurodegenerative biomarker profile in new onset IIH, and confirm previous findings relating to NfL. As NfL is currently the only CSF-biomarker with prognostic potential in IIH this is highly clinically relevant. Our approach diminishes confounding factors and bias e.g., effects of age, long-term chronic headache or large differences in disease duration, treatment response or weight changes. In Denmark there is access to high-quality medical care, free of charge, which serves to minimize delayed help seeking and social biases.
Our study has several limitations. The neuronal biomarkers investigated might increase following longer disease duration, as seen in NPH, where an increase in p-tau and a greater magnitude of the decrease in CSF Aβ-42 was seen in patients suffering from NPH more than one year (24). Normal NfL values in blood were assessed using serum (33) whereas our measurements were derived from plasma. A disparity, although unlikely, could exist. Furthermore, although samples were collected at diagnosis, our study has no firm information on how long patients had active disease prior to diagnosis. The study is underpowered to detect subtle differences in biochemistry or rare outcomes (e.g., blindness). We did not perform cognitive testing, prospective follow-up of headache history or serial biomarker measurements. It would be highly relevant to include this in future studies. Generalizability may be limited as patients with IIH were included at tertiary care centers.
Conclusion
We found elevated cNfL and pNfL in new-onset IIH compared to matched controls whereas there was no difference in any other neuronal biomarkers. cNfL levels were associated with severity of papilledema, visual field defects and OP at diagnosis. This profile suggests that elevated cNfL is related to optic nerve damage, and that NfL is a potential biomarker of disease severity in IIH. The ability to detect early elevation of NfL in newly diagnosed patients underscores the need for early treatment to prevent neuroaxonal damage and permanent loss of vision, once again disproving that IIH is a benign secondary headache disorder.
Key findings
Novel biomarkers are needed in the management of idiopathic intracranial hypertension (IIH). Neurofilament light chain (NfL) is elevated in IIH compared to controls, while other neuronal markers are unchanged. This isolated NfL elevation is associated with more severe disease, particularly optic nerve damage. Future studies addressing the use of neurofilament light chain as a prognostic biomarker in IIH are needed.
Contributor statement
KS and JJK contributed equally (joint first authorship).
JJK: Substantial contribution to conception and design of study, acquisition, analysis, collection and interpretation of data. Drafting and revision of paper.
KS: Substantial contribution to conception and design of study, acquisition, analysis, collection and interpretation of data. Drafting and revision of paper.
RHJ: Substantial contribution to conception and design of study, acquisition and interpretation of data as well as drafting and revision of paper.
TP: Substantial contribution to design, analysis and interpretation of data and revision of paper.
CSK: Substantial contribution to design, analysis and interpretation of data and revision of paper.
SGH: Substantial contribution to conception and design of study, interpretation of data and revision of paper.
SMH: Substantial contribution to data analysis, acquisition and interpretation as well as revision of paper.
EAW: Substantial contribution to data analysis, acquisition and interpretation as well as revision of paper.
LDM: Substantial contribution to data analysis, acquisition and interpretation as well as revision of paper.
DB: Substantial contribution to conception and design of study, acquisition and interpretation of data as well as drafting and revision of paper.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024241248203 - Supplemental material for Neurofilament light chain is elevated in patients with newly diagnosed idiopathic intracranial hypertension: A prospective study
Supplemental material, sj-pdf-1-cep-10.1177_03331024241248203 for Neurofilament light chain is elevated in patients with newly diagnosed idiopathic intracranial hypertension: A prospective study by Katrine Svart, Johanne Juhl Korsbæk, Rigmor Højland Jensen, Tina Parkner, Cindy Søndersø Knudsen, Steen Gregers Hasselbalch, Snorre Malm Hagen, Elisabeth Arnberg Wibroe, Laleh Dehghani Molander and Dagmar Beier in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KS: No conflicts of interest. JJK: Received funding from the Lundbeck Foundation, Rigshospitalet-Glostrup and Odense University Hospital. RHJ: Paid lectures for Pfizer, Eli-Lilly, Merck, TEVA, Novartis, Lundbeck and Allergan. Investigator in clinical trials with Eli-Lilly, Novartis and Lundbeck; Director of Danish Headache Center, Lifting The Global Burden of Headache and Founder of Master of Headache Disorders at University of Copenhagen. Recived research funding from University of Copenhagen, Rigshospitalet, Lundbeck Foundation, The Medical Society in Copenhagen, NovoNordisk Foundation and Tryg Foundation. TP: No conflicts of interest. CSK: No conflicts of interest. SGH: No conflicts of interest. SMH: No conflicts of interest. EAW: Medical Advisory Board for StatuManu ICP ApS. Received research funding from Øjenforeningen (Fight for Sight, Denmark) and the Synoptik Foundation. LDM: No conflicts of interest. DB: Presentations (TEVA, Novartis, Pfizer), Travel Support (Allergan, TEVA, Pfizer, Abbvie), Advisory Boards (Novartis, Lilly, Teva, Lundbeck, Pfizer, Abbvie) and Participations in clinical trials (TEVA, Lundbeck, Novartis, Lilly, Novo Nordic Foundation) outside the submitted work (other headache disorders), receiving grants from Odense University Hospital and Rigshospitalet, University of Copenhagen during the conduction of the work.
Ethic statement
The study was approved by the local ethical committee (Region of Southern Denmark, project ID: S-20170058) and followed the Helsinki declaration and Danish data regulations. Participants provided written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lundbeck Foundation [R-276-2018-403-4], Candys Foundation [grant number 2015-146], Rigshospitalet and Odense University Hospital. Foundation between Rigshospitalet and Odense University Hospital 2016 [R25 A1320], 2019 [69-A3346] and 2021 [123-A5083].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
