Abstract
Background
Functional anatomical research proposed the existence of a bilateral trigeminal ascending system although the anatomy trajectories of the trigeminothalamic connections cranial to the pons remain largely elusive. This study therefore aimed to clarify the anatomical distributions of the trigeminothalamic connections in humans.
Methods
Advanced deterministic tractography to an averaged template of diffusion tensor imaging data from 1065 subjects from the Human Connectome Project was used. Seedings masks were placed in Montreal Neurological Institute standard space by use of the BigBrain histological dataset. Waypoint masks of the sensory thalamus was obtained from the Brainnetome Atlas.
Results
Tractography results were validated by use of the BigBrain histological dataset and Polarized Light Imaging microscopy. The trigeminothalamic tract bifurcated into a decussating ventral and a non-decussating dorsal tract. The ventral and dorsal tracts ascended to the contralateral thalamus and ipsilateral thalamus and reflected the ventral trigeminothalamic tract and the dorsal trigeminothalamic tract, respectively. The projection of the ventral trigeminothalamic tract and the dorsal trigeminothalamic tract to both thalami confirm the existence of a bilateral trigeminothalamic system in humans.
Conclusions
Because our study is strictly anatomical, no further conclusions can be drawn with regard to physiological functionality. Future research should explore if the dorsal trigeminothalamic tract and the ventral trigeminothalamic tract actually transmit signals from noxious stimuli, this offers potential in understanding and possibly treating neuropathology in the orofacial region.
Introduction
In general, it is believed that each hemisphere of the human brain represents the contralateral side of the body, including the orofacial region. More specifically, trigeminal afferents synapse in the medullary trigeminal spinal nucleus and/or the pontine principal sensory nucleus (PSN), which together form the trigeminal sensory nuclear complex (TSNC) (1). From the TSNC, second-order neurons decussate as they ascend towards the ventroposterior medial nucleus in the contralateral thalamus from which thalamic radiations further project to cortical regions (2). However, a bilateral trigeminal system has been suggested by evidence derived from functional neuroimaging studies (3–5), animal-based tracing studies (6–8) and microscopical evaluation of the trigeminal fiber distribution in the human pons (9). The existence of a bilateral trigeminothalamic pathway, although new with regard to humans, has been well-known in animals (for an overview, see Henssen et al. (1)). Based on similar primate anatomical features, the two trigeminal pathways were named: 1) the ventral trigeminothalamic tract (vttt); and 2) the dorsal trigeminothalamic tract (dttt). The vttt sprouts from the ventral part of the PSN and the rostral trigeminal spinal nucleus, whereas the origins of the dttt lie in the dorsal PSN and the caudal trigeminal spinal nucleus. It is believed that the vttt forms a decussating ascending pathway and the dttt is a predominantly, non-decussating ascending system (1) (Figure 1). The course of the trigeminothalamic connections rostral to the level of the pons, however, have never been investigated anatomically in humans. Precise knowledge of the neuroanatomical pathways conducting nociceptive information remains of crucial importance for sophisticated neuromodulatory treatment of pain disorders. On a functional level, on the other hand, knowledge of a bilateral trigeminal projection system was found to have a significant impact on trigeminal pain alleviation by use of neuromodulation therapy (10).
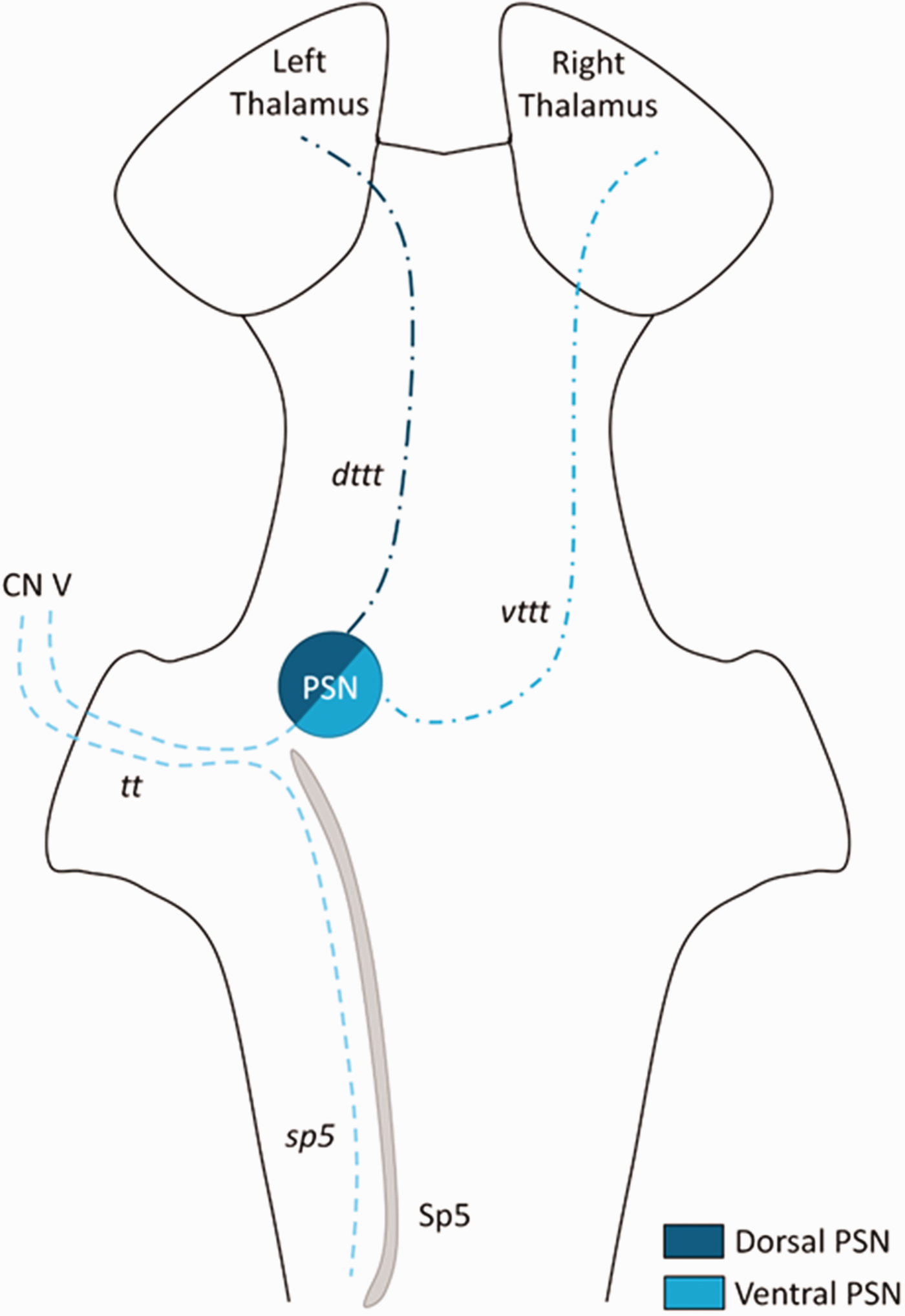
Schematic overview of the trigeminal system as proposed by Henssen et al. (1). CN V, trigeminal nerve (fifth cranial nerve); dttt, dorsal trigeminothalamic tract; PSN, principal sensory nucleus; Sp5, trigeminal spinal nucleus; sp5, trigeminal spinal tract; tt, trigeminal tract; vttt, ventral trigeminothalamic tract.
The armamentarium of the neuroscientist to investigate brain structure has expanded over the past decennia by use of magnetic resonance imaging (MRI), diffusion-tensor imaging (DTI), tractography (11) and digitized histology (12,13). DTI concerns a specific MRI protocol in which the diffusion of water is measured. It is based on the concept that water does not diffuse freely in all directions when restricted by micro-architecture of the brain (e.g. cell membranes, lipoproteins); this phenomenon is called diffusion anisotropy. This diffusion anisotropy is used in tractography when computer algorithms are used to track connections between different regions of interest (14–16). In general, two types of tractography methodologies exist: deterministic tractography and probabilistic tractography, each with its own strengths and limitations. Limitations of deterministic algorithms are the uncertainty of accuracy of the fiber orientation, susceptibility to noise and inter-researcher variability (16), although large tractography projects, including connectome mapping, preferably use deterministic tractography algorithms as these could result in a fully connected connectome (15–19).
This study set out to further elucidate the decussating and non-decussating trigeminothalamic connections in humans. First, histological data of the Big Brain Project (13) in the standard Montreal Neurological Institute (MNI) space was used to define the location of the trigeminal tract in the pons. Second, connectome-based tractography was applied to a diffusion tensor imaging (DTI) template of 1065 subjects, derived from the Human Connectome Project (HCP). Third, the connectome-based tractography results were verified by use of histological datasets of the whole human brainstem.
Materials and methods
Human Connectome Project
The HCP Consortium, led by Washington University, the University of Minnesota and Oxford University, is a (National Institute of Health) NIH-funded project aiming to construct an averaged human connectome in order to facilitate research on the relationship between anatomical connectivity and function. A neuroimaging database and behavioral database of healthy young adults has been constructed over the years. Every three months, imaging data have been released, freely available to the scientific community. All the images were constructed using a single customized Siemens 3T Skyra Connectome Scanner (20,21). The present study used a template of all data of the HCP (i.e., HCP-1065) for connectome-based tractography approach. The HCP project is supported by the National Institute of Dental and Craniofacial Research, the National Institute of Mental Health (NIMH) and the National Institute of Neurological Disorders and Stroke. Collectively, the HCP is the result of efforts of co-investigators from the University of Southern California, Martinos Center for Biomedical Imaging at Massachusetts General Hospital, Washington University, and the University of Minnesota.
Connectome-based tractography
The HCP-1065 template was created with the diffusion MRI data from 1065 subjects (575 female, 490 male, mean age 28.74 (range 22–37)) from the HCP (2017 Q4, 1200-subject release). Data was acquired using a multishell diffusion scheme and the b-values were 990, 1985 and 2980 s/mm2. The number of diffusion sampling directions was 90 for every b-value. The diffusion sampling length ratio was 1.7 and the output resolution was 1 mm. This specific template was chosen as it was constructed with data from the largest available population and has a resolution of 1.25 mm isotropic.
To address the reproducibility, the trigeminal tract on each side of the brainstem was manually and independently segmented by a junior medical doctor (C.P.) and a radiologist-in-training and neuroanatomist (D.H.). Segmentations were made in ITK-SNAP (www.itksnap.org) (22) using the BigBrain database (Figure 2) (13). When in doubt, a senior neuroanatomist was consulted. The two manually placed masks were compared and the overlapping region of interest was used as a seeding mask. The coordinates of each mask were linearly transformed to the HCP-1065 template. The masks within each side of the pons were used as seeding masks. Seeding masks were manually drawn in ITK-SNAP. Advanced deterministic fiber tractography was applied to the template using DSI studio (DSI Studio, freely available at http://brain.labsolver.org/diffusion-mri-templates). Streamlines were drawn from each seed-voxel (totaling of 50,000 streamlines per voxel). The anisotropy threshold was set by default at 0.07208. The angular threshold was, as default setting, randomly selected from 15 degrees to 90 degrees. The step size was selected from 0.5 voxel to 1.5 voxels. Only streamlines that reached -or passed through- the thalamic sensory nuclei were included to avoid the inclusion of other large dominating fiber pathways (e.g., the corticospinal tract, the middle cerebellar peduncle, the central tegmental tract). Additionally, tracks with length shorter than 30 mm or longer than 300 mm were discarded.
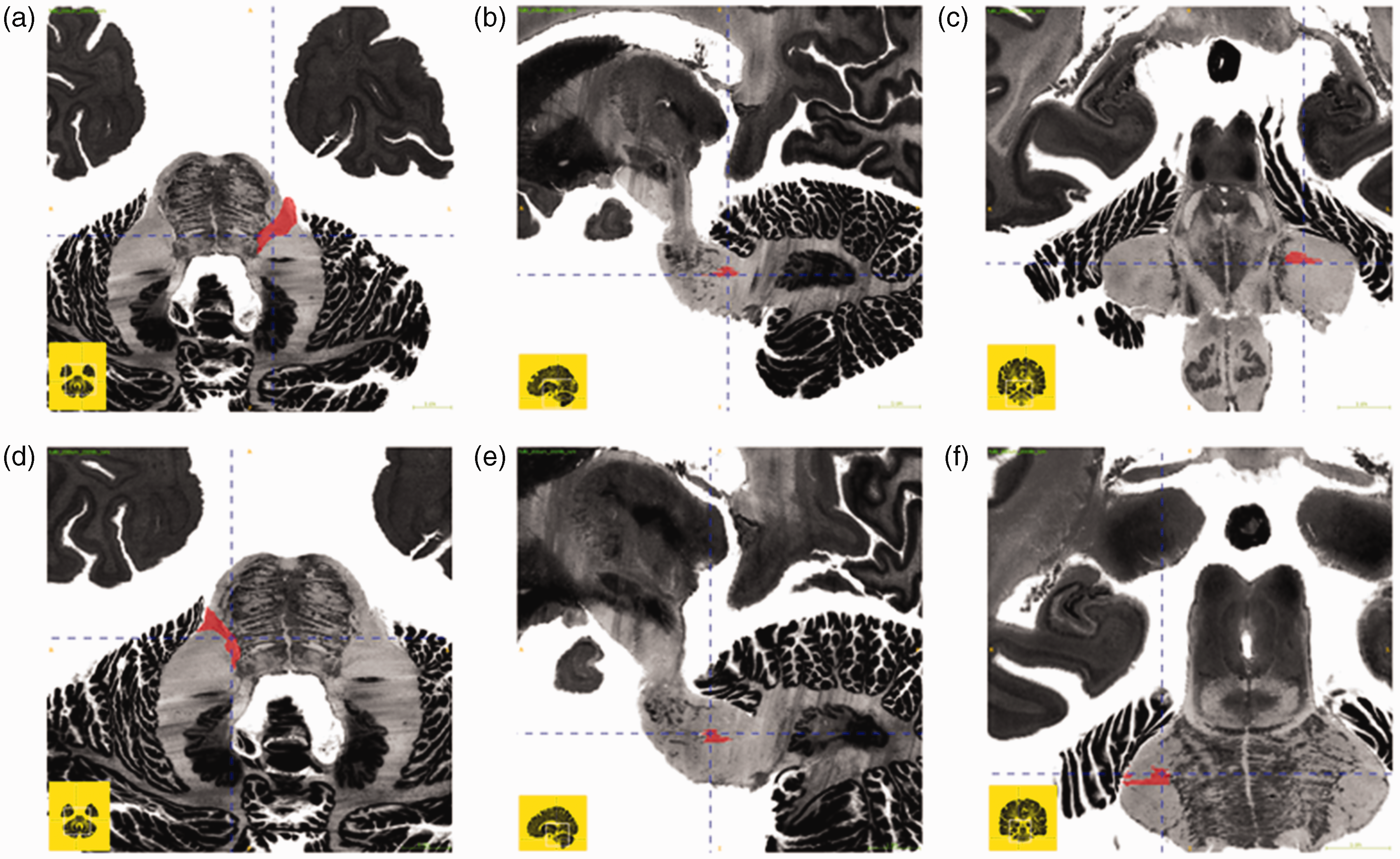
Mask of the trigeminal tract on each side of the pons of the BigBrain dataset (13). A–C, mask of the right trigeminal tract; D–F, mask of the left trigeminal tract; A;D, transverse section; B;E, sagittal section; C;F, coronal section.
To refine tractography results, a waypoint mask was added to the tractography methodology. We opted for a waypoint mask since the sensory nuclei of the thalamus serve as relay nuclei for the trigeminal fibers. Masks of the thalamic sensory nuclei (including both the ventral posteromedial nucleus (VPM) and ventral posterolateral nucleus (VPL)) were derived from the Human Brainnetome Atlas (23) (Tha_L(R)_8_3 Label ID(L): 235; Label ID(R): 236; MNI coordinates (L): −18, −23, 4; MNI coordinates (R): 18, −22, 3) (http://www.brainnetome.org/).
Histological validation
Tractography results in the course of fibers in the brainstem were validated by use of two, in paraffin embedded, post-mortem brainstems. Brainstems were sectioned on an LKB 2260 Macrotome (LKB Instruments, Bromma, Sweden). The knife was positioned at a 15-degree angle with respect to the sectioning plane. The tissue was serially sectioned at 4 µm thickness and every 15th slice was kept for staining, resulting in an interplane resolution of 60 µm. Each successful section was mounted and dried overnight in a stove at 37 degrees Celsius.
Then, sections were stained using two histochemical stains. The first stain concerned the Klüver-Barrera staining, which is a commonly used stain to observe myelin under light microscopy (24). The second stain was the Nauta silver stain, which can be used to visualize neurofilaments. This stain is used to observe neurodegenerative changes of white matter but can also be used to study myelinated and non-myelinated fibers (25,26).
Macrophotographs of the stained sections (referred to hereafter as the histological slices) were taken with a Canon EOS 550D camera with a Canon 100 mm autofocus lens to digitize the data.
Additionally, polarized light imaging (PLI) microscopy slices of three brainstems were used for further histological validation of tractography results. Each brainstem was sliced in one of the three orthogonal planes (i.e., axial, coronal and sagittal). Methodology of PLI as performed by our group has been described extensively elsewhere. In summary, PLI microscopy is especially useful for the differentiation of different fiber bundles with different orientation in densely packed areas such as the brainstem (27,28). One brainstem was used for PLI microscopy imaging. One in five 100 µm sections were mounted, coverslipped and imaged on an especially equipped microscope (Carl Zeiss Microscopy LLC, United States).
Anatomical findings are reported following the standardized Paxinos-Watson abbreviation system (29,30).
Ethical approval
Ethical approvals for the used datasets were acquired by the research groups that originally obtained these datasets (13,20,21). Furthermore, the post-mortem brainstems studied herein are part of the anatomical collection of the department of Anatomy of the Radboud university medical center, Nijmegen, Netherlands. All body donors signed a written informed consent during lifetime, permitting the use of their body and parts for scientific research and educational purposes. All protocols concerning the acquisition of data and tissue processing were approved by the CMO (Commissie Mensgebonden Onderzoek) region Arnhem-Nijmegen, Netherlands and are legislated under Dutch national law (BWBR0005009).
Results
BigBrain histological dataset: Localization of the trigeminal tracts in the human brainstem
In the BigBrain dataset, the trigeminal tracts were located in the ventrolateral aspect of the pons on each side. On the left, the x-coordinates ranged from −10.6 to −13.2, the y-coordinates ranged from −26.1 to −40.6, the z-coordinates ranged from −32.4 to −36.8. On the right, the x-coordinates ranged from −9.9 to −20.1, the y-coordinates ranged from −23.8 to −38.2, the z-coordinates ranged from −33.8 to −39.0 (Table 1; Figure 1).
MNI coordinates of the trigeminal tract in the brainstem derived from the BigBrain dataset.
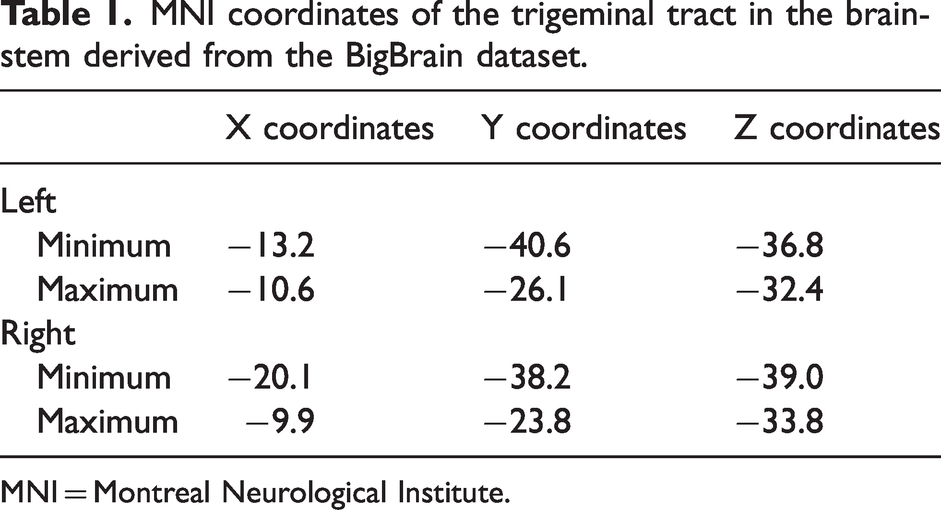
MNI = Montreal Neurological Institute.
Deterministic tractography results from the HCP-1065 dataset
From the created seeding mask, two streamline bundles were found to ascend to the diencephalic region in the HCP-1065 template. The first pathway ascended in the ventral aspect of the brainstem, just posterior to the position of the medial lemniscus. This tract decussated at the level of the pons to the contralateral sensory thalamic nuclei. Similar to anatomic features in primate brains, this course was consistent with the course of the vttt. The second ascending tract coursed more dorsally in the brainstem. This ascending tract ascended near the central tegmental tract and did not cross the midline. From the pons, this tract reached the ipsilateral sensory thalamic nuclei. The course of this tract was in agreement with the descriptions of the dttt in primates and other animals. As expected, most trigeminothalamic fibers crossed the midline as quantitative measurements depicted a ratio of 1:14 and 1:17 of tracts for the dttt:vttt on the left and right side. All quantitative measurement can be found in Table 2. Tractography results are depicted in Figure 3 for the mask of the right trigeminal tract and Figure 4 for the mask of the left trigeminal tract.
Quantitative tractography analysis.
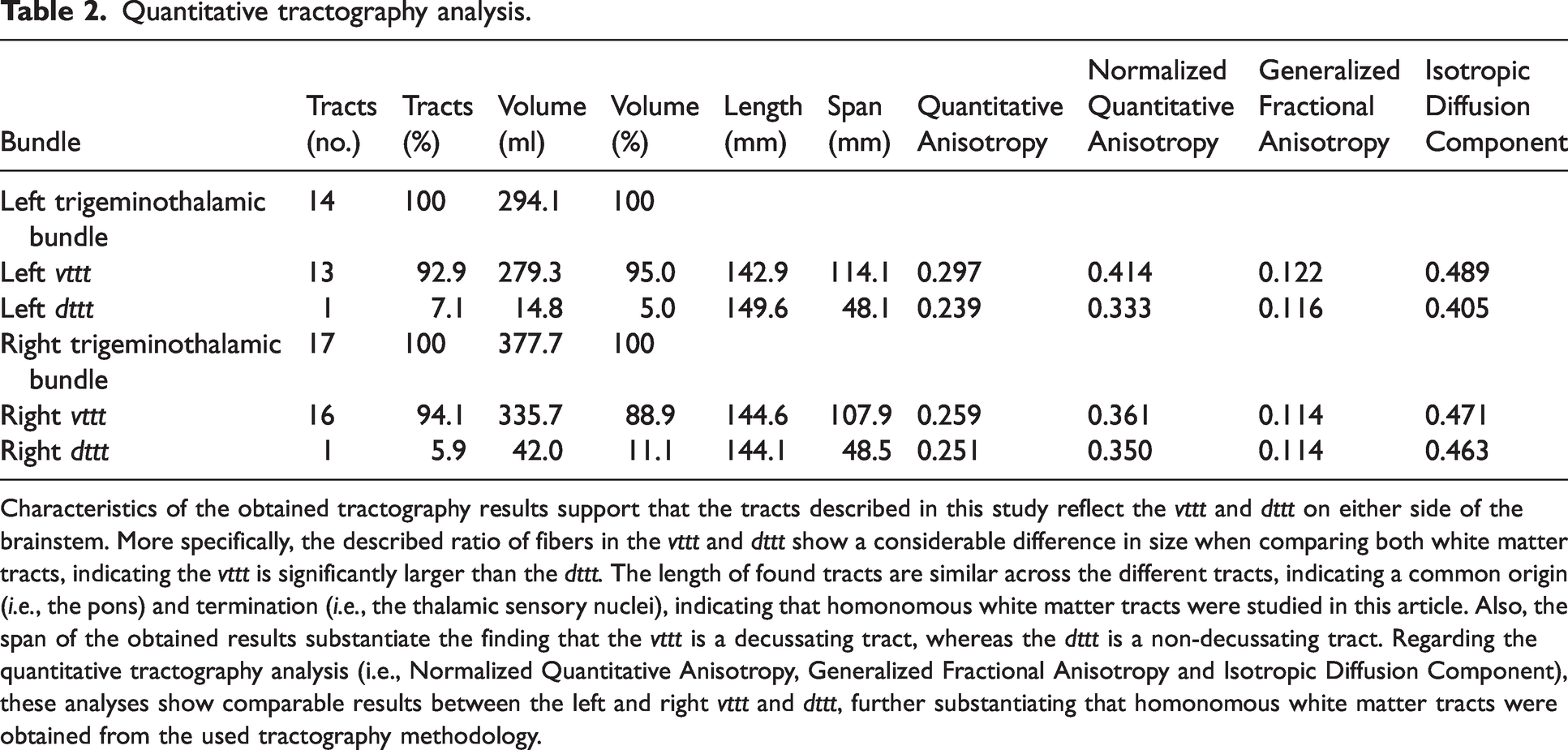
Characteristics of the obtained tractography results support that the tracts described in this study reflect the vttt and dttt on either side of the brainstem. More specifically, the described ratio of fibers in the vttt and dttt show a considerable difference in size when comparing both white matter tracts, indicating the vttt is significantly larger than the dttt. The length of found tracts are similar across the different tracts, indicating a common origin (i.e., the pons) and termination (i.e., the thalamic sensory nuclei), indicating that homonomous white matter tracts were studied in this article. Also, the span of the obtained results substantiate the finding that the vttt is a decussating tract, whereas the dttt is a non-decussating tract. Regarding the quantitative tractography analysis (i.e., Normalized Quantitative Anisotropy, Generalized Fractional Anisotropy and Isotropic Diffusion Component), these analyses show comparable results between the left and right vttt and dttt, further substantiating that homonomous white matter tracts were obtained from the used tractography methodology.
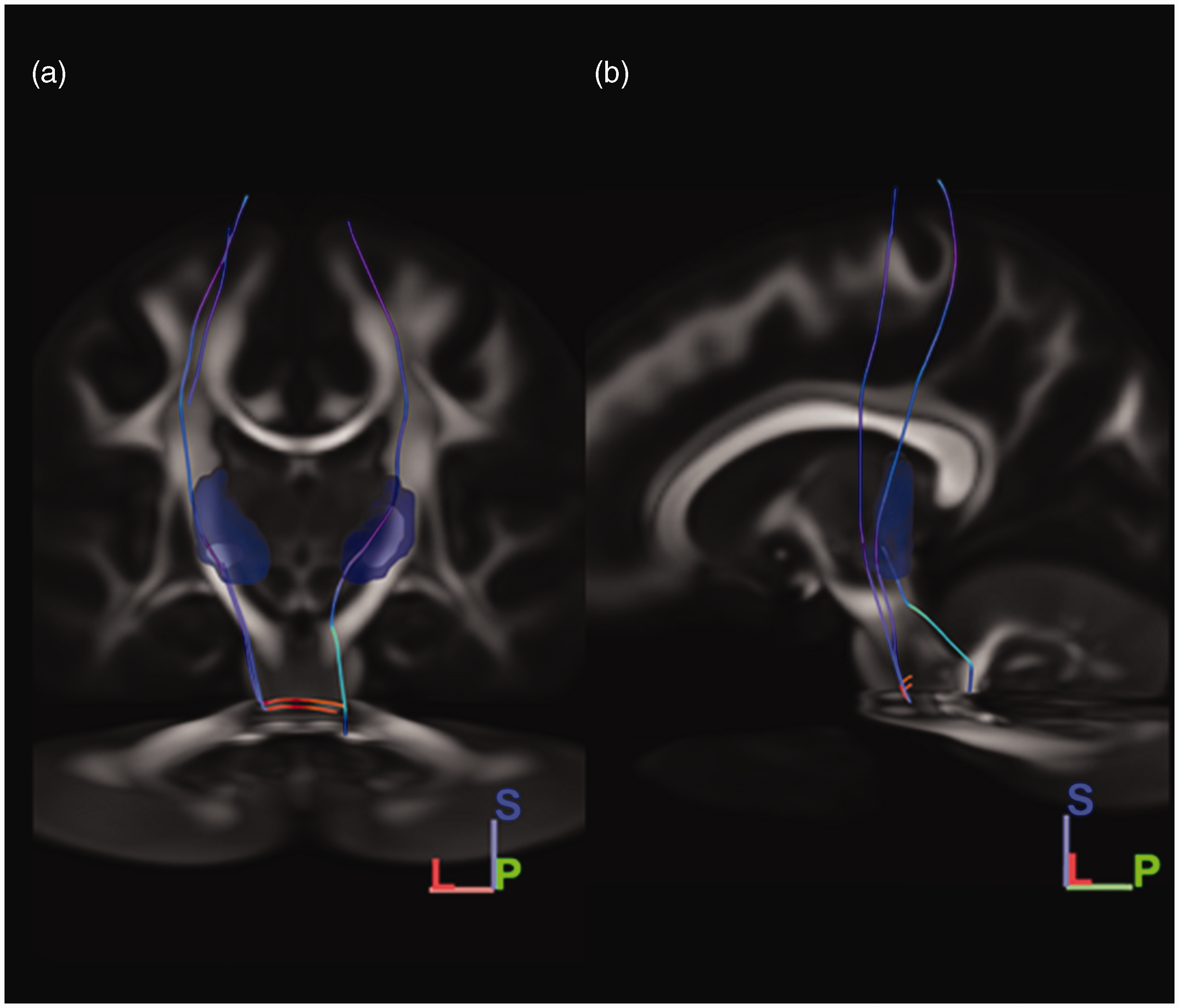
Tractography results when using the right trigeminal tract as seeding mask. A, Coronal view; B, Sagittal view; dttt, dorsal trigeminothalamic tract; vttt, ventral trigeminothalamic tract. Orientation is depicted in the right lower corner. L, lateral; S, Superior; P, posterior. From the created right-sided seeding mask, two fiber bundles were found to ascend to the diencephalic region in the HCP-1065 template. The first ascending pathway ascending in the ventral aspect of the brainstem, just posterior to the position of the medial lemniscus. This tract decussated at the level of the pons to the contralateral sensory thalamic nuclei and was considered to reflect the vttt. The second ascending tract coursed more dorsally in the brainstem. This ascending tract ascended near the central tegmental tract and did not cross the midline. From the pons, this tract reached the ipsilateral sensory thalamic nuclei. This tract was recognized as the dttt.
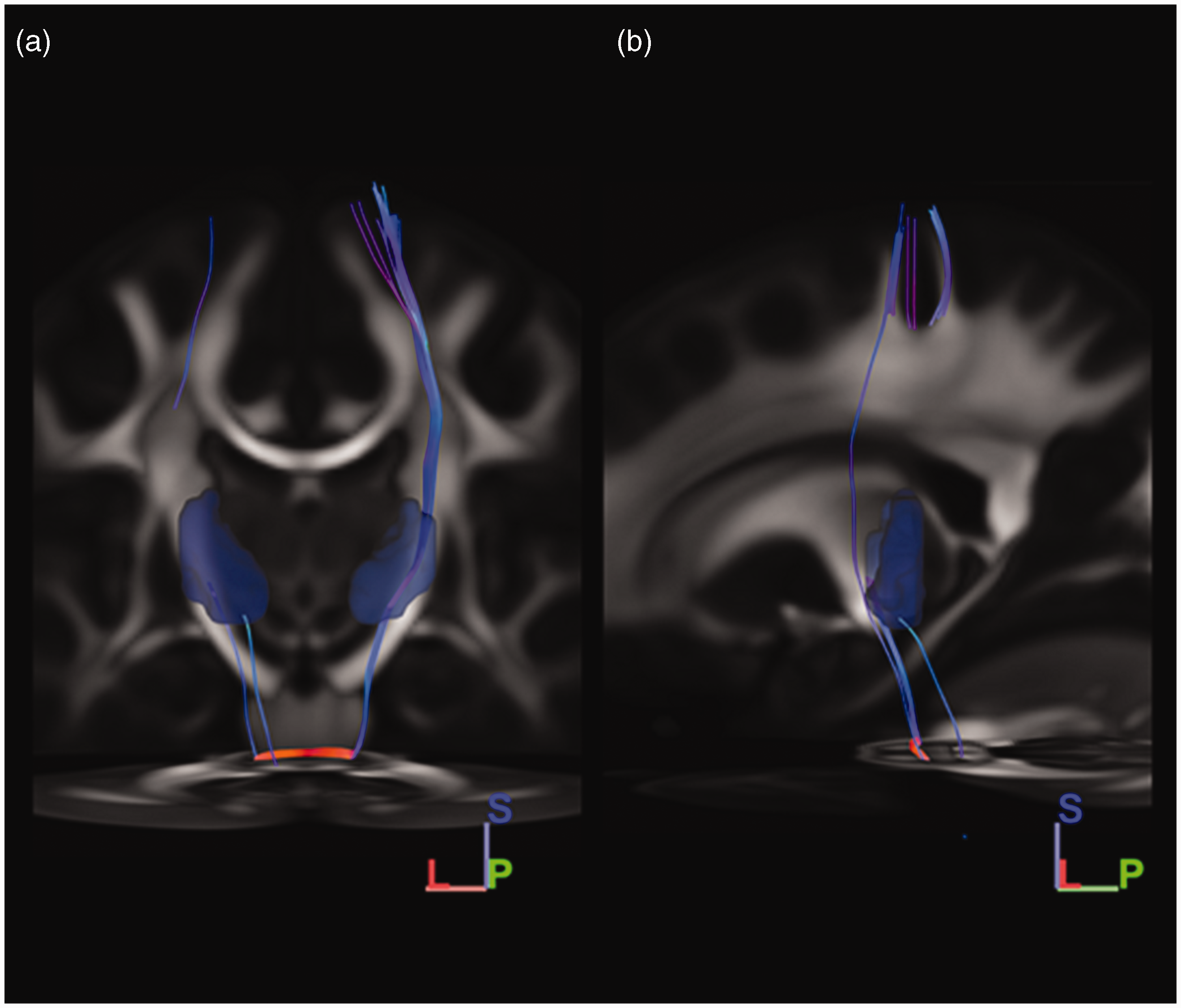
Tractography results when using the left trigeminal tract as seeding mask. A, Coronal view; B, Sagittal view; dttt, dorsal trigeminothalamic tract; vttt, ventral trigeminothalamic tract. Orientation is depicted in the right lower corner. L, lateral; S, Superior; P, posterior. Similar to the findings sprouting from the right-sided seeding mask, two streamline bundles were found to ascend to the diencephalic region in the HCP-1065 template when a similar seeding mask was placed on the left side. The first ascending pathway ascending in the ventral aspect of the brainstem, just posterior to the position of the medial lemniscus. This tract decussated at the level of the pons to the contralateral sensory thalamic nuclei and was considered to reflect the vttt. The second ascending tract coursed more dorsally in the brainstem. This ascending tract ascended near the central tegmental tract and did not cross the midline. From the pons, this tract reached the ipsilateral sensory thalamic nuclei. This tract was recognized as the dttt.
Histological validation
The vttt could be followed as a decussating fiber tract which runs just dorsal to the medial lemniscus. Thereby, the vttt was practically inseparable from the medial lemniscus (ml) on the stained sections. However, on the PLI microscopy images, the directionality and myelin density showed to be different between fibers running in the area of the vttt and the ml. In the midbrain, the vttt can be found just dorsomedial to the contralateral medial lemniscus where it remains to course regardless of the undulations of the medial lemniscus. Again, PLI microscopy images allow for differentiation between the two white matter bundles. These images confirm the course of the vttt just dorsal to the ml.
The smaller dttt was difficult to distinguish from the larger central tegmental tract on the stained sections. Based on information from anatomical atlases, the dttt could be identified as a small bundle of myelinated white matter which ascends in the dorsal part of the pons, just medial to the location of the central tegmental tract. In the midbrain, the dttt courses more dorsally to the central tegmental tract. At the level of the decussation of superior cerebellar peduncles (scp), the dttt can be distinguished as a separate fiber bundle in the dorsal part of the mid-pons, just dorsal to the larger central tegmental tract. Again, the PLI microscopy images help elucidating a small bundle of myelinated white matter courses in a different direction as compared to the adjacent central tegmental tract in the mid-pons and upper pons. At the level of the midbrain, the dttt could not be distinguished from the central tegmental tract with any certainty. Primarily based on the course of this white matter bundle as observed on the PLI microscopy images, this white matter tract seemed to course in the direction of the dttt in the mid-pons, upper pons and midbrain regions.
Histological images (both stained microscopy images and PLI microscopy images) partially validated the tractography results of a bilateral trigeminal projection system at the level of the brainstem. The stained microscopy images can be observed in Figure 5, whereas the PLI microscopy images can be seen in Figure 6. Please note that we decided to consequently show the vttt and dttt in the right sided brainstem regions, regardless of the decussating course (the vttt being a mainly crossing fiber bundle) or the non-decussating course (the dttt being a mainly ipsilateral projection system).
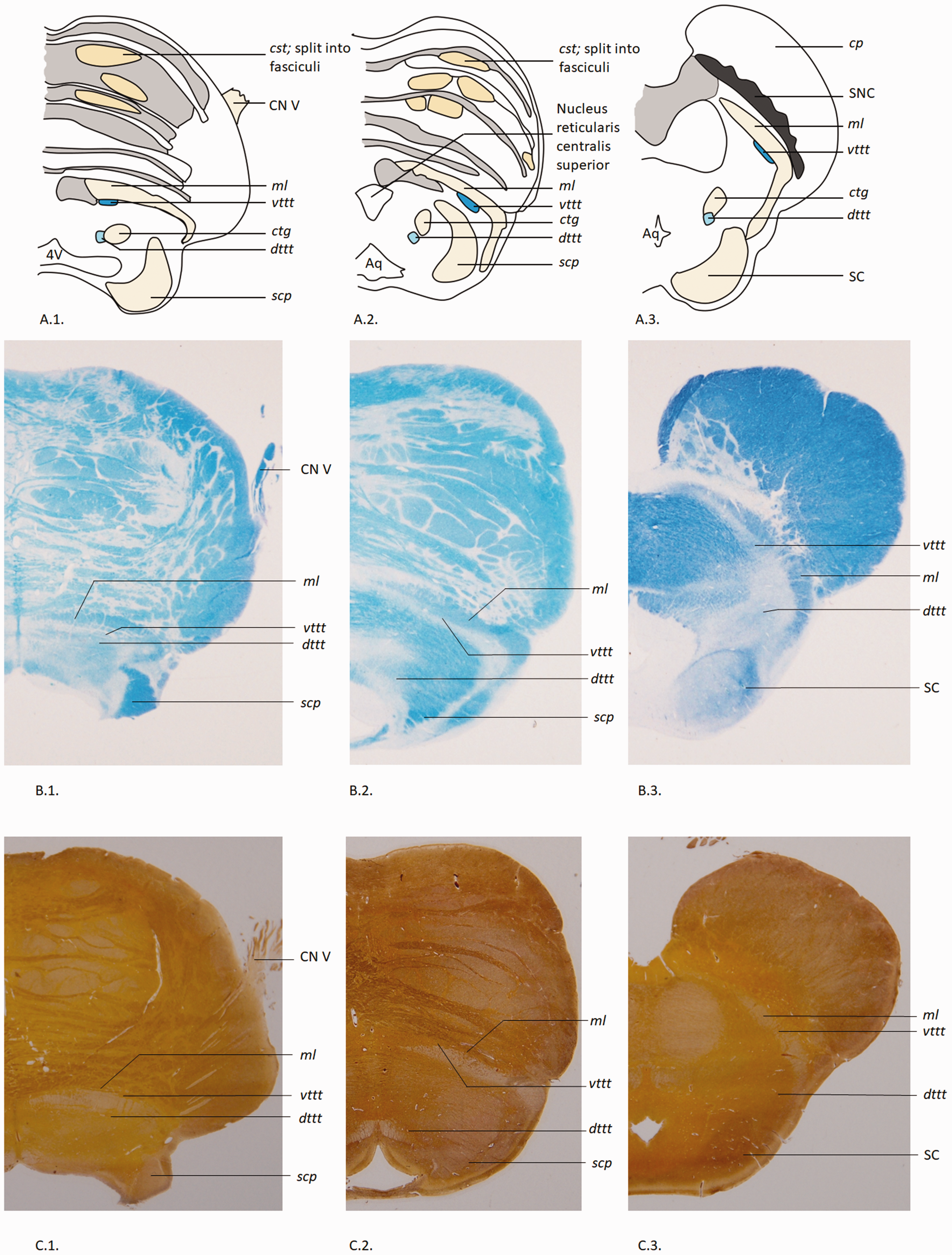
Schematic illustrations and histological sections of the vttt and dttt in the human brainstem. A, Schematic illustrations of axial brainstem anatomy at the level of 1) the trigeminal entry zone; 2) the upper pons; and 3) the midbrain. Adapted from the figures in Duvernoy’s atlas (40). B, Corresponding Klüver-Barrera-stained sections of the illustrations depicted in series A, with annotations.
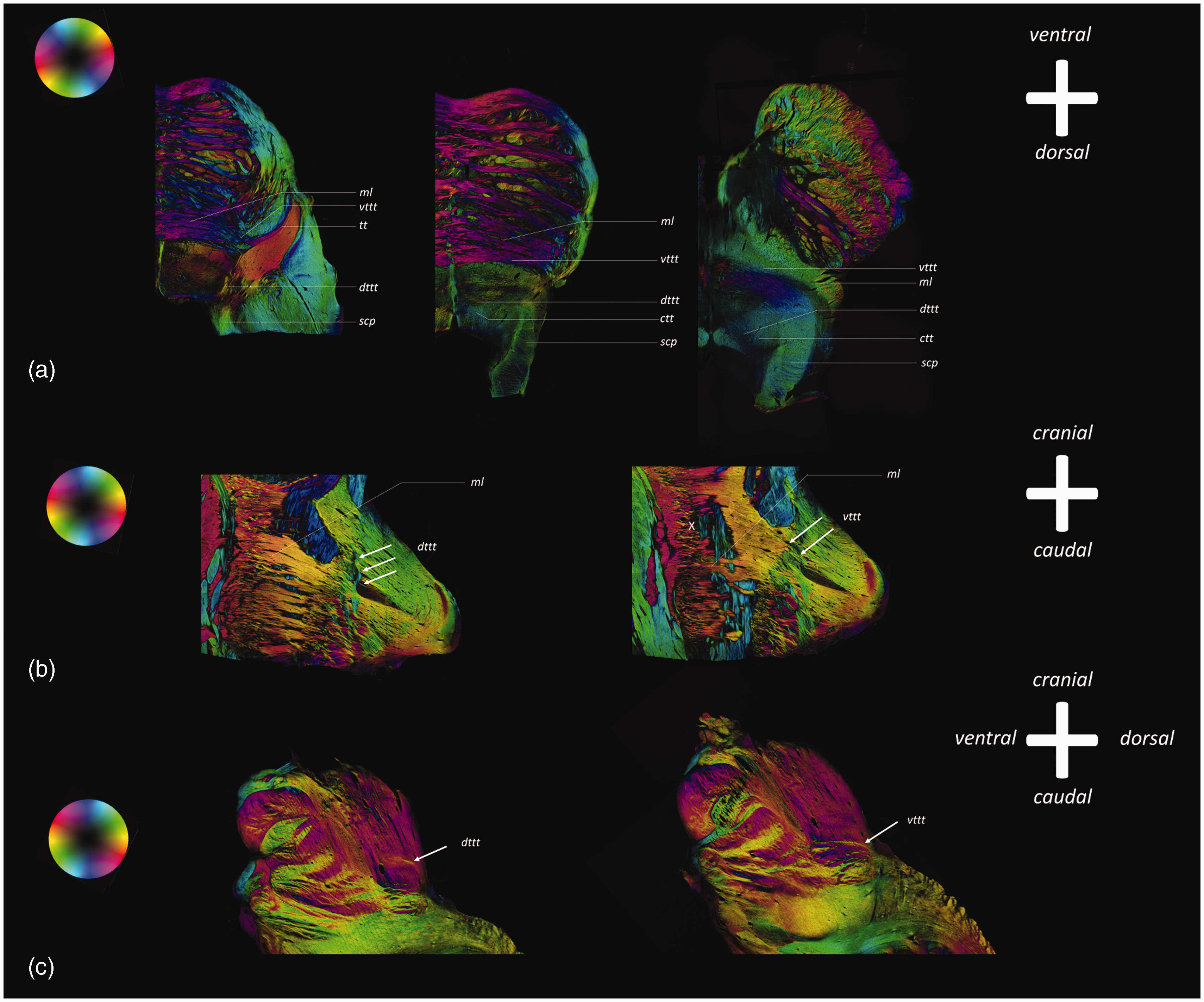
PLI microscopy images of the vttt and dttt in the human brainstem. cp, cerebral peduncle; cst, corticospinal tract; ctg, central tegmental tract; dttt, dorsal trigeminothalamic tract; ml, medial lemniscus; scp, superior cerebellar peduncle; SNC, substantia nigra, pars compacta; tt, trigeminal tract; vttt, ventral trigeminothalamic tract; X, decussation of white matter tracts. Axial PLI microscopy images show the tt coursing in between the fibers of the medial cerebellar peduncle, whereafter the tt bifurcates into a ventral projection system (vttt). Coronal images show how the vttt courses in the same direction as the ml as they course towards the midline of the brainstem. At the level of the pons, the fibers of both the ml and the vttt decussate towards the contralateral side (X). The vttt can be followed in its course cranially as a fiber pathway directly posterior to the ml. Axial PLI microscopy images also show the dorsal branch of the bifurcating tt, called the dttt. The dttt can be observed on coronal and sagittal sections as an ipsilaterally coursing fiber bundle. In its course cranially, the dttt can be observed to course in the dorsal aspect of the brainstem and can be followed up to the midbrain. Particularly the axial slices show how the dttt follows the course of the ctg.
Discussion
The present study shows that from each trigeminal nerve, a decussating and non-decussating trigeminothalamic tract ascends as assessed by using deterministic tractography on a population-averaged template from the HCP. These results were validated by use of histological datasets of the human brainstem. Although ex-vivo tractography results and histological data already showed a bifurcation of the trigeminal tract at the level of the pons (9), the current results provide further evidence that the pontine trigeminal tracts further ascend to the thalamic nuclei in a bilateral projection system. Furthermore, the histological data provided in this paper further substantiate the course of the vttt and dttt in the upper pons and the midbrain. The generally accepted view that afferent neurons from the trigeminal nerve synapse in the TSNC, cross the midline and project to the contralateral VPM in humans is thereby only partially valid. By visualizing the dttt and vttt, named after similar anatomic features in primates, this study shows the bilateral nature of the trigeminothalamic pathway from the trigeminal nerve to the thalamus for the first time in humans.
The representation of the ipsilateral PSN in the VPM of the thalamus in monkeys has been confirmed by multiple studies (6,31–33). Such studies could not be verified in humans due to technical and ethical limitations (34). A bilateral response to noxious orofacial stimuli in humans has, however, been reported to occur in the thalami, insular and primary somatosensory cortices when using functional MRI in humans (3,4). Transcallosal pathways were not regarded as a plausible explanation since nociceptive stimuli applied to a split-brain patient also caused bilateral pain perception (35). Another argument in favor of a bilateral system in humans is the study performed by Henssen et al. in 2019 (10). Clarification of the trigeminal projection system is believed to have consequences for the treatment of trigeminal neuropathies. A pilot study showed that bilateral transcranial magnetic stimulation was superior to unilateral stimulation in the treatment of trigeminal neuropathic pain (10). In addition, a recent meta-analysis of our group showed that trigeminal neuropathic pain does not respond favorably to invasive neuromodulation, whereas central trigeminal neuropathic pain patients experience significant pain relief (36). The present results provide further evidence that neuromodulation in trigeminal neuropathic pain patients should be carried out in a bilateral fashion as this will hopefully improve treatment effects. In addition, the current insights could aid in expanding the treatment armamentarium for diseases concerning the trigeminal nerve. Yet, no conclusions about the functionality can be drawn from the present results. We recommend further research to be performed to explore the functional properties of the dttt in order to evaluate the possible clinical implications of the findings presented in this study. One of the research methods could include functional MRI (fMRI) following a protocol as performed by Nash et al. (4). In their study, they injected the right masseter muscle and the overlying skin with hypertonic saline, inducing a noxious stimulus. Thereafter, participants were scanned using blood oxygen level dependent (BOLD) contrast using a 3T MR scanning system, demonstrating a bilateral fMRI-activation of the thalamus, S1 and S2 cortices (4). However, no replication studies have been carried out to the authors’ knowledge, demonstrating it to be an understudied field of research.
Strengths and limitations
One of the strengths of this study forms the combination of population-based tractography and histological validation using multiple datasets; this increases the validity of the present study, although this histological dataset only comprises one specimen. A limitation of the used methodology concerns the fact that tractography has various, well-known limitations as several validation studies confirmed that tractography techniques are plagued by both false-positive and false-negative results (37,38). However, a recent publication re-investigating the validity of tractography showed that prior anatomical knowledge in the form of manually placed or template-driven constraints can significantly improve the anatomical accuracy of tractography results. The same paper demonstrated that it is possible to achieve a high sensitivity and high specificity simultaneously, and concluded that current tractography algorithms, in combination with anatomically driven constraints, can result in reconstructions which very accurately reflect the ground truth white matter connections (39).
Conclusions
Using multiple datasets, spanning from histology to diffusion MRI data from 1065 subjects, the vttt and dttt may be visualized in humans. The vttt courses towards the contralateral thalamus, whereas the dttt ascends to the ipsilateral thalamus. Now that the anatomy of the trigeminal trajectories in humans has been further elucidated, functionality needs to be studied to ascertain the clinical relevance and to possibly further optimize neuromodulatory treatment strategies in orofacial pain.
Article highlights
This study shows a bilateral projection system of the trigeminal nerve via the ventral and dorsal trigeminothalamic tracts (vttt and dttt, respectively) in MRI data of 1065 subjects from the Human Connectome Project. When future research shows that the dttt and vttt actually transmit signals from noxious stimuli, this offers potential in understanding and possibly treating pathology in the orofacial region more effectively.
Footnotes
Acknowledgements
Data collection and sharing for this project was provided by the MGH-USC Human Connectome Project (HCP; Principal Investigators: Bruce Rosen, Arthur W. Toga, Van J. Weeden). HCP funding was provided by the National Institute of Dental and Craniofacial Research (NIDCR), the National Institute of Mental Health (NIMH), and the National Institute of Neurological Disorders and Stroke (NINDS). HCP data are disseminated by the Laboratory of Neuro Imaging at the University of Southern California.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
