Abstract
Background
The role of calcitonin gene-related peptide (CGRP) in the cyclic pattern of cluster headache is unclear. To acquire biological insight and to comprehend why only episodic cluster headache responds to CGRP monoclonal antibodies, we examined whether plasma CGRP changes between disease states (i.e. bout, remission and chronic) and controls.
Methods
The present study is a prospective case–control study. Participants with episodic cluster headache were sampled twice (bout and remission). Participants with chronic cluster headache and controls were sampled once. CGRP concentrations were measured in plasma with a validated radioimmunoassay.
Results
Plasma was collected from 201 participants diagnosed with cluster headache according to the International Classification of Headache Disorders, 3rd edition, and from 100 age- and sex-matched controls. Overall, plasma CGRP levels were significantly lower in participants with cluster headache compared to controls (p < 0.05). In episodic cluster headache, CGRP levels were higher in bout than in remission (mean difference: 17.1 pmol/L, 95% confidence interval = 9.8–24.3, p < 0.0001). CGRP levels in bout were not different from chronic cluster headache (p = 0.266).
Conclusions
Plasma CGRP is unsuitable as a diagnostic biomarker of cluster headache or its disease states. The identified reduced CGRP levels suggest that CGRPs role in cluster headache is highly complex and future investigations are needed into the modulation of CGRP and its receptors.
Introduction
Cluster headache (CH) is one of the most severe pain conditions among pain disorders and definitely among primary headaches (1). For most patients, CH is characterized by two disease states: the bout lasting weeks to months in which the attacks occur, and the attack-free remission lasting months to years. The third version of the International Classification of Headache Disorders, 3rd edition (ICHD-3) classifies episodic patients with annual remissions greater than three consecutive months (2). In the remission period, CH attacks cannot be provoked by pharmacological triggers (3). For 10–15% of patients with CH, the remission is absent; thus, they are classified as patients with chronic CH (4). Persons with episodic CH in bout and chronic CH both have similar attacks and are phenotypically similar (5). However, notable differences exist such as reduced effectiveness of medication and attack induction in chronic CH indicating separate states (3,6,7). These three disease states (i.e. bout, remission and chronic) represent a clear clinical distinction from migraine (3), although the pathophysiological substrate for the three disease states in CH is currently unknown.
Calcitonin gene-related peptide (CGRP) is a vasodilatory neuropeptide that is closely associated with the trigeminal vascular pain pathway. As in migraine, CGRP is implicated in CH pathophysiology because CH attacks can be provoked in bout by a CGRP infusion and episodic CH can be prevented by targeting the CGRP system (8,9). The elevated CGRP level during attacks is a consistent finding in saliva and plasma from the external jugular vein (10–15), which contrasts with the inconclusive results of CGRP levels in different CH states (16–18). However, none of the previous studies were sufficiently powered to detect an association or exploited the potential of the same person being in two states.
Therefore, we first aimed to investigate CGRP levels in plasma drawn from the antecubital vein in paired samples obtained prospectively in remission and in bout and, second, we aimed to compare the CGRP levels between participants with CH and controls. We hypothesized that plasma levels of CGRP depend on disease states and are higher compared to matched controls. Specifically, we hypothesized that CGRP levels were lower in remission compared to bout. We also hypothesized that plasma levels of CGRP correlate with attack frequency and intake of acute medication. Explicitly, we hypothesized that elevated CGRP levels are associated with high attack frequency, and the CGRP level is lowered if acute medication has been used in the last 24 hours prior to blood sampling. To test these hypotheses, we collected standardized plasma samples from 100 participants with episodic CH in a prospective study and, for comparison, we included 101 participants with chronic CH and 100 controls.
Methods
Participants
We recruited participants from the Danish Headache Center, Rigshospitalet – Glostrup, Denmark, between October 2018 and December 2021. Participants were diagnosed with CH according to the ICHD-3 (2) or were healthy control persons. Participants were aged 18–80 years. For all participants, exclusion criteria were chronic headaches (other than CH), known drug misuse, pregnancy or nursing, as well as severe somatic or psychiatric diseases.
Patients with episodic CH were defined as in bout when they experienced one or more CH attacks within one week, and in remission phase when they reported a minimum of 30 consecutive days without CH attacks. Participants with CH in their first bout were followed until remission occurred or the diagnosis of chronic CH could be made (follow-up period: one year). The use of stable CH preventive treatments was permitted, apart from a Greater Occipital Nerve block and oral steroids within the last 30 days before examination. To minimize selection bias, we invited patients with CH who were not followed by the Danish Headache Center but contacted the center to obtain oxygen treatment or advice during their bout.
The recruitment of control participants was conducted through the dissemination of notices on social media platforms, as well as the posting of physical notices in the hospital premises and its vicinity. Controls had no history of a primary or secondary headache except infrequent tension type headache (i.e. headache occurring on one day per month or less) or delayed alcohol-induced headache or prior acute headache attributed to infection such as influenza according to the ICHD-3 (2). Nor were they allowed to have a known first or second degree relative with a CH diagnosis. All controls were free of any type of headache for at least seven days before the blood sample collection. Control matching by age and sex was based on distribution in the episodic group in a 1:1 ratio (Table 1).
Baseline demographics.

Standard protocol approvals, registrations and patient consents
All participants gave written informed consent in accordance with the Declaration of Helsinki. The study was approved by the Regional Health Research Ethics Committee of the Capital Region (H-16048941) and the Danish Data Protection Agency.
Study design
Participants were included in the Danish Cluster Headache Biobank, which has a prospective observational case–control study design. The first study day for participants with episodic CH could be in remission or in bout, but, to complete the study, the second study day should be in the opposite state from the first study day. Controls and participants with established chronic CH completed the study after study day one (see supplementary Figure 1). Sampling was made in-between CH attacks. On all study days, participants were asked to adhere to a fasting period of at least eight hours for all forms of consumption, with the exception of water, for which a fasting period of only two hours was necessary.
CGRP measurements
Plasma CGRP concentrations were measured with a fully validated radioimmunoassay for human CGRP. The method was previously described in detail (16,19). The tracer was prepared by the method of Iodogeneral (Pierce, Rockford, IL, USA) (20) by iodination of [Tyr0] a-CGRP amide and purification by high-performance liquid chromatography (19). Samples, antibody and calibrators were incubated at 4°C for around 90 hours before the tracer was added. The subsequent incubation lasted 48 hours. Sac-Cel separation divided free and antibody-bound tracer. The limit of detection was 1 pmol/L. To minimize a batch effect, all analysis was consecutively carried out in winter of 2021.
Variables on study day
On study day one a baseline semi-structured interview was completed. Patients with CH had their diagnosis verified by a physician (AS, RHJ and ASP) or a specially trained medical student (AFP). All study day variables were based on self-reports in a structured interview (see supplementary eMethod).
Statistical analysis
Calculation of sample size is based on raw data from Snoer et al. (16) where the SD of the difference was 31.5. Here, we estimated that 100 patient pairs were needed to reach a power of 90% and a significance level of 0.01. To ensure to reach our recruitment target of 100, we included 129 patients with episodic CH to account for drop-out and transition to chronic state.
The primary endpoints were: (i) difference in plasma CGRP levels between groups (episodic CH in bout, episodic CH in remission, chronic CH and controls) and (ii) differences in plasma CGRP concentrations depending on attack frequency and acute medication within the last 24 hours.
Distribution of data was inspected visually for normality and with Bartlett’s test for variance. Criteria of normality were met for the paired comparison between episodic cluster headache in bout and remission; Thus, a parametric test was applied (paired t-test). The criteria of normality for the unpaired comparisons were not met in all groups; thus, non-parametric Mann–Whitney tests were applied to test differences in CGRP levels. A generalized linear model tested the effect of attack frequency on CGRP levels, adjusting for sex and acute medication during the 24 hours before sampling. Correlation between CGRP levels and bout/remission, time since last acute medication (in hours) and attack frequency in the last week up to sampling were calculated with the Pearson test. Model assumptions were checked and, if necessary, a logarithmic transformation was applied. After quality control, 397 samples were eligible for further analysis (Figure 1). If there was missing data in a predefined variable, a participant was excluded from the regressions. Statistical analyses were performed using R software, version 4.2.2 (R Foundation, Vienna, Austria) (21). We adjusted outcome p-values for multiple comparisons by the false discovery rate method. Adjusted p < 0.05 was considered statistically significant. Supervised machine learning tool was performed in QLattice based on the variables listed in Table 2 (22,23) (for further information, see supplementary, eMethod).

Flow chart of inclusion. Individuals were excluded from final analysis if they did not have a numeric value of CGRP (n = 1), were extreme outliers (defined as a CGRP value >1200 pmol/L, n = 1) or had no CGRP-value as a result of inappropriate tube handling (n = 2). After quality control, 397 samples were eligible for further analysis. To minimize batch effect, all analysis was consecutively carried out in winter of 2021. CH, cluster headache; cCH, chronic cluster headache; eCH, episodic cluster headache.
Clinical variables on the day of blood sampling.

Legend: Data presented as median with interquartile range (IQR) for numeric variables and as count with percentages for categorical variables. Missing values are indicated in brackets. Due to a protocol revision, the attack frequency was added later resulting in higher missing values.
Δ All patients with cluster headache in active phase were asked to retrospectively estimate their attack frequency in the preceding 24 hours and seven days.
◊ All headaches were mild and bilateral and was phenotypically classified as tension-type headache.
Results
We screened 310 patients with CH and 114 controls and 352 (83%) of these were included into the Danish Cluster Headache Biobank. Among the patients with CH, 24 (8%) were screening failures and 40 (13%) refused participation, absented or had a greater occipital nerve block done within 30 days prior to investigation (Figure 1). Among the controls, 10 (9%) were screening failures and three (3%) refused participation. In the group of patients with episodic CH included in bout (n = 113), 11 (10%) did not enter remission and instead transitioned into a chronic CH, and these were excluded from analysis. The first consecutive 100 participants with episodic CH who completed both study days along with first 100 controls and 101 participants with chronic CH were included.
Demographics and clinical parameters of participants
The mean ± SD age was 42.3 ± 11.9 years for participants with episodic CH and 46.7 ± 13.9 years for participants with chronic CH. The male-to-female ratio was 4:1 in the episodic group and 1.7:1 in the chronic group. Due to matching based on distribution in the episodic CH group, the male-to-female ratio was also 4:1 and the mean age was 41.4 ± 13.7 years in the control group. None received prophylactic treatment directly targeting the CGRP system as these are only approved for chronic migraine in Denmark. Summarized baseline demographics are listed in Table 1, and detailed information of study day characteristics are listed in Table 2 for the 301 participants included in the final analysis.
Levels of plasma CGRP are reduced in cluster headache and are associated with cluster headache states
Among the paired samples in episodic CH, we found that plasma levels of CGRP were higher in bout compared to in remission (paired t-test: mean difference: 17.1 ± 35.3 pmol/L, 95% confidence interval (CI) = 9.8–24.3, padj < 0.0001), equivalent to a mean reduction of 11.7% (Figure 2). Moreover, in individual participants CGRP levels in bout correlated with CGRP levels in remission (Pearson correlation: r = 0.17, 95% CI = 0.02–0.30, padj = 0.041). No difference in plasma CGRP levels were found between episodic CH participants in bout and participants with chronic CH (Mann–Whitney test: padj = 0.443) (Figure 3), although both had significantly lower plasma levels compared to controls (Mann–Whitney test: respectively, padj = 0.029 and padj = 0.0005) (Figure 3). Likewise, participants with episodic CH in remission had significantly lower CGRP plasma levels compared to controls (Mann–Whitney test: padj < 0.0001). A summary of exact CGRP levels is provided in Table 3.

Episodic cluster headache: paired comparison in between bout to remission. Plasma levels of calcitonin gene-related peptide (CGRP). Data presented as the mean ± SD (blue). Paired t-test: mean difference 17.1, 95% confidence interval = 9.8–24.3, padj < 0.0001. In the paired analysis, three extreme outliers are removed to adhere to the assumptions of the test, although the difference in between bout and remission remained significant with and without the outliers. ****p < 0.0001.
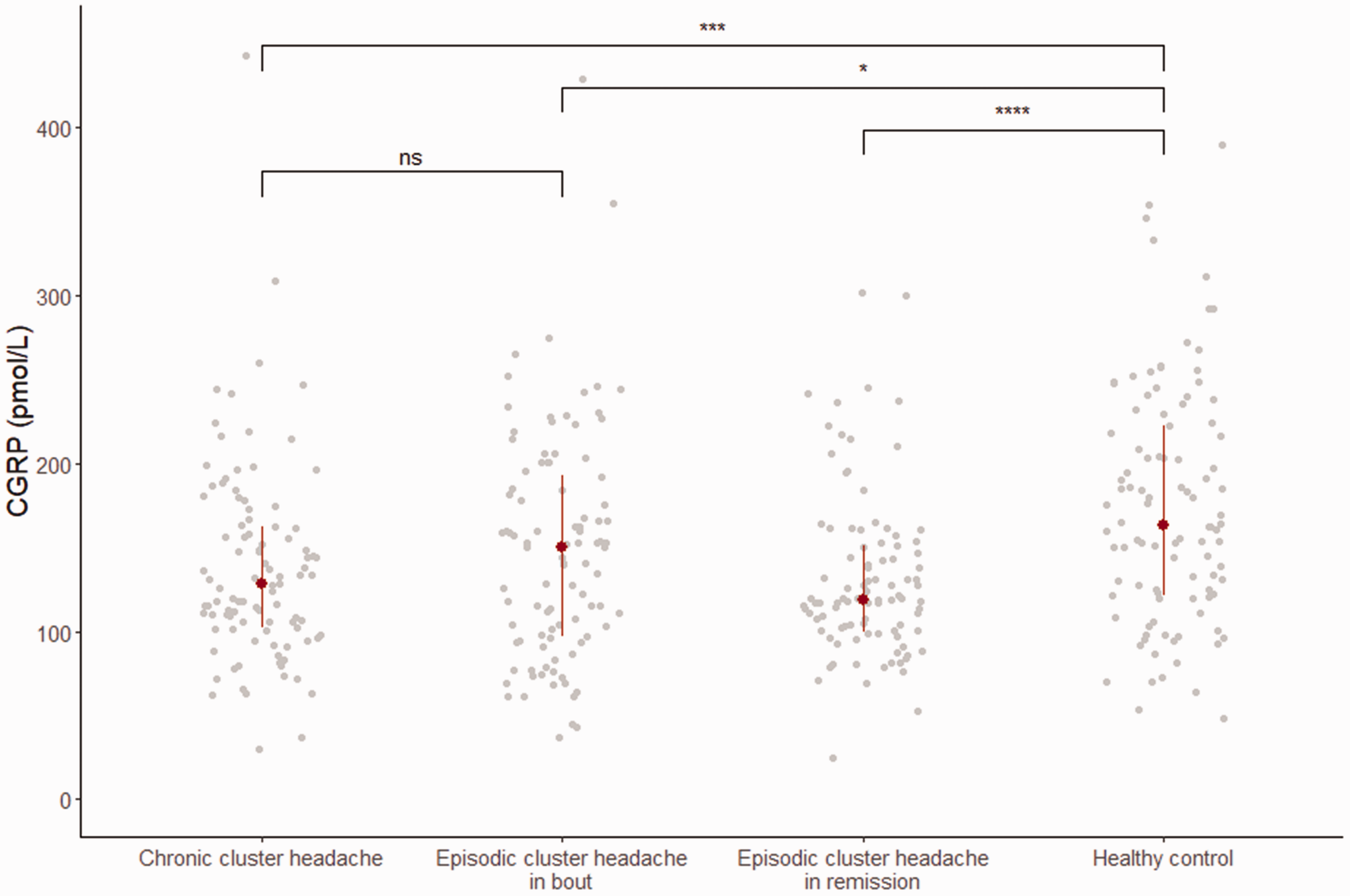
Reduced calcitonin gene-related peptide (CGRP) levels identified in cluster headache compared to controls. Plasma levels of CGRP. Data are presented as the median and interquartile range. A Mann–Whitney test was applied to test the difference in between the groups. *p < 0.05, ***p < 0.001, ****p < 0.0001. ns, not significant,
Concentration of CGRP in pmol/L.

No association between plasma CGRP and attack frequency or use of triptans or oxygen the last 24 hours
A generalized linear regression was used to test whether attack frequency, use of acute medication and diagnosis (episodic and chronic CH) predicted plasma CGRP levels. The model could not predict the CGRP level, by attack frequency (beta = –0.6, padj = 0.916), nor use of acute medication (beta = 1.2, padj = 0.916) within the last 24 hours (Figure 4) (missing: n = 11). CGRP levels did not correlate with attack frequency during the last week (Pearson test: r = –0.01, 95% CI = –0.15 to 0.12, padj = 0.916) or hours since last acute medication among patients with attacks within the last 24 hours (Pearsons test: r = –0.08, 95% CI = –0.35 to 0.21, padj = 0.829).

Calcitonin gene-related peptide (CGRP) levels cannot be predicted by attack frequency the last 24 hours. Scatter plot presented for the 197 patients with chronic cluster headache or episodic cluster headache in bout. The model presented is the unadjusted linear model.
To investigate whether a (non-linear) combination of parameters could explain CGRP levels, we predicted CGRP levels using all measured covariables with QLattice. We did not find a significant model predicting CGRP levels among all patients with CH, neither among patients with episodic, nor chronic cluster separately.
Discussion
The present study reports that people with episodic CH had lower levels of CGRP in their plasma during remission than during a bout. Furthermore, it shows that patients with CH, regardless of disease state, demonstrated lower, not higher, plasma CGRP levels compared to controls. An additional significant discovery was the absence of any discernible impact of attack frequency on plasma levels of CGRP. In general, our results support the notion that CGRP plays a role in the pathophysiology of CH; however, the role of CGRP appears to be part of complex signaling pathways and not a sole causal role. The role of CGRP may differ in cluster headache compared to migraine because we have identified lower CGRP levels in cluster headache, whereas, in migraine, CGRP levels have been identified to be higher compared to healthy controls (24).
In the present study, plasma CGRP levels were lower in all patients with CH compared to matched controls. This was in contrast to our hypothesis with the elevated level of CGRP being associated with CH. Three prior studies have compared CGRP levels in patients with CH vs. controls. The first study found elevated CGRP levels during CH attacks compared to controls (11) and thus cannot be compared with our results measuring CGRP levels in between CH attacks. The second study compared measurements from patients with CH to historical data from patients with migraine and controls finding elevated levels of CGRP associated with CH (16). The historical data is a critical point and only data from six controls were included; thus, given the enormous variance in CGRP measurements, this may have been a false positive finding. The third study found no difference between patients with episodic CH in bout, chronic CH and controls in plasma CGRP levels measured with an enzyme-linked immunosorbent assay (18). However, the study also found that CGRP levels in tear fluid were higher in participants with CH who had not used acute medication 48 hours before sampling compared to controls, but only a total of 17 patients were compared to 60 controls. We could not find evidence for plasma CGRP levels being associated with either attack frequency or acute treatment. These differences may be a result of methodological differences in the assays, or tear fluid may be more sensitive to detect fluctuations compared to plasma. We can also speculate that our overall lower level of CGRP could be a result of CGRP depletion in the trigeminovascular system. However, more research is needed to confirm this notion.
Our paired-sample analysis in episodic CH revealed that CGRP levels are higher in bout compared to remission, which confirms our hypothesis that CGRP levels vary across disease states. This is in line with three smaller studies with nine to 30 participants (12,13,25), whereas three other studies with 15 to 29 participants failed to detect a difference (10,16,17). Unexpectedly, the largest differences in CGRP levels in the present study were found between episodic patients in remission and the controls. Intuitively and based on previous data (10,16), we expected that CGRP levels in remission would be equal to those of controls, but our findings suggest that the CGRP signaling system is persistently disrupted in CH despite the cyclic presentation of attacks (29). In sum, the combined evidence supports that CGRP fluctuates according to disease states in CH.
The evidence is not causal, the individual variance is large and the association is far from linear, suggesting that the CH signaling system is highly complex and not solely dependent on CGRP. The high variance and lack of specificity make CGRP unattractive as a diagnostic biomarker for CH and disease activity. The observation that CGRP levels are higher in the control population compared to all CH states may be a further argument for the assumption that CGRP is not the key mediator in initiating CH attacks. More likely, CGRP is one among other players involved in the cascade of nociceptive processes that build up cyclic cluster attacks within the bout. Even though CGRP might just be a part of a cascade, it appears reasonable to conclude that CGRP is an important part, especially in episodic CH, given that the anti-CGRP monoclonal antibody (mAbs) galcanezumab is effective in episodic CH but not in chronic CH (9). Alternative signaling systems may also include other vasoactive peptides in the trigeminal autonomic reflex, such as pituitary adenylate-cyclase-activating polypeptide and vasoactive intestinal polypeptide, or may be influenced by epigenetics or currently unknown factors (26–28). The pathophysiology of CH is multifaceted and encompasses both an off-and-on switch of attacks and bouts, and so it may be plausible for different signaling pathways to interact in a complex manner.
Methodological considerations
This study holds several methodological strengths compared to previous reports on CGRP levels in CH; most importantly, the high number of participants and thorough inclusion and exclusion criteria. We acknowledge that a limitation of the present study is that CGRP is measured in plasma derived from the antecubital vein, whereas some previous studies have used blood from the external jugular vein. However, the main outflow from the central nervous system would require plasma derived from the internal jugular vein, cannulation of which is more invasive. In practice, the effects may be minimal because a study of spontaneous migraine attacks found no difference in plasma CGRP levels between blood from the external jugular vein and antecubital vein (29). The CGRP assay is a potential limitation because there are many different assays, but the assay applied in this study is fully validated for CGRP (19,20). CGRP exists in two isoforms (alpha and beta) and, although there are differences in expression, these are not exclusive, and data from knockout mice suggest typically co-expression (15). Therefore, we consider that our assay, which is validated for both alpha and beta CGRP, combined with our study design including both patients and controls, is a methodological strength. Recently, a study found that alpha CGRP, but not beta CGRP, is linked to chronic migraines (30). The main contributor to CGRP in the trigeminal ganglion is alpha CGRP (31). Given that we measure both alpha and beta CGRP, we may have underestimated the effect of alpha CGRP; however, CH and migraine are two separate diagnoses (3). The analytical variation in the present study was reduced by all samples being analyzed in the same batch of the assay. Importantly, the heterogeneity of assays renders direct comparisons of absolute CGRP concentrations between the assays almost useless. Another limitation is that patients were allowed to use acute and preventive medication. However, we found CGRP levels to be independent of both the use of oxygen and triptans, as well as verapamil. The high variance of CGRP measurements in plasma despite our large sample and efforts to pair and standardize sampling by fasting, tight timing, and detailed recording of medication and activity in the preceding 24 hours could be seen as a limitation. However, the variance is known from multiple previous studies, which merely underlines the need for a large sample size to avoid both false positive and false negative results. The data presented in the present study and previous studies (12,13,32) strongly suggest that the variance can be reduced by repeated measurements in the same individual. Future longitudinal studies are essential to understand the individual fluctuations in CGRP, as well as other potential biomarkers. Potentially, the sensitivity may also be increased by collecting paired samples of tear fluid (18) and saliva (15), as well as plasma.
Conclusions
In this large-scale and controlled study comparing well-classified patients with CH and matched controls, reduced CGRP levels are associated with CH in all states, including chronic, episodic CH in bout and episodic CH in remission. Additionally, the CGRP level was associated with the cyclic disease states of CH and found to be elevated in bout compared to remission in episodic CH. The high variance and lack of specificity make CGRP unattractive as a diagnostic biomarker for CH and for disease activity.
Clinical implications
Cluster headache is associated with reduced and not elevated plasma CGRP levels compared to controls independent of disease states (i.e. bout, remission, chronic). Increased levels of CGRP in plasma are associated with cluster headache in bout compared to cluster headache in remission. Attack frequency and acute medication did not predict plasma CGRP levels.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231223970 - Supplemental material for Reduced plasma calcitonin gene-related peptide level identified in cluster headache: A prospective and controlled study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231223970 for Reduced plasma calcitonin gene-related peptide level identified in cluster headache: A prospective and controlled study by Anja Sofie Petersen, Nunu Lund, Karl Meßlinger, Sarah Louise Christensen, Mads Barloese, Niklas Rye Jørgensen, Lisette Kogelman and Rigmor Højland Jensen in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231223970 - Supplemental material for Reduced plasma calcitonin gene-related peptide level identified in cluster headache: A prospective and controlled study
Supplemental material, sj-pdf-2-cep-10.1177_03331024231223970 for Reduced plasma calcitonin gene-related peptide level identified in cluster headache: A prospective and controlled study by Anja Sofie Petersen, Nunu Lund, Karl Meßlinger, Sarah Louise Christensen, Mads Barloese, Niklas Rye Jørgensen, Lisette Kogelman and Rigmor Højland Jensen in Cephalalgia
Footnotes
Acknowledgments
We thank Agneta Snoer for her part in initiating the Danish Cluster Headache Biobank and Adam Friis Pedersen, Mette Bisgaard, Marie-Louise Kulas Søborg and Kirstine Hartman Johansen for their contributions to data collection.
Author contributions
ASP, MB and RHJ were responsible for the concept and design of the study. ASP, NL, KM and SLC were responsible for the acquisition, analysis or interpretation of data. ASP was responsible for drafting a significant portion of the manuscript or figures. ASP, NL, KM, SLC, MB, NRJ, LK and RHJ were responsible for critical revision of the manuscript for important intellectual content. ASP and LK were responsible for the statistical analysis. ASP, NRJ and RHJ were responsible for administrative, technical or material support. MB and RHJ were responsible for study supervision.
Data availability
The Danish Cluster Headache Biobank contains sensitive information and can consequently not be shared in full form according to Danish data protection law. De-identified data that underlie the results of this article can be shared upon reasonable request.
Declaration of conflicting interests
ASP reports being a sub-investigator in trials and received a restricted research grant from Lundbeck (payed to the institution), which manufactures Eptinezumab, a monoclonal antibody targeting CGRP under investigation as a preventive treatment for cluster headache. KM reports no disclosures. SLC reports no disclosures. MB reports no disclosures. NRJ reports no disclosures. LK reports no disclosures. RHJ reports lectures for Eli-Lilly, which manufactures Galcanezumab, a monoclonal antibody targeting CGRP and FDA approved as a preventive treatment for episodic cluster headache. RHJ also reports lectures, acting as an investigator in clinical trials and receiving funding in the form of a restricted and an unrestricted research grant (paid to the institution) from Lundbeck, which manufactures Eptinezumab, a monoclonal antibody targeting CGRP under investigation as a preventive treatment for cluster headache.
Ethical statement
All participants gave written informed consent in accordance with the Declaration of Helsinki. The study was approved by the Regional Health Research Ethics Committee of the Capital Region (H-16048941) and the Danish Data Protection Agency.
Funding
This work was funded by a grant from Research foundation of the capital region of Denmark and TrygFoundation. The funders had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review or approval of the manuscript; and decision to submit the manuscript for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
