Abstract
Background
Postdromal symptoms, following headache resolution, are said to constitute a distinct phase of the migraine attack. We question the evidence for this, with regard both to the nature of such symptoms and to how often they are reported to occur.
Methods
We searched the Pubmed and Embase databases for relevant articles from their inception until 25 May 2023. We included observational studies recording the proportions of participants with migraine reporting one or more postdromal symptoms or specific individual symptoms. Two reviewers independently screened studies for relevance (agreeing on those to be included), extracted data and assessed risk of bias. Data were analyzed using random-effects meta-analysis to establish the proportions of those with migraine reporting one or more postdromal symptoms, whether among the general population or patients in clinic-based samples.
Results
Large majorities of participants in either case reported postdromal symptoms: 97% in the only population-based study, and a mean of 86% (95% CI: 71–94%) in four clinic-based studies. The most commonly reported specific symptoms were fatigue (52%; 95% CI: 44–60%), concentration difficulties (35%; 95% CI: 14–65%) and mood changes (29%; 95% CI: 9–64%), none of these being clearly described. These estimates could not be considered reliable: they were subject to substantial study heterogeneity, none of the studies applied International Classification of Headache Disorders definitions of postdromal symptoms, and all had high risk of bias.
Conclusion
Postdromal symptoms in migraine appear to be very commonly reported, but the data are unreliable with regard both to their nature and to how often they occur. Further studies are needed to conclude that they constitute a distinct phase of migraine.
Introduction
Migraine is often described in distinct phases: interictal, prodromal, aura and headache (ictal), with a less distinct postdromal phase (1). This term, poorly understood, was included in the International Classification of Headache Disorders, 2nd edition (ICHD-II) as an unspecified “resolution” phase (2). The latest (3rd) edition of ICHD (ICHD-3) provided a consensus-based definition: “A symptomatic phase, lasting up to 48 hours, following the resolution of pain in migraine attacks with or without aura” (3). It also, on limited evidence, described fatigue, mood changes and cognitive difficulties as the most common examples of postdromal symptoms (3). Nevertheless, the proportion of people with migraine who experience such symptoms, how often they occur, their nature and whether they represent a distinct phase of migraine all remain unclear.
Here, we present a systematic review and meta-analysis of the available observational data, and discuss methodological considerations and future directions for epidemiological investigations of these symptoms.
Methods
Terminology
We avoid the term “prevalence”, since the denominator, even in population-based samples, is those with migraine among the sample, not the sample as a whole. We recognise the ambiguity of “frequency” since this might imply occurrences per unit of time. In most cases we use “proportion” for the number reporting one or more postdromal symptoms expressed as a percentage of those with migraine. In the literature search and analyses, we use the terms “postdrome” and “postdromal” with whatever meanings were given to them by the original authors, but with comment later.
Literature search
We searched the Pubmed and Embase databases, using the terms ‘migraine’ AND (‘postdromal’ OR ‘postdromes’ OR ‘postdrome’), from database inception until 25 May 2023, with no language restrictions. Studies were considered eligible for meta-analysis if they were observational, included people with migraine (whether population- or clinic-based) and provided data allowing proportions to be estimated of those reporting one or more symptoms recorded as postdromal. Excluded were reviews, abstracts, conference presentations or proceedings, case reports, case series, and studies where data relevant to the meta-analysis could not be extracted. The reference lists of articles selected for full-text screening were checked for other potentially relevant references.
Data extraction
Two investigators (RHC and AKE) independently performed title, abstract and full-text screening for inclusion. The same investigators then, independently, extracted data from the included articles. Any discrepancies between the assessors were resolved by consultation between them and a third assessor (HA). We collected data on numbers and proportions of people with migraine reporting one or more postdromal symptoms. In making counts, we combined symptoms that were similarly described in different studies (e.g. “concentration difficulties” or “difficulties in concentrating”, and tiredness, asthenia or fatigue). Additionally, we collected data on study design, numbers of participants and population characteristics.
Risk of bias
Two investigators (RHC and AKE) independently assessed risk of bias in the included studies using the Joanna Briggs Institute’s Critical Appraisal Checklist for Studies Reporting Prevalence Data (4), a 9-item inventory addressing risks related to participant selection, representativeness of the population and statistical analysis, with studies scored as having high (<50%), moderate (50–69%) or low (≥70%) risk of bias according to the percentage of items without bias. Disagreements were resolved by consultation with a third investigator (HA), but studies with high risk of bias were not excluded from analysis since the data available were limited.
Outcome measures
The primary measure was the proportion, among participants with migraine, reporting one or more postdromal symptoms. The secondary measures were the proportions reporting specific individual symptoms.
Statistical analysis
We conducted meta-analysis of studies reporting our primary outcome measure. For the secondary measures, we conducted separate meta-analyses of studies (when there were at least three with ≥100 participants in total) reporting each specific individual postdromal symptoms. Separate meta-analyses were planned for clinic-based and population-based studies, and for adult and paediatric studies. A random effects model was used. Between-study heterogeneity was quantified using I2 (signifying the percentage of variation between studies attributable to study differences as opposed to expected random variation), with values >0.75 indicating considerable heterogeneity (5). Meta-regression on potential covariates was planned for variables reported in ten or more studies, in accordance with the most recent edition of the Cochrane Handbook for Systematic Reviews (5).
We performed statistical analysis with R version 4.1.0 using the ‘meta’ and ‘metafor’ packages.
Results
Search results
The database searches identified 84 unique records. We excluded 71 of these during title and abstract screening, and one during full-text review, and included 12 (6–17) in the qualitative analysis, with nine (6,7,10–14,16,17) potentially eligible for meta-analysis (Figure 1). The 12 studies included a total of 13,312 participants with migraine (6–17), of whom, in eleven studies, 74.6% were female (one study did not report the participants’ genders [14]).

Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline flowchart.
Study characteristics
An overview of the 12 studies is shown in Tables 1 and 2. Eleven were clinic-based (7–17) and one was population-based (6). Seven studies included only adult participants (6,8,9,12,13,16,17), four enrolled both adult and paediatric participants (7,10,11,14), and one (not included in the meta-analysis) only paediatric participants (15). Five studies (6,10,13,14,16) provided data on the proportions of participants with one or more postdromal symptoms (the primary outcome measure), but only the four that were clinic-based were included in the meta-analysis (10,13,14,16). Nine studies reported on individual postdromal symptoms (7–10,12–14,16,17), but only eight were eligible for meta-analysis (secondary measures) due to preselected cohorts (i.e. experiencing a pre – or postdromal phase was an inclusion criterion) (7,9,10,12–14,16,17). (Figure 2)
Overview of studies included in the meta-analysis.

ICHD: International Classification of Headache Disorders; MHD: monthly headache days; MMD; monthly migraine days; CI: confidence interval; PS: postdromal symptom(s).
Summary of study characteristics.

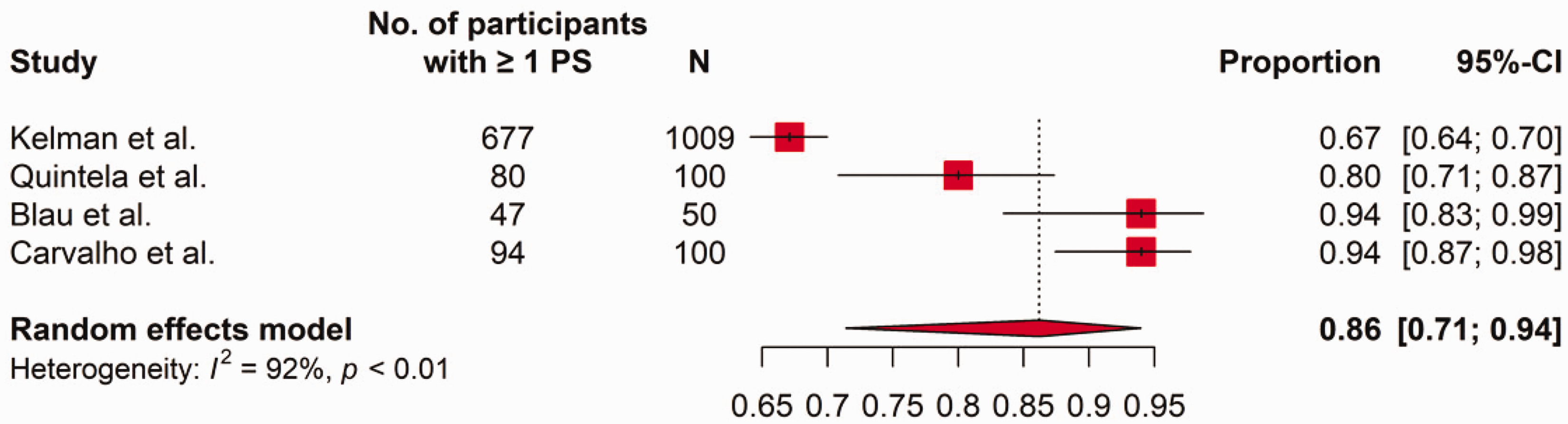
Forest plot of proportions of patients with migraine reporting one or more postdromal symptoms in clinic-based studies. CI: confidence interval; PS: postdromal symptom(s).
All 12 studies enrolled participants with migraine with or without aura. Four also allowed inclusion of participants with chronic migraine (7,10,16,17), while four excluded these (8,9,14,15) and four did not specify (6,11–13).
Six studies relied on self-completed questionnaires (6,8,12,14–16), while six used interviews to identify postdromal symptoms (7,9–11,13,17). Seven studies (6,9–11,13,15,16) recorded symptoms retrospectively and four prospectively (7,8,12,14). Nine studies used symptom lists in prompted questioning, incorporating a mean of 14.6 symptoms (range 3–28) (7,8,10,12–17); one study used only unprompted questioning (9); two studies did not specify whether prompted or unprompted (6,11).
Reporting of postdromal symptoms
None of the studies defined the postdromal phase of migraine in accordance with ICHD-3 (3). Moreover, symptoms were considered postdromal in five studies (6,7,11,12,16) even during residual, non-migrainous headache, but only after complete resolution of headache in seven others (8–10,13–15,17). The single population-based study, a multi-country online survey, enquired whether participants experienced a “postdromal phase after the attack”. The precise wording was not clear from the study publication, but examples were given such as “fatigue”, “hangover feeling” and “sleep helps” (6).
With regard to our primary outcome measure, almost all (97%) of the participants with migraine in the population-based study (N = 11,226) reported at least one such symptom. In the four clinic-based studies providing data (total N = 1259) (10,13,14,16), a mean of 86% (95% CI: 71–94%) of participants reported at least one postdromal symptom, but with I2 = 93%.
In the single paediatric study, also clinic-based, 91% of participants reported “postdromal” symptoms with onset before headache resolution, while 82% reported symptoms with onset after headache resolution (15). The study publication did not provide a clear account of the symptoms in either case.
With regard to secondary measures, eight clinic-based studies (7,9,10,12–14,16,17) provided data on a total of 75 individual postdromal symptoms, with sufficient data available for meta-analysis of ten of these (Table 3). The four most commonly reported were – in decreasing order – fatigue (by 52% of participants), concentration difficulties (35%), mood changes (29%) (none of these being clearly described) and yawning (22%), with all others reported by 15% or fewer (Table 3).
Proportions of participants with migraine reporting specific individual postdromal symptoms.

Risk of bias
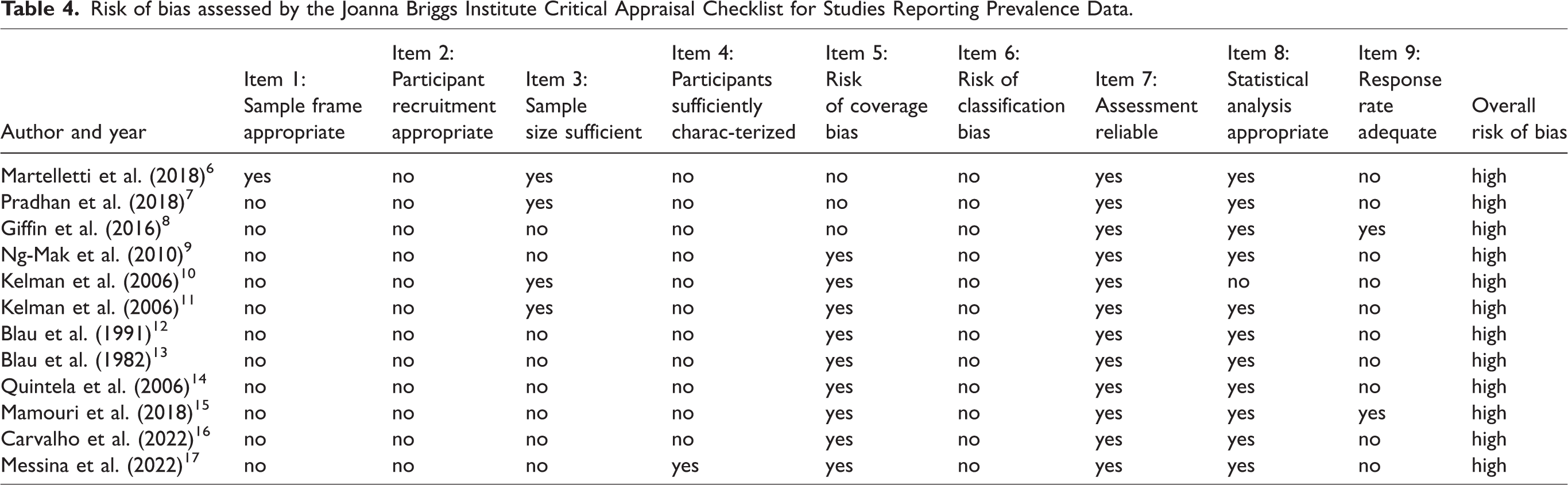
Our interrater agreement was 83% for the risk of bias assessment, which revealed high risk in all included studies. All relied on non-random sampling, and, except for one study, all failed adequately to characterize the study population (Table 4). In addition, eleven studies (7–17) used non-representative sampling frames, and eight had inadequate sample sizes (8–9,12–17). None used a validated method to record postdromal symptoms defined in accordance with ICHD-3.
Risk of bias assessed by the Joanna Briggs Institute Critical Appraisal Checklist for Studies Reporting Prevalence Data.
Discussion
This is the first meta-analysis providing an estimate of the proportion of people with migraine who report postdromal symptoms as a phase of the migraine attack. In the only population-based study, which adopted a very broad definition of postdromal symptoms (including, for example, “sleep helps”), such symptoms were reported almost universally (by 97% of participants) (6). In four clinic-based studies, a large majority (mean 86%) of patients reported such symptoms, but the high heterogeneity between these studies renders this estimate unreliable. The single paediatric study, also clinic-based, found a similar proportion (82%) reporting symptoms with onset after headache resolution (15). Thus, while the proportion is apparently very high, there is substantial uncertainty in estimating it.
With regard to individual symptoms, estimates are equally unreliable because of study heterogeneity (see Table 3), and, since they derive from clinic-based studies, cannot anyway be generalized to the broader population of people with migraine.
Methodological considerations
The dearth of evidence contributing to this meta-analysis was exacerbated by multiple differences between the studies in their approaches to assessment of postdromal symptoms, which may or may not have contributed to their heterogeneity (there were too few studies to perform meta-regression [5]). This heterogeneity is likely to have arisen from a number of methodological and clinical differences between the studies.
First, some studies introduced risks of acquiescence bias by prompting, whereas others did not. Among the former, lists of postdromal symptoms were of widely varying length. Studies with prompting (13,14,16) tended to find higher frequencies of symptoms, a factor very likely to contribute to an overall higher probability of reporting a postdromal phase.
Second, studies applied a range of definitions, none in accordance with ICHD. This was largely because “postdrome” was first formally defined in ICHD-3 in 2018 (3); nevertheless, two studies conducted after 2018 did not expressly use this definition either (16,17). A central element in this definition is that postdromal symptoms occur “following headache resolution”, yet several studies allowed residual, “non-migraine” head pain (6,10,11,13), introducing the risk of identifying symptoms associated with the headache phase (photophobia, phonophobia, nausea) as postdromal.
Third, casemix might have been an unrecognized influence, especially among the clinic-based studies. Findings from two studies suggested that participants with aura were more likely to report postdromal symptoms (10,14), and that those with frequent aura reported more postdromal symptoms (10). With regard to chronic migraine, headache frequency was associated with more postdromal symptoms in one study (16), but premonitory and postdromal phases, each of up to 48 hours’ duration, inevitably overlap when migraine days are ≥8/month (3). Several studies have noted similar symptoms (fatigue, yawning and neck pain) in both phases (7–9,14,17), while ICHD-3 states that “it is impossible to distinguish the individual episodes of headache in patients with such frequent or continuous headaches” (3). Four studies excluded chronic migraine (9,10,15,16), sensibly in our view. Differences between study populations in their proportions with aura or chronic migraine are likely to have influenced the proportions reporting postdromal symptoms. The studies may also have varied in terms of other clinical characteristics associated with postdromal symptoms, including the presence of trigger factors, occurrence of premonitory symptoms, the pain intensity of migraine headache, and vomiting (10). Variations in these factors may likewise have contributed to the substantial between-study heterogeneity.
Fourth is treatment effect, with two predictable consequences, each potentially leading to differences in reporting across studies. On the one hand, side effects of acute treatment may be misinterpreted as postdromal symptoms, and vice versa. On the other, effective treatment of headache may unmask associated (ictal) symptoms, which are then erroneously reported as postdromal. A review of pooled data from seven double-blinded, placebo-controlled trials with eletriptan and sumatriptan found that reports of somnolence increased after triptan use, but also in response to placebo (18).
These considerations, coupled with the high risk of bias in all studies, mean that the true proportion of people with migraine who experience postdromal symptoms remains unknown.
Comments on timing
Clearly, the reporting of symptoms considered to be postdromal is likely to be influenced by the duration of the period defined as postdromal. Additionally, the impacts on health, daily activities and quality of life of postdromal symptoms are determined not only by their frequency but also by their duration. It was recently proposed that defining duration as 24 rather than 48 hours would sufficiently capture the full impacts of postdromal symptoms (19). However, in our analysis, 27.5% of participants in the population-based study and 12–40% in the clinic-based studies reported postdromal symptoms lasting longer than 24 hours (8,10,16) (6% reported >48 hours in one study [16]). The issue arising is the distinction between postdromal and interictal symptoms, which should have a mechanistic basis rather than be determined by arbitrary definitions. Further studies are needed, particularly clinical studies of individual postdromal symptoms. Meanwhile, these data suggest that the ICHD-3 definition of 48-hour duration is the most appropriate, although it remains opinion-based.
A separate issue, which needs also to be the subject of clinical studies, is the time of onset: do postdromal symptoms commence during the headache phase (and survive beyond it), or after headache resolution? ICHD-3 is not clear on this, and, of the studies we analyzed, only the single paediatric study considered it (15). While a mechanistic basis is needed here also, this issue is central to whether or not “postdrome” exists as a distinct phase of migraine.
Comments on “postdrome” as a nosological concept
In the former case just referred to (postdromal symptoms commencing during but surviving beyond the headache phase), the definition of “postdrome” rests on the definition of a central (ictal) phase of migraine that begins and ends with headache, whereas this phase might more logically be considered to embrace not just headache but all other symptoms arising with it, not all of which may resolve as the headache resolves. This approach is being adopted in clinical trials that attach equal importance among outcome measures to headache and “most bothersome symptom” (20). In this approach, only symptoms with onset after headache resolution can be regarded as postdromal.
Whether such symptoms occur is not at present certain. If they do, they are only arbitrarily distinguished from interictal symptoms, unless it can be shown that there are ongoing mechanisms actively generating non-headache symptoms that persist for a period after headache resolution. Should this be the case, the definition of “postdrome” again rests on making headache central to the definition of the ictal phase of migraine.
Such a definition allows terminology that is descriptively convenient, and perhaps acceptable for this reason. But attributing centrality to headache sits uncomfortably with a definition of migraine with aura that does not require headache (2,3).
Future directions
Robust epidemiological data are needed, both to provide clear case definitions for experimental studies exploring the neurobiological underpinnings of postdromal symptoms and to estimate their contribution, if any, to overall disease burden. At present, the dearth of data limits our ability not merely to characterize postdromal symptoms with any accuracy but even to determine whether the term “postdromal phase” is appropriate. The need for future studies is clear, with agreed methodology for case ascertainment as a prerequisite.
The way forward requires expert consensus, especially on timing (onset and duration) of symptoms in relation to headache resolution. We recommend adoption for the time being of the ICHD-3 definition of “postdrome” (3). While 48 hours following headache resolution is opinion-based, any change would be arbitrary pending acquisition of the epidemiological evidence that is currently lacking. However, it appears essential to take explicit account of whether headache resolution is spontaneous or a consequence of acute treatment. If the headache phase is artificially truncated, symptoms might be deemed postdromal that would not be otherwise (any effect of preventative treatment is largely unknown).
With regard to case ascertainment, assuming this will usually be by questionnaire in epidemiological studies, the problem of prompting and its associated acquiescence-bias is a difficult one. We suggest open-ended questions initially, to be followed by a short prompting list that includes some highly unfeasible options. On the basis of our analysis, and until further data become available, we suggest including (from Table 3), as likely symptoms, only fatigue, concentration difficulties, mood change, yawning and photophobia. However, the first three of these are imprecise, and photophobia is notoriously difficult to express in lay terms. All may occur during the headache phase. It seems self-evident that studies should exclude chronic migraine: the concept of “postdrome” has no meaning without clear episodicity.
In due course, experts should consider whether any one or more symptoms are sufficiently specific to constitute (and define) a separate postdromal phase.
Ideally, and again in due course, epidemiological studies should endeavor to include symptoms after headache resolution in assessments of burden. Whether or not the existence of a distinct postdrome is established, and however it is defined, it is clear that not everyone living through a migraine attack recovers normal function immediately after headache resolution. As to assessing impacts of such symptoms on health, activity and quality of life, methodological guidelines will need to be developed. We recognize that, while questionnaires and interviews relying on recall may yield valuable information on the frequency of symptoms surviving or developing after headache resolution, prospective attack-by-attack recording, eliminating recall errors, is far better able to provide insight into the proportion of attacks in which these symptoms occur, when they commence, their duration, severity, and various impacts. Such benefits are negated, however, if studies are clinic- rather than population-based.
Finally, is the question of sample size. With regard to statistical power calculations, we have reported frequencies among those with migraine, not in broader populations, an important point in estimating likely prevalence, which determines the size of randomly selected population samples. Unknowns, for which allowance must be made, are the proportion among such samples who will reliably respond to enquiries of this sort and the factors that might influence this.
Strengths and limitations
We followed established methods for the literature search and meta-analysis. However, we were limited in the scope of the analysis, and in the conclusions we could draw, by the low number, poor quality and heterogeneity of the few studies, which precluded meta-regression and sensitivity analyses. We were obliged to combine prompted and unprompted symptoms, and could make no subgroup analyses according to definition of postdrome, method of enquiry or characteristics of study participants. It should also be noted that we excluded studies of specific subgroups of migraine (e.g. chronic migraine), since these subgroups could unduly alter the proportion of participants with postdromal symptoms. Lastly, the heterogeneity statistic, I2, which we report, may be biased in meta-analyses with relatively few studies (21).
Conclusions
The few published data indicate that symptoms in a 48-hour period after headache resolution – fatigue, concentration difficulties and mood changes in particular – are reported commonly by people with migraine, but they do not allow reliable estimates of the proportions who do so, either overall or with regard to specific individual symptoms. Neither are the data sufficient to conclude that a postdrome occurs as a definably separate phase of the migraine attack.
Further research, both epidemiological and clinical, is needed, but agreements on definition and methodology for assessment are required first. Any such agreements will be largely opinion-based initially, and will need to be reviewed as better data are gathered.
Key findings
Studies of migraine postdromal symptoms (those occurring within a defined period following headache resolution) are few, and characterized by variations in definition and methods of case ascertainment, and high risk of bias. The proportion of people with migraine experiencing such symptoms remains unknown, but may be high. In clinical samples of patients with migraine, fatigue (55%), concentration difficulties (35%), and mood changes (29%) are most frequently reported, but without clear definitions. Whether such symptoms constitute a definably separate postdromal phase of the migraine attack is, for now, uncertain.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HA reports personal fees from Teva, outside of the submitted work. MA reports receiving personal fees from AbbVie, Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals outside of the submitted work.
MA received institutional grants from Lundbeck Foundation, Novo Nordisk Foundation, and Novartis. MA reports serving as associate editor of Cephalalgia, associate editor of The Journal of Headache and Pain, and associate editor of Brain.
RHC, AKE, and TJS declare that they have no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
