Abstract
Background
This longitudinal cohort study aimed to investigate changes in migraine-related outcomes following COVID-19 infection and vaccination.
Methods
We identified 547 clinically diagnosed migraine patients from the Leiden Headache Center who kept a headache E-diary during the COVID-19 pandemic (February 2020 to August 2022). We sent a questionnaire to register their COVID-19 infection and/or vaccination dates. After applying inclusion criteria, n = 59 participants could be included in the infection analysis and n = 147 could be included in the vaccination analysis. Primary outcome was the change in monthly migraine days (MMD) between 1 month prior and 1 month post COVID-19 infection or vaccination. Secondary outcome variables were change in monthly headache days (MHD) and monthly acute medication days (MAMD).
Results
Vaccination against COVID-19 was associated with an increase in MMD (1.06; 95% confidence interval [CI] = 0.57–1.55; p < 0.001), MHD (1.52; 95% CI = 0.91–2.14; p < 0.001) and MAMD (0.72; 95% CI = 0.33–1.12; p < 0.001) in the first month post-vaccination. COVID-19 infection solely increased the number of MAMD (1.11; 95% CI = 0.10–1.62; p < 0.027), but no statistically significant differences in MMD or MHD were observed.
Conclusions
Our findings imply that vaccination against COVID-19 is associated with an increase in migraine, indicating a possible role of inflammatory mediators in migraine pathophysiology.
Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a single-stranded RNA virus and causes coronavirus disease 2019 (COVID-19). Although initially perceived as a disease mainly affecting the respiratory system, it became apparent during the pandemic that COVID-19 has a broader effect, among which are neurological manifestations including headache. 1 The COVID-19 pandemic also had a major impact on the care of people who suffer from migraine. Consequently, telemedicine became an effective way to ascertain continuation of headache care for patients. The Leiden Headache Center was already using telemedicine before the outbreak of the COVID-19 pandemic, including the use of validated headache E-diaries.2,3 E-diaries are a useful tool in migraine care and help to gain insight into attack frequency and treatment response.4,5 As a result of the Leiden Headache Center’s prospective E-diary data collection during the COVID period as part of regular care, we have a unique opportunity to investigate the effects of COVID-19 infection and vaccination on an individual level in migraine patients. At this point, there are still limited data about the effect of a COVID-19 infection on primary headache disorders. Headache in the acute phase after COVID-19 infection seems to be more common in people with a previous history of headache disorders and often resembles the features of that headache disorder. 6 Patients with a history of migraine self-reported to experience longer and more severe headaches attributed to COVID-19.7,8
In The Netherlands, vaccination against COVID-19 started in January 2021, with four available vaccine types. The aim of vaccines is to deliver or produce antigens derived from the pathogen to trigger immune responses without causing considerable disease manifestations. The immune system recognizes the vaccine as foreign and mounts a response similar to that of an infection, but, because the pathogen is not able to multiply, the body is able to build immunity to the pathogen without causing the disease. Viral vector vaccines, such as Ad26.COV2.S (Janssen, Beerse, Belgium) 9 and AZD1222 (AstraZeneca, Cambridge, UK) 10 , use viral DNA that is introduced into the target cells nucleus, where it is transcribed into mRNA. The mRNA strains introduced into the body will be translated and presented by the antigen-presenting cells (APC) to the Toll-like receptors (TLR), simulating a viral infection. Recognition by the TLR stimulates a T helper type 1 response by activating interferon (IFN)-1, which results in the engagement of several mediators such as cytokines and the complement system. 11 This leads to a strong antibody production as well as a T-cell response, demonstrated by an increase in IFNγ, tumor necrosis factor (TNF)-α and interleukin (IL)-2.11,12 The mRNA vaccines, such as BNT162b2 (Pfizer, New York, NY, USA/BioNTech, Mainz, Germany) 13 and mRNA-1273 (Moderna, Cambridge, MA, USA) 14 , use a different technique by solely using a RNA strand coding for the antigenic sequence of the spike protein (S-protein), modified to increase mRNA translation in vivo. 15 After mRNA translation, the newly synthesized S-proteins are presented on the surface of the APCs, activating the immune system cascade mentioned above. The mRNA vaccines thus bypass the need for viral DNA to be integrated into the nucleus, and instead directly provide the instructions for the body to produce the S-protein fragment of the SARS-CoV-2 virus.
Various side effects of the COVID-19 vaccines have been reported, among which injection site pain, swelling, fever and headache. 16 Headache is the most common neurological symptom reported, with an incidence of headache in the days after vaccination of >50%, and with rates varying between vaccine types and reports.9,13,17
Headache incidence after vaccination with Pfizer/BioNTech was significantly higher in patients with a migraine diagnosis compared to a healthy control group (69.2% and 37.9%, respectively, p = 0.012). 18 Other studies reported a higher pain intensity and duration of the post-vaccine headache in patients with a history of migraine for both Pfizer/BioNTech and AstraZeneca.19,20 Finally, a questionnaire study found migraine attacks to have a higher pain intensity, longer duration and reduced responsiveness to usually effective migraine attack medication after vaccination. 21 Important to note is that these studies should be interpreted with care because of their small retrospective nature and therefore a liability to recall bias.
Migraine attacks are considered to be the result of natural fluctuations in neuronal excitability and trigger factors. 22 Inflammatory mediators, such as peptides [predominantly calcitonin gene-related peptide (CGRP), substance P and neurokinin A], cytokines, metabolites (prostaglandins), nitric oxide and other molecules involved in the inflammatory response, have been implicated in the pathophysiology of migraine. These mediators are considered to play a role in the sensitization of trigeminal neurons, leading to the development of the migraine headache.23,24 However, the relationship between inflammatory mediators and migraine is complex and still not fully understood. Although the general population may experience an increase in other non-migrainous headache days post-vaccination, patients with migraine may have a higher impact, possibly leading to an increase in migraine or other headache days. 18 The objective of the present study was to determine whether COVID-19 vaccination or infection affects the number of monthly migraine days in individuals with migraine by merging E-headache diary information with the dates of infection and vaccination, which can be easily obtained through our national registration system.
Methods
For this longitudinal cohort study, we identified patients whose E-headache diaries were collected during the COVID-19 pandemic, covering a period between February 2020 and August 2022.2,4 Patients were included if they had a confirmed diagnosis of migraine at the Leiden Headache Center, were aged ≥18 years and filled out the E-headache diary for at least three subsequent months with at least 80% compliance per month. This study was approved by the medical ethics committee of the Leiden University Medical Center (LUMC) and all patients provided their informed consent.
During the study period, each morning at 08.00 h, an alert was sent to all patients, which could be accessed on a mobile device or a computer. The questionnaire contained from six to 31 questions, depending on the answers provided, about the presence of headache including its detailed characteristics and associated symptoms (one sided, throbbing, intensity, increasing with physical activities, photophobia/phonophobia, nausea, vomiting), presence of visual aura symptoms and their characteristics, use of acute medication, change in preventive medication, presence of menstruation, general wellbeing and pain coping. Questions covered the 24 h of the previous day (from midnight to midnight), enabling patients to enter data first thing in the morning. If the E-diary was not completed by the end of the afternoon, a reminder was sent. Entries could not be edited after completion of the questionnaire and questionnaires were time-locked after 48 h. Patients were encouraged to enter data daily. An automated algorithm based on ICHD-3 criteria verified for each day whether criteria for headache and migraine were met. 25 A headache day was defined as a day with headache symptoms lasting ≥1 h. Migraine days were defined based on detailed characteristics as described in the ICHD-3 criteria, including all days with triptan intake. 3 Additionally, days with visual aura symptoms lasting 5–60 min and days with triptan intake were considered migraine days. By definition, each migraine day was also considered a headache day.
An online questionnaire was sent to acquire information about a potential COVID-19 infection or vaccination. First, the patients were asked if they had noticed an increase in migraine days after infection or vaccination, both in the case of COVID-19 and for other pathogens, using a three-point scale. Second, patients were asked to report the number of COVID-19 infections, the method of testing and the corresponding date(s). Finally, they were asked to report the number and type of vaccinations and the corresponding date(s), which could be found in the “Corona check” app or website (https://coronacheck.nl), developed by the Dutch Ministry of Public Health, Welfare and Sport.
Statistical analysis
We integrated dates of infection and vaccination with the E-diary data, removing months in which ≥1 infection and/or vaccination date was present and months with ≤80% compliance. Baseline months were defined as 1 month (28 days) before the reported infection or vaccination date. Our primary outcome was change in number of monthly migraine days (MMD) between baseline month and one month (28 days) after the reported infection and/or vaccination date. Secondary outcome variables were change in monthly non-migrainous headache days (MHD) and monthly acute medication days (MAMD). Subgroup analyses for change in monthly triptan days and monthly analgetic days were performed. We predefined that, if a significant change in the outcome was found in the first month, we would investigate whether this change in month 1 was significantly declined in month 2. Additionally, we aimed to evaluate the effect of type of vaccination (mRNA or viral vector) on our outcome measures.
Baseline patient characteristics were described using descriptive statistics. Chronic migraine was defined as ≥15 headache days per month with ≥8 migraine days per month. For each outcome variable, a linear mixed model was fitted for the baseline month, and both the first (month 1) and the second month (month 2) after infection/vaccination. The patient was included as random effect to account for within-patient correlation. Possible confounding factors, including age, sex, body mass index, chronic migraine, changes in preventive medication and number of previous infections or vaccinations, were treated as fixed effects. We evaluated differences at a significance level of 5%, and examined the marginal effect on MMD. The marginal effect represents the mean difference in MMD between the baseline month, month 1 and month 2, while holding all other variables constant. All analyses were performed using RStudio (R Foundation for Statistical Computing, Vienna, Austria), version 4.0.5, and the lmer package was used to fit linear mixed-effects models.
Results
Out of 12,843 patients in the Leiden Headache Center database at the time of selection, we identified 547 clinically diagnosed migraine patients who fulfilled the initial inclusion criteria, out of whom 336 (61.4%) responded to the questionnaire. For the infection analysis, we excluded patients who did not experience any infection or had insufficient compliance in the 2 months surrounding the infection date, resulting in a representative group of n = 59 migraine patients with a combined total of 150 months of E-diary data (supplementary Figure 1). Concerning the vaccination analysis, 189 patients out of the group of 336 responders were excluded as a result of either lack of vaccination or insufficient compliance surrounding the vaccination dates, resulting in a representative group of n = 147 migraine patients with a combined total of 536 months of E-diary data (supplementary Figure 2). The mean age of responders to the questionnaire was 50.2 years and 81.3% were female. Baseline characteristics of the responders and those participants included in the infection or vaccination analysis are shown in Table 1. Of the 147 individuals in the vaccination group, the majority received solely one or more mRNA vaccines (73%), 16 participants (11%) exclusively received viral vector vaccines and 23 subjects (16%) received at least one vaccine of both types (supplementary Table 1).
Baseline characteristics of responders, included participants in the COVID infection analysis and included participants in the COVID vaccination analysis.
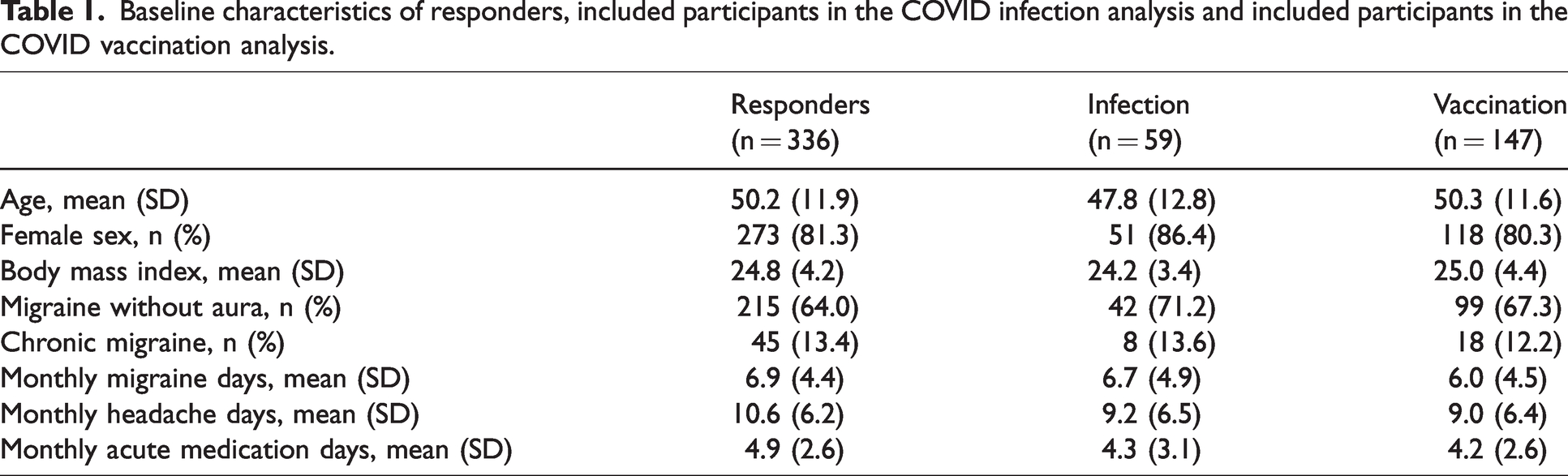
Patients self-reported perception
Data were collected through a one-time questionnaire from a total of 336 participants. Among the respondents, 164 patients (49%) with migraine reported a history of COVID-19 infection. Of these patients, 64.6% (n = 106/164) reported to have precepted no change in MMD, whereas 26.2% (n = 43) perceived an increase, 1.2% (n = 2) perceived a decrease and 7.9% (n = 13) were uncertain about this.
Regarding COVID-19 vaccination, 272 out of 336 (81%) patients with migraine reported having received a COVID-19 vaccine. Among them, 72.4% (n = 197) reported to have perceived no noticeable change in MMD, 12.9% (n = 35) perceived an increase, 3.7% (n = 10) perceived a decrease and 11.0% (n = 30) were uncertain.
Effect of COVID-19 infection
After correction for confounders, no difference in MMD was found in the first month (0.08; 95% confidence interval [CI] = –0.84 to 0.99; p = 0.983) after infection compared to the baseline MMD (Figure 1a). We also did not find a difference in MHD (0.85; 95% CI = −0.15 to 1.85; p = 0.112) but MAMD was increased in the first month after infection with 1.11 days (95% CI = 0.10–1.62; p = 0.027). Crude means without statistical modelling are visualized in Figure 1, adjusted estimates per outcome measure are shown in Table 2. Subgroup analysis showed an increase in days with analgetic intake in the first month after infection compared to baseline (1.18; 95% CI = 0.43–1.92; p = 0.002). However, this effect was not seen for medication days with triptan intake (–0.05; 95% CI = –0.53 to 0.43; p = 0.846). In addition, the increase in analgesic intake days was observed predominantly when combining non-migrainous headache days with non-headache days (0.79; 95% CI = 0.23–1.35; p = 0.006), but was not found for non-headache days alone (0.33; 95% CI = –0.07 fto 0.72; p = 0.104).

Crude mean ± SEM for migraine-related outcomes during baseline and first 2 months following infection. *p ≤ 0.05 in the linear mixed model. (a) Represents the mean change in monthly migraine days (MMD), (b) represents the mean change in monthly headache days (MHD) and (c) represents the mean change in monthly acute medication days (MAMD).
Change in monthly migraine days (MMD), monthly headache days (MHD) and monthly acute medication days (MAMD) for month 1 and month 2 post COVID-19 infection compared to the baseline month.

One month is defined as 28 days. Total months of E-diary data was n = 150 months from n = 59 participants.
Effect of COVID-19 vaccination
We found an increase in MMD in month 1 after vaccination compared to the baseline month (1.06; 95% CI = 0.57–1.55; p < 0.001) (Figure 2a). Analysis of marginal effects showed a significant difference in MMD between month 1 and month 2 post-vaccination (0.59; 95% CI = 0.00–1.17; p = 0.049), indicating that, in month 1, more MMD occurred compared to month 2 post-vaccination. Additionally, we found an increase in both MHD (1.52; 95% CI = 0.91–2.14, p < 0.001) and MAMD (0.72; 95% CI = 0.33–1.12; p < 0.001) in month 1 post-vaccination. Crude means without statistical modelling are visualized in Figure 2, adjusted estimates per outcome are shown in Table 3. Subgroup analysis showed an increase in both analgetic intake (0.49; 95% CI = 0.15–0.84; p = 0.005) and triptan intake (0.51; 95% CI = 0.22–0.80; p = 0.001) in month 1 after vaccination compared to the baseline month. Lastly, solely for the mRNA vaccination we found an increase in MMD in month 1 after vaccination compared to the baseline month (0.50; 95% CI = −0.54 to 1.55; p = 0.347) (supplementary Table 2). Analysis of marginal effects showed no evidence to suggest a difference between the effect of the mRNA vaccines and the viral vector vaccines on the number of MMD (0.50; 95% CI = 0.00–1.00; p = 0.35).

Crude mean ± SEM for migraine-related outcomes during baseline (28 days) and month 1 and month 2 following vaccination. *p ≤ 0.05 in the linear mixed model. (a) Represents the mean change in monthly migraine days (MMD), (b) represents the mean change in monthly headache days (MHD) and (c) represents the mean change in monthly acute medication days (MAMD).
Change in monthly migraine days (MMD), monthly headache days (MHD) and monthly acute medication days (MAMD) for month 1 and month 2 after COVID-19 vaccination compared to the baseline month.
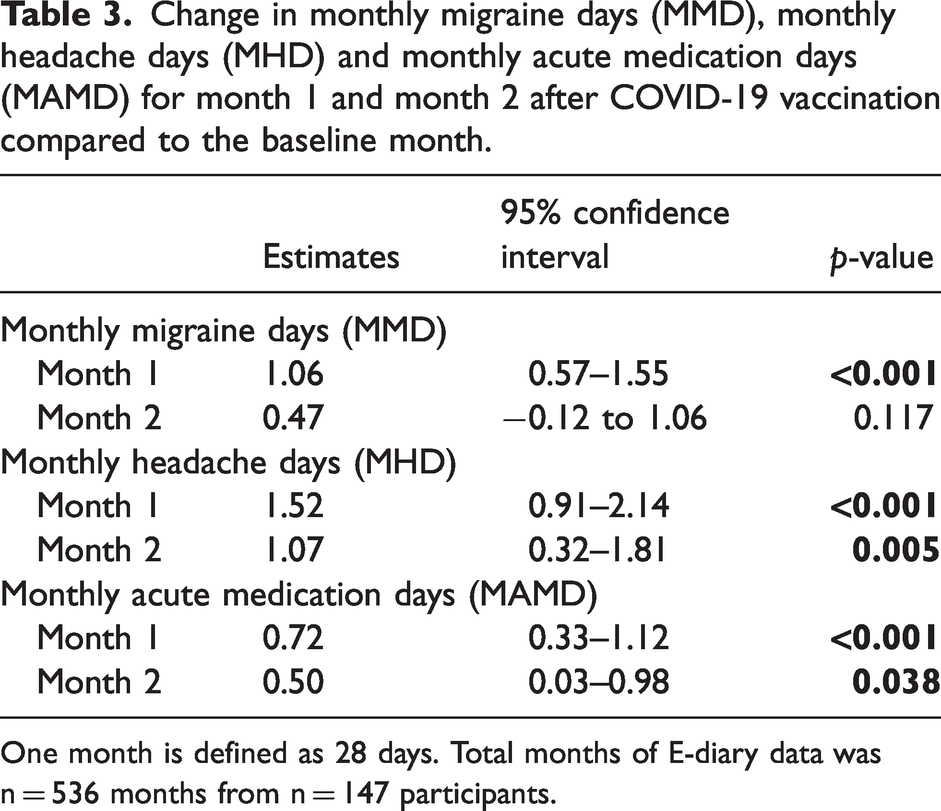
One month is defined as 28 days. Total months of E-diary data was n = 536 months from n = 147 participants.
Discussion
Our longitudinal cohort study examined the relationship between COVID-19 vaccination or infection and the frequency of migraine. We observed a rise in the number of MMD, MHD and MAMD during the first month after vaccination compared to the preceding month. Interestingly, there was a decrease in MMD during the second month after vaccination, indicating that the effect is temporary. These findings may suggest that the vaccine may decrease the threshold for migraine attacks, possibly because of an increase in circulating inflammatory mediators.
Inflammation plays a crucial role in migraine pathophysiology, as indicated by the increased prevalence of chronic inflammatory diseases such as multiple sclerosis and inflammatory bowel disease in patients with migraine. 26 During a migraine attack, plasma levels of inflammatory cytokines such as IL-6, IL-10 and TNF-α are increased.26,27 The release of neuropeptides, such as CGRP, pituitary adenylate cyclase-activating polypeptide, substance P and neurokinin-A, along with neurogenic inflammation, are also implicated in migraine pathophysiology.24,28,29 Activation of trigeminal nociceptors by inflammatory cytokines can lead to the release of these peptides, initiating a sterile meningeal inflammatory reaction. 30 This eventually leads to the release of nitric oxide, activation of glial cells and mast cell degranulation, resulting in sustained activation and sensitization of meningeal nociceptors, possibly contributing to migraine headache. 31 Apart from inflammation, inflammatory cytokines have also been implicated in modulation of the pain threshold and activation of trigeminal nociceptors. 24 Therefore, it is hypothesized that COVID-19 vaccination may initiate the release of inflammatory mediators, which in turn may trigger migraine headaches. 32
Our study has some limitations. First, the infection analysis was limited by a smaller sample size with less diary days and a low incidence of reported COVID-19 infections among our cohort of subjects with migraine. To ensure reliability of the results, we applied an E-diary compliance criterion of ≥80% per month, which further reduced our sample size. Despite this inherent limitation, the high compliance rate enabled us to precisely capture changes in migraine frequency and medication usage around the time of COVID-19 infection. However, this may explain why we found a significant increase in MAMD but not in MMD and MHD, because we had less statistical power compared to the vaccination data. Although the exact day and method of diagnosis of the infection may have differed between participants, leading to less accurate estimation exact infection date compared to the vaccination date, this potential limitation is mitigated by the fact that we analyzed the data on a monthly scale. By contrast, a strength of the vaccination analysis was that our participants could easily access their exact vaccination date(s) and type(s) through the “Corona check” app/website developed by the Dutch Ministry of Public Health, Welfare and Sport, ensuring accuracy. Despite the fact that regular care was given and treatment changes were made, we were still able to show an overall negative effect of COVID-19 vaccination. It is important to note that, although the effect size appears to be small, even subtle changes can have varying degrees of impact depending on the individual. Lastly, an additional limitation is the absence of validated questionnaires assessing disability, which might have offered a more comprehensive evaluation.
Discrepancy in our findings for vaccination in comparison with COVID-infection may also be a result of higher peak antibody levels following vaccination compared to natural infection. 33 However, it is important to note that individual antibody levels can vary depending on many factors, including the severity of infection. Interestingly, although COVID-19 infection did not lead to an increase in MMD, it did result in a higher number of MAMD. This could be a result of the increased use of pain medication to manage non-neurological or non-migrainous symptoms of COVID-19. Subgroup analysis indeed suggests that the increase in MAMD was driven solely by an increase in monthly analgesic days and not triptan days. Indeed, this increase in analgesic days was primarily observed on days with non-migrainous headache, indicating that the surge in analgesic intake is mainly associated with non-migrainous days. By contrast, after vaccination, both analgesic and triptan use increased in the first month, suggesting a stronger association with migraine. Moreover, the potential impact of analgesic use for non-headache purposes during the study period can be a possible explanation for the absence of an increase in MMD after COVID-19 infection. It is plausible that individuals resorted to analgesics to alleviate other COVID-19-related symptoms, which might have affected the occurrence of migraine attacks as a preventive measure.
A principal strength of this study is the use of our validated E-headache diary. As a result of its time-locked design patients are prevented from changing or delaying their input, thereby evading recall bias. 2 The automated algorithm ensures that every migraine day fulfilled necessary ICHD-3 criteria, consequently making the final results more reliable. 3 Moreover, all patients completed their E-diaries as part of their routine care or for other research purposes. At the time of data collection, none of them were aware that these E-headache diary data might be used for analyzing the effect of COVID vaccination or infection. This approach helped to minimize the potential nocebo effect with vaccination and negative expectations of patients. Furthermore, our detailed E-headache diary tool ensured compliance in the post-infection month, and the validated algorithm ensures that assessment is not based on patients own interpretation of their headaches.
As far as we are aware, there is currently no published research that has investigated the potential impact of COVID-19 infection or vaccination on the severity of pre-existing migraine in a longitudinal cohort study. Importantly, our vaccination analyses results are consistent with a retrospective questionnaire study that showed an increase in length and severity of headache attacks in patients with migraine in the first week after COVID-19 vaccination. 21 This study also reported that headaches were less responsive to usually effective pain medication, which is consistent with the increase in MAMD found in the present study. 21 Although the general population may experience mild and temporary headaches after vaccination, individuals prone to migraine, who are already more susceptible to headache attacks, might encounter a more pronounced impact. Our study offers valuable insights into how patients with migraine specifically respond to COVID-19 vaccination.
This follow up study shows that vaccination against COVID-19 is associated with an increase in migraine in the first month. COVID-19 infection solely increases the number of acute medication intake days. Our findings suggest that vaccination against COVID-19 is associated with an increase in migraine, which could indicate the involvement of inflammatory mediators in the pathophysiology of migraine and can impact the intrinsic threshold for upcoming attacks.
Clinical implications
This follow-up study suggest that vaccination against COVID-19 is associated with an increase in migraine. Our findings could indicate that the involvement of inflammatory mediators in the pathophysiology of migraine can impact the intrinsic threshold for upcoming attacks. COVID-19 infection solely increases the number of acute medication intake days.
Supplemental Material
sj-jpg-1-cep-10.1177_03331024231198792 - Supplemental material for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study
Supplemental material, sj-jpg-1-cep-10.1177_03331024231198792 for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study by Britt W. H. van der Arend, Mirthe M. Bloemhof, Alle G. van der Schoor, Erik W. van Zwet PhD, Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-jpg-2-cep-10.1177_03331024231198792 - Supplemental material for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study
Supplemental material, sj-jpg-2-cep-10.1177_03331024231198792 for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study by Britt W. H. van der Arend, Mirthe M. Bloemhof, Alle G. van der Schoor, Erik W. van Zwet PhD, Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024231198792 - Supplemental material for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study
Supplemental material, sj-pdf-3-cep-10.1177_03331024231198792 for Effect of COVID vaccination on monthly migraine days: a longitudinal cohort study by Britt W. H. van der Arend, Mirthe M. Bloemhof, Alle G. van der Schoor, Erik W. van Zwet PhD, Gisela M. Terwindt in Cephalalgia
Footnotes
Author contributions
BWHvdA, EWvZ and GMT contributed to conception and design of the study. BWHvdA, MMB, AGvdS and EWvZ contributed to acquisition and analysis of data. BWHvdA, MMB and GMT contributed to drafting a significant portion of the manuscript. All authors contributed to the critical review of the manuscript.
Declaration of conflicting interests
B. W. H. van der Arend and G. M. Terwindt report independent support from the Dutch Research Council (849200007) and the Dutch Brain Foundation (HA2017.01.05). G. M. Terwindt reports consultancy or industry support from Abbvie/Allergan, Lilly, Lundbeck, Novartis, and Teva, and independent support from the European Community, Dutch Heart and Brain Foundations, Dutch Research Council, and Dioraphte. E. W. van Zwet, M. M. Bloemhof and A. G. van der Schoor report no disclosures.
Ethical statement
This study was approved by the medical ethics committee of the Leiden University Medical Center (LUMC) and all patients provided informed consent.
Funding
This project was not funded by any of the grants indicated in the COI section.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
