Abstract
The quality of clinical trials is essential to advance treatment, inform regulatory decisions and meta-analysis. With the increased incidence of idiopathic intracranial hypertension and the emergence of clinical trials for novel therapies in this condition, the International Headache Society Guidelines for Controlled Clinical Trials in Idiopathic Intracranial Hypertension aims to establish guidelines for designing state-of-the-art controlled clinical trials for idiopathic intracranial hypertension.
Keywords
Introduction
Idiopathic intracranial hypertension (IIH) is characterized by raised intracranial pressure (ICP) and papilledema with the potential risk of permanent visual loss (1). The incidence rates of IIH have been correlated with country-specific World Health Organization obesity rates with a pooled incidence of 1.2 per 100,000 people (2). There is a peak incidence in women around the age of 25 years, reported in one study to be 15.2 per 100,000 (3). With increasing rates of obesity globally, there has been marked increase in the incidence of IIH observed around the world (2–4).
Headache is a predominant symptom (5,6). IIH has a detrimental effect on all aspects of the patient’s quality of life (QOL) which has been found to be predominately driven by headache (7–9). The current International Classification of Headache Disorders 3rd edition (ICHD-3) classifies headache attributed to IIH as a secondary headache disorder (10).
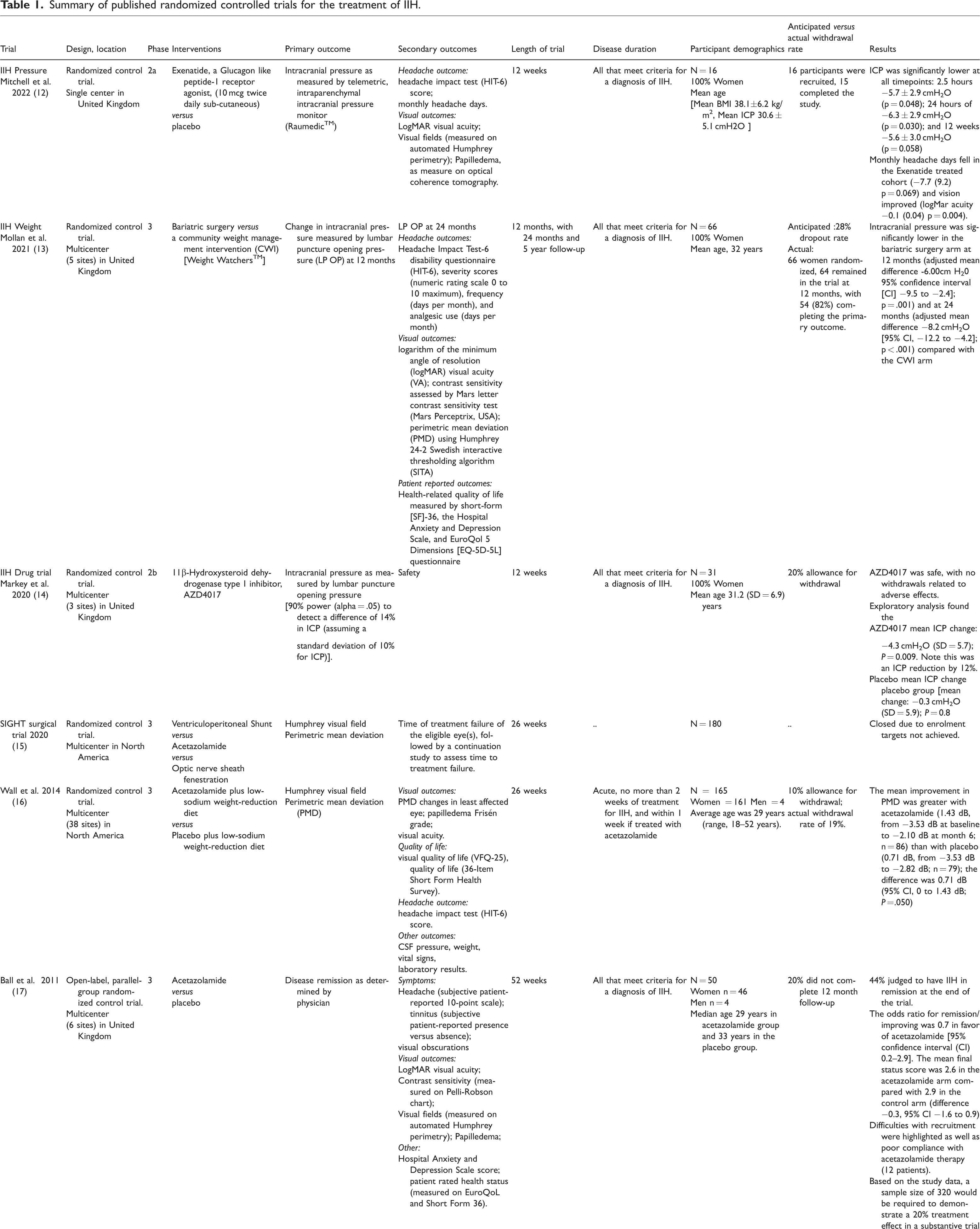
The 2015 Cochrane review concluded that there is a lack of evidence to guide management of IIH (11), as there are few published randomized clinical trials (RCTs) and only a few open label trials (Table 1 and 2) (12–20). To date, research has been sporadic due to the relative rarity of the disease but with the increasing incidence and prevalence of the disease there is an unmet medical need for effective acceptable interventions. The James Lind Alliance Priority setting Partnership, a National Institute of Health and Care Research supported initiative, was conducted in IIH and funded by IIHUK, a United Kingdom Charity for people living with IIH. This defined uncertainties in diagnosis and management that should be prioritized for research, as determined by patients, care-givers and health care physicians (21).
Summary of published randomized controlled trials for the treatment of IIH.
Summary of open label therapeutic and lifestyle interventions in idiopathic intracranial hypertension.

IIH is a disease that requires a multi-disciplinary approach and that poses challenges for trial design and choice of trial outcomes. IIH teams require a broad group of specialists, including ophthalmologists, neurologists, headache specialists, neurosurgeons, endocrinologists and dieticians (1,22). This also indicates the complexity of the disease management (23,24). The International Headache Society (IHS) has a track record of using an evidence-based approach to define the standards for the conduct and reporting of RCTs in headache disorders (25–31). Guidelines recommend standardized approaches to methodology and to define meaningful outcomes. Trial outcomes need to be sufficiently flexible to address the intervention type – valid by measuring what matters – reliable and clinically meaningful. Defining key trial outcomes is critically important for the ability to meta-analyze data from different trials. Regulatory bodies, such as the United States Food and Drug Administration and The Committee for Medicinal Products for Human Use at the European Medicines Agency, rely on the results of clinical trials to reach opinions as to whether or not to authorize various medicines and devices. These guidelines will therefore cover designs for both pharmacological treatments as well as for non-pharmacological treatments such as weight loss methods and surgery. Patient-reported quality of life and cost-effectiveness are important to support decisions regarding reimbursement. This guideline is designed to advance the quality of future IIH trials and will subsequently help improve finding meaningful treatments for people with IIH. Given that IIH is rarer in prepubertal children, and potentially has a different underlying pathogenesis (32), the present guidelines will primarily focus on those post-puberty and adults. The present guidelines will only address IIH and not secondary pseudotumor cerebri syndrome nor Idiopathic Intracranial Hypertension without Papilledema (IIHWOP) as these are rare conditions with likely alternative underlying pathophysiology (33–35).
Previous trials in IIH
There have been few RCTs in IIH and a small number of cohort studies (Table 1 and 2) (12–20). Choice of the primary outcome has varied between visual field status, as assessed using automated perimetry performed on the Humphrey field analyzer, and ICP as measured by lumbar puncture (Tables 1, 2 and 3). Note that, to date, there has been no RCT evaluating treatment for headache attributed to IIH, although an open-label study recently reported the use of a primary end point of change from baseline in monthly moderate and severe headache days at 12 months (18). Trials evaluating medicines often have different outcomes compared with surgical trials for interventions. A recent study on the design of trials and the common data elements reported in IIH, as identified by clinicaltrials.gov, highlighted the extreme heterogeneity in outcome reporting (36). Trial considerations for IIH require a systematic approach as this is a multispecialty disease where there still is uncertainty regarding treatment approaches depending on the leading specialty and/or disease severity. In recent years, a number of observational cohort studies have been published that are now helping to unravel the full disease spectrum, allowing more clarity on the sub-groups of IIH that may benefit from a particular intervention (37–40).
Potential primary outcome measures for trials evaluating people with IIH.

1.1. Selection of subjects
1.1.1. Idiopathic intracranial hypertension definition
Recommendations:
Eligible subjects should fulfil the internationally accepted diagnostic criteria for IIH (41). For trials in which the primary outcome is headache, eligible patients should fulfil the diagnostic criteria for headache attributed to IIH according to the most recent version of the International Classification of Headache Disorders (ICHD) of the International Headache Society (IHS).
Comments:
As papilledema is a key criterion for a definite diagnosis of IIH best practice would ensure that pseudopapilledema is excluded (1). As lumbar puncture opening pressure is a key criterion for a definite diagnosis of IIH, best practice would ensure that the left lateral decubitus position of the patient in the lumbar puncture procedure when the opening was recorded should be documented. There are a number of confounding factors in measuring lumbar puncture opening pressure accurately (41–43). For example, Valsalva maneuver (43), a curled body position and the use of sedation can increase ICP measurements. Whereas hyperventilation or repeated attempts may decrease the opening pressure artificially. Any controllable factors should be minimized where possible (1,22,41). Clinical trials for IIH should include all adults who meet these criteria, regardless of ethnicity or sex, to avoid population bias. Diagnostic criteria evolve with the advent of newer technologies and by prospective validation. Magnetic resonance imaging (MRI) by bespoke clinical protocols for raised ICP (44) or routine clinical protocols (45,46) have been evaluated and MRI findings have found to be predictive of raised ICP. Mallery et al. (45) found in a retrospective cohort that three of four MRI features of raised ICP distinguished between patients with IIH and controls with a moderate degree of sensitivity (64%) and high specificity (97%). Reduced pituitary gland height (PGH) was the most sensitive individual feature (80%) but had low specificity (64%); increased mean optic nerve sheath diameter (ONSD) was less sensitive (51%) and only moderately specific (83%) for identifying IIH without papilledema (IIH WOP). Flattening of the posterior globe (FPG) had low sensitivity (57%) but was the most specific individual feature (90%–99% specific). Venous imaging in this study was only performed in people with IIH and transverse sinus stenosis (TSS) was of moderate sensitivity (78%). Hoffmann et al. (44) found PGH to have a sensitivity of 88% and 76% specificity; ONSD with a sensitivity of 80% (left eye) and 72% (right eye), and 96% specific in either eye. FPG had a low sensitivity at 28% but was highly specific at 100%. TSS had a sensitivity of 36% and a specificity of 96%. Korsbaek et al. (46) in a prospective cohort found that three of four MRI features were found to distinguish between patients with IIH and non-IIH with a moderate degree of sensitivity (59.5%) and highly specific (93.5%).
1.1.2. Duration of disease
Recommendation:
The definition for the duration of disease should be clearly stated in the trial inclusion criteria. The history may be based on subject recall, medical records, or both.
At present, there is no consensus for defining duration of the disease. For example, onset of IIH could be defined as soon as the diagnosis is suspected from the history or as soon as the patient is found to have papilledema (Table 4).
Definitions of disease state.
1.1.3. Disease state
Recommendation:
The definition for the disease state should be clearly stated in the trial inclusion criteria. This should be based on history and examination from the medical records.
Comment:
At present, there is no consensus typology on the definitions for disease states (Table 4).
1.1.4. Age at entry to adult studies
Recommendation:
Adult subjects participating in clinical trials should be between >18 years and <60 years of age at entry.
Comments:
The age at which minors are able to provide informed consent for enrolment in research studies differs around the world. For example, The Medicines for Human Use Regulations prohibit children under the age of 16 years from giving consent to take part in a Clinical Trial of an Investigational Medicinal Product (CTIMP). However, both in Europe and in the United States regulations and law have set the age of 18 years for informed consent for research studies. Few adults will be excluded by the upper limit of this criterion, as IIH beginning after the age of 60 years is extremely rare and secondary pseudotumor cerebri syndrome is more likely to be diagnosed after this age (2–4).
1.1.5. Entry to trials involving adolescents
Recommendations:
Comments:
There are a number of different assessments that can be used to determine pubertal status (49). The second highest incidence of IIH is found in females over the age of 13 years. This was demonstrated in one study where incidence in women was highest in the 20- to 29-year age group (16.5 per 100 000 person-years) followed by the 13- to 19-year age group (8.7 per 100 000 person-years) and the 30- to 39-year age group (8.4 per 100 000 person-years) (3). IIH following onset of puberty has the same phenotype as the adult condition (32,50) Available treatment options are limited as there has been no prior RCT that has enrolled adolescents. It is therefore important to offer these patients access to novel treatment strategies but also to allow for adapted clinical trial rules, as for orphan diseases. Of note, in the European Union extrapolation of data from adult trials can be used as evidence for licensing of medicines in adolescents (51).
1.1.6. Sex
Recommendations:
Both females and males with IIH should be eligible to participate in IIH clinical trials. The trial design should account for the differences in prevalence of IIH between females and males. This could include stratification by sex or using sex as an interaction term within in the statistical analysis models. Whenever possible, a pre-specified sub-analysis could be considered to evaluate a possible sex difference in response.
Comments:
Sex should be recorded for clinical trial purposes, as the sex recorded at birth. This is important for sub-analysis where it is known that males have worse visual outcomes as compared to females (52). IIH is much more prevalent in women than in men, on the order of approximately 10 females to 1 male (2,3). Enrolling males requires caution for the exclusion of secondary causes (34). Special caution should be taken to avoid enrolling women who may be pregnant or breast feeding, unless they are the target of the trial. All fertile-age participants should practice effective contraception. Partners of fertile-age female participants should practice effective contraception. Those who may be transitioning between genders, and are undergoing hormonal therapies, such a testosterone replacement, who are subsequently diagnosed with raised ICP may be regarded as having secondary pseudotumor (53), and therefore not admissible to IIH trials.
1.1.7. Concomitant drug use
Recommendations:
The trial protocol should prespecify concomitant medications that are permitted at enrolment or during the trial and those that are not. Medicines that may influence the trial outcome should be stopped prior to the trial baseline visit with a wash out duration depending on the type of molecule.
Comments:
The mechanisms of absorption, distribution, elimination and metabolism of small molecule drugs differ significantly from biological agents. If the trial drug has a specific action that would confound the validity of the outcomes, such as ICP lowering, then any medicines that alter CSF dynamics (such as ICP lowering drugs, or other examples such as retinoids and indomethacin) must be carefully considered as to their acceptability for concomitant use. Concomitant medication use should be considered in the statistical analysis plan a priori and where applicable the analysis of participant data, when there is use of concomitant medication that lowers ICP, should be clearly described in the estimand intercurrent framework in the section describing intercurrent events. Any medicines that would not be acceptable for concomitant use should have a recommended wash out period of at least five and a half drug half-lives. An alternate option would be if concomitant medications are permitted, depending on their action, they could be stratified for at randomization. Specific medicines that could be stratified for include diuretics, glucocorticoids or other ICP lowering agents including topiramate depending on disease stages as indicated above (see section 1.2.4 Stratification). Depending on the investigational medicine product some concomitant medications such as simple analgesics should be recorded in order to avoid MOH but may need no stratification.
1.1.8. Comorbidities
Recommendations:
Patients with severely disabling concomitant disorders that may influence the conduct of a trial or the interpretation of its results, or that would be negatively impacted by the new treatment should be excluded. Patients who have been currently diagnosed with major depressive and/or generalized anxiety disorders, as defined by Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria should not be included (54). Patients suffering from a substance use disorder, as defined by DSM criteria (54), should be excluded.
Comment:
a. See section 1.3.2 on comorbid conditions in IIH.
1.2. Trial Design
Well-designed RCTs are widely regarded as the least-biased research design for evaluating new health technologies and therapies. Diseases may have different characteristics based on the participant’s background or geographical location, hence why multicenter trials may deliver results that are more applicable globally.
1.2.1. Masking
Recommendations:
Phase II and III efficacy trials of therapies for IIH should use a double-blind design and with parallel group comparison. Phase II and III surgical trials for IIH should mask the investigators where appropriate. Successful masking should be assessed at the end of the study for both patients and investigators involved in a trial (see masking assessment).
Comments:
Drugs intended for IIH can only be reliably evaluated in randomized, double-blind, placebo-controlled trials. Masking may not be required in long-term safety trials. Sham surgeries or comparative surgical trials should mask the investigators where possible. Data and laboratory analysis should be performed by masked investigators. Trial treatment and placebo should be administered in an appropriate format that allows maintenance of blinding. Typically cross-over studies risk unmasking and should generally be avoided.
1.2.2. Placebo
Recommendations:
Therapies under evaluation for IIH ideally should be compared with placebo in cases of IIH without fulminant disease (Table 4) in which the potential for visual loss with placebo is considered to be negligible. In cases of fulminant IIH and/or for surgical procedures the use of placebo is unethical and cannot be recommended. When two presumably active treatments are compared, a placebo control ideally should be included for assay sensitivity.
Comments:
The placebo effect is a genuine psychobiological phenomenon that affects the results of clinical trials across many conditions (55). The placebo effect has been studied in primary headache disorders and been noted to affect over 25% of individuals with respect to acute headache relief, with 6% becoming pain free. In addition, up to 25% of patients in such studies may have experienced an adverse event from placebo (56,57). Use of a placebo arm in the setting of carefully monitored clinical trials of consenting subjects outweighs the risk of approval of ineffective interventions. Given the significant risk of permanent visual loss in IIH, the requirement for the use of placebo should be restricted to trials of the milder spectrum of IIH only, with a protocol that includes careful and frequent ophthalmic monitoring.
1.2.3. Randomization
Recommendation:
Subjects enrolled in parallel-group trials should be randomized at the entry to the trial, except when considering adaptive randomization.
Comment:
True randomization is essential to avoid bias and, in large trials, to contribute to group matching. Block randomization using varying block sizes (e.g., block sizes of two, four, six, etc.) may be helpful in preventing investigators or participants from guessing treatment assignments.
1.2.4. Stratification
Recommendation:
Stratification should be considered when an imbalance between the treatment groups or an important factor may influence the results of a trial.
Comments:
Randomization alone may not ensure full comparability among subjects in different treatment groups, especially in smaller trials, and stratified randomization is sometimes used to circumvent potential imbalances. The Committee for Medicinal Products for Human Use recommends that stratification variables usually be included as covariates in primary analyses, regardless of their prognostic value (58). Some considerations for stratification of IIH trials will depend on the intervention and trial design but may include the following variables: Duration of disease Duration of treated disease Use of concomitant headache preventative medication Use of ICP lowering medications Use of concomitant diuretic medications Body weight or Body Mass Index d. Caution may be required, as if too many variables are stratified for there may be too many sub-groups which may increase the power calculation estimate and/or have a negative effect on the results of the study.
1.2.5. Intention to treat analysis
Recommendation:
RCTs of therapies for IIH should follow the principle of intention to treat, which implies that analyses should include all randomized subjects in the groups to which they were randomly assigned, regardless of treatment received.
Comments:
The intention to treat principle should be adhered to when the primary outcome is a variable that measures a change from baseline to any post-dose time point or to the end of the trial. When the primary outcome is defined as a rate of change, and the analysis will therefore imply a slope or rate of calculation, only subjects who have received at least one dose of the treatment and for whom at least one data point has been recorded should be included. Any per protocol analysis should be defined in the statistical analysis plan prior to initiating the trial.
1.3. Outcome measures
1.3.1. Phase 1 (pharmacokinetic trials)
Recommendations:
Endpoints within phase 1 trials will likely report the following outcomes in healthy volunteers and in specific populations: absorption distribution metabolism excretion Cmax (the maximum (or peak) serum concentration that a drug achieves in a specified compartment or test area of the body after the drug has been administered) area under the curve half-life in healthy subjects. b. Drug safety profile includes dose escalation in trials of healthy volunteers, safety signals supporting non-clinical findings and new safety signals in humans. c. Early phase trials prioritize safety.
Comment:
In IIH medication trials, consideration should be given to using healthy volunteer populations with a high body mass index (BMI), as the pharmacokinetics could be different from a person with a normal BMI.
1.3.2. Phase 2/phase 3 and above in IIH
Recommendations:
Early phase, such as phase 2, trials prioritize safety and initial indicators of efficacy. The primary outcome measure chosen depends on the target of the intervention (Table 3).
1.3.3. Clinical trials to evaluate interventions for headache
Recommendations:
The primary outcome for interventions that modify headache should be a headache outcome. Change in headache days; change in moderate to severe headache days; or responder rate should be a primary outcome. From these three endpoints, the two not selected as the primary endpoint should be considered as secondary endpoints. Guidance from the IHS for headache outcome measures have been documented extensively in other guideline documents and should be applied to IIH trials (25–31). Evaluations of efficacy should be based on information obtained from headache diaries. For multinational trials, diary design should be standardized, with translations adapted to the linguistic and sociodemographic characteristics of target population.
Comments:
Headache attributed to IIH should be defined according to the diagnostic criteria in the most recent version of the ICHD of the IHS. As people with IIH can demonstrate different headache characteristics each headache type that exists in a given participant should be phenotyped and documented. Tension-type headache and migraine are common and they can co-occur in people with IIH. Up to one third of people with IIH have a prior history of migraine (5,37) and nearly half of people with IIH have a family history of migraine (18). Consideration should be made as to whether participants with a past history of chronic migraine or chronic tension-type headache can be included. Participants with a concurrent history of chronic migraine or chronic tension-type headache at the time of IIH diagnosis and in whom preventative medicines were started prior to their diagnosis of IIH should be excluded. Those with post-IIH persistent headache may have both migraine-like and tension-type- like characteristics to their headache phenotype (5,59,60). Medication-overuse headache (MOH) due to frequent use of analgesics should be defined by the criteria of the most recent version of the ICHD of the IHS. Regular use of analgesic medications is recorded in up to half of IIH patients depending on the study and there is a high burden of opiate use (5,61). Both of which are likely due to the lack of targeted licensed treatments for IIH (60). Exclusion of people with MOH may restrict the eligible population for recruitment therefore MOH could be permitted if stratified for or could be dealt with by using the estimand intercurrent framework (see 1.5.3. Standard statistical methods). Analgesic usage should be recorded daily. The responder rate is calculated as a percent reduction from baseline in the number of headache days or number of moderate or severe headache days in each treatment period (31). Specific responder rate targets must be prospectively defined. When considering headache diaries in IIH, trials investing acute disease or surgical intervention should consider a reduced headache diary or a calendar, such as a 7-day calendar, as it is unethical to withhold treatment in these two acute scenarios.
1.3.4. Clinical trials to evaluate interventions for vision
Recommendation:
The primary outcome for interventions that modify visual function should be a visual outcome.
Comments:
Visual outcomes that can be considered are detailed in Table 3. Visual outcomes can be split into functional and structural outcomes. The most commonly used visual outcome is the Humphrey visual field mean deviation. It has several challenges as it dependent on both the technician and patient performance and is prone to variability and inaccuracy with up to one in five people having a performance failure (62,63). Many trials allow multiple attempts to allow for familiarization and learning (63,64). There is currently no consensus to the accepted level of improvement in mean deviation that is considered to be a clinically meaningful change in IIH. While the IIH treatment trial (IIHTT) observed a change in the mean deviation of 0.71 dB that was associated with significant improvement in visual quality of life and the short form 36 physical and mental component summary scores, papilledema and lumbar puncture opening pressure, it has yet to be replicated in other studies. Optical Coherence Tomography (OCT) imaging now provides multiple structural outcomes and is a valuable clinical tool used globally in ophthalmology clinics for the diagnosis and longitudinal monitoring of papilledema. It is a rapid, reproducible, non-invasive, and a highly adaptable technology (1,22). Measures have been shown to correlate well with ICP in the setting of RCT participants (65). There is currently no recommended accepted level of improvement in OCT measures that is considered to be a clinically meaningful change in IIH.
1.3.5. Clinical trials to evaluate interventions for ICP
Recommendation:
The primary outcome for interventions that modify ICP should be ICP.
Comments:
When the measurement is taken it should be time-stamped, as timing can influence symptoms and signs. The most common method of ICP measurement in IIH remains the opening pressure at time of lumbar puncture, with several well documented side effects (66,67). As there is high variability in the measure of opening pressure at lumbar puncture, the procedure should be standardized (42). The timing of lumbar puncture can impact the baseline headache diary assessment (67). Telemetric ICP monitors are available commercially and may provide accurate ICP measurements that can be measured at predefined time points. Invasive Continuous ICP monitors that have been previously used in an IIH trial are not currently available commercially (43). Intracranial ICP monitors by cables (often termed ICP bolt) can be used in-hospital or out of hospital for a few days, mostly for diagnostic purposes (68). They may not be suitable for clinical trials depending on the duration of the study.
1.3.6. Clinical trials to evaluate interventions for symptoms of IIH
Recommendations:
The primary outcome for interventions that modify symptoms of IIH could be the most bothersome symptom. The most bothersome symptom endpoint should be selected just prior to randomization and measured on a binary scale (present or absent).
Comments:
Defining a most bothersome symptom can be useful in trials in diseases with many different system symptoms like IIH. The use of the most bothersome symptom as a trial endpoint is an alternative to requesting demonstration of a positive treatment effect on all the IIH-associated symptoms. In general, most bothersome symptom requires larger sample sizes due to the need to consider the frequency of the symptoms. It is normally employed in trials involving acute headache therapy. Use of a time-locked recording device (e.g., an electronic diary) to record the most bothersome symptom throughout the duration of the trial is recommended.
1.3.7. Clinical trials to evaluate surgical interventions for IIH
Recommendations:
Comments:
Assessments of surgical procedures in IIH remain limited by the lack of consensus on how to best to define outcome measures. Often, complications or failure are used to define the best surgical intervention, particularly when evaluating two or more techniques. Avoiding surgical complications and revisions are not therapeutic goals in themselves. Intraoperative and post-operative adverse events should be separately reported (70). Secondary surgical end points that could be considered, in addition to the main outcome measures of headache, vision, and ICP measures in IIH trials.

Due to a lack of licensed treatments and the paucity of RCTs in IIH, there is no current consensus on the definitions of “medically refractory”, “drug-resistant” or “medically intractable”.
1.4. Secondary endpoints
1.4.1. Patient reported outcomes
Quality of Life is known to be affected by IIH (7–9), in particular, visual quality of life is known to be affected by IIH (8). There are two broad approaches to interpretation of quality-of-life changes in clinical trials: anchor-based and distribution-based. Anchor-based are those that rely on the distribution of changes and the effect size. Distribution-based are those that use an external anchor, such as patient judgments of change. Health-status questionnaires are routinely used in RCTs and are critical to fully understand overall treatment effectiveness and to establish the benefit of a given intervention over the standard of care. However, changes in scores on these tools may be difficult to interpret. The statistical significance of a change in any given score may be due to the sample size and may not indicate that the observed change is important.
Recommendations:
Visual quality of life tools also should also be used, where appropriate (Table 6).
Best practice would be to define the minimum change in scores on health status questionnaires that are considered important by patients or their clinicians a priori.
Comments:
There is no IIH-specific quality-of-life measure yet developed. There are two approaches to defining the minimum change: using an external reference e.g., a two-day change; or using distribution method such as standard deviation change. It should be noted that the Unites States FDA guidance recommends anchor-based methods and in some circumstances distributional methods (101). The Patient Global Impression of Change scale (PGIC) can be used to evaluate subject satisfaction as a secondary endpoint (93). A high proportion of people with IIH report migraine-like headaches (5,37,59). Therefore, the migraine validated quality-of-life tools may also be useful to deploy. Using tools that look over a short disease duration may be more appropriate for trials that involve acute cases. For example, the Migraine Functional Impact Questionnaire (MFIQ) evaluates the past seven days (82,83) compared with the Migraine Disability Assessment (MIDAS) questionnaire (86) evaluating over a four-week period. Tools that have been deployed in other migraine trials may be suitable for selection to allow for a comparison of improvement or lack thereof. These may include Migraine Disability Assessment questionnaire (86), The Headache Impact Test – (HIT 6) (89,90) or the headache under-response to treatment (HURT) questionnaire (91) (Table 6).
1.4.2. Considerations of confounding conditions that may affect the chosen end points in IIH clinical trials
Recommendations:
Depending on the trial, people with confounding conditions may need to be excluded from recruitment or the condition stratified for at randomization. Depression and anxiety levels should be recorded at the time of randomization and at the end of the double-blind treatment period. Validated scales for depression and anxiety should be used.
Comments on depression and anxiety
Depression and anxiety have been noted to be frequent in people with IIH (102–104). A recent study has suggested depression and anxiety burden in IIH is higher than in the general population, but not more common in IIH as compared to migraine. This may indicate that presence of headache as a symptom or migraine may be a potential driver for comorbid depression and anxiety in IIH (61). People with depression and anxiety diagnoses can be included if their symptoms are mild or moderate, properly treated and stable and if their mental status is monitored during the trial. Where there is doubt a professional opinion from a psychiatrist may be required. Validated scales for depression include: Patient Health Questionnaire-9 (PHQ-9) (80), Beck Depression Inventory (BDI) (81), Hospital Anxiety and Depression Scale (HADS) (76). Validated scales for anxiety include the Hospital Anxiety and Depression Scale (HADS) (72), State-train Anxiety Inventory (STA-I) (78), and the Generalized Anxiety Disorder (GAD-7) can be used (79).
Comment on psychiatric disorders:
In addition to depression and anxiety, people with IIH may have co-existing mental health diagnoses such as personality disorder, substance abuse, schizophrenia and bipolar disorder (104,105). People with IIH may be at risk of suicide and this has been documented in over 40% deaths in a United States IIH Registry (106). See section 1.1.8 Comorbidities.
Comments on obstructive sleep apnea (OSA):
A co-morbid relationship between IIH and OSA is well described. Nearly 50% of people with IIH and BMI ≥ 35 kg/m2 fulfil the diagnostic criteria for OSA. In the clinical setting, the most sensitive screening tool to identify OSA risk in IIH was the STOP-BANG questionnaire. Treating OSA in patients with IIH may improve papilledema (107,108). Polysomnography is required to confirm OSA. Presence of OSA may influence the visual and ICP endpoints. Patients with this diagnosis can be included, if their symptoms are actively managed and may need to be accounted for at statistical analysis. Where a person has untreated or unstable OSA it should be considered an exclusion to entry to the study. Where there is doubt a professional opinion from a respiratory sleep expert may be required.
Comments on cognitive performance:
Disturbances of cognitive performance have been formally noted as part of the IIH clinical phenotype (109). Studies have shown deficits in key areas such as memory, learning, visuospatial skills, concentration, language and executive function (109–112). In particular, deficits in reaction time and processing speed have also been demonstrated (113). Cognitive dysfunction has recently been shown to be reversible, both acutely after lumbar puncture and over time. Importantly, these authors also documented that cognition adversely impacted the visual field performance, which is often a key outcome measure in IIH trials (113). There are a number of potential factors that have been shown to influence cognitive function and that are likely to be relevant in IIH. These include obesity and the resulting pro-inflammatory state, headache, depression, sleep apnea, and hormonal dysregulation (113). If cognition is required to be evaluated then validated tests should be used.
Comment on polycystic ovarian syndrome (PCOS):
A higher portion of people with IIH have co-existing PCOS (114,115). This has been confirmed in a population study and people with IIH were compared with those with migraine or a general population without migraine (114). Patients with this diagnosis can be included if their symptoms are actively managed, stable (for >3 months) and may need to be accounted for at the statistical analysis.
Comments on optic atrophy:
In a trial with visual outcomes as key end points, optic atrophy should be excluded. Optic atrophy is defined as a pale optic nerve on clinical examination (fundoscopy). The global retinal nerve fiber layer measurement as determined by OCT imaging may be used to define optic nerve damage that could be used as an exclusion criterion. There is no consensus on what level of the global retinal nerve fiber layer measurement is defined as optic atrophy.
1.4.5. Adverse events
Recommendations:
Timely, accurate, and complete reporting and analysis of safety information from clinical studies are crucial for the protection of participants, investigators, and the sponsor, and are mandated by regulatory agencies worldwide. Adverse events (AEs) should be reported separately for the intervention and placebo arms. If a withdrawal from the trial is secondary to an AE (whether it is unrelated or related; expected or unexpected), it should be reported.
1.4.5.1. All adverse events
Recommendations:
AEs can be encountered in participants receiving investigational products. The AEs should be recorded. All AEs and special reporting situations, whether serious or non-serious, should be reported. Serious adverse events (SAEs), including those spontaneously reported to the investigator within 30 days after the last dose of study intervention, also should be reported. Abnormal pregnancy outcomes (e.g., spontaneous abortion, fetal death, stillbirth, congenital anomalies, ectopic pregnancy) are considered SAEs and should be reported. Disease-related events and disease-related outcomes that relate to the cause of death of a participant in a study within 12 weeks of the last study intervention, whether or not the event is expected or associated with the study intervention, should be considered an SAE. In addition, safety information that is spontaneously reported by an investigator beyond the time frame specified in the trial protocol should be evaluated.
1.4.5.2. Assessment of expectedness
The following categories, as outlined in Table 7, should be used to define the expectedness of the SAE.
Definition of the expectedness of a serious adverse event and the relatedness (causality) of the SAE.

1.4.5.3. Assessment of relatedness
The following categories, as outlined in Table 7, should be used to define the relatedness (causality) of the SAE.
1.5. Statistics
Recommendations:
A general description of the statistical methods to be used to analyze the efficacy and safety of clinical trial interventions should be stated a priori, and specific details should be provided in a Statistical Analysis Plan (SAP). The statistical hypothesis and definition of the primary end point should be stated, and a sample size determination should be published.
Comments:
Repeated measures of outcomes could be considered to increase power, particularly in a rare disease such as IIH. This requires a suitable analysis method to handle the serial correlation in outcomes, like hierarchical regression. To avoid mean reversion, it is recommended not to use screening measurements as baseline values when using repeated measures. Instead, it is recommended to use a separate assessment for baseline values.
1.5.1. Hierarchy of endpoints
Recommendations:
All endpoints need to be defined in the protocol. Hierarchy of endpoints may be adopted for trials investigating the efficacy of interventions for IIH.
Comment:
Creating a hierarchy of endpoints reduces the risk of type 1 error.
1.5.2. Sample size calculation
Recommendations:
A calculation of the optimum number of participants required to be ethical and provide scientifically valid results should be stated in advance. A feasibility pilot study may be required to determine the sample size.
Comments:
It is neither practical nor feasible to study the whole population in any study. Hence, a set of participants is selected from the population, which is less in number (size) but adequately represents the population from which it is drawn so that true inferences about the population can be made from the results. To avoid bias in interpreting results, the sample size should be determined before the start of a clinical study. If the study is underpowered (too few subjects in a study) the study then may not be able to detect the difference among trial arms. If the study is overpowered (too many subjects than required), more participants are subjected to the risk of the intervention, which is unethical and wastes resources. If the study population is biased towards a particular spectrum of the disease (e.g., mild versus severe visual loss) the results cannot be generalized to the population as the sample will not represent the whole disease spectrum.
1.5.3. Standard statistical methods
Recommendations:
Randomized controlled trials in IIH should follow the principle of intention-to-treat whenever possible to aim for the highest class of evidence. The statistical analysis plan should be included. An alternative analysis plan should be included if the distribution of data does not meet assumptions of initial planned analyses. Data primarily should be summarized using descriptive statistics. Continuous variables should be summarized using the number of observations, mean, standard deviation, median, interquartile range, minimum and maximum, as appropriate. Ordinal variables such as categorical values should be summarized using the number of observations and percentages as appropriate. The estimand framework has been recommended for use in clinical trials (116).
Comments:
Modern statistical approaches including hierarchical regression for repeated measures analysis may be helpful to improve the power of the trial in a rare disease, such as IIH, where there is a limited pool of patients feasible to recruit. The estimand summarizes what the outcomes would be in the same patients under different treatment conditions. The framework consists of the following five attributes: treatment, population, variable, population-level summary, and handling of intercurrent events (ICEs) (116–118). The attributes must be defined in advance, allowing design of a trial to estimate treatment effect.
1.5.4. Missing data reporting
Recommendations:
All IIH trials should report how they have minimized missing data and what strategy they have used to deal with missing data. Methods for handling missing data must be described. If multiple imputation methods are used they must be described. Last value-carried-forward is no longer the recommended method.
Comments:
Missing data are always going to occur despite best efforts for minimization. In an intention-to-treat analysis, all randomized participants have outcomes assessed and are analyzed in the group in which they were randomized (regardless of the actual intervention received). However, if participants drop out or miss visits, the intent-to-treat conclusions can be compromised (119). The prevention and treatment of missing data in clinical trials discusses strategies to limit missing data in trial design and the multiple methods employed to deal with missing data (120).
1.6. Trial registration
Recommendation:
Prior to initiation, all clinical trials should be pre-registered in a register acknowledged by regulatory authorities.
Comment:
Clinical trial registers such as clinicaltrials.gov, European Union Drug Regulating Authorities Clinical Trials (EudraCT) database (eudract.ema.europa.eu), clinicaltrialsregister.eu, or a similar regional or national official database should be used.
1.7. Recruitment
Recommendations:
Investigators should recruit widely from the population expected to use the treatment being evaluated. Subject participation in previous trials for IIH should be limited to two prior trials and recorded and presented in the publication. Recruitment strategies should be disclosed in the publication. A qualitative recruitment evaluation could be conducted as a Study Within A Trial (SWAT) within the first part of recruitment to the clinical Trial (host trial) to explore in depth the feasibility, acceptability, and appropriateness of the trial processes for participants and healthcare professionals.
Comments:
For example, as long as they meet eligibility criteria, all individuals being treated for IIH at specialty clinics and primary care facilities should be considered for enrolment in clinical trials. The inclusion of people who habitually participate in IIH clinical trials should be discouraged, to avoid bias. It is recommended that investigators establish a database of the number of IIH trials of any kind in which a particular subject has participated in the two years preceding a clinical trial. A SWAT evaluation is helpful in complex trials and will help develop optimum recruitment strategies. This pragmatic qualitative recruitment evaluation is aligned with the Medical Research Council (MRC) framework for evaluation of complex interventions (121).
1.8. Publication
Recommendations:
Publication of trial results is necessary and should include all primary and secondary efficacy endpoints and all safety data, whether positive or negative. Standardized reporting guidelines should be used for clinical trials.
Comments:
Before any trial-related activities are initiated, a Trial Steering Committee (TSC) should agree on timelines for publication and, if possible, include details in the protocol. At the initiation of the trial or prior to the end of recruitment, a design paper may be published. At the close of recruitment, a baseline-data publication may be considered and published. Authorship of trial-related publications should be based on the criteria of the International Committee of Medical Journal Editors (122). The Consolidated Standards of Reporting Trials (CONSORT) statement provides a minimum set of 25 items to be reported for all randomized trials. The original Consolidated Standards of Reporting Trials (CONSORT) statement published in 1996, was in response to inadequate reporting of randomized controlled trials being associated with bias in the estimation of treatment effect (123,124).
1.9. Conflicts of interests
Recommendation:
To maintain the credibility of a trial, authors must declare their conflicts of interest.
Comments:
A conflict of interest exists whenever professional judgment concerning a primary interest (e.g. subject wellbeing or the validity of research) may be influenced by a secondary interest (e.g. financial relationship to a trial sponsor). Financial relationships that represent potential conflicts of interest include employment, consultancies, research grants, fees and honoraria, patents, royalties, stock or share ownership, and paid expert testimony. Investigators should avoid agreements with sponsors, both for-profit and non-profit, that restrict access to trial data, limit its analysis and interpretation, or interfere with the independent preparation and publication of manuscripts.
b. Conflicts of interest also need to be disclosed for the investigator's immediate family (partner or spouse and offspring).
2.0. Independent data safety monitoring
Recommendation:
An independent Data Safety Monitoring Committee (DSMC), also named Data Safety Monitoring Board (DSMB), is recommended for all clinical trials.
Comment:
The DSMC should monitor safety data as an ongoing process in a clinical trial. The DSMC should have predefined stopping rules for feasibility, futility, and safety. Independent interim analysis by the DSMC should be considered for assessment of the pre-defined stopping rules.
2.0.1. Trial Steering Committee for industry-sponsored trials
Recommendation:
For industry-sponsored trials, the formation of a TSC comprised of academics, statisticians, and (if appropriate) company representatives is recommended.
2.0.2. Trial Steering Committee investigator-initiated trials
Recommendation:
For investigator-initiated trials (i.e., developed and sponsored by independent investigators or academia), a TSC is not necessary, but is best practice.
Comment:
Whether or not a TSC is formed, investigators and sponsors are responsible for all aspects of a clinical trial, including conception, design, operational execution, data handling, data analysis and interpretation, subsequent reporting and publication, and compliance with all local laws and regulations.
3. Post-approval registries
Recommendation:
The IHS recommends post-approval product registries (i.e., prospective open-label observational studies) to evaluate the use of newly approved treatments in clinical practice (125).
Comment:
Registries generate real-world data on long-term efficacy, tolerability, and safety. They also measure compliance and adherence. Registries for treatments also include individuals with relevant coexistent and comorbid diseases who were excluded from clinical trials on that basis.
4. Public and patient involvement
Recommendation:
Public and patient involvement and engagement (PPIE) should be part of all IIH clinical trials.
Comments:
PPIE plays an important role in the development and delivery of successful trials. It allows both patients and clinical researchers to understand the needs of one another and helps to ensure that the clinical trial is fit for purpose. Increasing the accessibility of PPIE in health research for people from diverse backgrounds is important for ensuring all voices are heard and represented. The public and patient role should include, but not be limited to, helping define the most relevant research question and trial outcomes; designing a trial appropriate to the needs and lifestyle of the patients; developing accessible and useful participant materials; conducting the trial in a participant friendly way; and dissemination of the trial results to maximize and awareness and ensure the adoption of trial results in clinical practice. Various countries have guidance on public and patient involvement in research. For example the Responsible Research and Innovation (RRI) is a European tool kit (European Union's Seventh Framework Programme for research, technological development and demonstration under grant agreement no. 61239) (126). There is a disease specific priority setting partnership which was funded by the United Kingdom (UK) charity, IIHUK. The James Lind Alliance, a UK National Institute for Health Research-supported initiative, IIH priority setting partnership identified existing gaps in knowledge that matter most to key stakeholders (patients, carers and clinicians) and ranked them to recommend the prioritization to funders and researchers (21).
5. Health economic analysis
Recommendation:
A health economic analysis could be made alongside clinical trials in IIH.
Comments:
Clinical trials evaluating medicines, medical devices, and procedures now commonly assess the economic value of these interventions. This information helps regulatory and reimbursement bodies who use this evidence of economic value alongside clinical efficacy to create policy. All health care resource use should be recorded, and the outcomes assessed using ‘natural units’ (e.g., change in vision, as measured by the visual field mean deviation) to form a cost-effectiveness analysis. The incremental cost-effectiveness ratio of an intervention compared with an alternative (such as standard of care) can be calculated and is the difference in costs divided by the difference in outcomes. The incremental clinical benefits, measured as Quality Adjusted Life Years (QALYs) also forms a cost-utility analysis. Clinical endpoints that focus on the impact of a treatment on how a patient feels, functions, and survives are the most useful for economic evaluation.
Methodology used for the use of these guidelines
A detailed search of the scientific literature was performed. This included all English language papers on PubMed, Cochrane and Google Scholar between inception until 1 December 2022. The search strategies combined free‐text and controlled vocabulary terms for IIH. Key words included: CONSORT; intracranial pressure; idiopathic intracranial hypertension; guidelines; headache; missing data; obesity; outcome measure; papilledema; pseudotumor cerebri; randomized control trial; vision and weight loss.
Three virtual meetings of the IIH Trial Guideline Subcommittee (TGS) took place to present the prior clinical trials and discuss outcomes that are important in this disease. The present guidelines were first drafted by the chairs of the Subcommittee and then presented to the entire Subcommittee. The document then was revised several times by members of the ad hoc subcommittee (e.g., bespoke meeting to discuss PROs). It was further revised based on the comments of the IIH subcommittee and IIH UK, a patient charity, by the writing committee. It next was sent for review to members of the IHS Standing committee, and revised further until an agreement was reached and the pre-final version was supported by all. This version was submitted to various stakeholders, including pharmaceutical and devices manufacturers, soliciting and incorporating their feedback on the expert analysis. IHS members then were invited to comment on the IHS website before final approval of the document by the IHS Board of Trustees was given. This process was as agreed by the IHS board.
The main purpose of this guideline was to highlight issues inherent in drug and surgical trials in IIH and to encourage investigators to tackle these problems during the design phase of the trial to improve the quality of controlled clinical trials. Recommendations are based on the clinical experience and research experience of the committee members.
Footnotes
Acknowledgements
The authors would like to thank Shelly Williamson and Clare Parr, representatives of IIHUK, a patient association, for their review of this guideline. IIHUK is a charity registered in England and Wales No. 1143522 and Scotland No. SCO43294.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the authorship and/or publication of this article:
SPM has received honoraria for speaker events from Heidelberg engineering; Chugai-Roche Ltd and Teva. Honoraria for advisory boards for Invex Therapeutics and Gensight. Consultancy fees Neurodiem and Invex Therapeutics. Research funding from the UK Space Agency.
HCD received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Lilly, Novartis, Pfizer, Teva, Weber & Weber and WebMD. The German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union support headache research by HCD. HCD serves on the editorial boards of Cephalalgia, Lancet Neurology and Drugs. HCD is a member of the Clinical Guidelines Committee of the German Society of Neurology and of the Clinical Trials Committee of the IHS.
NRM has received honoraria from the advisory board for Invex Therapeutics
CKMC reports no conflicts of interest.
AJS has received Honoraria for speaker events from Teva. Consulting fees from Invex therapeutics; salary and stockholding with Invex therapeutics. Received research funding from the Medical Research Council, National Institute for Health Research, Sir Jules Thorn Trust and the UK Space Agency.
RHJ has received honoraria for oral presentations from Lectures for Pfizer, Eli-Lilly, ATI, Merck, TEVA, Novartis, Lundbeck and Allergan. She is investigator in clinical trials with ATI, Eli-Lilly, Novartis and Lundbeck and Director of Danish Headache Center, Lifting The Global Burden of Headache and Founder of Master of Headache Disorders at University of Copenhagen. Received research funding from University of Copenhagen, Rigshospitalet, ATI, Lundbeck Foundation, The Medical Society in Copenhagen, NovoNordisk Foundation and Tryg Foundation.
CLF has received consulting fees for Invex therapeutics
PPR has received honoraria as a consultant and speaker for: AbbVie, Amgen, Biohaven, Chiesi, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva. Her research group has received research grants from Novartis, Teva, AbbVie, EraNET Neuron, RIS3CAT FEDER, AGAUR, ISCIII; has received funding for clinical trials from Alder, Amgen, Biohaven, Electrocore, Eli Lilly, Lundbeck, Novartis, Teva. She is the Honorary Secretary of the International Headache Society. She is the founder of ![]() . PP-R does not own stocks from any pharmaceutical company.
. PP-R does not own stocks from any pharmaceutical company.
KBD is on the data safety and monitoring committee for Invex trial.
RBL reports no conflicts of interest. MJ reports no conflicts of interest. MT reports no conflicts of interest. KB reports no conflicts of interest. MJD reports no conflicts of interest. CT has received received compensation for scientific advice and/or speaking activities for Allergan/Abbvie, Dompé, Eli Lilly, Novartis, Lundbeck, Teva and MDweb. GMT reports consultancy or industry support from Novartis, Lilly and Teva, Allergan/Abbvie, and Lundbeck and independent support from the European Community, Dutch Heart Foundation, IRRF, Dioraphte, and Dutch Brain Council.
Funding
KBD is supported in part by an Unrestricted Grant from Research to Prevent Blindness, New York, NY, to the Department of Ophthalmology & Visual Sciences, University of Utah.

