Abstract
Background
Previous findings indicate that the blink reflex is useful to distinguish between primary (classical/idiopathic) and secondary trigeminal neuralgia. No prior studies have investigated whether the blink reflex could identify differences in electrophysiological responses between classical and idiopathic trigeminal neuralgia. With this in mind, we investigated the blink reflex in a cohort of classical and idiopathic trigeminal neuralgia patients.
Methods
Participants were consecutively enrolled in the study. According to magnetic resonance imaging findings, the patients were subgrouped into either classical or idiopathic trigeminal neuralgia. Assessors were blinded to the subgroup and pain side, and the blink reflex was examined to assess R1 and R2 latencies, as well as the area under the curve.
Results
The study group constituted of 55 patients with primary trigeminal neuralgia: 25 patients with classical trigeminal neuralgia and 30 patients with idiopathic trigeminal neuralgia. None of the blink reflex latencies (R1 and R2) or the area under the curve significantly differed between the two subgroups when adjusted for age and sex (p > 0.05).
Conclusions
Our findings suggest that the blink reflex cannot be used to differentiate classical and idiopathic trigeminal neuralgia patients, and that both subgroups may share common pathophysiological mechanisms.
Introduction
Trigeminal neuralgia (TN) is the most frequent cranial neuralgia with a lifetime prevalence of 0.3% (1). Women are more often affected (60%) than men and the average age of onset is between 53 and 57 years of age (2). TN is characterized by intense, paroxysmal pain in one or more divisions of the trigeminal nerve and is triggered by innocuous stimuli (3). The diagnosis of TN is clinical and based on patient history and neurological examination (2). Although the neurological examination is usually normal, subtle sensory abnormalities can be found (4). In addition, magnetic resonance imaging (MRI) is used to exclude secondary TN and to subgroup TN patients (2).
In the most recent Third edition of the International Classification of Headache Disorders (ICHD-3), TN is subdivided into three subgroups based on aetiology: classical and idiopathic TN with or without concomitant continuous pain and secondary TN as a consequence of an underlying disease, such as a tumour, arteriovenous malformation or multiple sclerosis (5). According to the ICHD-3 guidelines, classical TN is diagnosed when evidence of neurovascular compression (NVC) is demonstrated on MRI or during surgery. In this subgroup, morphological changes, such as atrophy or displacement of the trigeminal nerve, are evident. However, neurovascular contact is either absent in idiopathic TN or neurovascular contact without morphological changes is present, and neither electrophysiological tests, nor MRI show significant abnormalities, according to the ICHD-3 (6).
Based on eight studies, the European Academy of Neurology concluded that abnormal trigeminal reflexes are useful indicators to distinguish secondary TN from classical and idiopathic TN, with a sensitivity of 80–93% and a specificity of 91–96% (7). However, the eight studies differed with respect to methods and use of diagnostic criteria (3,8–14). In 2010, Liao et al. (12) found that only three out of 49 patients with classical and idiopathic TN had abnormal blink reflex responses. In 1990, Cruccu et al. (9) demonstrated in a cohort that 100% (20/20) of the patients with secondary TN had abnormal trigeminal reflexes compared to only 6.7% (2/30) of the idiopathic TN patients.
Subsequent to the introduction of the anatomy-based diagnostic subgroups of classical and idiopathic TN in the ICHD-3, no studies have directly investigated whether the blink reflex responses differ between these two TN subgroups. Such a difference would imply that the degree of neurovascular contact affects electrophysiological responses, such as blink reflex latencies and the magnitude of the response measured as the area under the curve (AUC). Furthermore, the blink reflex could potentially be a tool to further elucidate possible differences in the pathophysiological mechanisms underlying classical and idiopathic TN (15). In the present study, we therefore aimed to investigate differences in blink reflex responses between classical and idiopathic TN patients. We hypothesized that patients with classical TN had prolonged latencies and reduced AUCs compared to idiopathic TN patients.
Methods
Participants
Patients diagnosed with classical or idiopathic TN at the Danish Headache Center, a tertiary medical referral center for headache and facial pain in Copenhagen, were included during a time period of 11 months from April 2021 until March 2022. The study protocol was approved by the relevant ethics committee and data protection agency. The study was conducted in accordance with the Declaration of Helsinki.
The inclusion criteria were: (i) age 18 years old or above; (ii) the ability to give signed informed consent; (iii) fulfilling the ICHD-3 diagnostic criteria for classical or idiopathic TN; (iv) evaluation by MRI scan according to neuralgia specific protocol for TN (16) a maximum of 2 years before or after the assessment of the blink reflex; and (v) having had a semistructured interview and neurological examination by independent assessors a maximum of 2 years before or after the assessment of the blink reflex. The exclusion criteria were: (i) incapacitated patients; (ii) previous neurosurgical treatment; (iii) bilateral TN; (iv) secondary TN; and (v) sensory deficits found at the clinical neurological examination.
Based on the MRI, the degree of neurovascular contact was evaluated by experienced neuroradiologists at the Department of Radiology at Rigshospitalet – Glostrup, and patients were subgrouped accordingly. An MRI showing no or simple neurovascular contact classified the patient as having idiopathic TN, whereas a severe contact with morphological changes or atrophical changes found on MRI, categorized the patient as classical TN (17).
Data collection
A medical laboratory scientist or nurse administered a semistructured questionnaire to assess the number of paroxysms, and the current dosage of medicine. The staff was blinded to the pain side and TN subtype. Patient data were obtained from the patient’s journal and included information about sex, age, MRI results and the occurrence of concomitant continuous pain.
The MRI protocol included sagittal T2-weighted turbo-spin-echo sequence covering the entire brain, axial thin section T2-weighted gradient and spin-echo sequence encompassing the brainstem and the proximal and posterior fossa, 3D time of flight magnetic resonance angiography and 3D high spatial resolution heavily T2-weighted sequence. Multiplanar reconstructions were performed of the cisternal part of the neurovascular contact strongly parallel to the trigeminal nerve in the axial and sagittal plane, perpendicular to the neurovascular contact in the coronal plane (18).
The blink reflex in the present study was measured by means of an electromyographic machine (Keypoint®; Natus Medical Inc., Middleton, WI, USA). The supraorbital nerve was stimulated transcutaneously at its emergence from the eyebrow arch (notch), indicating the path of the supraorbital branch of the ophthalmic division of the trigeminal nerve. The stimulation intensity was gradually increased according to a predefined staircase method using intensities of 3, 6, 9 and 12 mA; thus, four trials on each side. The duration of electrical pulses was 0.3 ms, bandwidth filter was 5 Hz to 3 KHz, and the time between consecutive stimuli was at least 45 seconds to avoid habituation (19).
With the patient in the supine position, the stimulating electrode was placed on the supraorbital notch, with the eyes open and patient relaxed. One electrode was placed on the inferior part of the orbicularis oculi muscle just below the lower eyelid, whereas another electrode was placed lateral to the eye as a reference. A ground electrode was placed on the forehead or the shoulder. The two components of the blink reflex [i.e. R1 (ipsilateral side) and R2 (ipsilateral and contralateral side) were recorded on the Keypoint machine, and the response was shown on a monitor, first with stimulation on the left side and then with stimulation on the right side. An experienced and blinded electromyographical assessor manually set the cursors for the onset of the blink reflex components in all patients.
Concerning the AUC, the areas for the left- and right-side responses were visualized and calculated using Mathlab® (MathWorks Inc., Natick, MA, USA). The onset of R2 constituted the starting point of the AUC, and the mark for no visible signal constituted the ending point. The area in between constituted the AUC in units [µV × ms], rectified and with a high pass filter of 25 Hz.
Outcome measures
The two primary outcomes were differences between classical and idiopathic TN for the following parameters: latencies for the R1 pain side and latencies for the R2 pain side. Latencies for the R1 non-pain side, latencies for R2 non-pain side, AUC for the pain side and AUC for the non-pain side were considered secondary outcomes.
Statistical analysis
Descriptive statistics on continuous normal data are expressed as the mean (SD). Categorical data are shown as frequency distributions in percentages. Differences between the two TN subgroups were investigated with the Mann–Whitney U test for continuous data that were not normally distributed, as well as Fischer’s exact test for binary data. To test differences between the pain and non-pain sides for blink reflex latencies and AUCs, the Wilcoxon signed-rank test was performed for the classical and idiopathic subgroups, individually.
For the blink reflex latencies and AUC, multiple regression was performed. R1 pain side, R1 non-pain side, R2 pain side, R2 non-pain side, AUC pain side and AUC non-pain side were defined as six individual response variables. Factors were classical versus idiopathic TN, sex and age. Assumptions for multiple regression were assessed by means of residual plots in SPSS (IBM Corp., Armonk, NY, USA). The analyses were conducted using SPSS, version 28.0. p < 0.05 (two-tailed) was considered statistically significant.
Results
Sixty-seven participants were consecutively enrolled in the study. In total, 12 participants were excluded after enrolment (Figure 1). Four participants lacked a MRI scan within 2 years of the investigation. Six participants lacked a blink reflex on any side (four had classical and two had idiopathic TN), one patient was diagnosed to have persistent idiopathic facial pain upon evaluation, and one patient decided to withdraw during the blink reflex procedure before sufficient data were obtained. Thus, the total study group consisted of 55 patients with primary TN. Blink reflex recordings for left side stimulations with 3, 6, 9 and 12 mA are shown in Figure 2 for a classical patient and for an idiopathic patient in Figure 3.

Flow chart of the inclusion process.

Blink reflex recordings for left side stimulations with 3, 6, 9 and 12 mA for a classical patient.
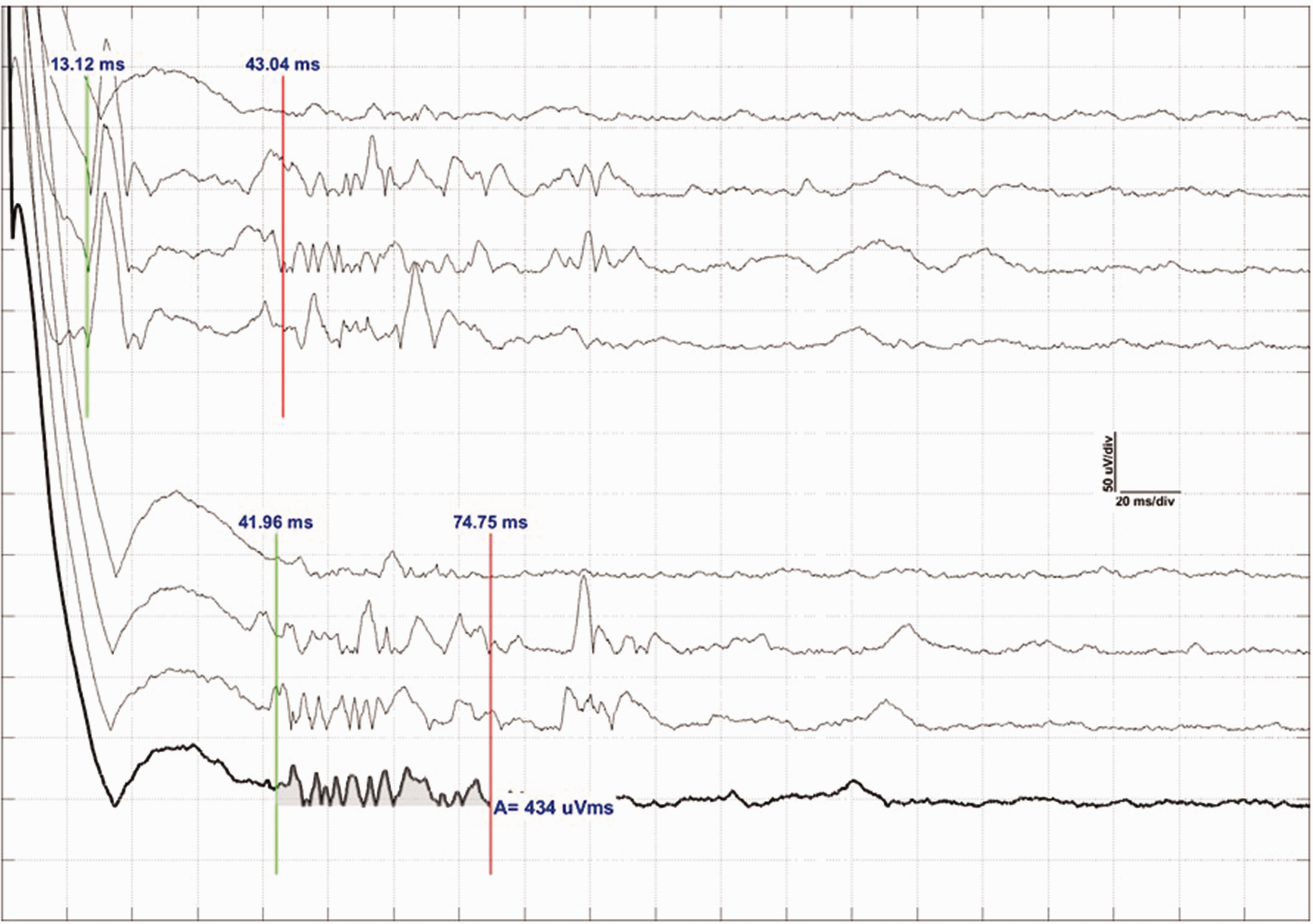
Blink reflex recordings for left side stimulations with 3, 6, 9 and 12 mA for an idiopathic patient.
Trigeminal neuralgia subgroups
The study group included 25 patients with classical TN (52% were women) and 30 patients with idiopathic TN (80% were women) (Table 1). Age was higher in the classical group (median age = 72 years/interquartile range = 20 years) than in the idiopathic group (median age 56 years/ interquartile range = 29.5 years) (p = 0.009). The idiopathic group included significantly more women than the classical group (p = 0.043). In total, 32% of classical TN patients and 57% of idiopathic TN patients experienced persistent continuous pain (p = 0.103). In the classical TN subgroup, 84% patients took medicine and, in the idiopathic TN subgroup, 67% patients were on TN medication (p = 0.215). All patients who received medicine experienced a reduction in pain intensity and frequency. Carbamazepine, oxcabazepine, gabapentin, pregabalin, lamotrigine, other medication or a combination of these were the medication types that patients in the study group were treated with (Table 2). Of the 25 patients with idiopathic TN, only one lacked neurovascular contact. Thus, 24 patients had neurovascular contact without morphological changes.
Demography and clinical characteristics.

CTN, classical TN; ITN, idiopathic TN; IQR, interquartile range; f, female.
Number and percentage of patients in the two groups who received medicine.

CTN, classical TN; ITN, idiopathic TN.
Blink reflex latencies
The R1 latency on the pain side was 10.5 ± 3.1 ms in classical TN and 11.7 ± 3.0 ms in idiopathic TN (p = 0.116) (Table 3). After adjusting for age (p = 0.556) and sex (p = 0.679), the TN subgroups did not significantly differ (p = 0.216) (Table 4). The R1 latency on the non-pain side was 11.2 ± 4.3 ms in classical TN and 10.4 ± 3.2 ms in idiopathic TN (p = 0.153). After adjusting for age (p = 0.854) and sex (p = 0.181), the TN subgroup did not significantly differ (p = 0.279). The R2 latency on the pain side was 40.3 ± 5.6 ms in classical TN and 38.1 ± 4.8 ms in idiopathic TN (p = 0.174). After adjusting for age (p = 0.325) and sex (p=0.838), the TN subgroup showed no significant difference (p = 0.336). The R2 latency on the non-pain side was 38.6 ± 4.6 ms in classical TN and 37.7 ± 4.7 ms in idiopathic TN (p = 0.240). After adjusting for age (p = 0.262) and sex (p = 0.951), the TN subgroup showed no significant difference (p = 0.799).
Blink reflex latencies and area under the curve.

p-values from results of adjustments in multiple regression.

Area under the curve
The AUC for the pain side was 1566.5 ± 865.2 µV × ms in the classical TN subgroup and 1903.7 ± 1003.5 µV × ms in the idiopathic TN subgroup (p = 0.302). After adjusting for age (p = 0.143) and sex (p = 0.058), this parameter did not differ by TN subgroup (p = 0.913).
The AUC for the non-pain side was 1806.1 ± 1453.3 µV × ms in the classical TN subgroup and 1600.7 ± 726.3 µV × ms in the idiopathic TN subgroup (p = 0.583). After adjusting for age (p = 0.694) and sex (p = 0.114), this parameter did not differ by TN subgroup (p = 0.232).
Pain versus non-pain side
R1, R2 and AUC did not differ between the pain side and the non-pain side in the classical subgroup (p > 0.300). In the idiopathic subgroup, R2 did not differ between the pain and the non-pain sides (p = 0.905). However, R1 (p = 0.040) and AUC (p = 0.033) differed between the pain side and the non-pain side in the idiopathic group. The mean R1 was longer and the mean AUC was greater on the pain side than on the non-pain side in the idiopathic TN subgroup (Table 5).
Mean latencies and SD of R1 and AUC on the pain side in the idiopathic group.

Discussion
Main findings
The present study is the first to investigate possible electrophysiological differences between classical and idiopathic TN. Our main findings were lack of differences in any blink reflex latencies or AUCs between classical and idiopathic TN patients. We also found no difference in any blink reflex latency or AUC in the classical group when comparing pain and non-pain side. In the idiopathic group, we found an increased R1 latency, and the AUC was greater on the pain side than on the non-pain side.
Nerve conduction in trigeminal neuralgia
The comparable and similar electrophysiological responses regardless of the degree of neurovascular contact suggest that the trigeminal nerve is likely not majorly and irreversibly affected in either classical- or idiopathic TN. The intactness of the nerve in terms of electrophysiologic response corresponds with the usual normal bedside sensory examination found in both groups (2). In support of the latter, TN patients were systematically investigated with quantitative sensory testing, and Younis et al. (20) found only slightly abnormal sensory findings. The present results may also explain why neurological examination is generally normal in patients with classical and idiopathic TN (21). Thus, our results are in agreement with those of another study showing that classical and idiopathic TN appear clinically similar (16). In this study, Maarbjerg et al. (16) compared the degree of NVC with clinical characteristics in 135 TN patients with classical (n = 71) and idiopathic (n = 64) TN and found no difference between classical and idiopathic TN patients with respect to the prevalence of concomitant persistent pain, autonomic symptoms, sensory abnormalities at bedside examination, response to sodium channel blockers, hypertension, other cardiovascular disease or family history of TN or facial pain.
Classical versus idiopathic trigeminal neuralgia – common pathology?
The absence of a difference in electrophysiological responses between the two groups could also imply that an overlap in pathophysiology between classical and idiopathic TN may exist. As a result of compression resulting in ectopic firing of the nerve fibers, demyelination is one of the prevailing theories about the development of classical TN (22). However, other unidentified factors may also cause local demyelination, especially in patients with idiopathic TN. It is possible that the blink reflex findings reflect that the classical TN patients in the present study might not have severe trigeminal nerve demyelination, despite severe neurovascular contact shown on MRI. Thus, the pathophysiology of the classical TN patients in this study might be caused by, for example, idiopathic inflammation or small fiber neuropathy rather than demyelination by neurovascular compression. A study using diffusion tensor imaging (DTI) indicated that the demyelination of the trigeminal root occur in idiopathic TN (17). Lutz et al. (23) investigated 61 patients with classical TN and 21 with idiopathic TN using advanced DTI to evaluate the influence of clinical and anatomical parameters on structural alterations. The study revealed significantly lower fractional anisotropy (FA) values, as a marker of demyelination, within the vulnerable zone of the affected trigeminal nerve. However, FA values did not differ between the classical and idiopathic TN patient groups. The lack of difference in FA values could explain the lack of differences and comparable latencies and AUCs within the two subgroups observed in the present study. Consequently, although classical and idiopathic TN reflect differences in MRI scans, a common pathophysiology shared by the two subgroups is likely to exist. These findings are also in accordance with recent data demonstrating that microvascular decompression can have a good and immediate effect in patients with idiopathic TN and neurovascular contact without morphological changes (24,25). However, the true and complex origin of idiopathic TN still remains unclear, and further longitudinal studies are needed to explore in this area.
Perspectives
In the present study, supraorbital nerve stimulation was performed by means of the blink reflex test. However, 60% have pain in the branch of V2 or V3 (26). Repeating the experiment and stimulation, the mandibular and maxillary branches of the trigeminal nerve could confirm or refute our findings further. Moreover, the use of the masseter inhibitory reflex likewise could complement our findings, and thus is relevant in future studies.
The findings of the present study could set the stage for a redefinition of idiopathic TN, according to the ICHD-3. Specifically, idiopathic TN, defined as a disorder where electrophysiological tests are normal cannot be endorsed because our findings demonstrate that electrophysiological tests do not differ between classical and idiopathic TN. Further studies are needed to clarify whether this recently introduced subdivision of TN, which was based on expert opinion, is scientifically sound.
Limitations
Considering the electrophysiological responses when differentiating the pain and the non-pain side in the two groups, our findings were numerically quite similar, which may be attributed to type 1 errors. The current findings may indicate both group patients had intact myelinated nerve (A-β) fibers function rather than similar pathophysiology. Thus, other electrophysiologic studies, including laser-evoked potentials (LEP) (27), contact heat evoked potentials (CHEPs) (28), nociceptive blink reflex (nBR) and pain-related evoked potentials (PREP) (29), and current perception threshold (CPT) (12) would be useful to investigate the conditions and possible difference of trigeminal nerve function in classical and idiopathic TN patients. If classical and idiopathic TN patients have similar LEP, CHEPs, nBR, and PREP results this could more strongly strengthen that patients with classical and idiopathic TN have identical conditions of A-β, A-δ and C-fibers, and probably also have similar pathophysiology.
Most patients were treated with antiepileptic drugs as preventive treatment for TN, which may have affected the responses. However, a post-hoc analysis adjusting for medicine intake did not result in any differences between the groups.
Conclusions
The present study showed no differences in electrophysiological responses between classical and idiopathic TN patients. The findings suggest that common pathophysiological traits between classical and idiopathic TN are likely to exist. Hence, using the blink reflex as a diagnostic tool to differentiate the two TN subgroups cannot be recommended.
Clinical implications
An investigation of the blink reflex was conducted in 25 classical and 30 idiopathic TN patients. Differences in electrophysiological responses were identified. R1 and R2 latencies and area under the curve were assessed. Classical and idiopathic trigeminal neuralgia may share common pathophysiological mechanisms.
Footnotes
Acknowledgements
We thank Merete Bak Bertelsen and Janne Jensen for their invaluable help with the data extraction. Additionally, we thank the Department of Public Health, Section of Biostatistics University of Copenhagen, for statistical analyses counselling.
Declaration of conflicting interests
The authors declare that there are no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
