Abstract
Background
The prevalence of cardiac cephalalgia is unknown and there is limited information about its clinical features. We aimed to assess the prevalence of cardiac cephalalgia, its clinical characteristics and associated factors.
Methods
We conducted a prospective study of patients with suspected acute coronary syndrome admitted to the Cardiology Service at Hospital Clínico Universitario Lozano Blesa, Zaragoza, Spain, over a one-year period. We interviewed patients within the first 24 hours of admission using a standardized case-report form to assess the presence of headache in relation to the acute coronary syndrome and its characteristics.
Results
We included 438 patients, 381 with confirmed myocardial ischemia. Prevalence of cardiac cephalalgia was 14.2% (n = 54). The most common features were frontal location, pressing quality and moderate intensity. Pain referred to the jaws (aOR 2.61; 95% CI 1.33–5.12; p = 0.005), palpitations (aOR 3.65; 95% CI 1.57–8.50; p = 0.003) and circumflex coronary artery as the culprit artery for the myocardial ischemia (aOR 3.8; 95% CI 1.07–13.74; p = 0.021) were related to cardiac whereas history of hypertension was inversely associated (aOR 0.37: 95% CI 0.18–0.74; p = 0.005).
Conclusion
The prevalence of cardiac cephalalgia was 14.2%. Our study provides valuable information about cardiac cephalalgia characteristics that suggest revision of current diagnostic criteria.
Introduction
Cardiac cephalalgia is included in the International Classification of Headache Disorders third edition (ICHD-3) (1) in the category of headache attributed to disorder of homeostasis. It is described as a ‘migraine-like headache, usually but not always aggravated by exercise, occurring during an episode of myocardial ischemia’ (1). Diagnostic criteria include the demonstration of myocardial ischemia and the evidence of causation (1) (Table 1). Despite various case reports being published since its first description (2), the prevalence of cardiac cephalalgia is unknown and information about its clinical phenotype is limited. Furthermore, current diagnostic criteria are based on case reports and on the opinion of experts (1).
ICHD-3 diagnostic criteria for cardiac cephalalgia (1).

Ischemic heart disease is frequent and constitutes the leading cause of death worldwide (3). Given that cardiac cephalalgia can be one of the manifestations of myocardial ischemia, a better understanding of this entity is necessary. Thus, the aims of our study were to assess the prevalence of cardiac cephalalgia in patients with myocardial ischemia admitted to a Cardiology Service and to describe its clinical features and associated factors.
Methods
Study design
We conducted a prospective study of consecutive patients admitted to the Cardiology Service at Hospital Clínico Universitario Lozano Blesa, a public third level hospital in Zaragoza, Spain, with suspected acute coronary syndrome (ACS) from 5th January 2021 to 5th January 2022. All patients admitted to the Cardiology Service with suspected ACS were evaluated for participation in the study.
The inclusion criteria were: 1) patients admitted to the Cardiology Service with suspected ACS; 2) older than 18 years; and 3) who agreed to participate and signed the informed consent form. The exclusion criteria were: 1) patients unable to communicate in Spanish; 2) unable to understand the study or to precisely describe their symptoms because of cognitive impairment or disorder of consciousness; and 3) who had already participated in the study.
Data were collected using a purpose-built standardized case-report form during face-to-face interviews performed by the research team (two neurologists (MPNP and SSL) and two neurology residents (JER and SBM)) within the first 24 hours of admission. We also analysed electronic medical records and the results of investigations performed during the admission with the collaboration of one cardiologist (PRM).
We obtained patients' baseline characteristics from clinical interviews and from patients’ electronic medical records. We included demographics (age and sex) and clinical variables. Clinical variables included prior medical history (diabetes, hypertension, smoking, hyperlipidemia, history of cerebrovascular disease, and history of coronary artery disease [CAD]); ACS characteristics (duration, presenting symptoms including chest pain, jaw pain, upper arm pain, nausea, sweating, dyspnoea, palpitations and syncope); acute investigations (12-lead ECG findings [normal, ST-segment depression and/or T-wave changes, ST-segment elevation or left bundle branch block] initial and maximum high-sensitivity troponin T [hs-cTnT] level); and acute treatment including IV thrombolysis, percutaneous coronary intervention and medical management. We also recorded the culprit coronary artery in patients who underwent percutaneous coronary intervention.
Prior headache history was also evaluated. Participants were classified by clinical interview as ‘migraine history’, ‘tension-type headache (TTH) history’, ‘trigeminal-autonomic cephalalgia history’ or ‘headache unspecified history’ based on ICHD-3 diagnostic criteria (1).
Cardiac cephalalgia assessment
We evaluated all headaches at ACS onset. We consider a headache as cardiac cephalalgia if ICHD-3 diagnostic criteria (1) were met.
For the demonstration of myocardial ischemia, criteria from the fourth universal edition of myocardial infarction (2018) (4) were used and patients were classified in four groups: ST-segment elevation myocardial infarction (STEMI), non-ST-segment elevation myocardial infarction (NSTEMI), unstable angina and stable angina.
To evaluate temporal relation, we recorded the time of headache onset in relation to the onset of ACS symptoms and we classified headache onset as simultaneously, before or after ACS symptoms. If the headache started after ACS symptoms, we paid special attention to exclude headache related to nitroglycerine or its derivates. We excluded headaches that started or worsened after the administration of nitroglycerine or derivatives of it.
For the evaluation of the four characteristics described in the diagnostic criterion C3 (1) we asked for the presence of accompanying symptoms (nausea, photophobia, phonophobia, conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, miosis, ptosis and eyelid oedema).
To assess pain intensity, we used the 11-point (0–10) numerical rating scale (NRS). 0 meaning “no headache at all” and 10 meaning “the worst possible headache”. We defined scores of 1 to 3 as mild, 4 to 6 as moderate and 7 to 10 as severe. We asked if the headache worsened by physical activity with three possible answers, “yes”, “no” and “don’t know”.
In patients who reported headache at ACS onset and who had headache history we asked if headache had the same characteristics as the pre-existing headache. If pre-existing headache occurred unaltered, the diagnosis of cardiac cephalalgia was excluded.
Statistical analysis
Descriptive statistics of demographic and clinical variables were conducted. We used frequencies and percentages to describe categorical variables. To present continuous variables means and standard deviations (SD) or median and interquartile range (IQR) were used depending on the normality of the data. To determine whether sample data were normally distributed, Shapiro-Wilk test was used.
To explore independent factors associated to cardiac cephalalgia a multivariate binary logistic regression analysis with enter method was performed. We included in our model the following variables: demographic variables (age and sex), prior medical history of hypertension and diabetes, prior headache history, presenting symptoms of the ACS, type of myocardial infarction (STEMI or NSTEMI) and culprit coronary artery. The adjusted odds ratio (aOR) was calculated with its 95% confidence intervals (95% CI). Complete-case analysis was used to manage missing data. A statistical significance was accepted if the P value was <0.05 (2-tailed). IBM SPSS version 22.0 software (Chicago, IL, USA) was used to conduct the statistical analysis.
Patient consent and registration
The study was approved by the Regional Research Ethics Committee of Aragon (CEICA) with the reference number PI20/524. All participants provided written informed consent before taking part into the study.
Data availability
De-identified participant data are available for other researchers under reasonable request.
Results
During the study period 538 patients were admitted to the Cardiology Service with suspected ACS. 100 patients met exclusion criteria and were excluded from the study. We initially included 438 patients with suspected ACS. After completing investigations during the admission, we identified 57 patients without confirmed myocardial ischemia: 41 patients (71.9%) in whom myocardial ischemia was ruled out, five patients (8.8%) where myocardial ischemia was suspected but required further investigations and was not confirmed during admission, and 11 patients with other causes of myocardial injury (six patients [10.5%] diagnosed with Takotsubo syndrome, four [7%] with myocarditis and one patient [1.8%] with myocardial injury after catheter ablation). Therefore, 381 patients with confirmed myocardial ischemia were included. Figure 1

Study flow diagram.
Prior medical and headache history and baseline characteristics
304 patients (79.8%) were male. Their mean age was 65.7 years (±12.9 years). Most patients had history of cardiovascular risk factors (n = 361; 94.8%) and 84 patients (22%) had history of headache. Diagnoses at admission were ST-segment elevation ACS in 169 patients (44.4%), non-ST-segment elevation ACS in 212 patients (55.6%).
After completing investigations during the admission, diagnoses at discharge were STEMI in 170 patients (44.6%), NSTEMI in 155 patients (40.7%), unstable angina in 52 patients (13.6%), stable angina in four patients (1%). Table 2 shows demographics, baseline and ACS characteristics of patients with myocardial ischemia with and without cardiac cephalalgia.
Demographics, baseline and ACS characteristics of patients with myocardial ischemia with and without cardiac cephalalgia.
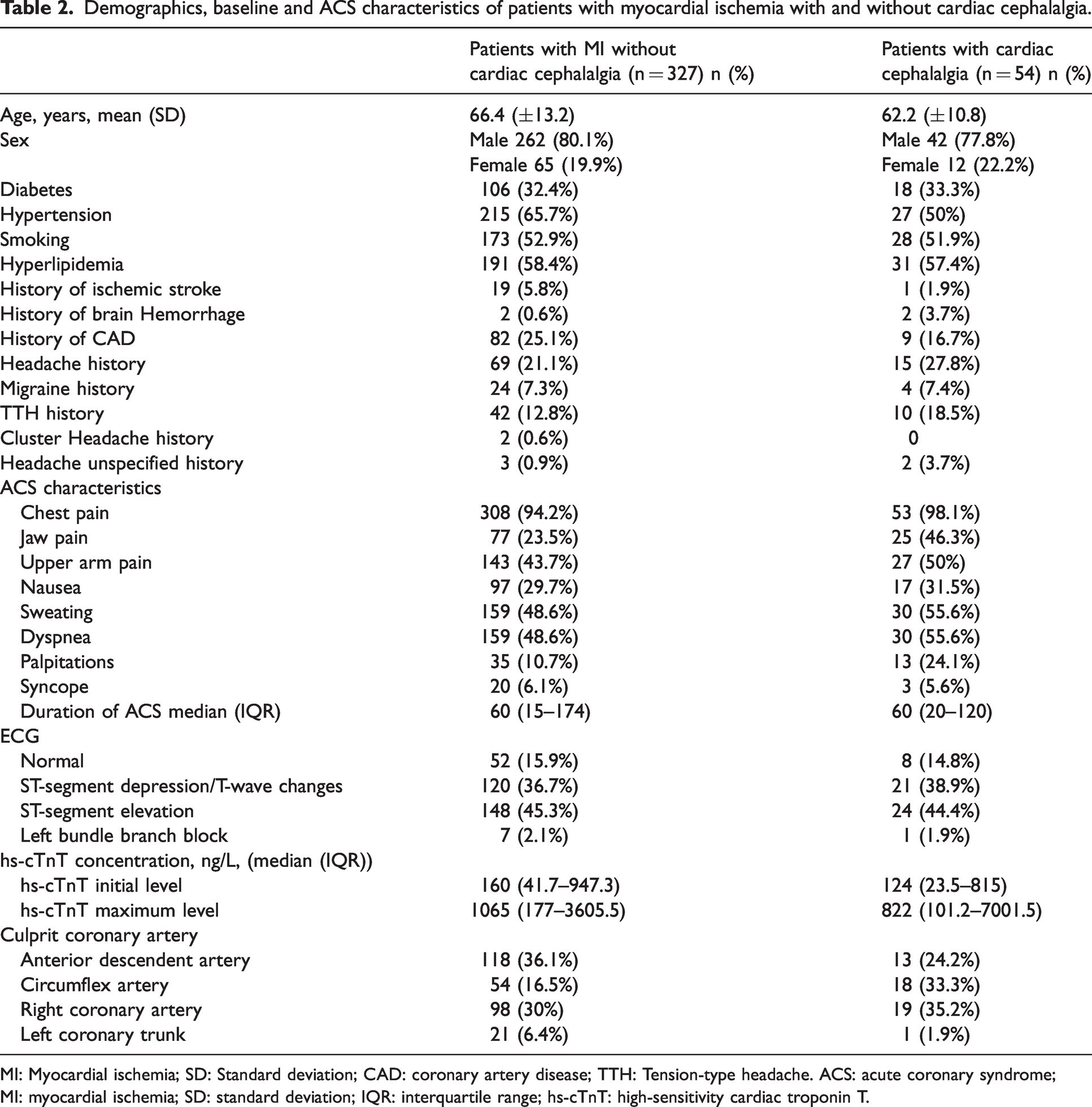
MI: Myocardial ischemia; SD: Standard deviation; CAD: coronary artery disease; TTH: Tension-type headache. ACS: acute coronary syndrome; MI: myocardial ischemia; SD: standard deviation; IQR: interquartile range; hs-cTnT: high-sensitivity cardiac troponin T.
Cardiac cephalalgia
Among 65 patients (14.8%) who reported headache in relation with ACS onset, 54 patients fulfilled cardiac cephalalgia diagnostic criteria. Among patients who did not fulfil these criteria, 10 patients did not have demonstration of myocardial ischemia and one patient presented his pre-existing headache unaltered (TTH). Therefore, prevalence of cardiac cephalalgia in patients with myocardial ischemia was 14.2% (54/381).
Most patients were male (n = 42; 77.8%) and their mean age was 62.2 years (±10.8 years). 25 patients (46.3%) were admitted with suspected ST-segment elevation ACS, 29 (53.7%) with non-ST-segment elevation ACS.
Regarding characteristics of cardiac cephalalgia, headache started simultaneously with ACS in most patients (n = 47, 87%), in six patients (11.1%) headache started before ACS symptoms with a median time of 15 minutes (IQR 18.7–48.7) and in one patient (1.9%) headache started 10 minutes after ACS symptoms. Median duration of headache was 60 minutes (IQR 30–120).
In most patients (n = 47, 87%) headache was bilateral, five patients (9.3%) reported unilateral left sided pain and two patients (3.7%) unilateral right sided pain. The most frequent locations of headache were frontal (n = 32, 59.3%) and temporal (n = 19, 35.2%). Regarding headache quality, headache was pressing in most cases (n = 36, 66.7%) and throbbing in six patients (11.1%). Median intensity in the NRS was 6 (IQR 4.7–7). A complete description of headache characteristics can be seen in Figure 2.

Characteristics of headache in patients with cardiac cephalalgia.
Factors associated with cardiac cephalalgia
Table 3 summarizes the results of the multivariate logistic regression. In total 380 cases were included in the analysis. This model was statistically significant (χ2 = 41.43; p = 0.002), explained 18.5% (Nagelkerke R2) of the variance in cardiac cephalalgia and correctly classified 84.6% of cases.
Results from the multivariate logistic regression model.

Bolded values are the statistically significant ones, but this can be modified.aOR: adjusted odds ratio; ACS: acute coronary syndrome; MI: myocardial infarction; STEMI: ST-segment elevation myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction.
Multivariate logistic regression analysis indicated that pain referred to in the jaws (aOR 2.61; 95% CI 1.33–5.12; p = 0.005), palpitations (aOR 3.65; 95% CI 1.57–8.50; p = 0.003) and circumflex coronary artery as the culprit artery for the myocardial ischemia (aOR 3.8; 95% CI 1.07–13.74; p = 0.021) were associated with cardiac cephalalgia. Hypertension was inversely associated with cardiac cephalalgia (aOR 0.37: 95% CI 0.18–0.74; p = 0.005).
Discussion
We found a prevalence of cardiac cephalalgia in patients admitted with myocardial ischemia of 14.2%. Although clinical characteristics of cardiac cephalalgia were variable, frontal location, pressing quality and moderate intensity were most frequently reported. Pain referred to the jaws, palpitations and myocardial infarction secondary to circumflex artery occlusion/stenosis were more frequent in patients with cardiac cephalalgia, whilst a history of hypertension was less common in these patients.
Prevalence of cardiac cephalalgia
We found a higher prevalence than expected based on previous studies that investigated headache as a presenting symptom of myocardial ischemia. Of 150 ambulatory patients with angina pectoris 6% reported headache as a presenting symptom but it was not the only manifestation in any case (5). Another study found headache as a presenting symptom in 5.2% of 1546 patients with myocardial infarction (6). In 3.4% headache was the main pain location (6). This higher prevalence might be due to the different research methodology employed in these studies.
Cardiac cephalalgia characteristics
We found that frontal and temporal location of cardiac cephalalgia were the most common in agreement with a previous study (7) while most previous case reports described an occipital location (8).
Cardiac cephalalgia is described as a migraine-like headache in the ICHD-3 (1), but our results showed that most patients presented pressing quality (66.7%). Headache quality information from previous case reports is scarce and description of quality is very variable (7–13). Throbbing and pressing pain were the most frequently reported in previous case reports (8) whereas an observational study found that throbbing quality was the most common (33.3%) (7). Contrary to previous case reports, that in most cases described severe headache (57.9%) (8–13), we found that in one-half of patients the intensity was moderate.
According to ICHD-3 criteria, two of the characteristics of cardiac cephalalgia are ‘accompanied by nausea’ (sub-criterion C3b) and ‘not accompanied by photophobia or phonophobia’ (sub-criterion C3c) (1). Our results showed that approximately one-half of patients with cardiac cephalalgia did not report any accompanying symptom and photophobia and phonophobia were reported by 14.8% and 13% respectively. Nausea was reported only by 18.5% of our patients with cardiac cephalalgia. This finding is in line with previous literature that showed a low frequency of accompanying nausea 23–36.6% (7–9). Two patients reported tearing but no other cranial autonomic symptoms. The presence of cranial autonomic symptoms has been described before in two patients with tearing and one patient with rhinorrhea (7).
In initial reports cardiac cephalalgia was described as an exertional headache. Nevertheless, several case reports showed headache at rest and some previous case reports did not specify if aggravation by physical activity occurred (8). We found that most patients (38.9%) did not know if headache worsened by physical activity.
All the patients in our study presented other symptoms related to the myocardial ischemia in agreement with a previous study where most patients presented cardiac symptoms, mainly chest pain (7). In up to 23.8% of previous case reports, cardiac cephalalgia was, however, the only manifestation of myocardial ischemia (8). Publication bias might be the most likely explanation for this discrepancy.
Factors associated with cardiac cephalalgia
Age at onset was similar to that described in prior case reports (62.3 years) (8) and in the observational study (64.6 years) (7).
Patients with cardiac cephalalgia had a lower prevalence of hypertension history (aOR 0.37: 95% CI 0.18–0.74; p = 0.005). These findings would be consistent with the phenomenon of hypertension-associated hypalgesia (14). Although its mechanisms remain to be elucidated, it is thought that high blood pressure (BP) might stimulate the baroreflex arch and lead to the inhibition of pain transmission (15). It has been shown that patients with hypertension and CAD had lower anginal pain perception during episodes of myocardial ischemia than patients with CAD without hypertension (16). Moreover, even though the relationship between migraine and BP is very complex, two prospective studies showed that high BP was related to a reduction in the risk of nonmigrainous headache 11 years later (17) and that increasing systolic BP and pulse pressure were associated with decrease in the prevalence of both migraine and nonmigrainous headache (18).
Interestingly, we found that the circumflex artery as the culprit artery was associated with cardiac cephalalgia (aOR 3.8; 95% CI 1.07–13.74; p = 0.021). These findings would be consistent with the convergence theory of cardiac cephalalgia (8,11,12). In accordance with this theory, cardiac cephalalgia would be a referred pain resulting from the confluence of cardiac autonomic fibers that transmit cardiac pain and somatic fibers in a set of neurons in the posterior horn of the spinal cord (8,11,12). In accordance with this hypothesis, ischemia of the inferior-posterior cardiac territory, mainly irrigated by the right and circumflex coronary arteries, would activate the cardiac parasympathetic afferent fibers that project towards the nucleus of the solitary tract and subsequently converge with somatic fibers coming from the cervical and trigeminal territory at the C1–C2 level (19–21). In contrast, ischemia in the anterior part of the left ventricle would activate sympathetic fibers which may converge in the upper thoracic and cervical spinal segments with the somatic stimuli from the chest and upper arm (19–21). Previous case reports have also reported occlusion/stenosis of the circumflex artery (8). Nevertheless, association between the culprit artery for the myocardial ischemia and cardiac cephalalgia have not been investigated before.
Other three pathophysiological mechanisms of cardiac cephalalgia have been proposed including vasodilation in brain arteries due to the release of inflammatory mediators, increased intracranial pressure secondary to a reduction of cerebral venous drainage because of the decrease of cardiac output and a third theory that states that headache could be the result of vasoconstriction of small intracranial arteries due to activation of the sympathetic system (8,11,12).
Jaw pain was more frequent in patients with cardiac cephalalgia (aOR 2.61; 95% CI 1.33–5.12; p = 0.005). Prior case reports have also reported pain referred to the jaws (22–25). According to previous studies, jaw and neck pain have been related to inferior myocardial infarctions (6) and they are less likely to be present in left anterior descending coronary occlusion (26). Hence, the association found between jaw pain and cardiac cephalalgia would also be in line with the convergence theory and with the distribution of sympathetic and vagal afferents in the cardiac wall (19–21).
Palpitations were also associated with cardiac cephalalgia (aOR 3.65; 95% CI 1.57–8.50; p = 0.003). Palpitations are a common symptom characterized by awareness of the heartbeat (27). Physiopathology of palpitations is unknown and they can be caused by cardiac and noncardiac etiologies (27–29). Palpitations have been previously described in prior case reports (8) and are also reported as a presenting symptom in ACS (30). Nevertheless, these findings should be taken with caution because palpitations can be described in multiple ways and can be influenced by multiple factors (29). Finally, it is also interesting that, despite the substantial overlap in symptoms of ACS, women with ACS have higher odds of presenting jaw pain and palpitations (30). However, we did not find sex differences in cardiac cephalalgia prevalence.
Implications for clinical practice
Our results showed that cardiac cephalalgia is not a rare presentation of acute myocardial ischemia and we provided further evidence to consider headache as a presenting symptom in these cases. We consider that this is an important finding to clinicians given that recognizing the first symptoms in patients with ACS is crucial.
Furthermore, compared to occlusions of other major coronary arteries, ECG detects acute occlusions of the circumflex artery only in 32 to 48% of patients which could delay treatment and result in worse outcomes (31). For this reason, the association found between circumflex artery stenosis/occlusion and cardiac cephalalgia could improve the diagnosis in these cases.
Finally, the association of cardiac cephalalgia with jaw pain and the circumflex artery stenosis/occlusion found in our study also contribute to the understanding of the possible underlying mechanisms of cardiac cephalalgia.
Strengths and limitations
The results of this study go beyond previous literature because, we prospectively assessed prevalence, clinical characteristics and factors associated with cardiac cephalalgia. We included a large number of patients with suspected ACS upon admission to a Cardiology Service. Trained physicians performed data collection using a purpose-built standardized case-report form.
Nonetheless, our study has some limitations. First, the lack of a control group does not allow us to rule out that headache could be influenced by other factors such as emergency room visit or admission. Second, we only included patients that were admitted for suspected ACS, but we did not include other patients with possible myocardial ischemia such as patients with stable angina who were managed in an outpatient setting. Therefore, this might misestimate cardiac cephalalgia prevalence and might limit the generalizability of the results. It is also possible that this group of patients with less severe cardiac symptoms might present headache as the only manifestation of myocardial ischemia. Third, despite interviews being performed within the first 24 hours of admission, information obtained may be subject to recall errors. Finally, there are some limitations of the generalizability of our findings given that we excluded patients that were unable to communicate in Spanish, understand the study or to precisely describe their symptoms.
Conclusions
Cardiac cephalalgia was present in 14.2% of patients admitted with myocardial ischemia. Our study provides valuable information about cardiac cephalalgia characteristics that suggest revision of current diagnostic criteria. We found that frontal location (59.3%), pressing quality (66.7%) and moderate intensity (50%) were the most common headache characteristics. Furthermore, accompanying nausea was reported only in 18.5% of patients and aggravation by physical activity was present in 31.5%.
Importantly, our results indicate that jaw pain, palpitations and circumflex coronary artery as the culprit artery for the myocardial ischemia were related to cardiac cephalalgia whilst history of hypertension was inversely associated.
Key findings
The prevalence of cardiac cephalalgia was 14.2%. The most common characteristics of cardiac cephalalgia were frontal location, pressing quality and moderate intensity. Jaw pain, palpitations and circumflex coronary artery as the culprit artery for the myocardial ischemia were associated with cardiac cephalalgia.
Footnotes
Acknowledgements
The authors thank the Cardiology Service of Hospital Clínico Universitario Lozano Blesa for their support and thank all the participants of the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
