Abstract
Background
Medication overuse headache shares several characteristics with substance use disorders. However, key features of substance use disorders such as increased impulsivity and alterations in reward processing remain little explored in medication overuse headache.
Methods
Temporal discounting and impulsive decision making behavior and the associated brain mechanisms were assessed in 26 chronic migraine patients with medication overuse headache and in 28 healthy controls. Regions-of-interest analyses were first performed for task-related regions, namely the ventral striatum and the ventromedial and dorsomedial prefrontal cortices. Resting-state functional connectivity between these regions were then explored. An additional 27 chronic migraine patients without medication overuse headache were included for comparison in the latter analysis.
Results
Patients with medication overuse headache showed steeper temporal discounting behavior than healthy controls. They also showed weaker subjective value representations in the dorsomedial prefrontal cortex, when accepting larger delayed rewards, and in ventral striatum and ventromedial prefrontal cortex, when accepting the smaller immediate reward. Resting-state functional connectivity was reduced among the valuation regions when comparing patients with medication overuse headache to the other two control groups.
Conclusions
Patients with medication overuse headache were characterized by altered processing and dysconnectivity in the reward system during intertemporal choices and in the resting-state.
Keywords
Introduction
In individuals with pre-existing primary headaches, excessive intake of acute abortive medication may lead to worsening of the headache causing the transformation, for example, from episodic to chronic migraine (CM). As overuse of acute abortive medication is one of the leading risk factors for headache chronification (1), it is important to understand why some patients progress to MOH while others do not. MOH shares several characteristics with substance use disorders (SUDs). About 70% of individuals with MOH fulfill the criteria for dependence (2–4). MOH may be associated with tolerance and withdrawal symptoms and a high relapse rate (4–6). Other features that remain little explored in MOH include the increased impulsivity and alterations in reward processing in the brain, both of which, like for SUDs (7,8), could constitute risk factors for progression and relapse.
Future rewards are typically devalued as a function of temporal delay. This behavior is referred to as temporal discounting of delayed rewards. Individuals with SUDs display steeper discounting behavior, that is, a preference for smaller but immediate rewards over larger delayed rewards (9,10). The steeper discounting behavior is largely independent of the type of drug used and applies to both drug and non-drug related rewards (9,11). Steeper temporal discounting is considered an indication of increased impulsivity and may underlie maladaptive behaviors in SUDs (11,12). Due to the similarities between MOH and SUDs, it is conceivable that MOH is also associated with steeper discounting not only to drug-related rewards but also to non-drug related rewards.
Temporal discounting behavior relies on reward valuation and cognitive control, among others (13,14). The valuation system of the brain assigns subjective value to choice options. Activity in core regions within this system, i.e. the ventral striatum (vSTR) and the ventromedial prefrontal cortex (VMPFC), increases with the magnitude of the subjective reward value (15). Attenuated vSTR and VMPFC activity has been observed in steeper discounting healthy individuals (16,17). Attenuated vSTR activity has also been observed in SUDs during the anticipation of monetary rewards (18). Furthermore, damage to the medial orbitofrontal cortex/VMPFC results in steeper discounting (19). Interestingly, MOH patients exhibit a specific functional connectivity pattern of the vSTR that allows for blind discrimination between MOH and non-MOH patients (20). MOH patients also exhibit altered VMPFC activity in a task involving decision making under risk (21) and glucose hypometabolism in the medial orbitofrontal cortex (22). Together these findings suggest a central role of these regions and their associated neural circuits in MOH. Cognitive control regions, such as the dorsomedial prefrontal cortex (DMPFC), exhibit a negative relationship with subjective reward value during temporal discounting (15) and are involved in the regulation of cravings, among others (23).
In the present study, temporal discounting and impulsive decision making behavior and the associated brain mechanisms were assessed in CM with MOH. Based on findings from this analysis, resting-state functional connectivity changes between the affected regions were then explored. As MOH shares features with SUDs, we hypothesized that relative to healthy controls, these patients would exhibit steeper discounting behavior and reduced task-related responses in core regions of the valuation network, i.e. vSTR, VMPFC, and in regulatory regions, i.e. DMPFC. We, furthermore, hypothesized that resting-state functional connectivity between the affected regions would be diminished in CM with MOH. To further examine the specificity of functional connectivity changes, CM with and without MOH were also compared.
Materials and methods
Overview
The main study consisted of CM patients with MOH and healthy controls undergoing task-based and resting-state functional magnetic resonance imaging (fMRI). However, due to a scanner replacement, it was not possible to complete data collection for CM patients without MOH. To further elaborate on our results from the main study, we included previously unpublished resting-state fMRI data from CM patients without MOH albeit without task-related data but from the same scanner.
Study population
In this cross-sectional study, CM patients with and without MOH were recruited from the Headache Clinic of Taipei Veterans General Hospital. Both diagnoses were made according to the criteria proposed by the International Classification of Headache Disorders, 3rd edition (ICHD-3) (24). Medication overuse was defined as the intake of ergotamine, triptans, or combination analgesics on ≥10 days/month or simple analgesics on ≥15 days/month for ≥3 months. All patients enrolled in the study fulfilled the following additional criteria for CM: (a) Had ≥15 days/month of headache in the preceding three months. (b) ≥8 of these days had to be with migrainous headache. (c) Utilized the same types and amount of acute medication that they had used prior to enrollment throughout the study period. (d) Refrained from taking acute medications 24 hours prior to scanning. (e) Had not previously received migraine prophylaxis. Healthy controls without any history of migraine or other primary headaches were recruited. All participants were without systemic diseases, psychiatric disorders, illicit substance abuse or alcoholism, or conditions incompatible with MRI. The study was approved by the Institutional Review Board of Taipei Veterans General Hospital (VGH IRB: 2018-02-011B) and all participants gave written, informed consent, in accordance with the Helsinki Declaration (6th revision), before undergoing any study-related procedures. Data was collected from March 2018 to March 2021.
The intertemporal choice task
The intertemporal choice task was divided into a behavioral session and an MRI session performed on the same day. Each session consisted of six runs, with 60 and 48 trials per run, respectively. The purpose of the behavioral session was to familiarize the participants with the task and to determine whether the choice behavior was stable across the two sessions. The trial sequence of the task is shown in Figure 1. To prepare the participants for the upcoming offer, each trial started with a warning cue, a yellow dot, displayed on a screen for 0.5 seconds. This was then replaced by the offer consisting of an implicit choice between a fixed immediate reward (200 NT$, not shown) or a delayed larger amount (e.g. 2000 NT$, delayed 180 days, displayed on the screen). The temporal delay and amount magnitude varied across trials (delay: 1–180 days; magnitude: 210–3000 NT$; see online Supplementary materials for details). The participants were required to respond to the offer by pressing one of two buttons corresponding to either “yes” or “no”. Following the response, the offer was replaced by the display of the selected response for 0.5 seconds. “No response” was displayed if participants failed to respond within three seconds. This screen was then replaced by a white fixation cross, indicating that the participant should wait for the next trial to begin. The fixation cross was displayed for a mean duration of four seconds (interval: two to six seconds) plus the remaining time from the response window (three seconds minus reaction time). The mean inter-trial interval was eight seconds but varied between six to 10 seconds. The assignment of left and right button presses to “yes” and “no” was counterbalanced across participants within each study group. Subject payment for participation was divided into payment for the behavioral session and MRI scanning and payment according to a randomly drawn trial from the task performed inside the scanner. The first payment was made available immediately at the end of the experiment. The amount and the delay time for the payment of the extra reward was selected according to the randomly drawn trial. This was done to ensure reliability of the discounting measures and to motivate participants to follow instructions.
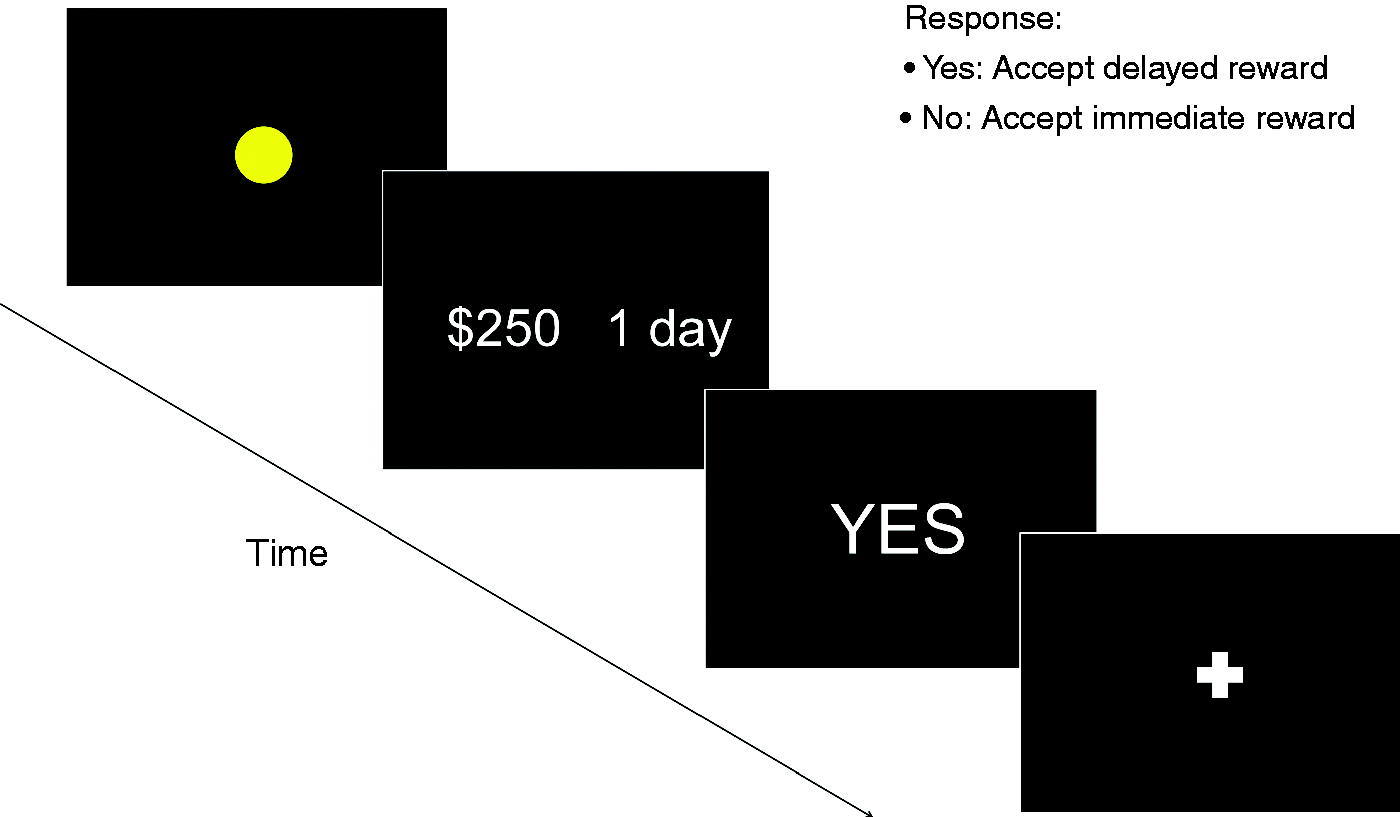
Trial sequence of the intertemporal choice task. The first screen (yellow dot) indicates that an offer is about to be made. The second screen presents the actual offer. Here the choice is between $250 received with a one-day delay or $200 received immediately (implicit choice). The third screen shows the response of the participant which is replaced by a fixation cross.
The decay in the delayed subjective value (SVdelay) over time was modeled by the following hyperbolic function: SVdelay = V/(1 + kD), where V is the amount or value offered (in NT$), D is the temporal delay (in days), and k is the estimated subjective temporal discount rate (25) (see online Supplementary materials for details). A larger k-value corresponds to a steeper discount rate and indicates an increased preference for immediate smaller rewards, i.e. increased impatience or impulsivity. Additionally, the impulsive choice ratio was calculated as the ratio of the number of rejected trials over the total number of trials (26). The k-value and impulsive choice ratio from the MRI session were used to assess temporal discounting and impulsive decision making behavior. Furthermore, for each trial in the MRI session the estimated subjective value of the chosen reward (SVdelay) was used to assess the amplitude of brain responses occurring at the time of the response to the offer, which is given as the onset time of the offer plus the reaction time.
Imaging protocol
Structural and functional imaging data were acquired on a 3T MR scanner (Trio, Siemens Medical Solutions, Erlangen, Germany) with a 32-channel head coil. To diminish motion artifacts during scanning, the participants’ heads were immobilized with cushions inside the coil. Initially, a high-resolution 3D Magnetization Prepared Rapid Acquisition Gradient Echo anatomical scan (repetition time/echo time/flip angle: 2530 ms/3.03 ms/7 degrees; field of view: 224 × 256 × 192; voxel size: 1 × 1 × 1 mm3) was acquired. This was followed by a resting-state functional scan acquired with a T2*-weighted gradient-echo Echo Planar Imaging sequence using a blood oxygenation level-dependent contrast (repetition time/echo time/flip angle: 2000 ms/30 ms/90°; matrix, 64 × 64 × 33; field of view, 220 × 220 × 132; voxel size, 3.44 × 3.44 × 3.4 mm3 with no slice gap). In total, 200 volume images were acquired. Before the resting-state scan, participants were instructed to fixate on a cross displayed on the screen inside the scanner, to refrain from moving the head during the experiment, to stay awake, and to indicate when the scan had ended by squeezing the bulb placed in their hand. The resting-state functional scan was followed by six task-related runs of the intertemporal choice task in CM patients with MOH and in healthy controls. Each run used the same acquisition parameters as the resting-state scan but with 192 images acquired per run.
Imaging data processing
All functional data were pre-processed with SPM12 (http://www.fil.ion.ucl.ac.uk/spm/software/spm12/). For each participant, the functional images were corrected for differences in slice acquisition time and then motion corrected by realigning all functional volumes to the first volume using a six-parameter rigid body transformation. These images were further co-registered to the individual anatomical image before being spatially normalized into standard space. Finally, functional images were spatially smoothed using an 8 × 8 × 8-mm3 full-width at half-maximum Gaussian kernel.
The task-related smoothed images entered into a first-level analysis in which the following events were modeled for each run: 1) onset times of accepted offers with durations equal to response times; 2) the regressor from 1) parametrically modulated (1st order/linear) by the magnitude of the estimated delayed subjective value of the offers (SVdelay from the above equation); 3) onset times of rejected offers with durations equal to response times; 4) the regressor from 3) parametrically modulated (1st order/linear) by the magnitude of the estimated delayed subjective value of the offers (SVdelay); and 5) the six head motion parameters. Serial correlations in the data were accounted for by an autoregressive AR(1) model. During model estimation, the masking value was lowered to 0.3, i.e. only voxels with a mean value larger than 30% of the global value were included. This was done to ensure that ventral striatal regions were not excluded from the analysis. Contrasts for the parametrically modulated accepted and rejected offers were then generated for all participants and analyzed separately. Initially, the main effects were examined in the full brain volume and across all participants (CM with MOH and healthy controls were combined into a single group). Clusters passing a voxel-wise threshold of P ≤ 0.005 followed by a cluster-wise threshold of P ≤ 0.05, corrected for the false discovery rate (FDR), were considered significant. To compare MOH patients with healthy controls, a regions-of-interest analysis was performed using three regions known to be involved in different aspects of SUDs and to encode the subjective value, i.e. vSTR, VMPFC and DMPFC. The three regions were defined according to masks provided by Bartra et al. (15) (see online Supplementary materials) and were based on a meta-analysis examining the neural correlates of subjective value. The MARSBAR toolbox was used to extract the individual mean effect sizes from each of the three regions. These values then entered into independent t-tests. To further examine whether headaches on the scan day influenced the between-group results, an additional linear regression analysis was performed in which the two groups entered as a binary independent variable, headache intensity on the scan day as a second independent variable and activity from the regions-of-interests as the dependent variable. For each of the analyses, between-group comparisons passing an FDR-corrected P ≤ 0.05 (three regions and two response types) were considered significant. Additionally, correlation analyses between individual effect sizes (beta values) and intake of abortive medication were performed for significant loci. Finally, whole-brain analyses were performed for between-group comparisons. Clusters passing a voxel-wise threshold of P ≤ 0.005 followed by an FDR-corrected cluster-wise threshold of P ≤ 0.05 were considered significant.
The smoothed resting-state functional images from all three groups of participants were further pre-processed using the DPARSF toolbox (v5.1; http://rfmri.org/DPARSF). Linear trends were removed and signals related to head motion parameters (Friston 24-parameter model), white matter and cerebrospinal fluid were regressed out. The resulting images were filtered with a band-pass of 0.01–0.1 Hz. A seed-to-seed correlation analysis was then performed. Seed-points were defined as spheres of 4-mm radii and included left vSTR (MNI coordinates: −10, 10, 2), right vSTR (10, 12, −4), VMPFC (−2, 42, −10), and caudal DMPFC (−4, 28, 44). The vSTR and VMPFC seeds were centered at peak activity within the masks applied to the task-based main effect analysis of accepted trials. The DMPFC seed was centered at the overlap between the negative effect of accepted trials and positive effect of rejected trials from the task-based main effect analyses as this locus may be involved in bidirectional modulation of the reward system (please refer to Supplementary materials for details regarding the coordinate extraction). Individual seed-to-seed correlation matrices were calculated and transformed to Z-scores using Fisher’s r-to-Z transformation. To compare MOH patients with healthy controls, independent t-tests were first performed for all pairs of seed-points (six inter-regional correlations). To further examine the influence of headache intensity on the scan day, linear regression analyses were also performed in which the two groups entered as a binary independent variable, headache intensity on the scan day as a second independent variable and pair-wise connectivity between regions as the dependent variable. Finally, for comparison between CM patients with and without MOH while accounting for differences in CM duration, linear regression analyses were performed in which the two groups entered as a binary independent variable, CM duration as a second independent variable and pair-wise connectivity between regions as the dependent variable. For each of these analyses, between-group tests passing an FDR-corrected P ≤ 0.05 (six inter-regional correlations per analysis) were considered significant. As the VMPFC emerged as a central hub, a seed-to voxel analysis was performed in SPM in which whole-brain Z-transformed maps were generated for the VMPFC seed and entered into independent t-tests for pair-wise comparison of MOH patients with the two other groups. CM duration was used as a covariate of no interest for the patient groups. Clusters passing a voxel-wise threshold of P ≤ 0.005 followed by an FDR-corrected cluster-wise threshold of P ≤ 0.05 were considered significant. Finally, associations between VMPFC connectivity and monthly intake of abortive medication were examine in the combined patient group and associations between VMPFC connectivity and the log-transformed subjective discount rate was examined in a group combining MOH patients and healthy controls. Loci from all whole-brain analyses were labeled using the Neuromorphometrics atlas under SPM12 and, additionally, the Automated Anatomical Labeling atlas 3 (27) for subregions within the thalamus.
Statistical analysis
Headache parameters obtained in both patient groups included intake frequency of acute pain medication (days per month), headache frequency (days per month), headache and chronic headache duration (years), headache intensity on the scan day, as well as mean and maximum headache severity (0–10 scale). Furthermore, smoking and alcohol consumption status and depression levels (Beck Depression Inventory; score range: 0–63) were assessed in all participants and the Leeds Dependence Questionnaire (score range: 0–30) was administered in all MOH patients and healthy controls. Demographic and behavioral data as well as region-of-interest data were analyzed using IBM SPSS (version 21.0). All data series were first checked for outliers using Grubb’s test. As the subjective discount factor k was not normally distributed, either non-parametric tests or a log10 transform were used. One-way ANOVAs, independent t-tests or chi-square tests were used for demographic data where appropriate. For demographic and behavioral data P < 0.05 (two-tailed) was considered significant.
Results
Demographics and behavior
Of the 81 participants included in the study, 26 had CM with MOH, 27 had CM without MOH and 28 were healthy controls. Demographics and clinical characteristics of the three groups are provided in Table 1. The three groups did not differ with respect to age, sex, depression scores, or alcohol and smoking habits. The two patient groups did not differ with respect to headache frequency, headache history, maximum and mean headache severity in the last year, or the presence of headache on the scan day. Furthermore, MOH patients were found to have moderate levels of substance dependence as assessed by the Leeds Dependence Questionnaire. As expected, MOH patients had a significantly higher intake of acute abortive medications than patients without MOH. The type of acute medication used included acetaminophen/caffeine, nonsteroidal anti-inflammatory drugs and ergotamine. None of the patients used narcotics. Finally, the two patient groups also differed with respect to the duration of CM.
Demographics of patients and healthy controls (mean ± SD).

Alcohol (none/infrequent/frequent) = alcohol consumption level, infrequent = less than once per week, frequent = more than once per week; Mean headache severity refers to the mean headache intensity over the last year. P-values in bold denotes a significant difference, i.e. P ≤ 0.05.
BDI, Beck Depression Inventory; CM, chronic migraine; HC, healthy controls; LDQ, Leeds Dependence Questionnaire; na, not applicable/not available.
*One-way ANOVA or independent t-test, when appropriate.
#Chi-square test.
For the intertemporal choice task, one MOH patient was excluded from further analysis due to an unreasonably short reaction time (0.16 sec or 3.7 standard deviations shorter than the mean) and inconsistent behavior between the two sessions, i.e. a shift from steep to no discounting behavior, suggesting improper execution of the task. For the remaining participants, the choice behavior was stable across the behavioral and fMRI sessions (r = 0.838). In the fMRI session, MOH patients did not differ from healthy controls with respect to reaction time across all trials or when accepted and rejected offers were considered separately (Table 2). The error rate did also not differ between the two groups. However, the impulsive choice ratio and the temporal discount rate were significantly higher in MOH patients suggesting increased impatience or impulsive choice behavior in this group. No associations were found between the log-transformed subjective discount rate and any of the headache parameters, i.e. intake frequency of acute medication, headache frequency, headache duration, chronic migraine duration, headache intensity on the scan day, or mean and maximum headache severity.
Task-related behavioral parameters.

Independent t-test were performed except for P-values denoted by * for which the Mann-Whitney U Test was used. Impulsive Choice Ratio = (# rejected trials)/(# total trials). k is the temporal discount rate. A higher k-value indicates steeper discounting or increased impatience/impulsivity. P-values in bold denotes a significant difference, i.e. P ≤ 0.05.CM with MOH, chronic migraine with medication overuse headache; fMRI, functional magnetic resonance imaging; HC, healthy controls, N, number of subjects.
Task-related imaging results
Five healthy controls and one MOH patient had excessive head movement during three or more runs and were excluded from further task-related imaging analysis. The main effect of temporal discounting (accepted offers) revealed a positive parametric effect of the estimated delayed subjective value in regions encompassing the VMPFC and bilateral vSTR as well as the posterior cingulate/precuneus among others (Figure 2A, online Supplementary Table 1). The medial superior frontal gyrus (caudal DMPFC) exhibited a negative parametric effect of the estimated delayed subjective value during temporal discounting. During impulsive decision making (rejected offers), a positive parametric effect was observed in the medial superior frontal gyrus (DMPFC), bilateral anterior insula, bilateral middle frontal and temporal gyri, and bilateral angular gyri while a negative effect was observed in the bilateral occipital and superior parietal lobes (Figure 2B, online Supplementary Table 1).

Regions in which the activation amplitude was parametrically modulated by the estimated subjective value. (a) The main effect of accepted offers included the bilateral ventral striatum (vSTR) and the ventromedial prefrontal cortex (VMPFC) for positive effects and the dorsomedial prefrontal cortex for negative effects and (b) The main effect of rejected offers included the DMPFC for positive effects, among others. Note that healthy controls and chronic migraine patients with medication overuse headache were combined in this analysis. Coordinates are provided in MNI space. Blue, negative effects. Red, positive effects.
Between-group differences in the parametric effect of the estimated delayed subjective value were examined in the vSTR, VMPFC and DMPFC. Using Grubb’s test, a single outlier was detected and removed from each of the data series from healthy controls in the vSTR and VMPFC for rejected offers and from MOH patients in the VMPFC for accepted and rejected offers as well as in the DMPFC for accepted offers. In MOH patients relative to healthy controls, significant reductions in activity (de-activations) were found in the DMPFC (independent t-test: P = 0.024, PFDR = 0.049) for accepted offers and in the vSTR (independent t-test: P < 0.001, PFDR = 0.005) and VMPFC (independent t-test: P = 0.006, PFDR = 0.019) for rejected offers (Figure 3). In the between-group regression analysis accounting for the intensity of the headache on the scan day, a reduction in activity was only found in the vSTR (P = 0.007, PFDR = 0.042) for rejected offers. No associations were found between activity in any of the significant loci and intake frequency of acute abortive medication.

Between-group regions of interest analysis for accepted and rejected offers. Significantly reduced responses were found in patients with medication overuse headache (MOH) relative to healthy controls (HC) for rejected offers in (a) the ventral striatum (vSTR) and (b) the ventromedial prefrontal cortex, and for accepted offers in (c) the dorsomedial prefrontal cortex. The three predefined regions were based on masks generated in the meta-review of Bartra et al. (15). *, P < 0.05 multiple comparison corrected using the false discovery rate (FDR). Error bars denotes standard deviations.
In the whole-brain analysis, significant differences between the two groups were not found for accepted offers. However, for rejected offers MOH patients had decreased activity relative to healthy controls in a cluster covering the left and right dorsal and ventral striatum and the thalamus (including the ventral anterior nucleus and the ventral lateral nucleus) (PFDR < 0.001, cluster extent = 1602 voxels) as well as in a cluster covering the precuneus and the left superior parietal lobe (PFDR = 0.021, cluster extent = 803 voxels) (see online Supplementary Figures 1A and 1B).
Resting-state functional connectivity
Resting-state functional connectivity analyses were performed for seed-points located in the left and right vSTR, the VMPFC and the DMPFC. The corresponding whole-brain seed-maps for healthy controls are shown in Supplementary Figure 2. When comparing MOH patients and healthy controls, decreased connectivity was found between the left and right vSTR (P = 0.001, PFDR = 0.006), the left vSTR and VMPFC (P = 0.002, PFDR = 0.008), the right vSTR and VMPFC (P = 0.006, PFDR = 0.018), and VMPFC and DMPFC (P = 0.011, PFDR = 0.026) (Figure 4). When accounting for headache intensity on the scan day, the decreased connectivity between the left and right vSTR (P = 0.008, PFDR = 0.024), the left vSTR and VMPFC (P = 0.016, PFDR = 0.032), and VMPFC and DMPFC (P = 0.007, PFDR = 0.042) remained significant while the connectivity between the right vSTR and VMPFC (P = 0.068, PFDR = 0.102) did not. Comparing patients with and without MOH, while accounting for differences in CM duration, decreased connectivity was found between the left and right vSTR (P = 0.001, PFDR = 0.006), the left vSTR and VMPFC (P = 0.016, PFDR = 0.048) and the right vSTR and VMPFC (P = 0.020, PFDR = 0.040) (Figure 4).
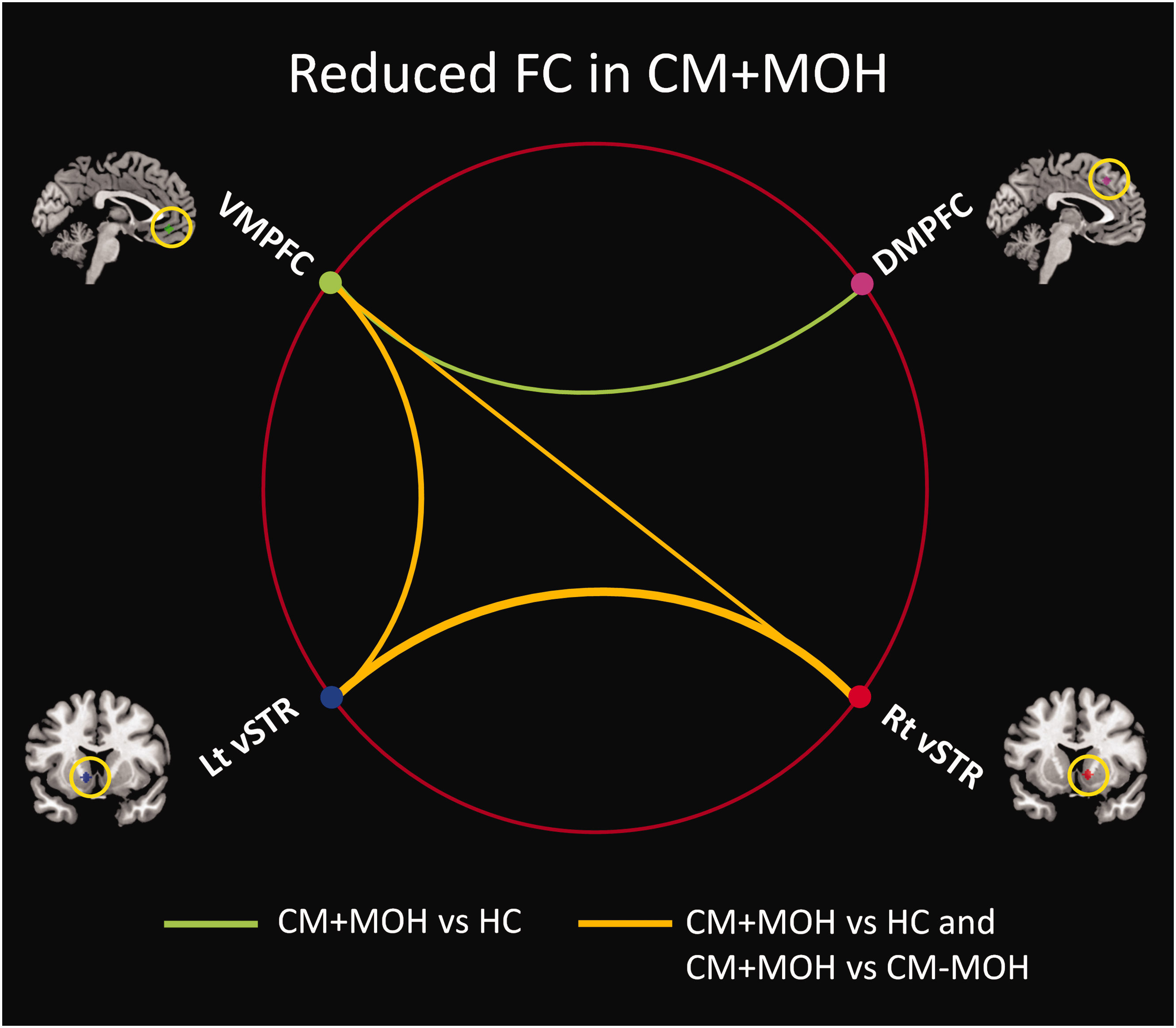
Resting-state functional connectivity between four regions of interest. Reduced functional connectivity (FC) was found in chronic migraine patients with medication overuse headache (CM + MOH) relative to healthy controls (HC) and chronic migraine patients without medication overuse headache (CM-MOH) between left (Lt) and right (Rt) ventral striatum (vSTR), between the ventromedial prefrontal cortex (VMPFC) and both Lt and Rt vSTR. Reduced connectivity was also found in CM + MOH relative to HC between the VMPFC and the dorsomedial prefrontal cortex (DMPFC).
As MOH patients had reduced functional connectivity between the VMPFC and vSTR and between VMPFC and DMPFC, we further examined the whole-brain network of the VMPFC seed-point using a seed-to-voxel analysis (Figure 5A, online Supplementary Table 2). Comparing MOH patients with healthy controls, reduced connectivity of the VMPFC was observed to vSTR and DMPFC, as expected, and to the thalamus, covering the ventral anterior nucleus and the ventral lateral nucleus, as well as to the bilateral ventral precentral and middle frontal gyri, among others. Reduced connectivity was observed to many of the same regions when comparing the two patient groups (Figure 5A, online Supplementary Table 2). Across the entire patient population, VMPFC connectivity to regions constituting the default mode network were positively associated with monthly intake of acute abortive medication. VMPFC connectivity to the bilateral vSTR as well as regions within the dorsal attention network were negatively associated with monthly intake of acute abortive medication (Figure 5B, online Supplementary Table 3). When considering MOH patients and healthy controls together, a negative association with the log-transformed subjective discount rate was found with connectivity to the DMPFC and several other regions engaged during rejected offers and within the DMPFC resting-state network (Figure 5C, online Supplementary Table 3, compare with Figure 2B and online Supplementary Figure 1).
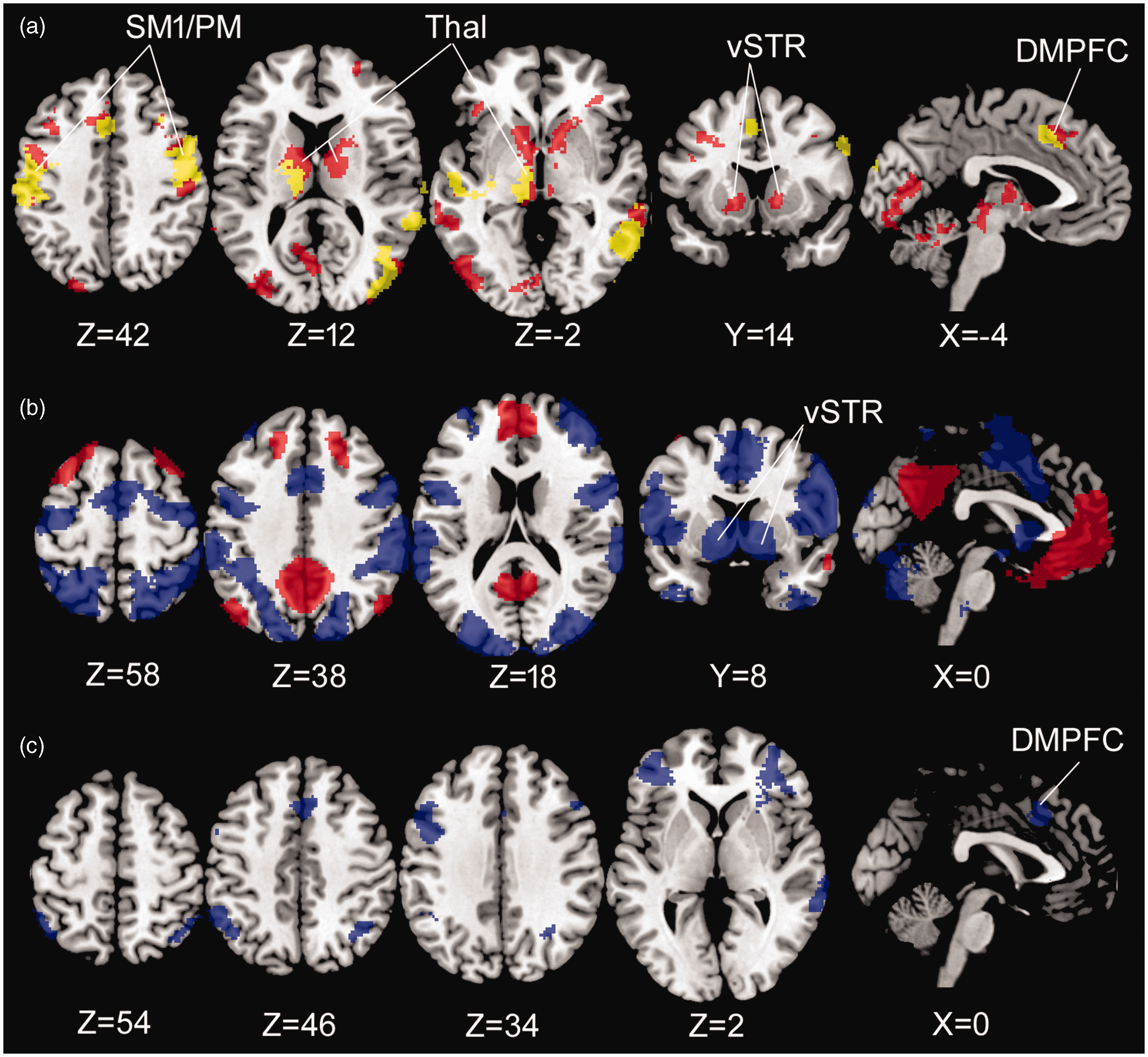
Whole-brain functional connectivity of the VMPFC. (a) Reduced connectivity in patients with medication overuse headache relative to healthy controls (red) and relative to patients without medication overuse headache (yellow). (b) Regions exhibiting positive (red) and negative (blue) associations with the monthly intake of acute abortive medication when combining patients with and without medication overuse headache. (c) Regions exhibiting negative associations with the log-transformed subjective discount rate when combining patients with medication overuse headache and healthy controls. DMPFC, dorsomedial prefrontal cortex; PM, premotor cortex; SM1, primary sensorimotor cortex; Thal, thalamus; vSTR, ventral striatum.
Discussion
This study addressed whether temporal discounting and impulsive decision making behavior and the associated brain mechanisms were altered in CM patients with MOH. Based on this, we further addressed whether alterations in resting-state functional connectivity was present within the involved brain networks in these patients. We first showed that MOH patients had an elevated subjective discount rate and impulsive choice ratio. We then showed that the encoding of the subjective value was reduced (increased deactivation) in the DMPFC during temporal discounting and in the vSTR and VMPFC during impulsive decision making in MOH patients. We further showed that resting-state functional connectivity in MOH patients was reduced among these regions and that the connectivity between the VMPFC and DMPFC was inversely related to the subjective discount rate and the connectivity between VMPFC and the bilateral vSTR was inversely related to the monthly intake of acute abortive medication.
Decision making behavior
In accord with our hypothesis, the temporal discount rate and the impulsive choice ratio were significantly increased in MOH patients relative to healthy controls. Increases in these measures are considered to reflect increased impulsivity/impatience (28,29). Previous studies, mainly using questionnaires for assessment, found no difference in impulsivity of MOH patients (30–32). However, self-report measures and task measures have been suggested to assess different aspects of impulsive behavior (29,33). For example, the intertemporal choice task may be less sensitive to biased self-perceptions but are limited to interpretations in the context of decision making (29). Nevertheless, as no relationship was found between the task-based measures of impulsivity and any of the clinical parameters, the associated behavior is likely not directly involved in disease progression. This is reasonable as impulsivity and poor self-control in context of decision making involves numerous processes not assessed by the task. It is noteworthy that, as we did not obtain information about the dosage or number of pills consumed per day of use, we cannot rule out that such measures could be associated with our task-related measures.
Parametric encoding of subjective value
The subjective value representation during temporal discounting (accepted offers) included the most commonly observed brain regions involved in valuation, i.e. vSTR, VMPFC and posterior cingulate/precuneus for positive effects and DMPFC for negative effects (15). We further showed that impulsive decision making (rejected offers) involved regions subserving executive function and salience detection (34,35), including the DMPFC, for positive effects. Such regions are often co-activated during cognitive tasks (35). Although we had hypothesized that MOH patients would exhibit reduced task-related responses in the vSTR, VMPFC, and in the DMPFC, we observed that this was only the case when the regions were not involved in the positive main effects analyses for accepted or rejected trials, respectively. This suggests that the two types of behavior were associated with a dysfunctional suppression of competing brain regions in patients with MOH during performance of the decision making task. Such a dysfunction may in part explain the altered assignment of subjective value and the steeper discounting behavior. Our results further suggest that the encoding of subjective value in the VMPFC and DMPFC was state-dependent as the difference in responses in these regions became non-significant when accounting for headache intensity. This is congruent with the VMPFC alterations during decision-making under risk being reversible when comparing non-detoxified with detoxified MOH patients having reduced headache intensity and frequency (21). Finally, for impulsive decision making, the whole-brain analysis further revealed increased deactivation in the thalamus, including the ventral anterior and ventral lateral nuclei. These thalamic subregions receive indirect input from vSTR and project to the VMPFC and are considered an integral part of a cortico-striatal-thalamic loop circuit subserving reward processing (36,37).
Resting-state functional connectivity
In the regions-of-interest analysis, reduced resting-state functional connectivity was found between the VMPFC and the bilateral vSTR and between the left and right vSTR when comparing MOH patients with either healthy controls or patients without MOH. The former comparison remained largely the same when controlling for the headache intensity. For the VMPFC, the seed-to-voxel analysis in the entire brain volume furthermore revealed reduced connectivity to the thalamus and caudal DMPFC when comparing MOH patients with either of the other two groups. This is in accord with the VMPFC receiving input from the thalamus and sending output to the vSTR (36,37). Furthermore, the VMPFC and the DMPFC have been found to be extensively interconnected in monkeys (38,39). Together these findings suggest the presence of functional dysconnectivity in the reward-related cortico-striatal-thalamic loop circuit that is specific to MOH patients. This dysconnectivity could either be in response to or lead to the increased medication intake as VMPFC connectivity to vSTR was progressively reduced with the severity of medication overuse, when considering both patient groups together. Interestingly, hypoconnectivity between VMPFC and vSTR has been linked to the risk of developing SUDs (40). Altered top-down modulation may also have contributed to the functional dysconnectivity of the reward circuit as VMPFC-DMPFC connectivity was reduced. The connectivity between the VMPFC and the caudal DMPFC was found to be inversely related to the temporal discount rate when considering MOH patients and healthy controls together. Steeper discounting behavior may thus be associated with reduced top-down influence from the DMPFC to the VMPFC. This is in agreement with the involvement of the DMPFC in executive functions such as cognitive regulation and response selection (35,41). Taken together, hypoconnectivity between the VMPFC and both vSTR and DMPFC may represent an imbalance between top-down cognitive control and bottom-up reward processes perhaps resulting in reduced resilience against medication overuse and a predisposition to relapse.
Limitations
The current study has limitations. First, we did not obtain task-related data in CM patients without MOH. Consequently, we were not able to show whether the steeper discounting behavior and task-related imaging results were specific to CM with MOH. Nevertheless, our study still provides the context for the MOH specific resting-state functional connectivity changes in the reward circuit. Furthermore, as we did not obtain alternative measures of impulsivity in our study, our behavioral results are limited to the interpretation within the context of intertemporal choices. Further studies of other aspects of impulsivity in patients with MOH are warranted.
Article highlights
CM with MOH is associated with a dysregulated reward system. Both intake frequency of acute abortive medication and impulsive choice behavior have important roles in this dysregulation. VMPFC-based mechanisms are central to this dysfunction. VMPFC dysconnectivity in the resting-state may have consequences for encoding of subjective value during specific decision-making behaviors.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231158088 - Supplemental material for An altered reward system characterizes chronic migraine with medication overuse headache
Supplemental material, sj-pdf-1-cep-10.1177_03331024231158088 for An altered reward system characterizes chronic migraine with medication overuse headache by David M Niddam, Shih-Wei Wu, Kuan-Lin Lai, Yun-Yen Yang, Yen-Fang Wang and Shuu-Jiun Wang in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DMN, SWU, KLL, YYY, and YFW report no disclosures. SJW has served on the advisory boards of Allergan, and Eli Lilly Taiwan. He has received speaking honoraria from the Taiwan branches of Pfizer, Elli Lilly and GSK and has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital, and Taiwan Headache Society.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Science and Technology (107-2314-B-010 -018 -MY3; 111-2314-B-A49-075-MY2), the Brain Research Center, National Yang Ming Chiao Tung University, and from the Ministry of Education, Aim for the Top University Plan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
