Abstract
Background
We aimed to assess the differences in quantitative sensory testing between chronic migraine and healthy controls and to explore the association between pain sensitivities and outcomes in chronic migraine following preventive treatment.
Methods
In this prospective open-label study, preventive-naïve chronic migraine and healthy controls were recruited, and cold, heat, mechanical punctate, and pressure pain thresholds over the dermatomes of first branch of trigeminal nerve and first thoracic nerve were measured by quantitative sensory testing at baseline. Chronic migraines were treated with flunarizine and treatment response was defined as ≥50% reduction in the number of monthly headache days over the 12-week treatment period.
Results
Eighty-four chronic migraines and fifty age-and-sex-matched healthy controls were included in the analysis. The chronic migraine had higher cold pain thresholds over the dermatomes of the first branch of trigeminal nerve and the first thoracic nerve (p < 0.001 and < 0.001), lower pressure pain thresholds over the dermatomes of the first thoracic nerve (p = 0.003), heat pain thresholds over the dermatomes of the first branch of the trigeminal nerve and the first thoracic nerve (p < 0.001 and p = 0.015) than healthy controls. After treatment, 24/84 chronic migraine had treatment response. The responders with relatively normal pain sensitivity had higher heat pain thresholds over the dermatome of the first branch of the trigeminal nerve (p = 0.002), mechanical punctate pain thresholds over the dermatomes of the first branch of the trigeminal nerve (p = 0.023), and pressure pain thresholds over the dermatomes of the first branch of the trigeminal nerve (p = 0.026) than the hypersensitive non-responders. Decision tree analysis showed that patients with mechanical punctate pain threshold over the dermatomes of the first branch of the trigeminal nerve > 158 g (p = 0.020) or heat pain threshold over the dermatome of the first branch of the trigeminal nerve > 44.9°C (p = 0.002) were more likely to be responders.
Conclusions
Chronic migraine were generally more sensitive compared to healthy controls. Preventive treatment with flunarizine should be recommended particularly for chronic migraine who have relatively normal sensitivity to mechanical punctate or heat pain.
Trial registration: This study was registered on ClinicalTrials.gov (Identifier: NCT02747940).
Keywords
Introduction
Patients with migraine who have ≥15 days with headache per month for ≥3 months, of which ≥8 days/month with migrainous features or respond to migraine-specific treatment, are diagnosed with chronic migraine (CM) (1,2). The treatment of CM is quite challenging, and it is difficult to predict the treatment outcomes. Identification of predictors of treatment outcomes is of clinical significance and importance. Factors reported to be associated with poor treatment outcomes include comorbid depression (3), medication overuse (3), longer disease durations of CM (4) or migraine (5), chronic headache (6–8), and signs of central sensitisation (i.e., increased pain sensitivity or allodynia) (9–13). However, most of these studies determined the presence or severity of pain hypersensitivity by questionnaires (6–8,11) rather than objective measurements. The quantitative sensory testing (QST) overcomes these problems by precisely controlled stimuli. A standardized protocol for the QST, including subject and investigator instructions and multi-centre reference data, is available from the German Research Network on Neuropathic Pain (DFNS); this test is currently one of the most commonly used methods to assess sensory thresholds (14–16). Previous studies, including ours, have shown that patients with migraine demonstrated altered pain thresholds compared to healthy controls (HCs), including cold-induced pain (17), heat-induced pain (18–21), and mechanical pain (17,22–24), especially in patients with CM. Whether the QST results before preventive treatment can predict the outcomes in patients with migraine deserves further investigations.
Flunarizine is recommended as the first-line prophylactic for migraine by the European Federation of Neurologic Societies and the Italian Guidelines for Primary Headache. It is also one of the most commonly prescribed preventive medications in Taiwan. On the other hand, topiramate is considered as the first-line preventive medication by the European Headache Federation, the European Academy of Neurology, and the American Headache Society guidelines. Of note, based on our previous study, flunarizine demonstrated a comparable treatment efficacy for CM but was much less likely to cause paresthesia as topiramate. It is estimated that paresthesia can develop in half of the patients on topiramate (25), which could be a problem for study measuring pain sensitivity.
Hence, we aimed to explore the association between pretreatment QST results and prognosis following preventive treatment in patients with CM. We also compared the differences in pain sensitivity between CM and HC. We hypothesized that 1) CM patients were more sensitive than HC and 2) CM patients who are more sensitive to pain stimuli before intervention would be more refractory to preventive treatment. Flunarizine was chosen as the preventive agent because our prior study showed that it was as effective as topiramate and, unlike topiramate, did not cause paresthesia.
Methods
Participants
This prospective study was part of an integrated project of multimodal decoding of CM that recruited both CM patients and HCs. The baseline assessments, including the QST, electroencephalography, magnetic resonance imaging, heart rate variability, and biochemical assessments, were performed in order to explore the potential treatment outcome predictors. In this paper, only the results regarding the QST are reported. Newly diagnosed preventive-naïve CM patients were recruited from the headache clinic of a tertiary medical centre from May 2016 to December 2019. The diagnosis of CM was based on the criteria of the International Classification of Headache Disorders, third edition, beta version (ICHD-3β) (26). The demographics and clinical profiles of the participants were collected by self-administered questionnaires, which were later verified during face-to-face interviews by headache specialists. The CM patients had to fulfill the following inclusion criteria to be recruited: a) willing to participate in the study, b) between 20 and 60 years old, c) diagnosed with CM, and d) occurrence of migraine before the age of 50 years old. The exclusion criteria for the CM patients included: a) acute headache with a history of <1 month, b) secondary headache disorder except for medication overuse headache, which was allowed in this study, and c) history of migraine preventive medication use before enrollment. HCs, matched by age and sex, were enrolled from the nearby neighborhood and university if they did not have: a) ≥1 headache episode/month, b) history of moderate or severe headaches, c) history of myofascial pain syndrome involving at least two body parts, or d) history of migraine or fibromyalgia. Both patients with CM and HCs were excluded if they had: a) history of major systemic illness, such as uncontrolled hypertension, diabetes, chronic kidney disease, autoimmune disease, or malignancy, b) history of neurological disorders that may alter sensation, such as stroke or peripheral neuropathy, c) history of psychiatric illness, for example anxiety disorders and major depression or regular use of psychotropic agents, such as antidepressants, antipsychotics, or sedatives, d) daily consumption of >20 cigarettes, e) pregnancy or lactation, f) any obvious infection or inflammation over a period of at least one month before enrollment, and g) any localized inflammatory process or other active pathologies involving the forehead or the arm where QST was to be administered. The sample size was based on a previous study that revealed significant heat pain threshold (HPT) differences between CM and HC with 20 subjects in each group (19). The responder rate and drop-out rate were estimated at ∼30% each. Therefore, the total sample for CM group was estimated to be at least 96 subjects to reach statistical significance.
The study protocol was approved by the Institutional Review Board at the Taipei Veterans General Hospital (IRB-TPEVGH No. 2015-11-002B). All participants provided written informed consent prior to participation. The study procedures followed the Declaration of Helsinki. Data from this study are available from the corresponding author upon reasonable request.
Clinical assessment
The specifically designed questionnaires included several instruments to measure psychological disturbances, stress, and functional disabilities related to headache attacks. The Hospital Anxiety and Depression Scale (HADS) (27) was used to evaluate the anxiety and depression levels of the subjects. The Perceived Stress Scale (PSS) (28) assessed the life stress of the subjects. The Migraine Disability Assessment (MIDAS) (29) evaluated the disability caused by migraine for the CM patients. The HCs completed all of the baseline assessments with the same procedures and questionnaires except for the headache diary and headache-related questionnaires, as those with a history of headaches were excluded from the study.
Patient treatment
Eligible CM patients were asked to keep a headache diary after enrollment to document their baseline headache profiles, including headache frequency, medication use, headache intensity (11-point numeric rating scale), and headache duration (hr/day) for 4 weeks, which constituted the prospective baseline period. Only acute abortive medications were allowed during the 4-week baseline period. A headache day was defined as a calendar day with a headache lasting for >4 h or a calendar day with acute medication use for a headache. All patients were treated with flunarizine (5 or 10 mg/day), as prescribed by the patient’s neurologist for migraine prophylaxis. The use of acute medications, such as simple analgesics, nonsteroidal anti-inflammatory drugs, triptans, and ergots, was permitted during the study period and was recorded in the headache diary. These patients were followed up regularly in our outpatient department for 12 weeks and were asked to keep their headache diary during this study period.
QST
The QST was performed one day during the baseline period and was rescheduled if acute medications had been used for any reason within 48 hours before the assessment. The assessor recorded the headache status of the subject right before the QST assessment. The participants lay comfortably in the supine position during the assessment. The left supraorbital (i.e., the first branch of the trigeminal nerve dermatome, V1) and medio-ventral forearm (i.e., the first thoracic nerve dermatome, T1) areas were targeted since no side-to-side differences were found in previous studies (30,31) and in our pilot study. There were breaks lasting >5s between successive stimuli. Most stimulus parameters were copied from the DFNS protocol (14), except that all stimuli were given as ramps (method of limits) in order to make them more comparable to each other. The testing and analytic methods of HPT, cold pain threshold (CPT), mechanical punctate pain threshold (MPT), and pressure pain threshold (PPT) were the same as our previous study (24).
Outcome measures
Patients who provided at least one post-treatment outcome (at least 4 weeks after treatment began) were included in the analysis. The change from baseline in the number of monthly headache days (MHDs) across the 12-week treatment period for each patient was calculated. Treatment response was defined as a ≥50% reduction in the number of MHDs following preventive treatment. The subjects included in the analyses had to complete the diary for at least 14 days every four weeks during the study period. The MHD was calculated based on the percentage of headache days of the documented days. The last observation carried forward (LOCF) method was used for those missing diary information for weeks 5–12 after the initiation of treatment.
Statistical analyses
The data were presented as means ± standard deviations or numbers (percentages). All statistical analyses were performed using SPSS version 22.0 (IBM, Armonk, NY, USA). Independent t-tests, or Mann–Whitney U tests for non-normally distributed variables, were used to compare the differences between the HCs and CM patients or between the responders and non-responders. One-way analysis of variance (ANOVA) tests with post-hoc least significant difference (LSD) test was used for the comparison between HC, responders, and non-responders. Categorical variables were compared using the chi-square test. Paired-sample t tests, or signed tests for non-normally distributed variables, were used to compare the QST results and the change of headache days within groups. The chi-squared automatic interaction detection (CHAID) decision tree was applied to the significant variables with ≥10 cases/parent node and ≥2 cases/child node. The maximum tree depth was automatically limited at 3 for the CHAID methods. CHAID was published by Kass (32) in 1980 and is utilized for prediction, classification, and recognition of interconnection among variables. It encompasses built‐in significance testing with the consequence of using the most significant predictors instead of the most explanatory ones. Due to its usefulness, CHAID has been commonly adopted in studies aiming to identify predictors. Cohen’s d or Cohen’s f was used to denote the effect size of the comparisons, and the z scores of the QST parameters of the CM patients, based on the results of the HCs, were calculated. All tests were two-tailed, and the significance level was set at p < 0.05.
Results
Study participants and baseline characteristics
We included 114 preventive-naïve CM patients, and 108 of them completed the baseline diary and assessment. Among these 108 CM patients (11 males, 97 females, mean age: 38.6 ± 11.3 years old) who had completed the baseline diary and received flunarizine (mean dose: 6.6 ± 2.4 mg/day) for migraine prevention, 84 (6 males, 78 females, mean age: 38.3 ± 11.5 years old) had at least one post-treatment outcome assessment and completed their headache diary during the first four weeks after the initiation of treatment. Thus, 84 CM patients were included in the outcome analysis, and 24 CM patients (drop-out rate = 22%) were excluded from the analysis due to incomplete post-treatment information. In total, the MHD of nine subjects were carried forward based on the MHD of weeks 1–4, and those of another seven subjects were carried forward based on the MHD of weeks 5–8. Sixty-eight of the CM patients completed their headache diary for 12 weeks. In addition, 50 HCs (8 males, 42 females, mean age: 37.4 ± 9.2 years old) completed the QST assessments and the questionnaires (Figure 1).

The flow chart of the study.
The demographics were comparable between the CM patients (n = 84) and the HCs (n = 50). As typical for chronic pain, the CM patients had more symptoms of anxiety (HADS-A: 8.3 ± 3.9 vs. 3.9 ± 3.4, p < 0.001, Cohen’s d = 1.20), depression (HADS-D: 6.4 ± 3.4 vs. 2.7 ± 2.9, p < 0.001, Cohen’s d = 1.17), and higher levels of perceived stress (PSS: 26.2 ± 8.1 vs. 21.2 ± 9.1, p = 0.001, Cohen’s d = 0.58) (Table 1); however, none fulfilled the criteria of anxiety disorder or major depression. Upon sensory testing, the CM patients had higher V1 and T1 CPTs (V1: 21.0 ± 7.6 vs. 15.2 ± 7.9°C, p < 0.001, Cohen’s d = 0.75; T1: 20.4 ± 8.2 vs. 13.4 ± 9.8°C, p < 0.001, Cohen’s d = 0.77), lower V1 and T1 HPTs (40.7 ± 3.8 vs. 43.8 ± 3.4, p < 0.001, Cohen’s d = 0.86; T1: 40.5 ± 3.7 vs. 42.2 ± 3.8°C, p = 0.015, Cohen’s d = 0.45), and lower T1 PPTs (238 ± 75 vs. 284 ± 97 kPa, p = 0.003, Cohen’s d = 0.53) compared with the HCs. (Supplementary Table 1)
Demographic data and headache profiles of patients with chronic migraine and healthy controls.

*p < 0.05.
†calculated with the Mann–Whitney U test.
aDisease duration stands for the years since migraine onset.
bHeadache intensity assessed with 11-point numeric rating scale.
CM: chronic migraine; HC: healthy control; BMI: body mass index; HADS-A: Hospital Anxiety and Depression Scale-Anxiety Scale; HADS-D: Hospital Anxiety and Depression Scale-Depression Scale; hr: hours; MHD: monthly headache days; MIDAS: Migraine Disability Assessment; MOH: medication overuse headache; NA: not applicable; PSS: Perceived Stress Scale; QST: quantitative sensory testing; yrs: years.
Adverse events
Throughout the study period, no subject developed any severe adverse events. Thirty-one out of 84 (37%) subjects reported at least one of the following adverse events. The most common adverse event was somnolence or fatigue (23%), followed by body weight gain (10%), and dizziness (2%). Mood change, constipation, skin rash, and dry mouth were each reported by one subject. The incidence of adverse events was similar to our previous study using flunarizine (33).
Treatment outcome in CM patients
After treatment with flunarizine, 24 CM patients (29%) had a ≥50% reduction in the number of MHDs across the 12-week treatment period and were categorized as responders. The baseline demographics and clinical profiles were comparable between the responders and non-responders, including the proportion of the presence of migraine during QST assessment (Table 2). Both the responders (20.9 ± 6.3 vs. 7.2 ± 3.4 days, p < 0.001, Cohen’s d = 2.71) and non-responders (22.0 ± 5.9 vs. 17.4 ± 7.2 days, p < 0.001, Cohen’s d = 0.70) had fewer MHDs across the 12-week treatment period. Moreover, significantly fewer MHD were found in the responders compared to the non-responders at the end of the first (9.8 ± 5.9 vs. 19.8 ± 7.3 days, p < 0.001, Cohen’s d = 1.51), second (6.3 ± 3.1 vs. 17.0 ± 7.6 days, p < 0.001, Cohen’s d = 1.84), and third (5.4 ± 3.1 vs. 15.2 ± 8.4 days, p < 0.001, Cohen’s d = 1.55) month of treatment.
Demographic data and headache profiles of responders and non-responders.

†calculated with the Mann–Whitney U test.
aDisease duration stands for the years since migraine onset.
bHeadache intensity assessed with 11-point numeric rating scale.
HADS-A: Hospital Anxiety and Depression Scale-Anxiety Scale; HADS-D: Hospital Anxiety and Depression Scale-Depression Scale; hr: hour; MHD: monthly headache days; MIDAS: Migraine Disability Assessment; PSS: Perceived Stress Scale; MOH: medication overuse headache; nR: non-responders; QST: quantitative sensory testing; R: responders; yrs: years.
Pain thresholds as outcome predictors
QST findings in responders did not differ from HC, while non-responders exhibited significant pain hypersensitivity compared to HC in all parameters except for T1 MPT (Table 3). Compared with the non-responders, the responders had higher V1 HPTs (42.7 ± 4.1 vs. 39.9 ± 3.5°C, post hoc p = 0.002, Cohen’s f = 0.52), V1 MPTs (123 ± 55 vs. 96 ± 34 g, post hoc p = 0.027, Cohen’s f = 1.52), and V1 PPT (182 ± 64 vs. 150 ± 59 kPa, post hoc p = 0.026, Cohen’s f = 1.47) at baseline, and there was a nonsignificant trend toward higher T1 HPTs (41.7 ± 3.6 vs. 40.0 ± 3.7°C, post hoc p = 0.066, Cohen’s f = 0.32) and T1 PPT (265 ± 68 vs. 228 ± 75 kPa, post hoc p = 0.035, Cohen’s f = 1.48) (Table 3).
Quantitative sensory testing results of the responders, non-responders, and healthy controls.

*p < 0.05.
†not normally distributed, calculated with log transformed data.
CPT: cold pain threshold; HC: healthy control; HPT: heat pain threshold; MPT: mechanical punctate pain threshold; nR: non-responders; PPT: pressure pain threshold; R: responders; T1: first thoracic nerve; V1: first branch of trigeminal nerve.
Decision tree analysis was carried out to identify outcome predictors, and the three QST parameters mentioned above, namely V1 HPT, V1 MPT, and V1 and T1 PPT, were included in the analysis. It was found that V1 MPT > 158 g (p = 0.020) or V1 HPT > 44.9°C (p = 0.002) was associated with a favorable treatment outcome (Figure 2). The overall accuracy was 80%, and the sensitivity, specificity, positive, and negative values for having a treatment response were 87%, 63%, 85%, and 65%, respectively (Table 4).
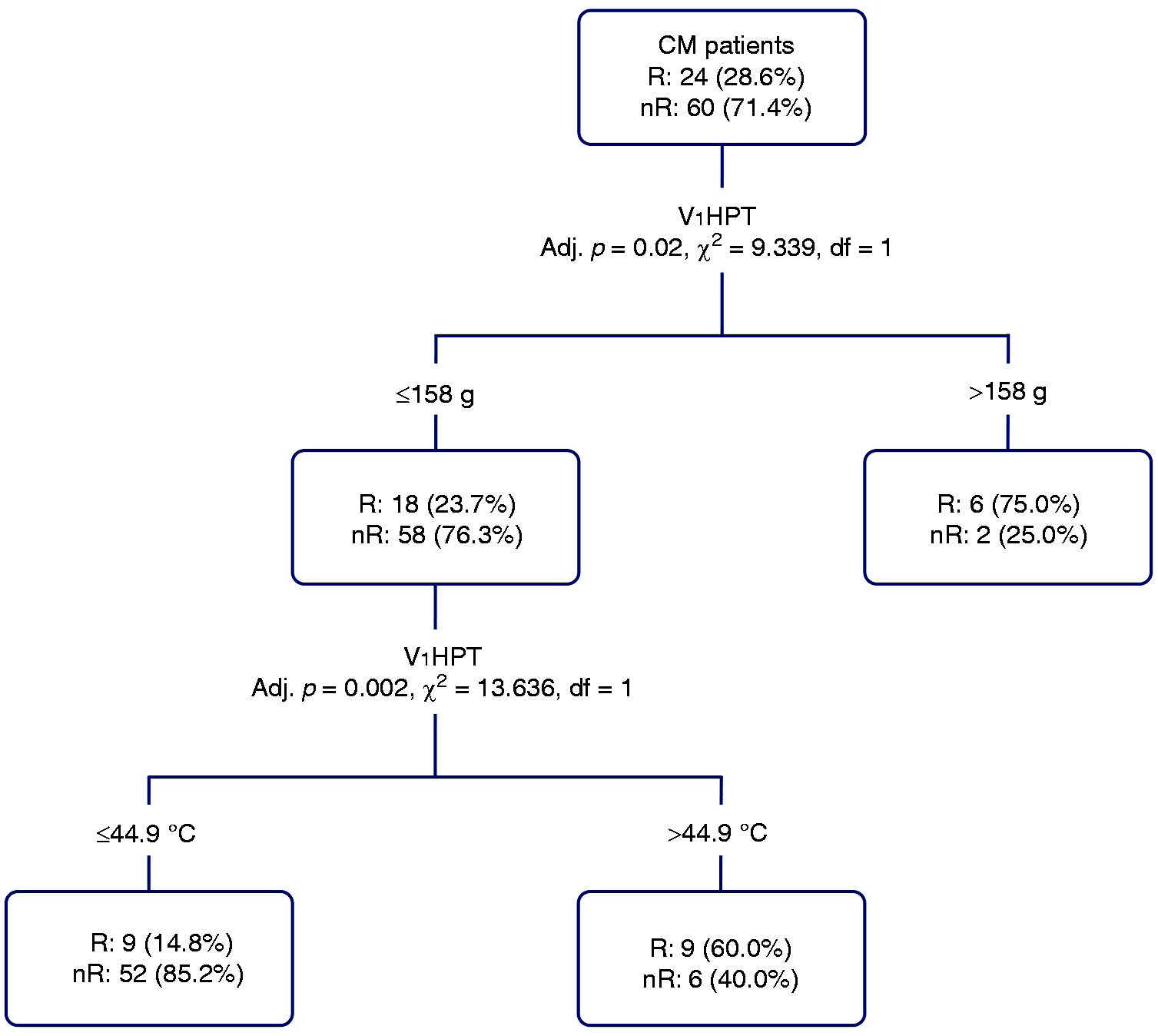
The responders and non-responders were classified based on HPT and MPT with decision tree analysis CM: chronic migraine; R: responders; nR: non-responders; V1: first branch of trigeminal nerve; MPT: mechanical punctate pain threshold; HPT: heat pain threshold.
The confusion matrix of the prediction.
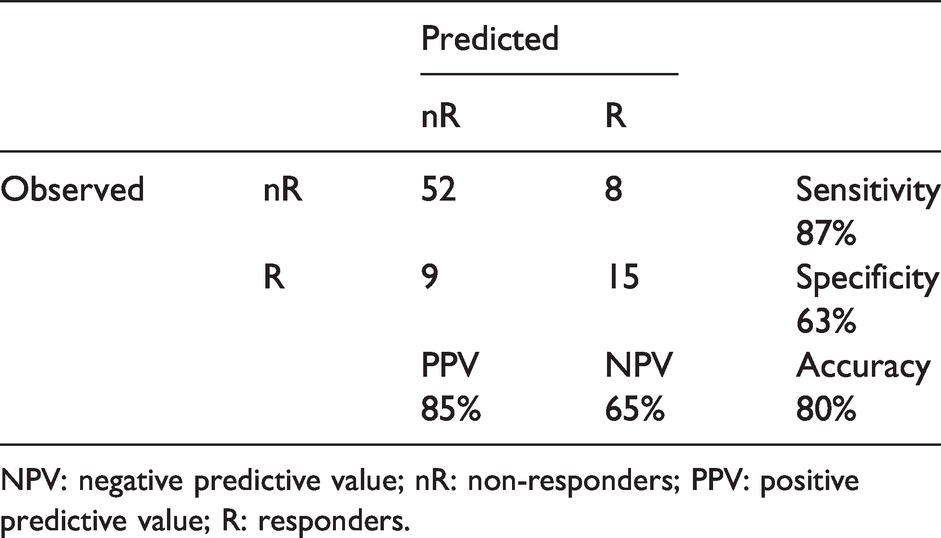
NPV: negative predictive value; nR: non-responders; PPV: positive predictive value; R: responders.
Discussion
This prospective study showed that the CM patients with lower pretreatment MPTs or HPTs in the V1 dermatome were less responsive to preventive treatment with flunarizine than patients with a normal pain sensitivity. These findings indicate an association between increased pain sensitivities and less favorable outcomes following preventive treatment.
One of the most important strengths of this study was the prospective nature of the study design, which reduced recall bias. Second, the data were of high quality and reliability, since the clinical information was confirmed by headache specialists and the QSTs were carried out in a standardized fashion. Besides, the headache frequency was also prospectively recorded with headache diaries. Third, the preventive treatment was identical across the CM patients, and the timing of prescription was also defined according to the protocol. These factors may minimize the fundamental differences among different preventive medications. Fourth, none of our CM patients had received prior treatment, and none of them had any comorbid major psychiatric illness. Therefore, confounding biases resulting from prior treatment or QST changes due to psychiatric disorders were minimized.
In the current study, the responders had higher V1 HPTs, V1 MPTs, and V1 PPTs at baseline, indicating the presence of relatively normal pain sensitivities in these individuals. Although a previous review (34) has shown inconclusive results for the comparison between episodic migraine (EM) and CM, some (35–37) revealed higher pain sensitivity in CM compared to EM, including our previous study (24) which demonstrated that the EM patients had higher V1 MPTs (115 ± 41 vs. 95 ± 35 g) than the CM patients. In fact, the V1 MPTs of the responders (123 ± 55 g) in the present study were similar to those of the EM patients, whereas the non-responders had V1 MPTs (96 ± 33 g) similar to those of the CM patients in our prior study involving an independent cohort (24). Furthermore, there was a similar trend in V1 HPTs. This finding suggests that even though the responders fulfilled the diagnostic criteria of CM based on the clinical criteria and had headache profiles indistinguishable from those of the non-responders, the disease process of chronification or central sensitisation might be less severe. More importantly, the QST is an objective measure of pain sensitivity that reflects the severity of the underlying pathophysiology. Based on decision tree analysis, the CM patients with V1 MPT ≤158 g (z-score: 1.5) or V1 HPT ≤44.9°C (z-score: 0.3), i.e., with an increased pain sensitivity at baseline, were more likely to become non-responders. Such an approach could have the potential to serve as a prognosticator for CM patients or even as an indicator in clinical decision-making as to whether choosing a different preventive agent or even combination therapy initially may be necessary.
The QST was designed to assess the function of nociceptive Aδ fibers and nociceptive C fibers. The slowly increased heat stimuli (1°C/s) selectively activate the C fibers (38), while pinprick or punctate stimuli activate the Aδ fibers. The decreased pain thresholds in non-responders indicate hyperalgesia to pinprick, which is a sign of central sensitisation (13,39,40). Many previous studies have proposed that sensitisation of dural and meningeal trigeminal nociceptors (first-order), trigeminal nucleus caudalis (second-order), and thalamus (third-order) are the underlying mechanisms of increased cephalic and extracephalic pain sensitivities, i.e. cutaneous allodynia (9). In the current study, the decreased pain thresholds in non-responders may indicate the long-term potentiation of the spinal dorsal horn and abnormal neuronal excitability in the trigeminal nucleus caudalis (41–44) possibly suggesting that more severe central sensitisation could make the patients more refractory to migraine preventive treatment. In addition, it has been demonstrated that the impaired descending nociceptive inhibition may decrease pain thresholds as in the case of the pre-ictal stage of migraine attacks (40,45,46), which is one of the possible mechanisms of central sensitisation. In the current study, stimuli to trigeminal and spinal dorsal horn neurons were precisely calibrated, and there was no evidence of peripheral sensitisation, and therefore, it is likely that the observed phenomenon of mechanical punctate hyperalgesia could be attributed to central sensitisation. Therefore, when treating CM patients with lower V1 MPTs or V1 HPTs, treatments targeted at endogenous pain controls, e.g., serotonin and norepinephrine reuptake inhibitors (47,48) and neuromodulation techniques (49), should be considered as the priority.
This study had some limitations. First, this was a single-arm open-labeled exploratory study, and the efficacy of flunarizine could have been overestimated. However, the reduction in the number of headache days (9.3 ± 7.4 days/4 weeks) was of a similar magnitude (4.9 ± 3.8 days/2 weeks) as reported in a previous randomized controlled trial (33). Second, the results of the current study were derived from preventive treatment with flunarizine among treatment-naïve patients only; whether the findings could be generalizable to patients treated with other preventive drugs, such as topiramate, awaits further study. Third, the MPT was measured by using an electronic von Frey device in the present study rather than a traditional von Frey monofilament. Even though the traditional von Frey filament set was commonly used in previous studies, the validity and reproducibility of electronic von Frey devices have been demonstrated in prior studies (24,50). Fourth, the intention-to-treat analysis was applied in this study and the nature of this analysis may have downplayed the preventive effect of our treatment and further resulted in a lower responder rate in this study. However, the purpose of this study was to explore whether QST results can be used to predict the preventive treatment outcomes instead of to evaluate the efficacy of flunarizine alone. Fifth, in the current study, the term central sensitisation was used in compliance with the definition of the International Association for the Study of Pain (13), the exact underlying mechanism is uncertain. Although we can exclude peripheral sensitisation (first-order neurons) as a cause of the observed mechanical punctate hyperalgesia, we cannot distinguish where in the central nervous system the central sensitisation is taking place (second-order, third-order neurons or beyond). Lastly, we did not control the diet, exercise, and sleep throughout the study period that might potentially contribute to the treatment outcomes. However, all the subjects were advised to keep their routine lifestyle during the study period.
Conclusion
In this prospective hospital-based clinical study, CM patients were found to be more sensitive to cold and heat pain over both V1 and T1 dermatomes, and to pressure pain over T1 dermatome compared to HC. Moreover, it was demonstrated that CM patients with higher (i.e., closer to normal) V1 HPTs or V1 MPTs were more likely to have a favorable outcome following preventive treatment with flunarizine. This finding indicates that these patients had lower pain sensitivities and possibly less severe central sensitisation mechanisms; therefore, they had a better treatment response. Future studies should be conducted to assess whether the current non-responders can benefit from prophylactic medication targeting endogenous pain controls, e.g., serotonin and norepinephrine reuptake inhibitors, or from nonpharmacological prophylaxis.
Clinical implications
CM patients with higher (closer to normal) heat pain thresholds or mechanical punctate pain thresholds over the supraorbital area were more likely to have a favorable outcome following preventive treatment with flunarizine. The cutoff values for treatment responders are >44.9°C for the heat pain threshold or >158 g for the mechanical punctate pain threshold, respectively.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221080572 - Supplemental material for Pain sensitivities predict prophylactic treatment outcomes of flunarizine in chronic migraine patients: A prospective study
Supplemental material, sj-pdf-1-cep-10.1177_03331024221080572 for Pain sensitivities predict prophylactic treatment outcomes of flunarizine in chronic migraine patients: A prospective study by Li-Ling Hope Pan, Yen-Feng Wang, Yu-Hsiang Ling, Kuan-Lin Lai, Shih-Pin Chen, Wei-Ta Chen, Rolf-Detlef Treede and Shuu-Jiun Wang in Cephalalgia
Footnotes
Acknowledgments
We would like to thank the study participants for actively participating. This work was supported by the Brain Research Center, National Yang Ming Chiao Tung University, from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education of Taiwan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Ministry of Science and Technology of Taiwan (MOST 108-2314-B-010-023-MY3, 110-2321-B-010-005, and 111-2321-B-A49-004 to SJ Wang, and 109-2314-B-075-054 to YF Wang) and Taipei Veterans General Hospital (VGH 108-C-092, VGH 109-C-096, and VGH 110-C-111 to YF Wang). RD Treede was supported by DFG (SFB 1158).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
