Abstract
Objective
To investigate how cluster headache preventatives verapamil, lithium and prednisone affect expression of hypothalamic genes involved in chronobiology.
Methods
C57Bl/6 mice were exposed to daily, oral treatment with verapamil, lithium, prednisone or amitriptyline (as negative control), and transcripts of multiple genes quantified in the anterior, lateral and posterior hypothalamus.
Results
Verapamil, lithium or prednisone did not affect expression of clock genes of the anterior hypothalamus (
Conclusion
Data suggest that cluster headache preventatives act upstream or downstream from the hypothalamus. Our findings provide new insights on hypothalamic homeostasis during cluster headache prophylaxis, as well as neurochemistry underlying cluster headache treatment.
Introduction
Cluster headache (CH) has a prevalence of 1 in 1000 and a male/female ratio of 2.5–3.5. Rhythmicity represents one of the most enigmatic feature of CH, and a great deal of effort has been directed at deciphering the underlying molecular mechanisms. Nevertheless, the neurobiology of the circadian/circannual pattern of CH and its relationship with attack pathogenesis still waits to be deciphered (1). Because of the key role of the hypothalamus in human chronobiology, as well as in the pathogenesis of primary headaches in general, it has been repeatedly proposed that this brain region is causally involved in CH rhythmicity and pain (2). Accordingly, several fMRI studies report hypothalamic activation during CH attacks (3). Further confirming a pivotal role of the hypothalamus in the etiopathogenesis of CH, deep brain stimulation with electrodes implanted in the posterior hypothalamus reduces both severity and frequency of attacks in CH patients refractory to pharmacological therapy (4).
Verapamil, lithium, and corticosteroids represent the mainstay of CH prophylaxis, but how they function as preventatives is unknown. In this regard, it is worth noting that from a pharmacodynamic perspective, CH preventatives do not show any apparent, mutual mechanism of action. Specifically, verapamil is a calcium channel blocker approved as an antiarrhythmic, lithium is an inositol phosphatase/glycogen synthase kinase inhibitor used as a mood stabiliser, and corticosteroids are transcription-regulating, immunomodulatory drugs. Notwithstanding the lack of similar pharmacodynamics, to our knowledge the question as to whether these drugs share a similar impact on hypothalamic functions is still unanswered. We also reasoned that a deeper understanding of the pharmacological mechanisms underlying CH prophylaxis can help developing innovative therapeutics, for which patients are in greet need.
Hence, in an attempt to gather information on CH pathogenesis and molecular mechanisms underpinning prophylaxis, in the present study we investigated whether oral treatment with verapamil, lithium carbonate or prednisone alter expression profiles of genes involved in hypothalamic functioning, circadian clock and sleep regulation in mice.
Materials and methods
Animals
All animal care and experimental procedures were performed according to the European Community guidelines for animal care (European Communities Council Directive 2010/63/EU) and were approved by the Committee for Animal Care and Experimental Use of the University of Florence. Male C57Bl/6 mice 20–25 g were purchased from Charles River (Milan, Italy) and housed in conventional units (six per cage) with free access to food (Harlan Global Diet 2018, Harlan Laboratories, Udine, Italy) and water. Mice were maintained on a 12 h light/dark cycle at 21°C room temperature for 15 days before drug exposure. As for the power analysis, the experimental hypothesis of our study prompted us to consider the effects of verapamil, lithium or prednisone on expression genes involved in hypothalamic functioning as the parameter of reference. Unfortunately, we have been unable to find studies reporting this information. This precluded the possibility of defining an
Quantitative PCR
Total RNA was isolated from hypothalamic regions using Trizol Reagent (Life Technologies, Monza, Italy). One µg of RNA was retrotranscribed using iScript (Bio-Rad, Milan, Italy). RT-PCR was performed as reported (5). The primers used are reported in Figure 2.
Statistical analysis
Data are expressed as mean ± SEM. To test the difference between more than two groups ANOVA plus Tukey’s post hoc test (according to Kolmogorov–Smirnov test for normality) was used. Differences were considered to be significant with a
Results
Effects of CH preventatives and amitriptyline on hypothalamic gene transcripts
In order to adopt a clinically-relevant treatment protocol, mice were exposed to daily, oral drug administration for 15 days, a length of time sufficient for a CH preventative to prompt a therapeutic effect. Similarly, drugs were administered according to routinely adopted treatment schedules. Specifically, according to human-to-animal dose conversion guidelines and chronic treatment (6), verapamil and lithium carbonate were administered three times per day (8:00 am, 1:00 pm and 8:00 pm) at 3.4 and 13 mg/kg, respectively, whereas prednisone was dosed at 3 mg/kg at 8.00 am. An additional group of mice was exposed to oral amitriptyline (1 mg/kg at 8:00 pm), a migraine preventative altering some hypothalamic functions here adopted as a negative control. Collectively, drug doses were about three-fold higher than those routinely adopted in patients, in keeping with standard conversion from human to mouse chronic dosing. Drug treatments were well tolerated, showing no effects on body weight and food/water consumption (not shown). As far as clock genes of the anterior hypothalamus are concerned, we found that none of the CH preventatives altered transcript levels of
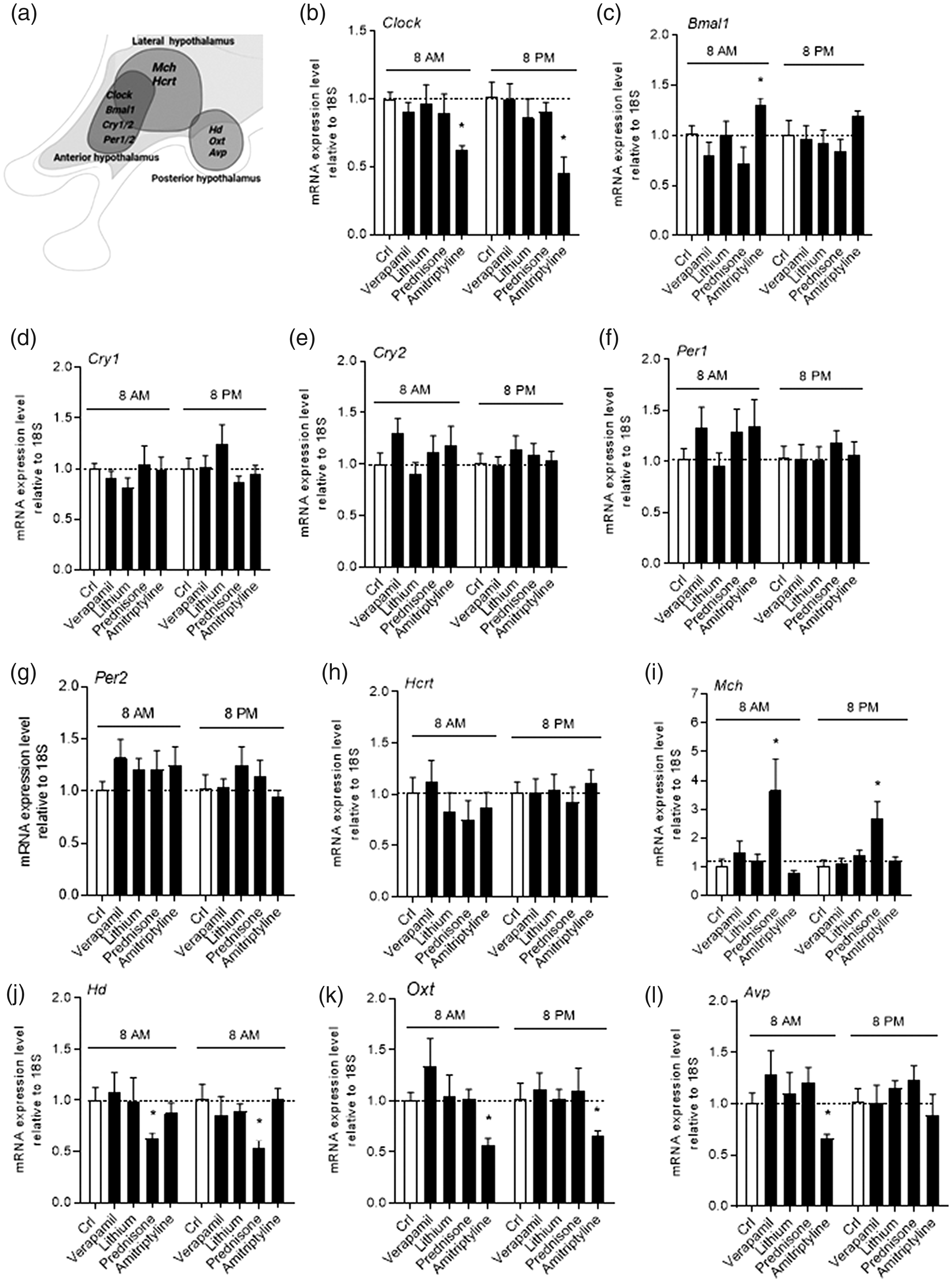
Effects of verapamil, lithium, prednisone, and amitriptyline on gene transcripts involved in hypothalamic chronobiology. The hypothalamic regions (anterior, lateral and posterior) analysed and the respective genes is shown in (a). The impact of a chronic (15 days), oral treatment with verapamil (3.4 mg/kg, three times per day), lithium carbonate (13 mg/kg, three times per day), prednisone (3 mg/kg) or amitriptyline (1 mg/kg) on transcript levels of

Primers used are shown in this table.
Discussion
Even though pathophysiology of CH is largely unknown, both clinical evidence and functional neuroimaging point to a key role of the hypothalamus in disease pathogenesis. Given the preventative nature of verapamil, lithium, and prednisone in CH therapy, it makes sense that these drugs act within the CNS to reduce attack frequency. Indeed, all the three preventatives are known to cross the blood-brain barrier and affect CNS functioning. We reasoned that the three drugs may share the ability to alter hypothalamic homeostasis thereby altering the expression of genes involved in circadian rhythm and sleep/wake cycle. Interestingly,
In conclusion, our data suggest that CH preventatives act upstream or downstream from the hypothalamus, possibly targeting signalling pathways independently involved in disease pathogenesis. However, given that the present findings only provide evidence for lack of diurnal variation in hypothalamic gene expression, further studies are needed to rule out that CH preventatives prompt circadian alterations showing a different timescale.
Article highlights
It is currently unclear whether cluster headache (CH) preventatives modify hypothalamic transcriptional homeostasis. We report that prolonged exposure to verapamil, lithium or prednisone does not affect expression of mouse hypothalamic genes regulating chronobiology. CH preventatives may act upstream or downstream from the hypothalamus.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Health Project 2018 Region of Tuscany, and PRIN2017 Italian Ministry of the University and Research (2017JL8SRX8).
