Abstract
Abstract
Purpose
Previous studies on brain morphological alterations in chronic cluster headache revealed inconsistent findings.
Method
The present cross-sectional explorative study determined telencephalic and cerebellar cortex thickness alterations in a relatively wide sample of chronic cluster headache patients (n = 28) comparing them to matched healthy individuals.
Results
The combination of two highly robust state-of-the-art approaches for thickness estimation (Freesurfer, CERES), strengthened by functional characterization of the identified abnormal regions, revealed four main results: chronic cluster headache patients show 1) cortical thinning in the right middle cingulate cortex, left posterior insula, and anterior cerebellar lobe, regions involved in nociception's sensory and sensory-motor aspects and possibly in autonomic functions; 2) cortical thinning in the left anterior superior temporal sulcus and the left collateral/lingual sulcus, suggesting neuroplastic maladaptation in areas possibly involved in social cognition, which may promote psychiatric comorbidity; 3) abnormal functional connectivity among some of these identified telencephalic areas; 4) the identified telencephalic areas of cortical thinning present robust interaction, as indicated by the functional connectivity results, with the left posterior insula possibly playing a pivotal role.
Conclusion
The reported results constitute a coherent and robust picture of the chronic cluster headache brain. Our study paves the way for hypothesis-driven studies that might impact our understanding of the pathophysiology of this condition.
Introduction
Cluster headache (CH) is a primary headache disorder characterized by episodes of short-lasting unilateral and severe craniofacial pain associated with trigeminal autonomic symptoms, such as lacrimation, ptosis, conjunctival injection, and facial sweating. About 90-95% of patients suffer from the episodic form of the disorder (eCH), characterized by periods of multiple daily episodes lasting weeks or months (in-bout phase cluster period) followed by attack-free periods (out-of-bout phase remission period). If the symptomatic period does not remit within 12 months, the disorder is considered chronic (cCH) (1).
Together with human hormonal and animal models studies (2), neuroimaging promoted a paradigmatic shift from peripheral pathogenic hypotheses towards a central pathogenic hypothesis, which emphasizes a possible key role of the hypothalamus (3), and concomitant maladaptation in other pain-processing brain regions (4–7). The anatomical rearrangement of the hypothalamus in CH patients was first described by the seminal work of May et al. (8) Although these results were not replicated in most of the subsequent investigations (4,6,9), a more recent study demonstrated enlargement of the anterior portion of this structure in both eCH and cCH patients, suggesting a role in CH pathophysiology for the suprachiasmatic nucleus, the most important brain biological clock, and the paraventricular nucleus, a modulator of autonomic and nociceptive activities (10).
Also not fully consistent are the results concerning changes in grey matter (GM) volume or thickness of pain processing regions, in particular across different CH phases (i.e., out-of-bout and in-bout) in eCH. During the out-of-bout phase, GM abnormalities were detected in the prefrontal and frontal areas (4,7,11), parietal lobe (4,11), temporal lobe, and insula cortex (4,6). Partially overlapping, GM alterations were observed during the in-bout phase specifically in frontal (12), insular (6), and temporal (6,10) regions. GM changes of extra pain-processing regions were also revealed, pointing to widespread neuromorphological changes in CH. In particular, the alterations within visual areas have been repeatedly reported in morphological (4,7) and resting-state functional (rs-fMRI) (5,13) studies.
Despite the importance of understanding the neuropathological bases of CH chronicization, brain morphological investigations of the chronic condition are still very scarce and revealed conflicting results. On one side, Naegel et al. (6) reported reduced GM volumes in core pain processing regions, such as the right (R) anterior cingulate cortex, the R somatosensory cortex, and the left (L) anterior insula cortex (IC), and in extra-pain processing regions, such as the L hippocampus, the L inferior temporal gyrus, the R orbitofrontal cortex, and the R occipital lobe. Moreover, these authors observed increased GM volumes in the R posterior insula, the cerebellum, and the sensorimotor and visual areas suggesting wide brain morphological rearrangements. On the other side, Arkink et al. (10), reported highly regional-specific increased GM volumes in the anterior section of the hypothalamus and the middle frontal gyrus, a region involved in the modulation of pain (14).
Inconsistent findings between these two studies may be explained in terms of the use of voxel-based morphometry (VBM) (15). One crucial theoretical assumption of VBM is that individual brain differences and anatomical correspondence of brain areas are maintained during the spatial normalization process (16). However, this assumption is challenged by the spatial normalization inaccuracies and the use of different normalization templates which strongly affect the results (16). Moreover, VBM lacks a clear in-vivo or ex-vivo histological neurobiological validation in humans (16). Differently, the Freesurfer’s (http://surfer.nmr.mgh.harvard.edu) (17) and the CERES’s automated pipelines (https://www.volbrain.upv.es) (18) are two robust, accurate, and widely accepted methods (19,20)
In an attempt to fill the gap among the reported inconsistencies and to overcome the limitations of VBM, we investigated telencephalic and cerebellar cortical thickness abnormalities in a relatively wide sample of cCH patients compared to healthy individuals (CTRL) employing Freesurfer’s and CERES’s algorithms. Moreover, we supported our findings functionally characterizing the identified abnormal regions employing resting-state functional connectivity (rs-FC) maps in a subsample of the same participants and a large independent sample of healthy individuals (21).
Materials and methods
Participants
cCH patients were consecutively recruited among cCH individuals hospitalized to treat the recurrent CH attacks and assessed at the Fondazione IRCCS Istituto Neurologico C. Besta in Milan.
All patients were diagnosed with cCH by two expert neurologists (ML and AP) according to the International Classification of Headache Disorders, 3rd edition criteria (ICHD-3) (1). Patients with a concomitant diagnosis of other primary or secondary headache disorders, cardiovascular diseases, diabetes mellitus, hypertension, or a history of psychiatric conditions were excluded from the study. Age and sex matched CTRL individuals reporting no history of primary headache or chronic pain and no history of neurological or psychiatric disorders were recruited as controls. To test between-groups differences on demographic variables, Kruskal–Wallis and Chi-square tests were used.
All participants provided written informed consent according to the Declaration of Helsinki before the study inclusion. This study was approved by the Fondazione IRCCS Istituto Neurologico C. Besta ethics committee.
MRI assessments
All participants were scanned in the attack-free state with a 3T magnetic resonance imaging (MRI) system (Achieva, Philips Healthcare BV, Best, NL) using a 32-channel head coil. As part of the MRI protocol, high-resolution structural 3D T1-weighted (T1w) image (TR = 9.86 ms, TE = 4.59 ms, FOV = 240 × 240 mm, voxel size = 1 mm3, flip angle = 8◦, 185 sagittal slices) was acquired in each participant. To explore the functional connectivity (rs-FC), a rs-fMRI sequence (TR = 2800 ms, TE = 30 ms, α = 70°, 2.5 mm isotropic voxel size, matrix size 90 × 95, 50 slices with 10% gap acquired in ascending order, volumes number = 200) was acquired in the subsample of cCH and CTRL individuals. All MRI images were visually inspected by expert neuroradiologists (LC and MGB) to exclude apparent brain abnormalities or artifacts affecting the quality of the exam.
Structural MRI data analyses
We examined the presence of cerebral structural abnormalities in cCH patients investigating the cortical thickness of telencephalic regions and cerebellar lobes, using automated, well-validated, and robust tools (respectively, Freesurfer and CERES) (17–20).
Telencephalic cortical thickness analyses
We performed surface-based analyses, employing Freesurfer version 6 (http://surfer.nmr.mgh.harvard.edu). Cortical model reconstructions were performed using the recon-all algorithm applied on T1w images. The segmentation outputs were visually checked and manually corrected by an expert operator (CP) blind to the diagnosis (cCH or CTRL) to remove the pial-white boundary surfaces segmentation inaccuracies. The resulting surfaces were subjected to a vertex-wise between-group analysis to test for telencephalic cortical thickness differences between cCH and CTRL individuals. Age and gender were used as covariates in the analysis and the results were considered significant with a vertex-wise threshold of p < 0.001 (uncorrected) and a cluster-wise threshold of 30 mm2. Significant clusters were anatomically labelled according to the Destrieux Cortical Atlas (22). Moreover, in the cCH group, we tested the effect of disease duration on the cortical thickness of the abnormal areas that emerged in the vertex-wise between-group analysis. To this aim, for each patient, we extracted the mean cortical thickness of each significant cluster and we performed the linear regression analyses employing the disease duration as explanatory variable. The results were considered significant for a threshold of p < 0.05.
Cerebellar cortical thickness analyses
We computed the cerebellar cortical thickness using the automated pipeline CERES software (18) on T1w images, obtaining, in every single subject, the mean cortical thickness of the bilateral anterior (I, II, III, IV, and V lobules), posterior (VI, VII, VIII, IX lobules, and crusI and II), and flocculonodular (X lobule) lobes. The computed values were employed to test the differences between cCH and CTRL individuals in each lobe. Shapiro-Wilk test showed that the data were normally distributed and Barrett test revealed that the between-group variance was not statistically different; therefore, ANCOVAs were performed using age and gender as covariates. Moreover, similar to the telencephalic cortical thickness, we employed regression analyses to test for the effects of disease duration on each significant area that emerged in the between-group comparisons. Results were considered significant for a threshold of p < 0.05.
Functional characterization of the abnormal cortical telencephalic regions
Methodological details of this paragraph are reported in Supplementary Materials.
To provide a functional characterization of the identified clusters of cortical abnormalities, we used two different and independent approaches. First, in the sub-sample of the investigated CTRL individuals, we explored the rs-FC of the identified regions of cortical abnormalities. Then, in the subsample of cCH patients, we explored whether the rs-FC of these regions was atypical compared to CTRL participants. Using CONN toolbox (www.nitrc.org/projects/conn), after an extensive and state-of-the-art preprocessing of rs-fMRI data, we built 10mm-spherical regions of interest (ROIs) centered in the identified regions of abnormalities. Then, we performed a ROI-to-ROI analysis. One sample t-tests were used to identify significant rs-FC among the identified regions in CTRL and cCH individuals. A one-way ANCOVA was performed to analyze between-group differences using age and gender as covariates. The results were considered significant for p < 0.05 FDR-corrected.
Secondly, we explored whether the identified regions of cortical abnormalities were reciprocally functionally connected in an independent and large sample of 1000 healthy individuals (23,24). Employing the Neurosynth platform (https://neurosynth.org), we generated rs-FC maps seeded in each peak coordinate of the identified regions (24). Then, we determined whether each 10-mm spherical ROI centered in these regions belonged to any of the produced rs-FC maps and whether they were reciprocally connected. In the reciprocally connected regions, we parsimoniously identified the possible cognitive processes in which they are involved and that might be impaired or, at least, more prone to abnormalities in CH pathophysiology. Using NIMARE, we identified which terms, related to cognitive processes (21), were mainly used in the fMRI studies (present on Neurosynth Platform) reporting activity in the identified regions of abnormality. Then, we identified the ‘terms’ shared by the clusters of abnormalities resulting reciprocally connected.
Results
Twenty-eight cCH patients (23 men, 5 women; mean age ± SD: 45 ± 11.7 years; age range: 28–70 years; see Table 1) were enrolled. All patients, except for two, were taking prophylactic treatment (Supplementary Table 1). Twenty-eight sex and age-matched CTRL individuals (23 men, 5 women; mean age ± SD: 45 ± 10.1 years; age range: 29–64 years) were enrolled. No between-group differences were detected in terms of age and sex (Table 1). A subsample of cCH patients (n = 19, 2 women; mean age ± SD: 46 ± 10.9 years) and CTRL individuals (n = 18, two women; mean age ± SD: 45±10.7 years) underwent also rs-fMRI acquisitions.
Demographic and clinical data.

cCH, chronic cluster headache group; CTRL, control group; bilat, bilateral; L, left; M, mean; MIN-MAX, minimum-maximum values; n.a., not applicable or not assessed; R, right; SD, standard deviation; ys, years.
*Referred to the week before the MRI examination.
Telencephalic cortical thickness results
The between-group vertex-wise analysis showed that cCH patients compared to CTRL individuals present reduced cortical thickness in clusters located in the posterior part of the R midcingulate cortex (MCC), in the anterior part of L superior temporal sulcus (STS), in the L collateral sulcus/lingual sulcus (CLS), and the left posterior insula cortex (postIC), while increased cortical thickness in a cluster located in the R inferior temporal gyrus (ITG), (see Figure 1 and Table 2). No significant correlation between cortical thickness and disease duration was observed in these regions (Supplementary Table 2).

Vertex-wise between-group analysis: significant clusters of reduced cortical thickness in cCH patients compared to CTRL participants expressed as z-values (p < 0.001, cluster-wise threshold 30 mm2), displayed on FreeSurfer’s semi-inflated cortical surfaces. The color bar indicates the significance levels of the clusters. Significant clusters of vertex-wise between group analysis: boxplot reported mean thickness in groups and results of t-test comparing CH and CTRL mean thickness; the results were considered significant with a p < 0.001. Abbreviations: cCH = chronic cluster headache patients, CTRL = control individuals, L = left, R = right, A = anterior, P = posterior, MCC = midcingulate cortex, STS = superior temporal sulcus (STS), CLS = collateral sulcus/lingual sulcus (CLS), postIC = posterior insula cortex (postIC), ITG = inferior temporal gyrus.
Vertex-wise between-group analysis: significant clusters of cortical thickness abnormalities in cCH patients compared to CTRL (p < 0.001, cluster-wise threshold = 30 mm2). Significant regions were labelled according to the Destrieux Cortical Atlas. Coordinates of the peaks of the observed cortical abnormalities were transformed from MNI305 to MNI152 using the matrix provided in https://surfer.nmr.mgh.harvard.edu/fswiki/CoordinateSystems.
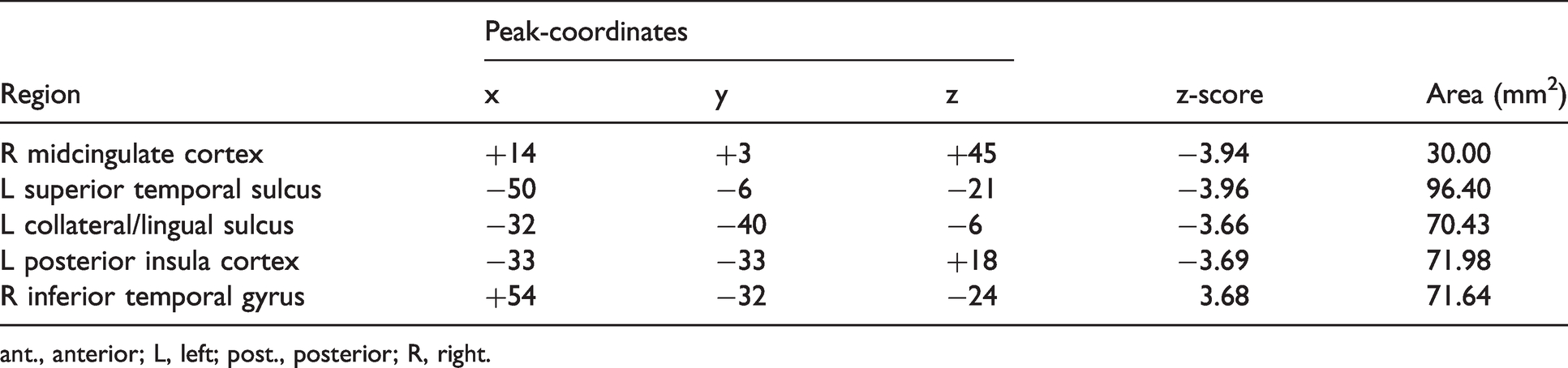
ant., anterior; L, left; post., posterior; R, right.
Cerebellar cortical thickness results
The ANCOVA showed that, compared to CTRL individuals, cCH patients presented a significant thinning of the cerebellar anterior lobe (Table 3). No significant correlation between the mean cortical thickness of this cerebellar lobe and disease duration was observed (Supplementary Table 2).
Mean and standard deviation (in mm) of cerebellar lobe cortical thickness and results from the between-group comparison (cCH vs. CTRL individuals) using ANCOVA (p < 0.05).

cCH, chronic cluster headache patients; CTRL, control individuals.
Functional characterization of the abnormal cortical telencephalic regions
The analyses of the ROI-to-ROI rs-fMRI data showed that CTRL presented robust FC among the investigated ROIs. Specifically, the L postIC ROI appeared to be functionally connected with every other investigated ROIs (positive FC with R MCC, L STS, L CLS; negative FC with R ITG). Also, the L CLS and the L STS ROIs appeared to be functionally connected (positive FC). In addition, in cCH patients, the L postIC ROI was the only region functionally connected with all the other investigated ROIs; however, a positive FC was observed with the R MCC, L STS, while a negative FC with the L CLS and R ITG. The between-group analysis results showed that, compared to CTRL individuals, cCH, patients presented a reduced FC between the L CLS and both the L postIC and the R MCC ROI (Table 4 and Figure 2).
Results from one-sample t-tests and ANCOVA. One-sample t-tests were used to test separately in the cCH and CTRL groups significant functional connectivity among the 10mm ROIs built in peak coordinates of the identified regions of abnormalities in cCH patients. ANCOVA, using age and gender as covariates was employed to test for between-group differences (cCH > CTRL). Results were considered

cCH, chronic cluster headache patients; CLS, collateral sulcus; CTRL, control individuals; ITG, inferior temporal gyrus; L , left; MCC, midcingulate cortex; postIC, posterior insula cortex; R, right; STS, superior temporal sulcus.
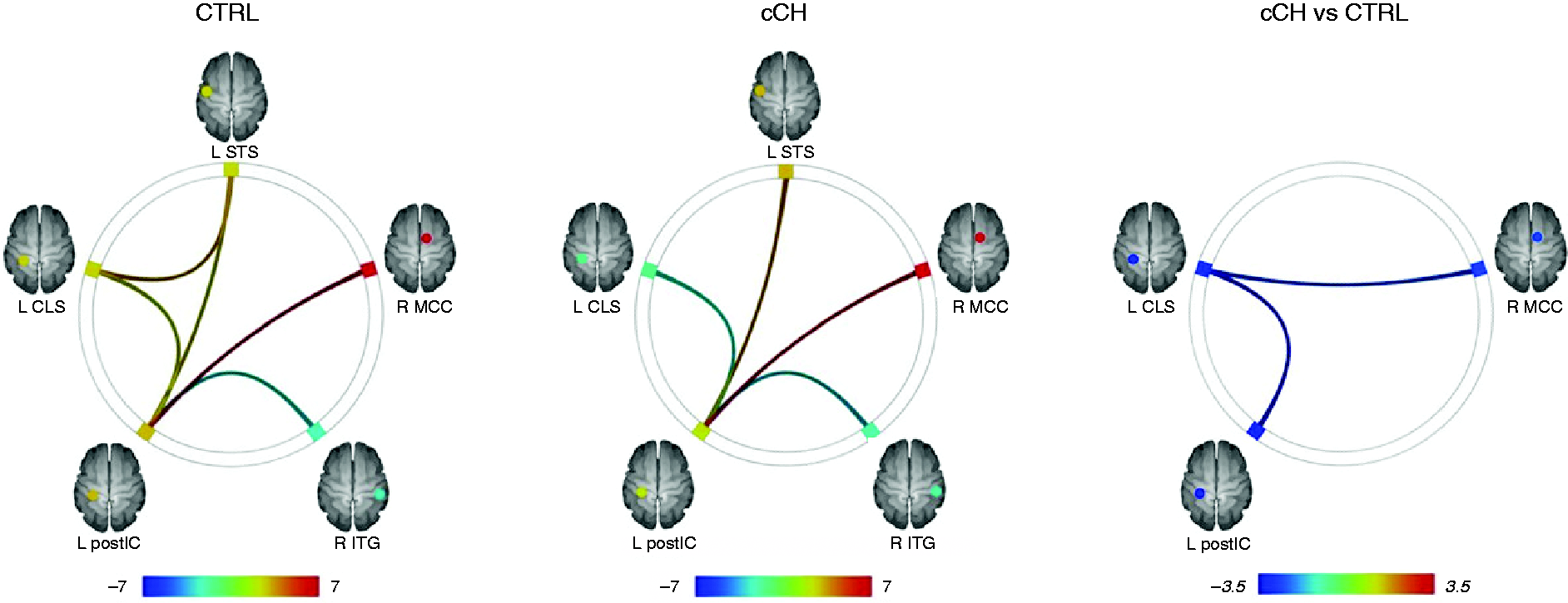
Rs-fMRI results (CONN): Connectome ring representations of the significant ROI-to-ROI functional connectivity of the identified regions of cortical abnormalities in CTRL individuals, cCH patients, and for the differences between the two groups (cCH vs CTRL). Significant results were reported using p < 0.05 FDR-corrected. The color bar represents T values. Abbreviations: cCH = chronic cluster headache patients, CTRL = control individuals, L = left, R = right, A = anterior, P = posterior, MCC = midcingulate cortex, STS = superior temporal sulcus (STS), CLS = collateral sulcus/lingual sulcus (CLS), postIC = posterior insula cortex (postIC), ITG = inferior temporal gyrus.
The analysis conducted employing the independent and large sample of healthy individuals (see supplementary Table 3) showed that the R MCC and L postIC were reciprocally functionally connected. Similarly, the L CLS and L STS, while no reciprocal connection emerged for the R ITG.
Capitalizing on the possible reciprocal connections, we observed that the shared ‘terms’ between the R MCC and L postIC ROI were related to pain processing (common terms: ‘noxious’, ‘nociceptive’) (Supplementary Table 4), while the shared terms between L STS and L CLS ROI were related to the so-called ‘social brain’ (25,26) and, specifically, to autobiographical memories (common terms: ‘remembered’, ‘autobiographical memory’, ‘autobiographical’, ‘remembering’). Notably, this couple of ROIs also presented the common term ‘construction’, mainly related to papers referring to the construction of memories (e.g. MULLALLY) (Supplementary Table 5).
Discussion
The present study capitalized on two robust and extensively validated methods (Freesurfer, CERES) to determine telencephalic and cerebellar cortex structural abnormalities in the chronic form of CH. Our results were strengthened by the functional characterization of the identified regions in a subsample of the same individuals and, using an unbiased large-scale dataset approach, in an independent sample of healthy subjects (21).
Our main results showed that cCH patients, compared to CTRL individuals, present a reduced cortical thickness in clusters located in the R MCC, the L post-IC, and the anterior lobe of the cerebellum, regions playing a key role in pain processing (14), and the L STS and L CLS, extra-pain processing areas, possibly part of the ‘social brain’ (26). A region of cortical thickening was observed in the R ITG. Remarkably, the rs-FC maps acquired in the subsamples of the same cCH and CTRL individuals showed that the identified telencephalic regions are functionally connected in CTRL subjects, as well as in cCH patients, albeit differently, with the L postIC appearing to be a central hub of this circuit. More importantly, our results showed that, in cCH patients, some of these regions present abnormal rs-FC (specifically between the L CLS and both the R MCC and the L antIC).
Notably, the employed tools (Freesurfer and CERES) outperform the widely employed VBM methods described in CH literature. Specifically, Freesurfer was shown to provide a robust quantification of brain volumetry with high reproducibility (27,28), and, more importantly, it was validated against histological measurements in post-mortem brain (29) and in histological specimens resected from in-vivo brains (19). Additionally, CERES was proven to be an accurate and reproducible tool for the segmentation and parcellation of the cerebellum (30) and, compared to automated state-of-the art tools, was shown to present better overall performances (20).
Cortical thinning of R MCC, L postIC
The identified clusters of cortical thinning in R MCC and L postIC are quite inconsistent in respect to the previous findings in cCH patients. Naegel et al. (6) reported reduced volume of the R posterior-anterior cingulate cortex (in a region close to the one we observed) and increased volume in the R postIC. Conversely, Arkink et al. (10) did not detect any sign of abnormalities in the insula or cingulate cortex. Similarly, in eCH patients, volume alterations in the insula and cingulate cortex were detected in different areas compared to our findings (4,12), while decrease of cortical thickness was reported by Seifert et al. (11) in different regions (i.e. angular gyrus and the precentral gyrus contralaterally to the headache side).
It is well accepted that R MCC and L postIC play an important role in the experience of pain (31). These are core-areas of the spino-thalamic-cortical tract, one of the two main ascending systems (the other being the spino-parabrachialamygdalar pathway) that convey and process nociceptive information. Accordingly, the identified clusters of cortical thinning in the R MCC and the L postIC appeared to be robustly functionally connected in both cCH patients and CTRL individuals investigated for this study and in the large independent rs-fMRI dataset (24).
The MCC has been implicated in a wide range of cognitive functions associated with nociception including monitoring, cognitive conflict, reward, and emotional and aversive stimuli processing (32). The posterior MCC (where we mainly detected the cortical thinning – see Figure 1) is more specifically involved in the reflexive spatial orientation of the head and body towards salient sensory stimuli (33).
The postIC codes the intensity and the location of the nociceptive inputs (34). This key role in the sensory processing of nociception is also underlined by acute painful sensations induced by its stimulation (35). From this perspective, our results indicate abnormalities in areas mainly involved in the sensory-motor processing of nociceptive information traveling along the spino-thalamic pathway. In support of this hypothesis, the application of the decoding algorithm across all studies present in the Neurosynth database showed that these two regions shared ‘terms’ (a proxy of cognitive processes) related to pain processing (shared term: ‘noxious’). Remarkably, in a recent murine study, the development and maintenance of nociceptive hypersensitivity were shown to be mediated by the MCC-post-IC pathway (36). It is tempting to speculate that the observed reduced cortical thickness of MCC and postIC in cCH patients might indicate a dysfunctional adaptation of this network that might be critical also in CH chronicization. Intriguingly, these two regions are also part of the central autonomic system, as shown by a robust fMRI meta-analysis (37) in agreement with the trigeminal autonomic symptoms characterizing the CH attacks.
Cortical thinning of L STS, L CLS and R ITG
Different from our findings (i.e. cortical thinning of a cluster in L STS), specific GM reductions of the L anterior STS were not reported in previous studies on CH. However, a reduced GM volume was reported in the left inferior temporal gyrus in cCH patients (6) similarly (although in the opposite direction) to the cortical thickening we observed in the R ITG. The anterior section of STS was shown to be a fundamental hub of the so-called ‘social brain’ (26), a wide network also comprising the lateral fusiform gyrus, the medial prefrontal cortex, the precuneus, the posterior STS, and the amygdala. Supporting this notion, the results from the application of the decoder across the whole Neurosynth database revealed that studies presenting fMRI activity in this area show a very high probability of presenting terms referred to as ‘social brain’ and, in particular, to autobiographical memories processes. Reduced cortical thickness was also observed in the L CLS, which appeared to be functionally connected with L STS in healthy individuals, as indicated by the ROI-to-ROI analyses conducted in our subsample of CTRL subjects and by the seed-based analyses conducted in a large independent rs-fMRI dataset (24).
The application of the decoder across the whole Neurosynth database showed that these two regions shared ‘terms’ (a proxy of cognitive processes), mainly related to the ‘social brain’ and specifically to aspects related to autobiographical memories (shared terms: ’autobiographical’, ’autobiographical memories’, ‘reconstruction’ of memories, ‘remembering/remembered’). We speculate that the cortical thinning observed in cCH patients, in clusters located in L STS and L CLS, might indicate an impairment of the ‘social brain’ network, which vulnerability might support the most common psychiatric comorbidities observed in CH (38) (e.g. depression, anxiety, and aggressive behavior).
Cortical thinning of the anterior lobe of the cerebellum
We identified a significantly reduced cortical thickness of the anterior lobe of the cerebellum of cCH patients in comparison to CTRL individuals. Although our results confirmed abnormalities of the cerebellum in cCH patients, it is important to note that they are inconsistent with Arkink et al. (2017) (10), who did not report cerebellar alterations, and Naegel et al. (2014) (6), who showed a cluster of increased grey matter in the cerebellum of cCH patients compared to CTRL participants, cluster that disappeared employing a more stringent threshold. The anterior lobe of the cerebellum is mainly involved in sensorimotor process (39) but a role in nociception, even though still to be clarified, was also confirmed: acute nociceptive stimulation is processed in a somatotopic way with pain anticipation observed in almost overlapping cerebellar anterior areas (40).
Functional characterization of the abnormal cortical telencephalic regions
Our results in the functional domain strongly support the anatomical findings and the above inferences. First, we found that all the observed telencephalic regions of abnormalities appear to be (directly or indirectly) functionally connected in the investigated sample of CTRL individuals. Specifically, the L postIC seems to be the central hub of this network with robust positive FC with R MCC, L STS, and L CLS and negative FC with R ITG. Moreover, a strong positive FC was also observed between the L STS and L CLS. Importantly, these results are robustly supported by the analyses conducted on rs-FC maps from a large independent sample of healthy individuals, indicating that the clusters in R MCC and L postIC are reciprocally functionally connected, as well as the clusters in L STS and L CLS. Notably, the rs-FC maps employed for this analysis only report correlated FC, therefore, the absence of the evidence of reciprocal FC for the R ITG is in line with the identified negative FC observed between this region and L postIC in the investigated sample of CTRL individuals. Second, our results showed that also in cCH individuals, the L postIC is the main hub of this extended functional network, being positively or negatively functionally connected with all the other clusters of abnormalities. This suggests that the L postIC might be the key-region in determining or modulating the observed anatomical abnormalities. Remarkably, the between-group comparison showed that cCH patients present a reduced FC between the L postIC and L CLS and between the L CLS and R MCC in agreement with the reduced cortical thickness of these regions.
Notably, we did not perform the rs-FC analysis of the anterior lobe of the cerebellum because in some of the investigated patients it was not inside the FOV of the rs-fMRI sequence and because the conducted analysis did not produce localized regions of abnormalities, preventing us from investigating this region with the Neurosynth database and to perform the decoding.
Limitations
Two main limitations suggest caution in the interpretation of these data. The first is related to the ongoing pharmacological treatment of the investigated cCH patients that might impact the brain structure. For ethical reasons, we did not ask the patients to discontinue their medications to perform the washout. The second limitation is related to the fact that we did not match the cCH and CTRL individuals for smoking habits. Heavy smoking habits are consistently reported in CH; however, the relationship between the two is still not understood. It is well acknowledged that smoking-dependent individuals present morphological abnormalities of several brain regions (41). Overlapping with our results, a recent meta-analysis showed that smokers, in comparison to non-smokers, present reduced GM volumes in the L postIC (41). It is important to note that the effects of tobacco smoking on the brain might be exacerbated in older individuals, further complicating the CH pathophysiology.
Conclusion
Our study reports four important findings. First, we showed that cCH patients present cortical thinning in brain regions mainly involved in pain processing (R MCC and L postIC and anterior lobe of the cerebellum) and possibly in autonomic function (R MCC and L postIC). Whether these abnormalities are the origin or the consequence of CH or of chronicization of pain in cCH should be a major focus of future studies.
Second, we showed that cCH patients present abnormalities in areas belonging to the ‘social brain’ (L STS and L CLS), which in principle might support the psychiatric comorbidity frequently observed in cCH individuals (38). Future studies should assess whether, in cCH individuals, the ‘social brain’ is impacted from the behavioral and neural point of view. Third, we showed that all the areas of telencephalic cortical abnormalities are functionally connected in the healthy brain and cCH individuals; remarkably, the L postIC appears to be the central hub of the observed anatomical and functional anomalies. Forth, cCH individuals present abnormal FC among some of these areas. The reported results constitute a coherent and robust picture of the cCH brain. Our study paves the way for hypothesis-driven studies that might involve both cCH and eCH patients and that, in principle, might impact our understanding of the pathophysiology of this condition.
Article Highlights
The combination of two highly robust state-of-the-art approaches for thickness estimation of telencephalic and cerebellar cortex showed that chronic cluster headache patients present: cortical thinning in key-areas of the spino-thalamic-cortical tract involved in the sensory-motor aspects of nociception (right middle cingulate cortex and the left posterior insula) cortical thinning in areas possibly involved in social cognition (left anterior superior temporal sulcus and the left collateral/lingual sulcus).
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211058205 - Supplemental material for Chronic cluster headache: A study of the telencephalic and cerebellar cortical thickness
Supplemental material, sj-pdf-1-cep-10.1177_03331024211058205 for Chronic cluster headache: A study of the telencephalic and cerebellar cortical thickness by Greta Demichelis, Chiara Pinardi, Luca Giani, Jean Paul Medina, Ruben Gianeri, Maria Grazia Bruzzone, Benjiamin Becker, Alberto Proietti, Massimo Leone, Luisa Chiapparini, Stefania Ferraro and Anna Nigri in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Italian Ministry of Health, research grant RF-2016-02364909.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
