Abstract
Background
Gut microbiota disturbance is increasingly suggested to be involved in the pathogenesis of migraine but this connection remains unsubstantiated. This study aimed to investigate whether the gut microbiome influences migraine-related hyperalgesia.
Methods
Nitroglycerin-induced hyperalgesia was evaluated in mice with different gut microbiota statuses as follows: Specific pathogen-free mice; germ-free mice; specific pathogen-free mice treated with antibiotics to deplete the gut microbiome (ABX mice); and germ-free mice transplanted with the gut microbial profile from specific pathogen-free mice (GFC mice). Moreover, nitroglycerin-induced hyperalgesia was compared between recipient mice transplanted with gut microbiota from a patient with migraine and those that received gut microbiota from a sex- and age-matched healthy control.
Results
In specific pathogen-free mice, a decreased mechanical threshold in the hind paw, increased grooming time, increased c-Fos expression level and decreased calcitonin gene-related peptide expression level as well as increased tumor necrosis factor-α concentration in the trigeminal nucleus caudalis were observed after nitroglycerin administration compared with saline treatment. However, increased basal sensitivity and higher basal concentrations of TNF-α in the trigeminal nucleus caudalis were observed in germ-free and ABX mice, while no significant difference in hyperalgesia was observed between the nitroglycerin group and saline group in germ-free and ABX mice. Moreover, significant hyperalgesia was induced by nitroglycerin administration in GFC mice. The mice transplanted with the gut microbial profile from a patient with migraine had more severe nitroglycerin-induced hyperalgesia than the mice receiving microbiota from a matched healthy control.
Conclusion
Our findings highlight the involvement of the gut microbiome in normal mechanical pain sensation and pathogenesis of migraine.
Introduction
Activation of the trigeminovascular system is considered to be an important component in the pathogenesis of migraine but contributors to its susceptibility are unclear. Although 38 genomic loci have been identified (1), a meta-analysis of twin studies estimated that the heritability of migraine was approximately 42% (2), suggesting that environmental factors also contribute. A growing body of research has indicated the potential role of the gut microbiota in the pathogenesis of migraine. Migraine is often accompanied by gastrointestinal (GI) symptoms, including nausea, vomiting, and bowel disturbances (3). Dietary triggers, such as dairy products and chocolate, are commonly reported by patients with migraine (4). In addition, migraine occurs at a higher rate in patients with GI disorders such as cyclic vomiting syndrome, abdominal migraine, and inflammatory bowel disease (5,6). In addition, some studies have shown the beneficial effects of probiotic supplementation on migraine headaches (7). These studies suggest that gut microbiota disturbance may be involved in the occurrence of migraine.
The gut microbiome comprises the community of microorganisms (i.e. bacteria, archaea, fungi, and viruses) colonizing the GI tract, and its homeostasis is crucial for maintaining host health-related processes, such as the digestion of dietary macronutrients, the production of nutrients, and the maintenance of the immune system (8). In addition, the gut microbiome and brain interact bidirectionally via the vagus nerve, enteroendocrine signalling, immune system crosstalk and neurotransmitters; this system is termed the microbiota–gut–brain axis (9). Increasing clinical and preclinical evidence suggests that microbiota–gut–brain axis disturbance contributes to the occurrence of neurologic disorders, such as anxiety, autism, Alzheimer’s disease, and Parkinson’s disease (10). In addition, previous studies have shown that the gut microbiome impacts pain perception. Inflammatory pain induced by carrageenan, TNF-α, and IL-1β and chemotherapy-induced pain were both reduced in germ-free (GF) mice that were raised without intestinal flora (11,12). In contrast, visceral sensitivity was increased in GF mice (13). The potential influence of microbiota–gut–brain axis disturbance on migraine pain has been increasingly suspected (14–16), but there has been a lack of clear evidence.
Therefore, the present study aimed to determine whether gut microbiota can influence migraine-related pain. We compared nitroglycerin (NTG)-induced migraine- related hyperalgesia in mice with or without gut microbial colonization and then built a human microbiota-associated (HMA) model via faecal microbiota transplantation (FMT) to determine whether susceptibility to migraine-like pain could be induced in mice via FMT from a patient with migraine.
Methods
Animals
To determine whether the gut microbiota influences migraine-related hyperalgesia, four groups of adult male C57BL/6J mice with different gut microbiota statuses were used in this study as follows: Specific pathogen-free (SPF) mice; GF mice; SPF mice treated with antibiotics to deplete the gut microbiome (ABX mice); and GF mice colonized with the gut microbiota from SPF mice (GFC mice). To further support the idea that the gut microbiota is involved, the guts of GF mice were transplanted with the gut microbiota from a patient with migraine (M_mice) or a sex-and age-matched healthy control (H_mice) (Figure 1). All animals were allowed to acclimatize for 1 week before the experimental procedures and were randomly assigned to experimental groups with the rand() function of Excel software. SPF mice were purchased from SIPEIFU (Beijing) Biotechnology Co., Ltd., and GF mice were bred in flexible film isolators at the Institute of Laboratory Animal Science, Chinese Academy of Medical Sciences & Peking Union Medical College. Mice were maintained under a 12-h light-dark cycle in a temperature-controlled (22–25°C) environment with free access to food and water. All experimental procedures were approved by the Ethics Committee of the Chinese PLA General Hospital (ethics no. S2018–126–01, clinical registration no. ChiCTR1800019629), and followed the ethical guidelines of the International Association for the Study of Pain in conscious animals (17). All efforts were made to minimize animal suffering.
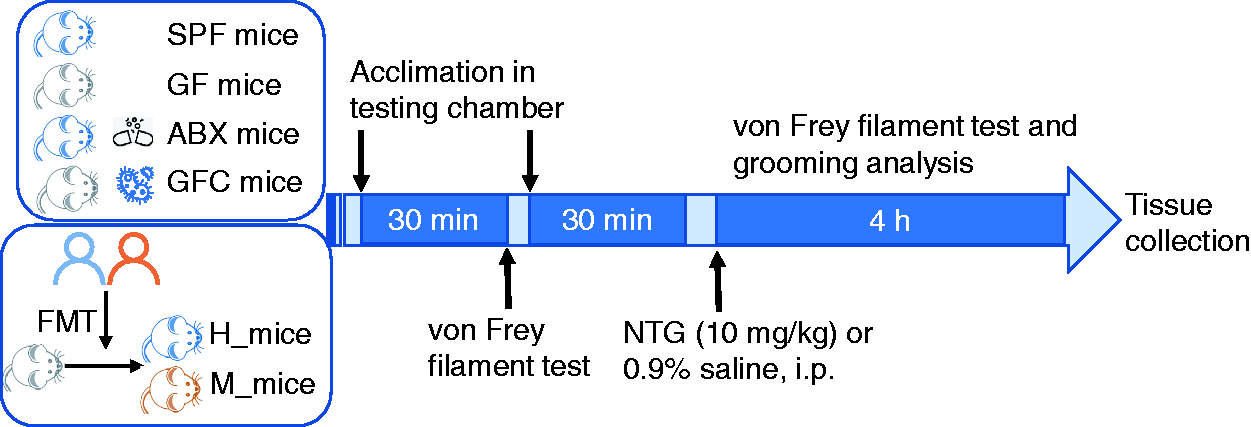
Schematic of the experimental outline.
Behavioural tests
The behavioural tests were performed between 9:00 am and 3:00 pm. Eight- to 10-week-old mice were placed in a testing chamber made of crystal acrylic plates (8 × 8 cm) with a wire mesh surface. The mice were allowed to acclimate for 30 min in the testing chamber before testing to reduce stress (18). To determine the mechanical pain threshold, a series of ascending von Frey monofilaments (North Coast Medical Co., Gilroy, CA, USA) was used (18). The withdrawal mechanical threshold was defined as the filament gauges (g) at which the mouse exhibited a positive response to at least three of six applications. A positive response was indicated by withdrawal of the paw from the stimulus or by paw licking. The NTG experiment was then performed 30 min after basal mechanical threshold determination.
NTG (10 mg/kg, Beijing YiMing, Beijing, China) or the same volume of 0.9% saline vehicle was administered via intraperitoneal (i.p.) injection. NTG was prepared from a stock solution of 5.0 mg/ml nitroglycerin in 30% alcohol, 30% propylene glycol, and 0.9% saline. Because there was no significant difference in the mechanical thresholds observed in mice treated with 0.9% saline or vehicle (6% propylene glycol, 6% alcohol, 0.9% saline) (19), the control used in the present study was 0.9% saline. The mechanical thresholds were measured at 30-min intervals for 4 h after administration. For the grooming time analysis, each mouse was videotaped by an overhead camera throughout the 4-h test, and the time spent on body grooming and face grooming was analysed manually. Face grooming was identified as grooming the craniofacial area with the forepaw(s) or a hind paw. Body grooming was defined as licking of the forepaws, belly, genitals or tail or grooming any area of the body with the forepaw(s) or a hind paw. The investigators were blinded to the treatment during behavioural measurements and analysis.
Antibiotic treatment
Eight-week-old SPF mice were given a cocktail of ampicillin (100 mg/kg), neomycin (100 mg/kg), metronidazole (100 mg/kg), and vancomycin (50 mg/kg) orally once daily for 2 weeks, which was adapted from previously described methods (20). The mice were transferred to flexible film isolators with autoclaved food and autoclaved water 1 week before antibiotic treatment until the day of NTG administration. 16S rRNA gene sequencing was used to confirm bacterial clearance.
FMT
The FMT method was adapted from a previous report (21). Faecal samples were freshly collected from SPF mice or human donors, suspended in 10 ml/1 g saline solution containing 30% glycerol and stored at −80°C. On the transplantation day, the suspension was warmed in a 37°C water bath for 30 min and filtered through sterile gauze. Six- to 8-week-old GF mice were colonized with 300 µl of suspension administered once via oral gavage. Two weeks later, faecal pellets of the recipient mice were collected for validation of colonization status by 16S rRNA analysis, and an NTG experiment was then performed.
16S rRNA gene sequencing
Bacterial genomic DNA was extracted from frozen mouse faecal samples using a QIAamp PowerFecal Kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. The quality of the DNA was checked by 1% agarose gel electrophoresis. 16S rRNA gene V3-V4 sequencing was performed on the MiSeq platform at Allwegene Technology Co. Ltd (Beijing). Raw sequence reads were screened using Trimmomatic (version 0.36). Then, qualified reads were clustered into operational taxonomic units (OTUs) with 97% similarity. The Ribosomal Database Project (RDP) Classifier tool was used for taxonomic classification (22). A Venn diagram was constructed in R to analyse the numbers of common and unique OTUs between groups. To estimate the bacterial diversity, the Shannon index, indicating α-diversity, was calculated using Mothur 1.31.2 software. The primary biomarkers contributing to differences between two groups were assessed with linear discriminant analysis effect size (LEfSe) combined with a nonparametric Wilcoxon rank-sum test.
Immunofluorescence staining
After adequate anaesthesia with 1% pentobarbital sodium (80 mg/kg), the mice were perfused intracardially with 20 ml of precooled 0.9% saline and then with 20 ml of 4% paraformaldehyde (PFA)/0.1M PBS. Brain tissue was removed and fixed in 4% PFA for 12 h at 4°C, dehydrated in 30% sucrose at 4°C, embedded in Tissue-Tek OCT Compound (Sakura Finetek, Torrance, CA, USA) and sectioned at 20 μm using a Leica CM1950 cryostat. The sections were permeabilized in 0.5% Triton X-100, blocked with 10% goat serum (ZLI-9005; ZSGB-BIO) at room temperature for 1 h, and incubated overnight at 4°C with diluted primary antibodies against c-Fos (1:1000, ab222699; Abcam) or calcitonin gene-related peptide (CGRP) (1:200, 24112, ImmunoStar). Sections were then incubated for 2 h at room temperature with anti-rabbit secondary antibodies conjugated to Alexa Fluor 488 (1:2000, ab150077, Abcam). Images were acquired using a microscope (DP73; Olympus, Tokyo, Japan). c-Fos expression was quantified by counting total c-Fos-positive cells from five sections from the trigeminal nucleus caudalis (TNC) area (three visual fields per section). The %area in the TNC for CGRP quantification was measured with ImageJ, and all results of the NTG group were normalized to those of the control group, which were processed simultaneously.
Enzyme-linked immunosorbent assay (ELISA) assay
Tissues were collected and stored at −80°C until they were used for the assay. TNF-α levels in the TNC tissue homogenate were determined with an ELISA kit (Elabscience Biotechnology Co., Ltd., Wuhan, China) according to the manufacturer’s instruction.
Statistical analysis
Sample size was determined with previous studies (23,24), and post hoc power analysis with G*power (3.1) (25) was performed to confirm that the power (1-β) was up to 0.8. Statistical analysis was performed using GraphPad Prism software (GraphPad Prism 8). Data were assessed for distribution normality and plotted in the figures as the mean ± standard error of the mean (SEM) or as a box diagram. For parametric data, a two-tailed unpaired Student’s t-test was used to analyse the difference between two groups. One-way ANOVA with Dunn’s or Holm-Sidak’s multiple comparisons test was performed when one variable was evaluated, and two-way repeated ANOVA with Sidak’s multiple comparisons test was used for comparisons of two variables between two or more groups. For nonparametric data, the Mann-Whitney U test was used to analyse the difference between two groups; p < 0.05 was considered significant.
Results
1. Increased basal hyperalgesia, but no further hyperalgesia was induced by NTG administration in mice without gut microbial colonization
To determine the influence of the gut microbiome on NTG-induced hyperalgesia, we determined the mechanical threshold and grooming time induced by NTG or 0.9% saline administration in SPF and GF mice. We first examined the basal mechanical threshold in the hind paws. The basal mechanical threshold (Figure 2(a)) was lower in GF mice than in SPF mice. After NTG administration, SPF mice showed a significantly decreased mechanical threshold at 1.5 h and 2 h compared with saline treatment (Figure 2(b)). In addition, in SPF mice, an obvious resting state was observed in the first 30 min after NTG treatment, during which body grooming time was significantly decreased. There were significant increases in body grooming times during the 1–1.5 h after NTG administration (Figure 2(c)). In contrast, the mechanical threshold and grooming time in GF mice were not significantly different between the NTG group and saline group (Figure 2(b),(c)). To confirm the hyporesponsiveness to NTG administration in GF mice, we detected c-Fos and CGRP expression in the TNC to determine whether the trigeminovascular system was activated. The number of c-Fos-ir cells was significantly increased and the area % of CGRP-ir signalling was significantly decreased after NTG administration compared with saline administration in SPF mice, while there was no significant difference in signalling between the NTG- and saline- treated GF mice (Figure 2(d)–(f)).

Increased basal hyperalgesia but no significant differences in hyperalgesia induced by NTG administration compared with saline in mice reared devoid of microbial colonization. (a) The basal mechanical thresholds in the hind paw. Compared with SPF mice, the basal mechanical threshold was lower in GF mice (*p = 0.0211) and ABX mice (****p < 0.0001). The mechanical threshold was lower in ABX mice than in GF mice (**p = 0.0059). Kruskal–Wallis test with Dunn’s multiple comparisons test. n = 20, 20, 16 per group. (b) A decreased mechanical threshold was induced by NTG administration in SPF mice but not in GF mice. The SPF mice showed significantly decreased mechanical thresholds at 1.5 h and 2 h after NTG administration compared with mice treated with saline. Two-way repeated ANOVA, time F (6, 63) = 5.241, p= 0.0002; Treatment F (1, 63) = 15.26, p = 0.0002; **p = 0.0029 (1.5 h), **p = 0.0019 (2 h). No significant difference was found in GF mice between the NTG and saline groups. (c) Body grooming time induced by NTG administration was altered in SPF mice but not GF mice. The SPF mice showed significantly decreased body grooming time during the first 30 min (***p = 0.0003), and increased body grooming time during 1–1.5 h (*p = 0.0289). Two-way repeated ANOVA with Sidak’s test, time F (4.495, 80.91) = 5.882, p = 0.0002, treatment F (1,18) = 5.527, p = 0.0303. n = 10 per group. (d) Increased c-Fos expression in the TNC was induced by NTG administration in SPF mice but not in GF mice. Unpaired t-test, **p = 0.0054, n = 4 per group. (e) The area % of CGRP-ir in the TNC decreased significantly with NTG administration compared with saline treatment in SPF mice (*p = 0.0191) but not in GF mice. Unpaired t-test, n = 4 per group. (f) Representative immunofluorescence images of CGRP expression in the TNCs of SPF and GF mice treated with saline vehicle or NTG. Scale bar: 100 µm. Data are presented as the mean ± SEM.
Moreover, we investigated whether NTG administration could induce significant hyperalgesia in antibiotic-treated SPF mice to deplete the gut microbiota. 16S rRNA sequencing showed that the α-diversity was significantly decreased after antibiotic treatment (Figure 3(a)). The ABX mice showed a lower basal mechanical threshold than SPF and GF mice (Figure 2(a)). No significant difference was observed in mechanical threshold (Figure 3(b)), c-Fos expression or CGRP expression in the TNC between those mice with NTG administration and those with saline treatment (Figure 3(b)–(e)). These results suggest that the depletion of the gut microbiome induced basal mechanical hyperalgesia and that no further hyperalgesia was induced by NTG administration in those mice.

No significant differences in hyperalgesia induced by NTG administration compared with the administration of saline vehicle in mice with gut microbiome depleted by antibiotic treatment. (a) Apparent decreased α-diversity (as assessed by the Shannon index) induced by 2 weeks of antibiotic treatment. Bf- ABX means before antibiotics treatment. Mann–Whitney test, ***p = 0.0002, n = 8 per group. The data are expressed as median + min-to-max values. (b) No significant alteration in the mechanical threshold induced by NTG administration compared with saline administration in ABX mice. Two-way repeated ANOVA with Sidak’s test, n = 8 per group. (c)–(e) No significant alteration of c-Fos expression (c) or area % of CGRP-ir (d) in the TNC induced by NTG administration compared with saline administration in ABX mice. Unpaired t test, n = 4 per group. (e) Representative immunofluorescence images of CGRP expression in the TNC of ABX mice treated with saline vehicle or NTG. Scale bar: 100 µm. (f) The concentration of TNF-α in the TNC determined by ELISA. The concentration of TNF-α was increased after NTG administration compared with saline treatment in SPF mice (*p = 0. 0.0222, unpaired t-test). No significant difference was found between the saline group and the NTG group in GF and ABX mice. The TNF-α concentration of saline group: GF mice > ABX mice > SPF mice. (one-way ANOVA with Tukey’s test, *p = 0.0473, GF vs. ABX; **p = 0.0097, SPF vs. ABX; ***p = 0.0002, SPF vs. GF). n = 4 per group. Data are presented as the mean ± SEM.
Inflammatory pathways are initiated by NO released from NTG-activated trigeminal fibres in an NTG mouse model (26). To gain insight into whether the inflammatory pathway is activated in mice without gut microbial colonization, we determined the concentration of TNF-α in the TNC tissue using ELISA. TNF-α concentration was increased after NTG administration compared with saline treatment in SPF mice. However, no significant difference was found between the saline group and NTG group in GF and ABX mice. In addition, the TNF-α concentration of the saline group was higher in GF and ABX mice than in SPF mice (Figure 3(f)).
2. Restoring the microbiota of GF mice reverses basal mechanical hyperalgesia and hyporesponsiveness to NTG administration
To determine whether hyporesponsiveness of GF mice to NTG administration could be reversed by gut colonization, we transplanted gut flora from SPF mice into GF mice (referred to as GFC mice). Two weeks later, the GFC mice had a gut microbiota composition similar to that of the SPF donor mice (Figure 4(a),(b)). The basal mechanical threshold was higher in GFC mice than in GF mice (Figure 4(c)). After NTG administration, a reduced mechanical threshold in the hind paw at 1 h (Figure 4(d),(e)) after NTG administration was observed compared with the saline group. No significant difference was observed in c-Fos expression in the TNC (data were not shown). The area % of CGRP-ir signalling in the TNC was significantly lower after NTG administration than after saline administration in GFC mice (Figure 4(f),(g)). The response to NTG injection in GFC mice was similar to that in SPF mice, suggesting that the colonization of the gut microbiome reverses hyporesponsiveness to NTG in GF mice.
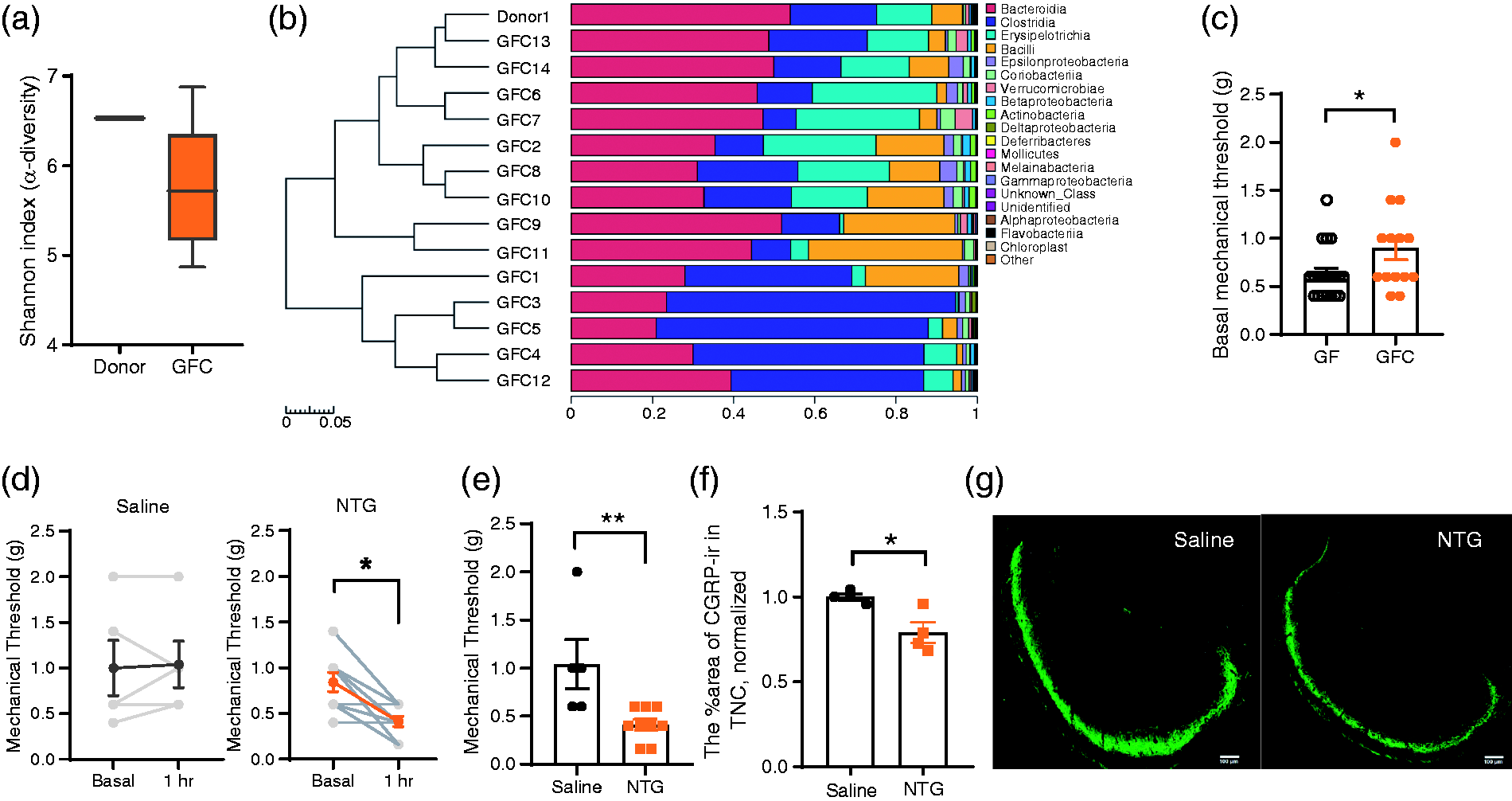
NTG administration induced significant hyperalgesia compared with administration of saline vehicle in GF mice receiving the gut microbial profile from SPF donor mice. (a) α-diversity analysis (as assessed by the Shannon index) of SPF donor and recipient GFC mice. The data are expressed as median + min- to- max values. (b) A heatmap reflecting the scaled relative abundance of bacteria and hierarchical cluster analysis of samples. The top 20 in class level are included. (c) Increased basal mechanical threshold after gut microbial colonization. *p = 0.0493, Mann–Whitney test, n = 20, 14 per group. (d) Decreased mechanical threshold induced by NTG administration not by saline treatment between baseline and the 1 h timepoint in GFC mice. Wilcoxon matched-pairs signed rank test, *p = 0.0156, n = 9 pairs. (e) Lower mechanical threshold at 1 h after NTG administration than saline administration in GFC mice. Mann–Whitney test, **p = 0.007, n = 5,9 per group. (f) The area % of CGRP-ir in the TNC was significantly lower after NTG treatment than after saline treatment in GFC mice, and representative immunofluorescence images are shown in (g). Scale bar: 100 µm. Unpaired t test, *p = 0.0154. n = 4 per group. Data are presented as the mean ± SEM.
3. Susceptibility to migraine-like pain was induced in mice via FMT from a patient with migraine
Because of basal hypersensitivity, it was difficult to confirm that NTG-induced hyperalgesia was affected by the gut microbiota. To further investigate the causality of gut microbiota in the pathogenesis of migraine, we colonized the guts of GF mice with microbiota samples from a representative male patient with migraine via FMT. The patient was selected based on the gut microbiota composition and a sex- and age-matched healthy control was selected (Table S1). After 2 weeks of colonization, 16S rRNA sequencing showed different OTU numbers (Figure 5(a)) and primary genera (Figure 5(c)) between M_mice and H_mice, and the Shannon index, indicating α-diversity in M_mice was lower than that in H_mice (Figure 5(b)). The basal mechanical threshold was slightly lower in M_mice than H_mice but with no significance (Figure 5(d)). After NTG administration, a decreased mechanical threshold (Figure 5(e), (f)) and more face and body grooming time during 1–1.5 h (Figure 5(g), (h)) were observed in M_mice not in H_mice. These findings indicate that NTG-induced hyperalgesia was more obvious in recipient mice colonized with gut microbiome sample from a migraineur than in those colonized with sample from a matched healthy control and that susceptibility to migraine-like pain could be transmissible by FMT.

NTG administration induced more severe hyperalgesia in GF mice receiving the gut microbiota from a patient with migraine than in those that received gut microbiota from a healthy control. (a) A Venn diagram of the numbers of OTUs. The numbers in the hollow circle represent the OTUs in recipient mice transplanted with the gut microbial community of a healthy control (H_mice). The numbers in the filling circle represent the OTUs in recipient mice transplanted with the gut microbial community of a patient with migraine (M_mice). (b) α-diversity analyses (as assessed by the Shannon index) of H_mice and M_ mice. The Shannon index of the H_mice was higher than that of the M_mice (unpaired t-test, n = 9 per group, ****p < 0.0001). (c) LEfSe of species with significant abundance differences between the M_mice and H_mice. The linear discriminant analysis (LDA) threshold was set as 3. (d) The basal mechanical threshold of H_mice and M_mice. Mann–Whitney test, p = 0.1534, n = 9 per group. (e) Variations of mechanical threshold after NTG administration of M_mice and H_mice. Results are expressed as Δwithdrawal threshold (in g) calculated by subtracting zero-time threshold from the time threshold. (f) Decreased mechanical threshold after NTG administration in M_mice not in H_mice. Paired t-test, **p = 0.0021, n = 9 per group. (g),(h) More body and face grooming time during 1–1.5 h after NTG administration in M_mice than H_mice. Unpaired t-test, **p = 0.0055, *p = 0.0402, n = 9 per group. Data are presented as the mean ± SEM.
Discussion
In recent years, gut microbiota disturbance has been speculated to be involved in the pathogenesis of migraine, but the association remains unsubstantiated. In this study, we found increased basal mechanical sensitivity and no further hyperalgesia induced by NTG administration in mice without gut microbial colonization. Restoring the gut microbiota reversed basal mechanical hyperalgesia and hyporesponsiveness to NTG administration. Moreover, susceptibility to migraine-like pain was successfully reproduced from patient to mice via FMT.
In this study, our results showed basal mechanical hyperalgesia and higher concentrations of TNF-α in the TNC of mice without microbial colonization. In a study focusing on visceral pain and microbiota, increased gene expression level of TNF-α was also observed in the spinal cord of GF mice (13). The increased cytokine level in the spinal cord may facilitate the transmission of sensory information from the hind paw and contribute to basal mechanical hyperalgesia. This finding suggests that gut microbiota is required for normal pain sensation. We also noticed a lower basal mechanical threshold in mice treated with antibiotics to deplete the gut microbiota than in mice reared devoid of microbial colonization. The inconsistent degree of basal hyperalgesia may be due to chronic GF rearing and acute antibiotics exposure. Besides, a study by Vuong et al. (27) reported that short-term depletion of the maternal gut microbiome during pregnancy to mid-gestation caused decreased mechanical sensitivity in adult offspring due to deficient thalamocortical axons, suggesting that the maternal microbiota modulates the sensitivity of offspring. The GF mice we used in this study were adult offspring of GF mice with long-term microbial depletion. Therefore, the relatively less basal hyperalgesia in GF mice than ABX mice may be the compromise of decreased mechanical sensitivity caused by absence of maternal microbial colonization and the increased mechanical sensitivity caused by the absence of its own gut flora. The specific mechanism for how depletion of gut microbiota influences the host sensitivity needs further research.
Our results showed that no further hyperalgesia was induced by NTG administration in mice without gut microbial colonization. Because of basal mechanical hyperalgesia, it remains to be determined whether depletion of the gut microbiota reduces NTG-induced hyperalgesia. A study reported no local (intestine), remote (lung), or systemic inflammatory response or lethality in GF mice but marked inflammatory responses and lethality in SPF mice after intestinal ischaemia and reperfusion injury (28). This finding suggests that that the lack of intestinal microbiota was accompanied by a state of inflammatory hyporesponsiveness. Inflammatory hyporesponsiveness may contribute to hyporesponsiveness to NTG administration in GF mice, but further research is needed to verify this hypothesis.
HMA models are considered a foundation for addressing the causal relationships between altered microbial profiles and host pathology (29). In our study, a susceptibility state to NTG-induced migraine-like pain was transmissible from patient to mice via FMT. This study provides direct evidence for the causality of gut microbiota disturbance in the pathogenesis of migraine. In addition, because the gut microbiota is affected by sex differences (30,31), to avoid the potential influence of sex, the gut microbiota of a male patient was transplanted to the gut of GF male mice in our study. After 2 weeks’ colonization, we also noticed that the α- diversity in M_mice was lower than in H_mice, which was consistent with our clinical findings that the α-diversity was decreased in patients with migraine compared with healthy controls (unpublished). The involvement of gut microbiota disturbance in migraine pain may also provide new insights into the familial aggregation of migraine (32). A large population-based cohort study revealed a stronger association between maternal migraine and offspring migraine than paternal migraine and offspring migraine (33). The potential modulations of maternal gut microbiota dysbiosis on foetal neurodevelopment (34,35) may contribute to the pathogenesis of offspring migraine and this may be one of the mechanisms of stronger association between maternal migraine and offspring migraine.
In the present study, our results showed an important role of the gut microbiota in host basal mechanical sensitivity and its involvement in the pathogenesis of migraine. However, the pathways by which the gut microbiota acts on migraine-related pain have not yet been defined and further research is needed.
Conclusion
The present study revealed the dual effects of the gut microbiome on host basal sensitivity as well as migraine-related hyperalgesia in mice. Further studies focusing on the specific mechanism are needed to ultimately develop novel therapies for the prevention or treatment of migraine.
Article highlights
Depletion of the gut microbiome increased basal mechanical sensitivity in mice. Restoring of the gut microbiota reversed hyporesponsiveness to NTG administration in mice without gut microbial colonization. NTG administration induced more severe hyperalgesia in GF mice receiving the gut microbial profile from a patient with migraine than those that received gut microbiota from a healthy control.
Footnotes
Acknowledgement
The authors are grateful to Senior Technologist Zhu Hua and her colleagues at the Institute of Laboratory Animal Science, Chinese Academy of Medical Sciences & Peking Union Medical College for helping breed germ-free mice.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grants 81600952, 81671077, 81771180, 81771200, 81901134, and 81901145), Beijing Natural Science Foundation Essential Research Project Z170002.
