Abstract
Abstract
Objective
The aim of the TOP-PRO-study, a double-blind randomized controlled trial, was to assess the efficacy (non-inferiority) and tolerability of propranolol compared to topiramate for the prevention of chronic migraine.
Background
Except for topiramate, oral preventive treatment for chronic migraine lacks credible evidence.
Methods
Chronic migraine patients aged above 18 years and less than 65 years of age, not on any preventive treatment were randomly allocated to receive topiramate (100 mg/day) or propranolol (160 mg/day). The primary efficacy outcome was the mean change in migraine days per 28 days at the end of 24 weeks from baseline. A mean difference of 1.5 days per four weeks was chosen as the cut-off delta value. Multiple secondary efficacy outcomes and treatment emergent adverse events were also assessed.
Results
As against the planned sample size of 244, only 175 patients could be enrolled before the spread of the corona virus disease-2019 pandemic and enforcement of lockdown in India. Of the 175 randomized patients, 95 (topiramate 46 and propranolol 49) completed the trial. The mean change in migraine days was −5.3 ± 1.2 vs −7.3 ± 1.1 days (p = 0.226) for topiramate and propranolol groups respectively. Propranolol was found to be non-inferior and not superior to topiramate (point estimate of −1.99 with a 95% confidence interval of −5.23 to 1.25 days). Multiple secondary outcomes also did not differ between the two groups. Intention to treat analysis of 175 patients and per-protocol analysis of 95 patients yielded concordant results. There was no significant difference in the incidence of adverse events between the two groups.
Conclusion
Propranolol (160mg/day) was non-inferior, non-superior to topiramate (100mg/day) for the preventive treatment of chronic migraine and had a comparable tolerability profile.
Trial Registration: Clinical Trials Registry-India CTRI/2019/05/018997)
Introduction
Chronic migraine (CM) is defined as headache occurring on ≥15 days/month for more than 3 months, having features of migraine headache on at least 8 days/month (1). A systematic review of 12 population-based studies has estimated the prevalence of CM to be 1.4 to 2% (2). CM is a highly disabling condition (3). The mainstay of treatment of CM is preventive therapy. However, the treatment options in CM are limited. Food and Drug Administration (FDA) approved repeated multiple injections over the face, neck, shoulder, and head of Onabotulinum toxin A (OBT-A) for prevention of CM based on Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) trials (4). Recently, monoclonal antibodies against calcitonin gene-related peptide (CGRP) have shown good efficacy in CM (5). However, both these treatments are expensive.
None of the oral established agents for prevention of episodic migraine (EM) have undergone robust clinical trials in CM except for topiramate (6). Two pivotal randomized controlled trials (RCT) by Silberstein et al. (7) and Diener et al. (8) in 2007 comparing topiramate with placebo in CM had shown good efficacy at a dose of 100 mg/day although treatment-emergent adverse events (TEAE) were high (75–85%) and led to treatment withdrawals in 10.9% and 18.8% of patients respectively. Propranolol has been inadequately studied for the treatment of CM in three studies conducted in 1980, 1983, and 2009 (9–11). It is noteworthy that during 1980s, the concept of CM was rudimentary and proper definition of CM was non-existent. The results of the first two studies were negative (9,10). However, in the third study done in 2009, using ICHD-2 criteria for CM, propranolol alone and its combination with nortriptyline were significantly better than nortriptyline alone in terms of 50% responder rate (defined as ≥50% reduction in headache days from baseline) and reduction in mean number of headache days/month (11). Silberstein et al. studied the role of adding propranolol to topiramate in patients of CM who did not benefit from topiramate alone (12). They did not find any significant difference between those receiving topiramate alone versus topiramate with propranolol. On the other hand, propranolol has class I evidence of efficacy for the prevention of episodic migraine (EM) (13). It is cheap and has an excellent safety profile. Further, in a head-to-head comparative trial of topiramate with propranolol for EM, both have been found to be equally effective (14). Hence, an RCT, called TOP-PRO study was conducted to assess whether propranolol (160 mg/day) is as efficacious (non-inferior) and safe as topiramate (100 mg/day) for the prevention of CM.
Methods
Study participants
Inclusion criteria
All patients above 18 years and less than 65 years of age, male or female who fulfilled the diagnostic criteria laid down by ICHD 3 for CM1 (with or without aura) and were not on any preventive treatment for CM were included.
Exclusion criteria
All patients with the clinical phenotype of CM but on further investigation, found to have a secondary cause for their headaches were excluded. Pregnant women, patients with known allergies against propranolol and/or topiramate, patients with a history of bronchial asthma, chronic obstructive pulmonary disease, diabetes, bradycardia or heart blocks, congestive heart failure, glaucoma, renal stones, dementia, psychosis and severe depression were excluded from the study.
Exit Criteria
The participants who developed systolic blood pressure (SBP) less than 90 mm Hg and bradycardia i.e., heart rate less than 60/min., or developed serious adverse effects (SAE) or intolerability to the drugs or those who were not willing to continue with the study.
Trial design
This interventional, double-blind RCT with a non-inferiority design was conducted in a tertiary care headache clinic at Govind Ballabh Pant Institute of Postgraduate Medical Education and Research (GIPMER), New Delhi, India from May 2019 to February 2021. The trial design is shown in Figure 1. Consecutive preventive drug-naive CM patients fulfilling inclusion and exclusion criteria were screened for a period of four weeks. Patients did not receive any preventive medications during the screening period and only acute treatments were allowed. During the screening period, patients were required to fill a paper headache diary mentioning the days with or without headache, the days with or without migraine, visual analog scale (VAS) score for each day of migraine or headache and the days with or without acute migraine treatment (AMT) (analgesics, triptans). A complete set of routine investigations including electrocardiogram (ECG), urine pregnancy test (for reproductive age group females), and MRI (if required based on the presence of any red flags) were done during this period. Headache days, migraine days, AMT days, and VAS scores were recorded in the headache diary during the screening period from which baseline averages of these measures were calculated. Mean headache impact test-6 (HIT-6) scores and mean migraine-specific quality of life (MSQOL) scores were also generated at the end of the baseline period.

Trial design.
Randomization and blinding
Following the screening period of four weeks, the screen failures were excluded and the rest of the patients were randomly allocated to topiramate (Drug A) and propranolol group (Drug B) based on a computer-generated randomization chart generated by a neurology consultant (DC). Sequentially numbered opaque sealed envelopes were prepared and 270 labels were prepared for each drug labeled Drug A or B. They were put into envelopes numbered from 1 to 270 according to the randomization chart by a neurology resident (DD). Envelopes were attached to the patient's proforma. Patients were recruited sequentially and were given enrolment numbers starting from 1 which were mentioned on their outpatient cards. Proforma carrying the same number as the patient enrolment number was then opened and the patients were assigned to receive drug A or B based on their randomization by second neurology resident (AM). Drugs were supplied in identical boxes labeled A or B with 100 tablets each of the minimum dose of each drug (25 mg for topiramate and 40 mg for propranolol) and distributed to the patients by another neurology resident (LB). Neither the investigator nor the patient was aware of the composition of drugs A and B. The entire double-blind trial period consisted of 24 weeks with initial 4 weeks of dose titration. After 4 weeks of titration, the patients received either 160 mg of propranolol (80 mg twice a day) or 100 mg of topiramate (50 mg twice a day). The dose schedule is shown in Table 1. No other preventives were allowed as concomitant medication.
Dosing schedule of the investigational drugs.

This study was conducted in accordance with the International Council for Harmonisation Good Clinical Practice regulations and guidelines (15). The final study protocol, informed patient consent forms and accompanying materials were reviewed and approved by the institutional ethics committee.
Primary and secondary outcomes
The primary efficacy outcome was mean change in migraine days per 28 days (migraine day was defined as a calendar day when the patient reported ≥4 continuous hours of headache meeting ICHD-3 criteria for migraine) at the end of 24 weeks. Secondary efficacy outcomes were mean change per 28 days at the end of 24 weeks in headache days (headache day was defined as a calendar day [00:00 to 23:59] when the patient reported ≥4 continuous hours of headache episode in the diary) and achievement of more than 50% reduction in headache days compared to baseline. Additional secondary efficacy outcomes included mean change per 28 days at the end of 24 weeks in AMT days, average VAS score, and HIT-6 score. Physician’s global impression of change (PGIC) score was also calculated and the means were compared between the two groups at the end of 24 weeks. Treatment-emergent adverse events (TEAE) including SAE were documented.
Patient evaluation
Patients were evaluated as per a detailed structured proforma covering all the aspects of clinical characteristics of headache including detailed family history. A specific list of prodrome, postdrome, triggers, and cranial autonomic symptoms (CAS) were assessed for presence or absence. Cephalic allodynia was confirmed by a simple question requiring a “yes/no” response as to whether the patients experienced pain on touching their head or not, during or after a headache attack. Patients were classified as with co-existent medication overuse headache (1) (MOH) as defined by ICHD-3 in the two treatment groups. Clinical examinations, including pulse and blood pressure measurements, were done on each visit and average headache days, migraine days, VAS score, MSQOL score, AMT days and HIT-6 score per 28 days was estimated. Adverse events, if any were captured. The study drugs were tapered and stopped at the end of 28 weeks. Patients who were still symptomatic and wished to continue the treatment were given the choice of treatment as per the standard therapy including topiramate or other oral preventive drugs such as flunarizine, divalproate, or amitriptyline, or greater occipital nerve blocks or OBT-A after excluding any specific contraindications.
Statistical analysis
Sample size calculation
This study compared the effectiveness of propranolol (160 mg/day) versus topiramate (100 mg/day) at the end of 20 weeks after 4 weeks of titration (24 weeks of drugs in total). The key outcome parameter was mean change in migraine days (per 28 days) at the end of 24 weeks compared to baseline. The predetermined hypothesis was H1: propranolol is not inferior to topiramate (non-inferiority analysis). The control drug was topiramate. H1 was tested in accordance with consolidated standards of reporting trials (CONSORT) recommendations for non-inferiority analysis by both intention to treat (ITT) and per-protocol (PP) analysis (16). The last observation carried forward (LOCF) was used for ITT analysis. A mean difference of 1.5 days per four weeks was chosen as the cut-off delta value. The expected treatment effect difference between the active drug and control drug had been estimated from a previous CM trial comparing topiramate with placebo (8). Considering the 1:1 randomization ratio, with a power of 80% and a one-sided 95% CI, a sample size of 244 (122 in each group) was estimated. Keeping 10% loss to follow up, 135 subjects were planned to be enrolled in each group.
Demographic characteristics were summarized using descriptive statistics. Categorical data were summarized as frequencies and percentages. Continuous data were summarized as means. The primary and secondary efficacy (except >50% responder rate and PGIC score) outcomes were analyzed using a linear model with baseline value as a covariate and treatment as factor. Based on observations per 28 days data during the double-blind treatment phase, pair-wise comparisons of topiramate versus propranolol were obtained. To study the homogeneity between the two groups for each outcome parameter, Levene's test of equality of error variances (that tests the null hypothesis that the error variance of the dependent variable is equal across groups) was performed. The Cochran-Mantel-Haenszel pairwise test was used to assess treatment differences in the proportion of patients in the two groups who achieved >50% reduction in headache days compared to baseline at the end of 24 weeks. Mean PGIC scores at the end of 24 weeks were compared using an independent sample student-t-test. For non-inferiority analysis, point estimates of the primary outcome, that is, mean change in migraine days per 28 days at 24 weeks of the control drug (topiramate) minus test drug (propranolol) (C-T) with 95% confidence interval (CI) were obtained. The level of significance was set at p < 0.05. The patient incidence of TEAE was reported using the system organ class and preferred term by treatment group. Separate summaries were provided for adverse events leading to discontinuation of treatment, TEAE, and treatment-related SAE.
Clinical Trials Registry-India (No and Date): CTRI/2019/05/018997 on 8/5/2019
Results
The study was conducted from 13th May 2019 to 19th March 2020. During this period, 376 CM patients attended the headache clinic. Out of these, 215 patients fulfilled the inclusion and exclusion criteria and underwent a screening period of four weeks. There were 40 screen failures. The remaining 175 patients were randomized into two groups, 93 received drug A (topiramate), and 82 received drug B (propranolol). The CONSORT flowchart of the study is depicted in Figure 2. All 175 randomized patients were analyzed by ITT analysis. 95 patients completed the double-blind protocol and were analyzed by PP analysis. 80 patients discontinued treatment at various stages of the double-blind study period. The details of randomization, discontinuation profile and the reasons for discontinuation are shown in Supplementary Table 1. A large number of patients could not complete the double-blind phase due to the coronavirus disease 2019 (COVID-19) pandemic and lockdown. Until November 2019, the discontinuation rate was very low (6/97; 6.2%) but greatly increased following the onset of the pandemic (74/78; 94.9%). Overall, the major reasons for discontinuation from the study were COVID-19 related lockdown and restrictions (67.5%), lack of efficacy (13.8%), drug-related side effects (6.3%), no reasons cited (6.3%), and lost to follow-up (6.3%). The investigators had to stop further recruitment of participants for the study after 19th March 2020 as India enforced one of the strictest lockdowns from 23rd March 2020 onwards.
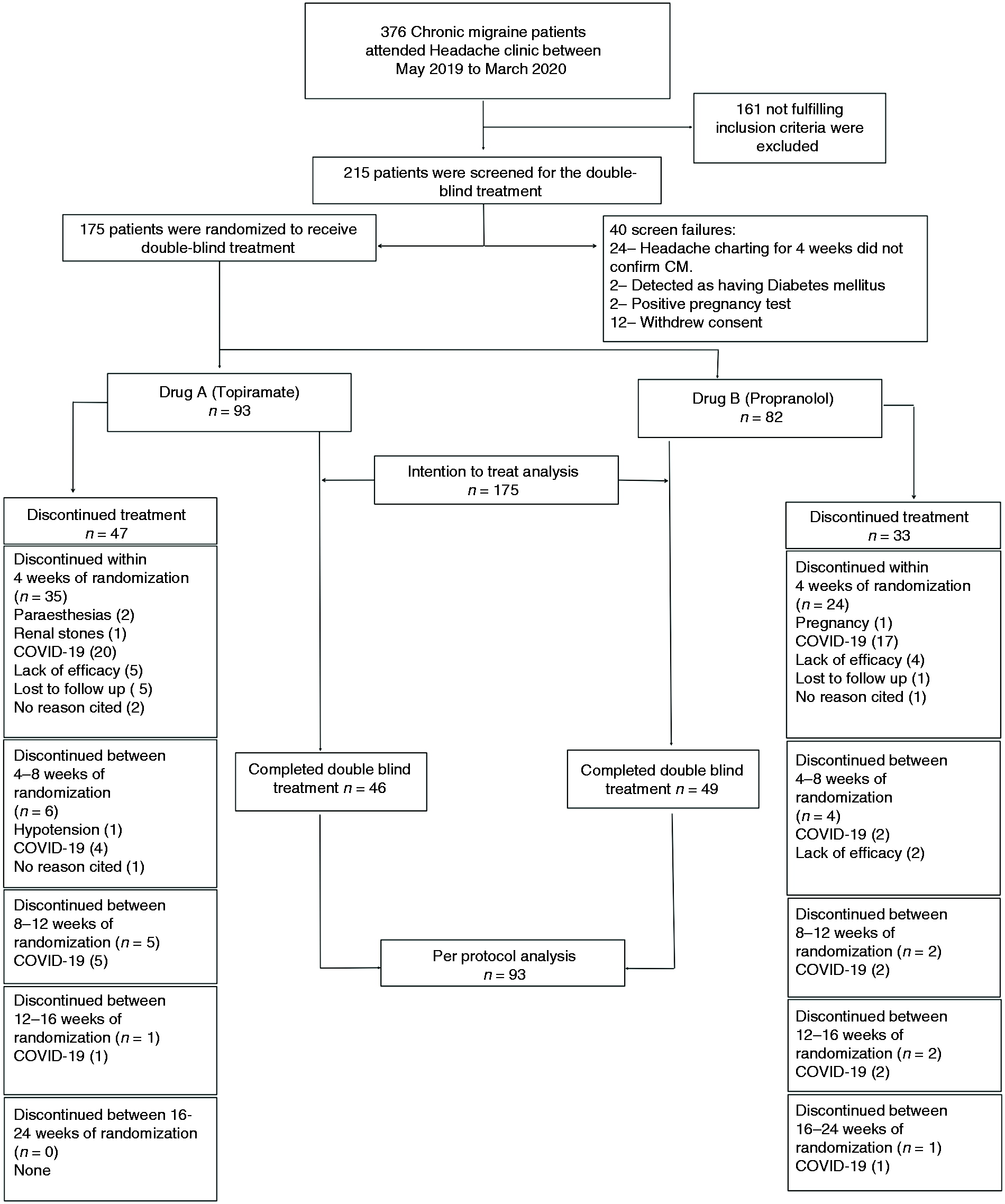
Flow of the patients through the study (figures represent the number of patients).
Demography and headache characteristics: The mean age for patients in the topiramate group was 32.2 years while for patients in the propranolol group, it was 33.9 years. The topiramate group included three males and 90 females while the propranolol group included five males and 77 females. The mean duration of migraine and CM for patients in the topiramate group was 6.1 and 1.3 years while for patients in the propranolol group, it was 5.9 and 1.0 years, respectively. Positive family history of migraine was present in 33.3% of patients in the topiramate group and 28% of patients in the propranolol group. Aura (visual only) was present in 6.4% of patients in the topiramate group and 2.4% of patients in the propranolol group. The headache features of the two groups are compared in Table 2. Lacrimation and facial sweating were the most common cranial autonomic symptoms and fatigue was the most commonly reported premonitory as well as postdrome feature in both groups. Triggers were present in 88.2% of patients in the topiramate group and 87.8% of patients in the propranolol group. Exposure to bright lights (including bright sunlight), noise and travel, were the most commonly reported triggers in both groups.
Demography and baseline headache characteristics.

At baseline, mean migraine, headache, and AMT days per 28 days were 17.3 ± 6.7 vs 17.6 ± 6.6 days; 25.6 ± 4.1 vs 24.6 ± 4.4 days and 16.2 ± 8.9 vs 14.7 ± 8.1 days for patients in the topiramate group and propranolol group respectively. Mean VAS score, HIT-6 score, and MSQOL score per 28 days was 6.0 ± 1.8 vs 5.6 ± 1.8, 63.6 ± 4.5 vs 63.10± 4.6 and 64.0 ± 7.8 vs 62.3 ± 7.7 for patients in the topiramate group and propranolol group respectively. Medication overuse headache was present in 55.9% of patients in the topiramate group and 47.6% in the propranolol group. None of these parameters except mean headache days differed significantly between the two groups (Table 2).
Non-inferiority analysis
Non-inferiority analysis for the primary endpoint i.e., change in mean migraine days per 28 days at the end of 24 weeks after adjusting for baseline migraine days in the regression analysis for the ITT group showed a point estimate of −1.71 with a 95% CI of −3.82 to 0.39 days (p = 0.109). A similar analysis for the PP group showed a point estimate of −1.99 with a 95% CI of −5.23 to 1.25 days (p = 0.226). Thus both ITT and PP analyses showed non-inferiority (and non-superiority) of propranolol over topiramate (Figure 3).

Non-inferiority analysis with point estimates and 95% CI ( ITT and PP analyses).
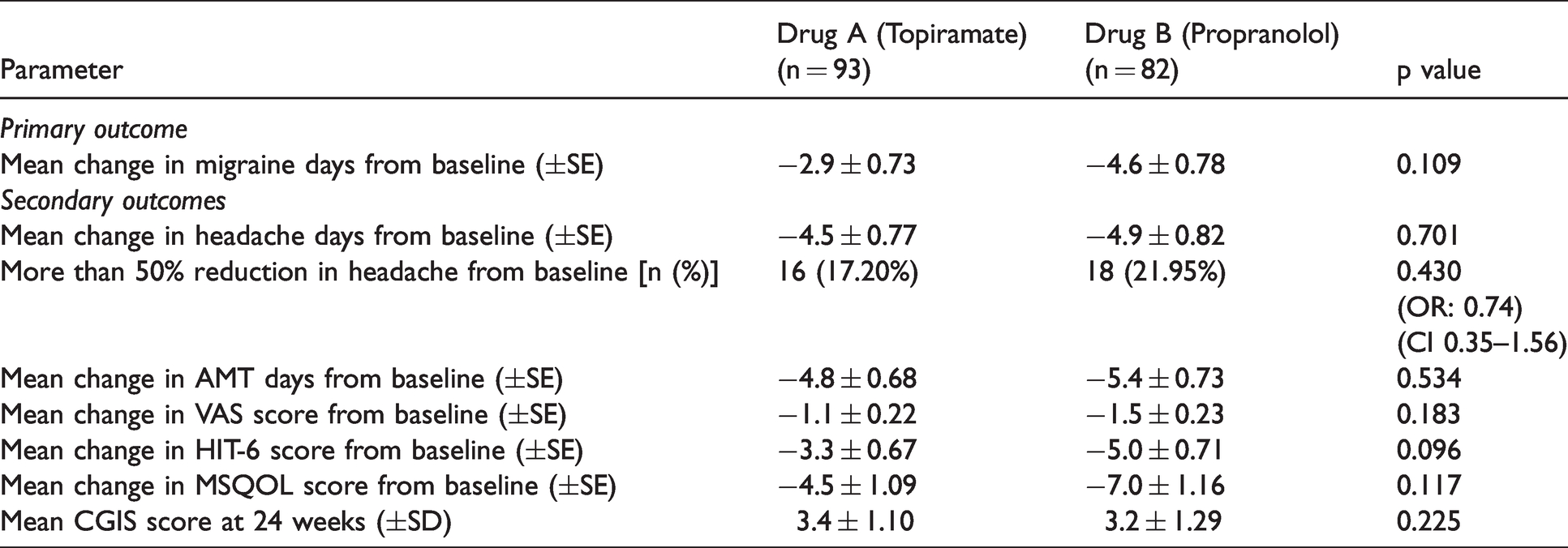
ITT analysis: All the 175 patients who were randomized were analyzed. At the end of 24 weeks, the mean change in migraine days per 28 days was −2.9 ± 0.7 days for the topiramate group while it was −4.6 ± 0.8 days for the propranolol group. The difference was not statistically significant (p = 0.109). The secondary endpoints also did not differ significantly between the two groups at 24 weeks. These results are depicted in Table 3. Presence of MOH had no impact on the primary outcome in both the groups. In the topiramate and propranolol groups, mean change in migraine days per 28 days in patients with MOH as compared to those without MOH at the end of 24 weeks were −2.8 vs −2.9 days (p = 0.929) and −4.3 vs −5 days (p = 0.640) respectively.
Comparison of primary and secondary outcomes of efficacy (ITT analysis) (n = 175).
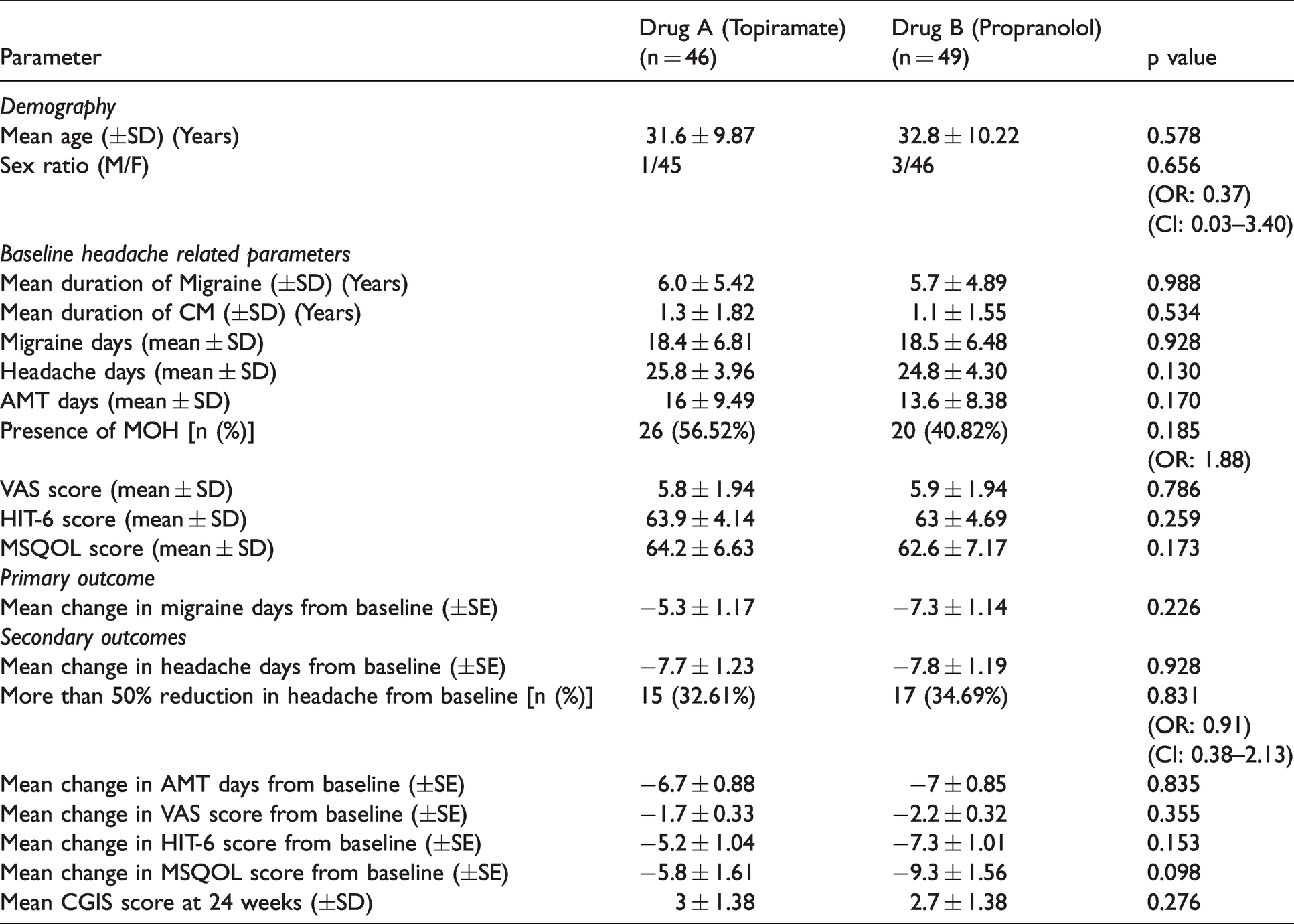
PP analysis: 95 patients completed the double-blind treatment period of 24 weeks and were included in the PP analysis. All of them reached the target dose of propranolol and topiramate. Similar to ITT analysis, the patients who received topiramate (46) were matched with those who received propranolol (49) in terms of mean age at presentation, sex ratio, mean duration of migraine and CM, presence or absence of aura, baseline mean migraine days, and headache days, and AMT days per 28 days at the baseline. They were also matched for mean VAS, HIT-6, and MSQOL scores at the baseline (Table 4). At the end of 24 weeks, the mean change in migraine days per 28 days was −5.3 ± 1.2 days for topiramate while it was −7.3 ± 1.1 days for propranolol. The difference was not statistically significant (p = 0.226). The secondary outcomes also did not differ significantly between the two groups at 24 weeks. These results are depicted in Table 4. Presence of MOH had no impact on the primary outcome in both the groups. In the topiramate and propranolol groups, mean change in migraine days per 28 days in patients with MOH as compared to those without MOH at the end of 24 weeks were −5.5 vs −4.9 days (p = 0.843) and −6.4 vs −8 days (p = 0.499) respectively.
Comparison of baseline demography, headache parameters and primary and secondary outcomes of efficacy in PP completers (n = 95).
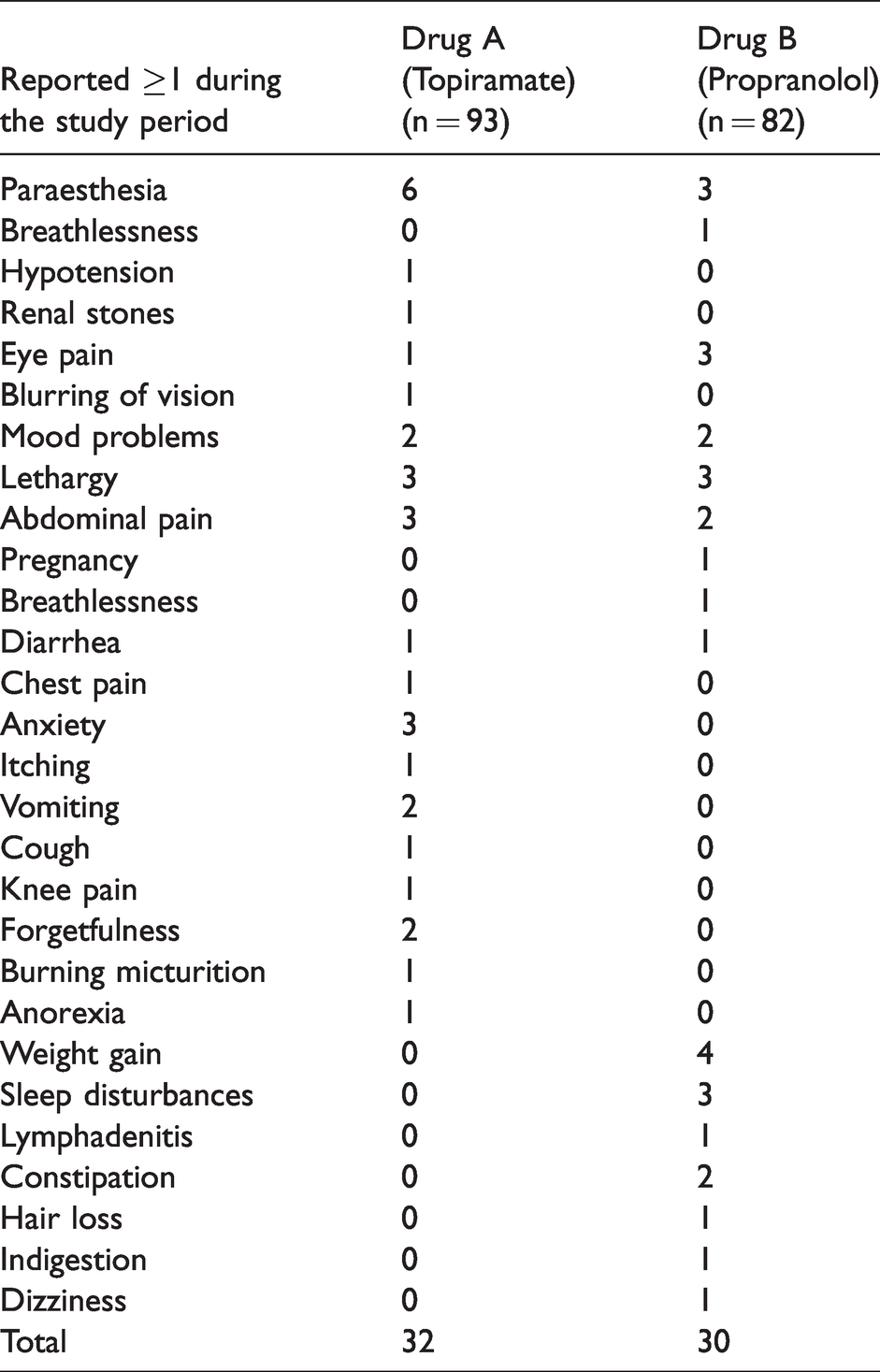
Adverse events
There were 26 types of TEAEs reported by the patients. Overall, TEAEs were reported by 35.4% of patients (62/175; 32/93 in the topiramate group and 30/82 in the propranolol group) (Table 5). Only 1 SAE was reported. 1 patient from the propranolol group had an unplanned pregnancy while on treatment. She underwent medical termination of pregnancy within eight weeks as the couple had already completed their family. The procedure was uneventful. The patient exited the study.
Comparison of treatment emergent adverse effects profile.
Discussion
We found that there was no statistically significant difference in the primary as well as multiple secondary outcomes assessing the efficacy of propranolol (in the maintenance dose of 160 mg/day given for 20 weeks after a titration period of 4 weeks) compared to topiramate (in the maintenance dose of 100 mg/day given for 20 weeks after a titration period of 4 weeks) as a preventive treatment for CM.
We used a robust protocol that included standardized outcome measures as suggested by the International Headache Society (IHS) for conducting an RCT in CM (17). We chose to assess the outcome at 24 weeks because it provides more stable estimates of outcome measures, increases the power of the trial and the efficacy of many preventive treatments accrues gradually with some medications requiring up to 24 weeks before their full potential is realized (17).
Randomization and blinding were adequately achieved in our study. Our institute runs a tertiary care headache clinic and a large number of CM patients of varied severities are referred here from Delhi and nearby states. The referral system is entirely random and the patients are referred from private practitioners and other government primary and secondary care hospitals or clinics. The consecutive recruitment of such patients in this study thus ensured a representative group of CM patients likely to be encountered by the physicians in the community, thereby reducing the selection bias. Our patient population was similar in terms of headache characteristics compared to previously reported CM cohorts (18–20). However, our patients were a little younger (mean age 33.1 ± 9.5; median 31 years), mostly consisted of females (95.4%) and had shorter duration of CM (mean 1.18 ± 1.48; median 5 years) compared to those reported in the previous studies (4,7,18). Both the treatment groups were matched at the baseline with respect to age, sex, and headache parameters such as mean migraine days, AMT days, HIT-6 scores, VAS scores, and MSQOL scores. The proportions of MOH were also similar. Hence both groups were comparable. The maintenance dose of propranolol (160 mg/day) and topiramate (100 mg/day) was chosen based on previous studies for adequate dosing of these two drugs (14). A fixed maintenance dose was kept to reduce the confounding effects of inadequate and variable doses.
Our results must be viewed with certain caveats, the most important of which is the inability to reach the target sample size due to unprecedented COVID-19 pandemic and lockdown and high rate of dropouts. For an estimated sample size of 244, we managed to randomize 175 patients out of which 95 completed the full study protocol. It was a long study with an overall study duration of about eight months. We lost many randomized patients who were due for follow-up in the subsequent months due to their inability to report to the study center. Until about November 2019, the discontinuation rate was very low (6/97; 6.2%) but greatly escalated following the onset of the pandemic (74/78; 94.9%). Hence, we had to stop the study prematurely because there was no realistic chance of continuing with further recruitment and regaining contact with the patients lost during the pandemic and lockdown. Thus, our overall discontinuation rate of 45.7% (80/175) though seems to be high can largely be explained by the negative impact of the COVID-19 pandemic. It is also noteworthy that Silberstein et al. reported a discontinuation rate of 44.5% in their RCT of topiramate for CM (7).
Both ITT and PP analyses showed similar results i.e., there was no significant difference between primary and secondary outcomes between the patients receiving topiramate versus those receiving propranolol. For the ITT analysis, we had to include the worst baseline headache parameters using the LOCF method in a substantial number of patients (59/175; 33.7%) who were lost to follow-up following the onset of COVID-19 pandemic and lockdown. Despite this, the results were similar to the efficacy estimates done by the PP method. Further in absolute terms, the reduction in mean migraine days in the case of topiramate in our study was comparable to the estimates by previous RCTs. For example, Silverstein et al. (7) reported a mean reduction of −5.6 days for topiramate (vs −4.1 days for placebo) at 16 weeks whereas Mathew et al. (21) reported a mean reduction of −5.3 days for topiramate (vs −7.4 days for OBT-A) at the end of 24 weeks. These compare well to our estimate of −5.3 days for topiramate (vs −7.3 days for propranolol) at the end of 24 weeks in PP completers. Unfortunately, no such previous estimates are available for propranolol. Interestingly, a study by Silberstein et al. (12) that tried to find out the role of propranolol as an add on to topiramate in patients of CM who did not respond to topiramate alone found that the mean reduction in migraine days at the end of 24 weeks to be −4.0 days for the dual treatment vs −4.5 days for topiramate alone. The difference was non-significant. We found a better response of −7.3 days for propranolol (vs −5.3 days for topiramate). However, the CM populations were different as mentioned above.
PGIC scores were also comparable between the two groups at the end of 24 weeks thereby suggesting that the investigators also did not find any clinically meaningful difference in terms of global outcome. It is also noteworthy that 43.5% of CM patients in the topiramate group and 44.9% in the propranolol group at the end of 24 weeks had a reduction in migraine frequency to less than 15 days per 28 days thereby suggesting a reversal to EM phenotype. These reversal rates are better than the population-based estimates reported by Manack et al. (22) for the American migraine prevalence and prevention study (AMPPS) cohort which could be explained by the fact that in our case, the intervention was done under a controlled setting of an RCT.
The statistical analysis for non-inferiority also yielded concordant results. We chose a mean difference of 1.5 days per four weeks as the cut-off delta value. Delta is defined as the ‘smallest value that would be a clinically significant effect’ (16). Various approaches such as 95%–95% (fixed margin approach) and the synthesis approach has been advocated for calculation of inferiority margin but there is no definite guideline for choosing its value and the choice is largely based on clinical judgment and statistical considerations (23). Our choice was based partly on our clinical impression that most CM patients would appreciate at least one and half additional migraine-free days per four weeks as a clinically meaningful gain, and partly on power analysis to ensure a reasonable probability for avoiding ‘inconclusive’ results for definite clinically insignificant differences. The Committee for Proprietary Medicinal Products guidelines formally state that ‘in a non-inferiority trial, both the ITT and PP analyses have equal importance, and that their results should lead to similar conclusions for a robust interpretation’ (24). Accordingly, CONSORT guidelines strongly recommend providing the details of both estimates in clinical trials, because when ‘ITT and PP provide identical conclusions, the confidence level of the investigator for the study results is augmented’ (25). Since, both ITT and PP analyses showed that propranolol is non-inferior, non-superior to topiramate for prevention of CM, our study results become meaningful despite failing to achieve the target sample size and having sizeable attrition during follow-up.
Topiramate and propranolol were well tolerated by the patients and TEAE was comparable between the two groups. Overall, TEAEs were reported in 35.4% patients (34.4% in the topiramate group vs 36.6% in the propranolol group). These were, however, mild and no SAEs were reported except for one patient in the propranolol group becoming pregnant. Compared to previous studies (7,8,12), lesser adverse events were reported by our patients in both groups. The possible reasons might have been the slow dose escalation protocol in which the dose was slowly built over a period of four weeks. It is well known that the slow titration of the preventive drugs for migraine decreases the incidence of TEAE (26). Another possible reason might have been the fact that 34.4% of patients randomized to topiramate and 28% of patients randomized to propranolol discontinued treatment within four weeks of randomization due to reasons other than adverse events and thus precluded the capture of the adverse events. No new adverse events (that were not described in the past trials and clinical experience) were identified.
It is noteworthy that none in the propranolol group discontinued due to TEAE as compared to four (4.3%) in the topiramate group. This finding assumes importance when viewed in the context of the second International Burden of Migraine Study (IBMS) study that reported poor acceptability of oral preventive drugs by CM patients (27). In this study, it was found that the anti-epileptics (including topiramate) (53.2%) and antidepressants (45.5%) had a higher prevalence of side effects as compared to β-blockers (including propranolol) (36.5%), although the efficacy was slightly lower for the latter. In another study by Bigal et al., propranolol was found to have the highest patient satisfaction levels among all the drugs used in CM (28). It was well tolerated by 80% of patients and improved headache frequency in 70% of patients.
The mechanism of action of both these drugs in CM remains unknown. The pathophysiology of CM is itself still poorly understood (29). Atypical pain processing by reduced inhibition of pain by descending pain modulatory pathways (30,31), central sensitization due to recurrent migraine attacks (32), cortical hyperexcitability (33), and increased neurogenic inflammation as evidenced by higher CGRP levels (34) have all been implicated in its pathogenesis. Functional connectivity changes and structural changes in the brain have been described in CM patients (35,36). Despite the lack of clear understanding of the pathogenesis of CM and the exact mechanisms of action of these drugs, both of them seem to work for CM patients as shown by our study.
Although used in migraine prevention for more than 50 years, propranolol did not undergo a rigorous trial of non-inferiority with another oral drug for CM so far. This is possibly due to the fact the drug is cheap, non-patented and therefore investing in a clinical trial may not be considered a lucrative option for the pharmaceutical industry. However, the patient’s experience of use of propranolol in CM has been good (28) and a recent meta-analysis by Jackson et al. found that propranolol is likely to reduce headache frequency by more than 50% in CM patients (relative risk: 2.0, 95% CI: 1.0–4.3) (37).
The major limitation of our study as discussed above is that we could not achieve the target sample size and had considerable attrition due to the effects of the COVID-19 pandemic and lockdown. Secondly, the majority of our patients consisted of females with relatively shorter duration of CM. Therefore, the results of our study may not be generalizable to male CM patients and those with a long duration of CM. Thirdly, topiramate because of its peculiar side effect of causing paraesthesias might have caused unblinding in some patients. However, we informed every patient of all the possible side effects that can be seen with the use of either of the drugs at the time of randomization. Further, paraesthesias were reported only by a few patients in the topiramate group and even by some patients in the propranolol group. Hence, we think that it did not have any significant impact on the blinding. Finally, we are cognizant of the fact that without a placebo arm in an efficacy trial, the observed decrease in migraine days per 28 days by both the drugs can be due to a time effect. We did not use a placebo group for ethical reasons as topiramate is already accepted as a standard of preventive care for chronic migraine patients and also our hypothesis was comparison of relative efficacy of propranolol and topiramate and not absolute efficacy.
Conclusion
To conclude, this study found no significant differences in terms of efficacy and TEAE between the topiramate and propranolol when used as a preventive treatment for CM by both ITT and PP analysis. Although propranolol performed better compared to topiramate across all the outcome parameters in numerical terms, the difference was not statistically significant. ITT and PP analyses for non-inferiority found that propranolol was non-inferior, non-superior to topiramate. Despite the limitations discussed in the text, this RCT for the first time provides evidence of non-inferiority of propranolol over topiramate for the preventive treatment of CM.
Clinical implications
In terms of primary outcome of efficacy, that is, mean change in migraine days per 28 days at the end of 24 weeks, propranolol (160mg/day) was found to be non-inferior, non-superior to topiramate (100mg/day) for the preventive treatment of CM. There was statistically no significant difference in multiple secondary outcomes between the patients who received propranolol versus those who received topiramate. The adverse effects were mild and both propranolol and topiramate had comparable tolerability profiles.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211047454 - Supplemental material for TOP-PRO study: A randomized double-blind controlled trial of topiramate versus propranolol for prevention of chronic migraine
Supplemental material, sj-pdf-1-cep-10.1177_03331024211047454 for TOP-PRO study: A randomized double-blind controlled trial of topiramate versus propranolol for prevention of chronic migraine by Debashish Chowdhury, Luv Bansal, Ashish Duggal, Debabrata Datta, Ankit Mundra, Anand Krishnan, Arun Koul and Anu Gupta in Cephalalgia
Footnotes
Acknowledgment
The authors thank Intas pharmaceuticals Ltd. for providing the drugs free of cost for the conduct of this trial.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
