Abstract
Background
Adult abrupt severe non-traumatic headache (thunderclap) is often related to serious underlying etiologies such as subarachnoid hemorrhage. However, data are sparse regarding thunderclap headache in the pediatric population.
Objective
The aim of the study was to evaluate the prevalence, characteristics and causes of thunderclap headache in the pediatric and adolescent population, aged 6–18 years, presenting to a pediatric emergency department.
Methods
The electronic database of a tertiary care pediatric emergency department was searched for children presenting with acute headache during 2016–2018. Headache severity was defined by pain scales, either a visual analogue scale or by the Faces Pain Scale–Revised.
Results
Thunderclap headache was diagnosed in 19/2290 (0.8%) of the included patients, all of them with a pain score of 10/10. All the patients had a benign course. Primary headache was diagnosed in 15/19 (78.9%), six patients had migraine and eight were diagnosed with primary thunderclap headache. Four of the 19 patients were diagnosed with secondary headache: three with infectious causes and one with malignant hypertension.
Conclusions
Thunderclap headache is rare among children and adolescents presenting to the emergency department. This headache is generally of a primary origin. Extensive evaluation is still needed to rule out severe diagnosis problems.
Introduction
Serious intracranial causes in acute non-traumatic headache at an adult emergency department are not rare, and account for 19.4% of adults admitted to the emergency department with acute non-traumatic headache (1). Abrupt onset, awakening pain, fever, worst headache ever according to the patient, alteration of consciousness, and localizing neurological deficits have been identified as significant predictive factors for a serious intracranial cause in these patients (2). Among adults who presented to the emergency department with acute headache, 23% had a pain score of 10/10 (2).
Adult thunderclap headache is characterized by peak intensity occurring within 1 min (3). This headache is considered a red flag that may indicate a serious intracranial process, and should prompt evaluation (4). In a study in Sweden, the population-based incidence of thunderclap headache in adults was estimated as 43/100,000; 11% of the patients were diagnosed with subarachnoid hemorrhage, and for most of the remaining, underlying causes were not identified (5).
In contrast to adults, 80% of children and adolescents presenting to pediatric emergency departments with non-traumatic headaches have at least one red flag that may indicate an underlying severe cause of the headache. However, only 1% of them were found to have intracranial abnormalities (6).
Only a few case reports and no extensive studies have been published of the prevalence and causes of pediatric and adolescent thunderclap headaches.
The first aim of this study was to describe the prevalence, demographic and clinical characteristics, and the causes of abrupt severe non-traumatic headache (thunderclap) in pediatric and adolescent patients attending a pediatric emergency room in a tertiary care facility. The second aim was to compare demographic and clinical characteristics between patients with abrupt severe headache (thunderclap) and patients with non-thunderclap headache at the same severity of pain score.
Methods
Patients and settings
A retrospective study was conducted of patients presenting during 2016–2018 to the emergency department in Schneider’s Children Medical Center, a tertiary hospital in Israel. The emergency department is the largest in the country. Pediatric patients aged 0–18 years are admitted.
Study eligibility criteria included: Age 6–18 years with acute headache and severe or very severe pain, defined as a score of 8, 9 or 10 on the 10-point pain scale or its equivalent on face scale routinely used in our emergency department. Patients younger than age 6 years were not included because of the possibility that they did not fully understand the pain scale used.
Study exclusion criteria were: Insufficient data, leaving the emergency department before investigation, a history of brain tumor, a head trauma in the 3 preceding months, a return visit to the pediatric emergency room with an acute headache during the 3 years of the study period, active oncologic disease, a cerebral shunt, a diagnosis of trigeminal neuralgia and a stabbing headache.
The study was approved by the Research Ethics Board of Rabin Medical Center (approval no. RMC-19-704). Due to the retrospective study design, the committee waived the need for informed consent.
Data collection
The computerized database was scanned for referrals whose chief complaint was headache. YL, an expert pediatrician, reviewed the files and selected those that met study eligibility criteria. Demographic, clinical and laboratory data were obtained for each patient, together with self-reported pain scores. Patients with abrupt severe (thunderclap headache) and with severe non-thunderclap headache were identified. In addition, for each child included, medical records were reviewed of outpatient visits in pediatric neurology clinics, and in pediatric headache clinic, and by ambulatory primary physicians, at least 1 year after the emergency department visit.
Emergency department protocol for pain assessment and management
According to our local protocol for assessment and management, a triage nurse evaluates any patient who reports pain (including headache) at admission to our pediatric emergency department. A pediatric pain scale suitable to the patient’s age and development is used. We use a visual analogue scale (VAS) and the Faces Pain Scale–Revised (7–8). The VAS has been validated for children aged 7 years and older (7–10). It has increments of color intensity and numeric increments. The scale entails the following explanations: 0 means the absence of pain; 1–4 indicates gradual increments of pain intensity according to number and color; 6 and 7 indicate stronger pain with increments of the numbers; 8 and 9, severe pain; and 10 represents very severe, intolerable (unbearable) headache. The nurse uses the Faces Pain Scale–Revised instead of the VAS if the child is deemed not able (in terms of cognitive development) to respond to the VAS, does not understand the explanation of the VAS or does not have numeric identification abilities.
For children younger than age 7 years, the nurse shows a graphic re (7–10). This is a self-report measure for evaluating acute pain in children (7), with strong validity and reliability for ages 4 to 17 years (7). The scale is scored with six faces that show increasing pain, from no pain to severe pain, to unbearable pain (9).For this study, we considered a score of 10 on the VAS scale as equivalent to an assessment of very severe pain on the Faces Pain Scale–Revised; and a score of 8–9 on the VAS as severe pain on the Faces Pain Scale–Revised (10). A high correlation (0.79) has been demonstrated between the VAS and the Faces Pain Scale–Revised (9).
Final diagnosis of headache
Primary and secondary headaches were classified retrospectively, according to the criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) (3).
During the study period, patients were diagnosed by consultant pediatric neurologists in the emergency department or the hospitalization department, or during follow-up in the pediatric headache clinic after clinical parameters and revising imaging and lumbar puncture results. CTs were performed during emergency department follow-up evaluations, according to physicians’ decisions, and MRI scans were performed, if needed, during hospitalization.
Thunderclap headache assessment
For each of the patients included in the cohort, the authors reassessed whether the headache was thunderclap or another diagnosis.
The ICHD-3 (3) adult criteria for thunderclap headache are as follows: A. Severe head pain fulfilling criteria B and C; B. Abrupt onset, reaching a maximum intensity in <1 min; C. Lasting ≥5 min; and not attributed to another disease.
The diagnostic criterion that was used in an extensive review of 2345 adults with thunderclap headache was a “sudden and severe headache” (11). We adopted this approach and defined thunderclap headache in this study as a severe to very severe headache, with abrupt onset, lasting more than 5 min and reaching maximal intensity immediately at onset of the headache. From our clinical experience, children are not able to precisely describe a headache as less than 1 min, as in the ICHD-3 criteria, but rather describe a headache as immediate or very quick, or abrupt, reaching maximal intensity.
Idiopathic thunderclap headache was diagnosed according to ICHD-3 criteria (3). The criteria of primary thunderclap headache are met after excluding organic causes. This implies normal brain imaging, including the brain vessels (MRI or CT) and/or normal cerebrospinal fluid (3).
For the purpose of this study, blood pressure was adjusted retrospectively for gender, age and height according to the 2017 American Academy of Pediatrics guidelines (12).
Headache evaluation
During review of the medical records, information was collected about two relevant brain imaging tests: a) Head CT – in our institution, this exam is done during the emergency department visit, if the treating physician considers that urgent imaging is needed; b) brain MRI – this test is performed during hospitalization, according to the decision of the attending physicians. Lumbar puncture is performed according to the decision of physicians in the emergency room, to rule out increased intracranial pressure, meningitis and encephalitis. All patients with severe headache are followed for at least 1 year in a pediatric headache clinic or pediatric neurology clinic.
Statistical analysis
Data were analyzed using BMPD software (13). Continuous variables were calculated as means and standard deviations, and compared between groups, using analysis of variance. Parameters that did not have a Gaussian distribution were compared between groups using the nonparametric Mann–Whitney U-test. Discrete variables were calculated as numbers and percentages, and compared between groups using Pearson’s chi-square test. A p-value of ≤0.05 was considered significant. Odds ratios (OR) and 95% confidence intervals (CI) were determined. The effect size for continuous variables was calculated with Cohen’s d. Stepwise logistic regression was used to identify the variables found to be significant at p ≤ 0.1 on univariate analysis. We also included age and gender. Odds ratios (OR) and 95% confidence intervals (CI) were determined.
Power analysis to calculate sample size was not performed due to the lack of other studies of thunderclap prevalence in pediatric patients, and the assumption that this is a very rare diagnosis in this age group.
Results
Figure 1 describes the study design and patient diagnoses. During the 3-year study period, 104,086 children and adolescents aged 0–18 years were admitted to the pediatric emergency department; of them, 3112 (3%) presented with acute headaches. A total of 2290 children, aged 6–18 years (mean 13.3 ± 3.26) were admitted with a chief complaint of headache, and reported their level of pain according to one of the two scales used. For 355 of them, the pain of the headache was severe or very severe (8–10). After excluding 72 patients from the analysis according to study criteria, 283 remained with severe to very severe headache. These comprised: 86 patients with a pain score of 10, including 19 with thunderclap headache, and 197 with a severe pain score of 8–9 according to the VAS or its equivalent on the Faces Pain Scale–Revised scale. Headache pain scale 10 according to the VAS scale or its equivalent on the Faces Pain Scale (including thunderclap headache) accounted for 3.7% of all the children with acute non-traumatic headache aged 6–18 years admitted to the pediatric emergency department during the study period.

Flowchart of the study depicts the distribution of the cohort.
Of the 283 patients with severe to very severe headache (pain scale 8–10), 11 had severe causes, and only one had a thunderclap headache.
Severe diseases were diagnosed in the five patients with very severe headache, pain scores of 10 (Table 1). One had thunderclap headache and a hypertensive crisis (Table 1). Among the six patients with severe headache pain scores of 8 or 9 on the VAS, or the equivalent on the Faces Pain Scale; orbital cellulitis was diagnosed in one, encephalitis in two, aplastic anemia in one and multiple sclerosis in two. During the study period, only one patient was diagnosed with intracranial bleeding (due to bleeding to a cavernous hemangioma) and one with sinus vein thrombosis. None of these had pain scores of 8, 9 or 10, or thunderclap headache.
Secondary causes of very severe headache (thunderclap vs. other very severe headaches with pain score 10) and life-threatening diseases.

*Severe life-threatening diseases
Abrupt and severe thunderclap headache
The 19 patients with thunderclap headache comprised 6.7% of the 283 patients with severe to very severe headache (pain score 8–10), and 0.8% (19/2290) of all the children aged 6–18 years admitted to the emergency department with a chief complaint of a headache. All the patients diagnosed with thunderclap headache had a pain score of 10 – very severe pain. Their mean age was 14.13 ± 4.12 years (median 16 years, range 6.5–17.9 years). Ten (52.6%) were females. All of them had normal neurologic examinations and normal fundoscopy; none had nuchal rigidity or encephalopathy (these parameters precluded a lumbar puncture examination).
Nine patients with thunderclap headache had reported headache in the past, four had been diagnosed with migraine and five could not define their past diagnosis of headache. We found no medical information history regarding headache of the other 10 patients with thunderclap. None of the patients with either thunderclap headache or a headache pain score of 10 without thunderclap died during the study follow-up period or was admitted to the pediatric intensive care unit or had intracranial bleeding; all had a benign course.
Table 2 compares demographic and clinical characteristics between patients with thunderclap headache and patients with other headaches with a pain score of 10. Compared to patients with non-thunderclap headache and a pain score of 10, those with thunderclap headache were more likely to experience alleviation in their pain before discharge from the emergency room (89.5% versus 62.7%, p = 0.03; odds ratio (OR) 5.06, 95% confidence interval (CI) 1.04–48.08), to have shorter duration of headache attack (9.1 ± 8.7 vs. 85.9 ± 12.1 h, p < 0.001, Cohen’s d – 0.895) and to have primary compared to secondary headache, 78.9% vs. 40.3%, p = 0.01. (OR 1.322, 95% CI 0.98–1.79). Fifteen (78.9%) of the patients with thunderclap were diagnosed with primary headache: Six (31.6%) migraine, eight (42.1%) primary thunderclap headache and one headache not otherwise specified. All eight patients with primary thunderclap headache had normal MRI or CT. Four patients with thunderclap headache were diagnosed with secondary headache (Table 1). Eleven patients with primary thunderclap headache did not undergo neuroimaging. This was due to physicians’ decisions in the emergency department, either following rapid improvement during patients’ stay in the emergency department or the identification of an underlying benign infectious cause of the headache. Primary headache was more frequent among patients with thunderclap than among patients with a pain score of 10 but without thunderclap headache: 78.9% (15/19) vs. 40.3% (27/67), p = 0.01. Most patients (40/67, 59.7%) with a pain score of 10 but without thunderclap headache had a secondary headache (Table 1). Twenty-seven (68%) of the 40 patients had infectious causes (upper respiratory infection, sinusitis, meningitis, pneumonia or encephalitis).
Characteristics of patients in the pediatric emergency department with thunderclap headache and with another very severe headache (pain score = 10).
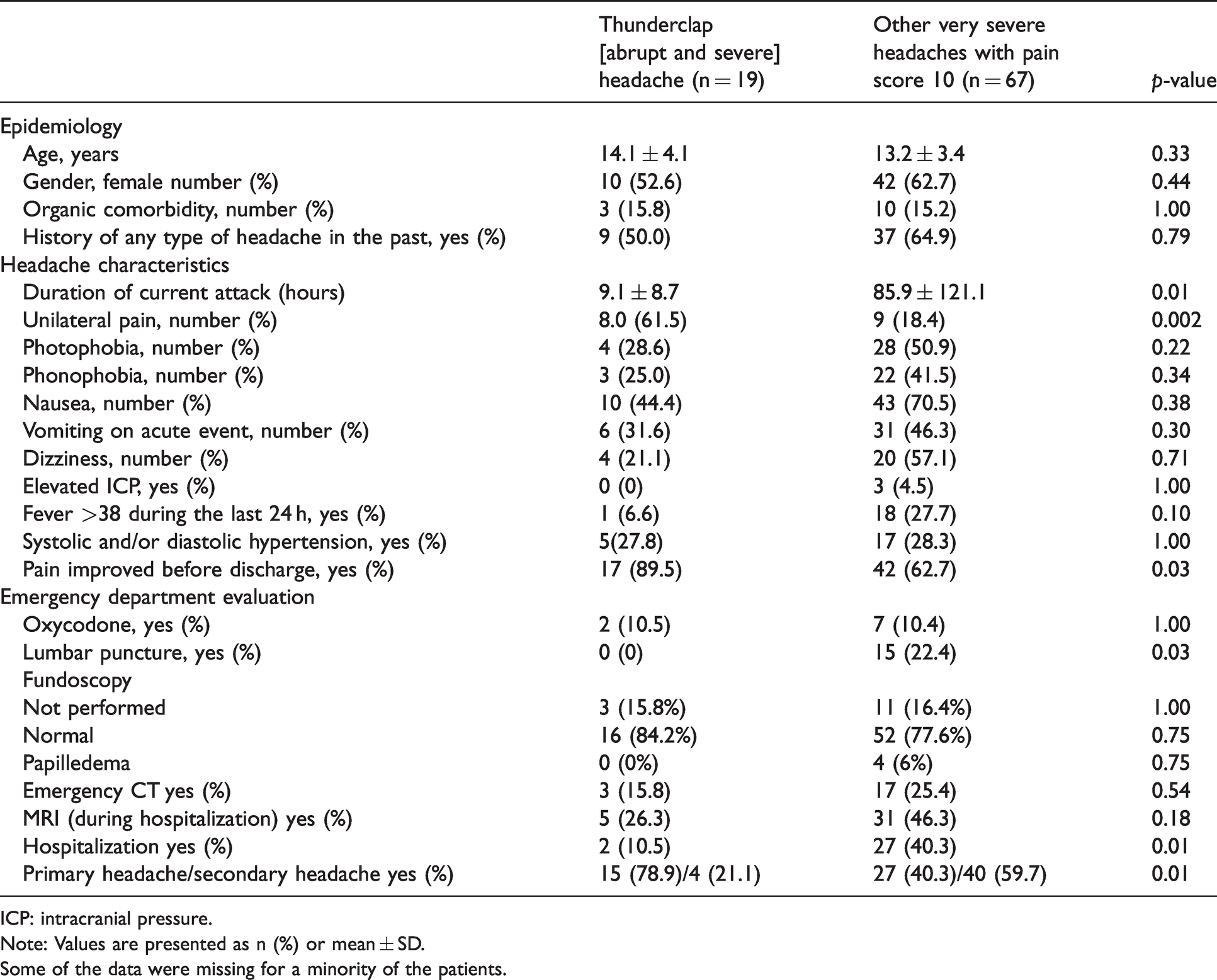
ICP: intracranial pressure.
Note: Values are presented as n (%) or mean ± SD. Some of the data were missing for a minority of the patients.
None of the patients with either thunderclap headache or a headache pain score 10 without thunderclap died during the study follow-up period or was admitted to the pediatric intensive care unit or had intracranial bleeding; all had a benign course.
Logistic regression
Table 3 shows the results of the logistic regression analysis. Two variables were found to be significantly associated with thunderclap headache in patients who attended the emergency department: Primary versus secondary headache, and headache improvement before discharge from the emergency room.
Logistic regression: Factors associated with headache in patients with thunderclap headache vs. patients with headache pain score 10 attending a pediatric emergency department.

Area under the curve = 0.73.
Missing data and excluded patients
Of patients with severe and very severe headache pain, 72 were excluded from the analysis according to the study criteria: Lack of information (25); exit from the emergency department before investigation (2); the experience of a recent head trauma (17); the presence of a cerebral shunt (10); an established diagnosis of a brain tumor (6), active oncologic disease (6) or trigeminal neuralgia (2); or returning admission to the emergency department with acute headache during the study period (4).
Data of some parameters were missing for some patients. No significant differences were observed in missing data between those with and without thunderclap headache: Systolic/diastolic pressure, 1 vs. 7, p = 0.68; a history of headache, 1 vs. 0, p = 0.23; unilateral pain, 6 vs. 18, p = 0.77; photophobia, 5 vs. 12, p = 0.51; nausea, 2 vs. 7, p = 1.0; dizziness, 10 vs. 32, p = 0.79.
Discussion
We report that among pediatric patients aged 6–18 years admitted to the emergency department with major complaints of acute headache, 3.7% reported a maximal pain intensity of 10/10. All 19 (0.08% of all the children with non-acute headache) diagnosed with thunderclap headache had a pain score of 10 – very severe pain. Most patients (42%) with thunderclap headache had primary headache; of the minority with secondary headache, most had infections causes. All the patients had a benign course.
Our 42% rate of idiopathic thunderclap headache is higher than that reported among adults with thunderclap headache (11). In a large systemic review of 213 studies including 2345 adult patients, 119 causes of thunderclap headache were reported (11). For 632 patients (27%), the cause was neurovascular, including vasoconstrictive syndrome; 252 (10.7%) of those with thunderclap headache had intracranial hemorrhage due to several causes; ruptured aneurysm and intracranial hemorrhage were found in only 18/2345 (0.7%) and idiopathic thunderclap was diagnosed in 459 (19.5%) (5).
Yang and Fuh (14) reported the following frequent causes for adult thunderclap headache: subarachnoid hemorrhage, reversible cerebral vasoconstriction syndrome, cervical artery dissection, cerebral venous sinus thrombosis, cerebral infarction, intracerebral hemorrhage, spontaneous intracranial hypotension, intracranial infection, and pituitary apoplexy. None of our patients presented with symptoms related to any of these diagnoses.
A recent publication (15) reported that thunderclap headache caused by adult subarachnoid hemorrhage is rare and is manifested by a dramatic clinical presentation, with a high mortality rate and the necessity of early imaging (15).
In their review of childhood headache in the emergency department, Raucci et al. (16) delineated causes of thunderclap pediatric headache. These included cervical artery dissection that is manifested by acute onset headache associated with neck pain and/or tenderness, and with ipsilateral supraorbital, auricular or mandibular pain, and very frequently with focal neurologic symptoms. None of our patients presented with these clinical symptoms.
Pediatric venous sinus thrombosis may present as thunderclap headache; possible triggers are coughing, sneezing and change in position. Other symptoms associated with this condition are vomiting, diplopia and papilledema and convulsions. None of our children had these symptoms, and all of them recovered in a short time. Reversible cerebral vasoconstriction syndrome is a clinical condition of transient dysregulation of the cerebral vascular tone, which has also been cited as a possible cause of pediatric thunderclap headache and has been associated with an altered level of consciousness and/or focal neurological symptoms. Neuroimaging is mandatory with these symptoms (16). None of our patients presented with them.
A possible explanation for the higher rate of idiopathic thunderclap headache among children may be related to a generally lower prevalence of neurovascular conditions compared to adults. The pediatric stroke rate (hemorrhagic and ischemic) in one region in the United States was reported as 4: 100,000 and remained stable over 17 years (17). Among adults, the rate of stroke was reported as 240: 100,000 in France and reached 600: 100,000 in Russia (15). In adults, neurovascular diseases are mostly related to diabetes, dyslipidemia, hypertension, smoking and atrial fibrillation – conditions that are rare among pediatric patients compared to adults (18). In children, frequently identified ischemic stroke etiologies are arterial dissection, sickle cell disease, and meningitis; common hemorrhagic stroke etiologies are arterial venous malformation and rupture due to aneurysmal subarachnoid hemorrhage (17). The different methodologies of the two studies (systemic review in adults vs. our retrospective study) may also be related to the higher neurovascular causes of thunderclap headache in the latter.
None of our patients had subarachnoid hemorrhage. This contrasts with a prospective study (5) of 137 adults in the emergency room with thunderclap headaches, inferring an incidence of 43/100000. Of the 137 patients, 11.3% had subarachnoid hemorrhage. Other causes of thunderclap headaches in adults were intracerebral hematoma, aseptic meningitis, cerebral edema and sinus vein thrombosis (5). All our patients had a benign course; this also characterizes most adults with thunderclap headache for which no specific cause was identified (5). In a prospective study of 29 regional hospitals, 15% of 847 admitted to adult emergency departments with acute headache presented with thunderclap headache. Eighteen (2%) patients were with acute headache subarachnoid hemorrhage, but the authors did not report if they presented with thunderclap headache (4).
No extensive report of thunderclap headache in pediatric emergency rooms has been published. Ito et al. reported on two ruptured intracranial aneurysms, one of them presenting as thunderclap headache (19).
Nam et al. (20) reviewed 23 pediatric and adolescent patients diagnosed with intracranial aneurysm in a tertiary medical center during 22 years; of them, 60% presented with sudden severe headache (thunderclap headache). Due to the rarity of cerebral aneurysms and neurovascular diseases in pediatric patients (19), the absence of this diagnosis among our patients who arrived with thunderclap headache is not surprising. The prevalence of a pain score of 10/10 is about 4% of our population aged 6 years and older admitted with an acute headache to our pediatric emergency department in a tertiary center. This is lower than reported among adults (up to 22.7%) (2). The reason could be that serious etiologies to headaches are more common among adults. In addition, adults are more familiar with headaches, so they may more likely present at the emergency department only when a headache is severe; for children, parents usually decide whether to present to the pediatric emergency department with less severe headache. In our study, only 283 of 2290 patients had severe to very severe headache according to pain scales, and others had less severe headaches.
The higher prevalence of very severe headache (score 10) in adult patients is most probably not related to higher pain perception in adults. In the meta-analysis of Stefan Lautenbacher et al., the pain threshold was shown to increase with age (21). Accordingly, we would have expected a higher rate of severe headache in children versus adults. This supports the reliability of the lower rate of very severe headache, scored 10, in our cohort compared to studies in adults.
In our pediatric cohort, cerebrovascular causes were not considered the etiology for any of the patients. In contrast, the prevalence of neurovascular causes of adult thunderclap was 20.1% (11). Eight (42%) of our patients were classified as having idiopathic thunderclap headache. Among adults with idiopathic headache, thunderclap was reported in 20% (11).
Six of our patients with thunderclap headache were diagnosed with migraine. Torriero et al. (22) reported that for children aged 6 years and younger, the duration of migraine is shorter than 1 h. This raises the possibility of a higher rate of undiagnosed migraine in the primary thunderclap headache group. For only three of our children with thunderclap headache who were aged 6–8 years, the duration of the thunderclap was longer than 1 h. During follow up of at least 1 year, none of these had recurrent headache; this precludes diagnosis of migraine, presented thunderclap headache] according to ICHD-3 criteria (at least five attacks are needed).
Three of our patients were diagnosed with upper respiratory infection and sinusitis (Table 1). During long-term follow up, no other diagnosis such as primary headache was recorded. In a systematic review (11), viral infection and rhino-sinusitis were reported in 73 (3%) of adults with thunderclap headache. The higher rate of infectious causes in pediatric thunderclap headache in our study may be related to a higher rate of infectious diseases among pediatric patients.
Limitations
This study has a few limitations: First is its retrospective design. The reporting of pain scores by different triage nurses may have biased the results of the pain scores obtained. This is despite the reliability attributed to the visual analogue scale and Faces Pain Scale–Revised for assessing pain in the pediatric emergency department setting (7). In addition, the study was performed in a tertiary care pediatric medical center that serves as a referral center for more severe cases, and this may have created a selection bias.
A limitation of the study is the absence of neuroradiological investigations in 11/19 patients with thunderclap headache. However, all the patients who did not improve during their stay in the emergency department or hospitalization, and without an identified reason for their severe headache, had normal neuroimaging and were diagnosed as having primary thunderclap headache. None of the patients had abnormal neurologic examinations, nuchal rigidity or encephalopathy that prompted the physicians in the emergency department or hospital units to perform neuroimaging. Since the routine follow-up of these patients was 1 year, a severe secondary thunderclap headache was likely not missed. Another limitation is that the study was conducted in a single center, limiting the generalizability of the findings.
Conclusion
Thunderclap headache in pediatric and adolescent patients referred to a tertiary pediatric emergency room is very rare, and is associated with a pain scale of 10. Rapid resolution of headache with normal neurologic examination and normal neuroimaging, if indicated, are related to benign course. According to our experience, as reported in this single-center study, causes for thunderclap headache in children are related to primary headache. However, this does not preclude the need for extensive evaluation to rule out underlying severe diagnoses.
Clinical implications
Thunderclap headache was very rare among patients ages 6–18 years attending a pediatric emergency department in a tertiary center. In our cohort, most patients with thunderclap had primary headache or infectious causes, and they all had a benign course. Our experience from a single center should not preclude extensive evaluation to rule out underlying severe diagnoses.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
