Abstract
Background
The prevalence of migraine and non-migraine headache declines with age.
Methods
Data from the third visit (2011–2015) of the population-based Heinz Nixdorf Recall study were analysed (n = 2038, 51% women, 65–86 years). Possible risk factors for headache activity (obesity, education, smoking, sports, alcohol, partnership status, living alone, having children, sleep quality, depression, hypertension, diabetes mellitus, stroke, coronary heart disease, medication), and headache symptoms were assessed. We estimated the lifetime prevalence and the prevalence of current active headache of migraine with and without aura, and non-migraine headache. The associations between possible risk factors and headache activity (active vs. inactive) were estimated by age and sex-adjusted odds ratios and 95% confidence intervals (OR [95% CI]) using multiple logistic regression.
Results
The lifetime prevalence of migraine was 28.6% (n = 584). One hundred and ninety-two (9.4%) had still-active migraine, 168 (3.5%) had migraine with aura, and 416 (5.9%) had migraine without aura. One hundred and sixty-eight (8.2%) had “episodic infrequent migraine, 0–8 headache days/month”, 10 (0.5%) had “episodic frequent migraine, 9–14 headache days/month”, and five (0.2%) had “chronic migraine, ≥15 headache days/month”. Overall, 10 (0.5%) had “chronic headache, any headache on ≥15 days/month”. Female gender and younger age were the most important associated migraine risk factors. Depression (1.62 [1.06; 2.47]) and poor sleep (1.06 [1.00; 1.12]) were associated with migraine and headache activity in general. Antihypertensives were associated with headache remission (0.80 [0.64; 1.00]). Additionally, undertaking less sports (0.72 [0.51; 1.03]) was associated with higher migraine activity.
Conclusions
Headaches and migraines are not rare in the older population. They are related to mood and sleep disturbance, and migraine even to less physical activity. Antihypertensives are related to headache remission.
Background
Primary headache disorders are among the most common neurological conditions in the world and are the third most disabling problem (1). The prevalence of most common primary headaches such as migraine and tension-type headache peak at the age of 20–40 years and decline with older age (2). Perhaps therefore, most epidemiological studies have targeted populations aged between 18 and under 65 years of age (3). Some others have investigated children and adolescents, and few have focused on elderly populations. A survey based on 35 different studies from all over the world found that, for headache in general, the prevalence was on average 46% but variation was immense, between 1% and 87%. The lifetime prevalence of headache based on 14 studies was 64% on average, varying between 8% and 96% (4). Although it is generally accepted that headache prevalence decreases with age (4–6), the exact reasons for this are still unknown (4,7). Migraine is the second most common primary headache (8). Based on several studies from European countries and North America, the prevalence of migraine in the adult general population was estimated to be between 12% and 25% for women and between 5% and 9% for men (4,9–11). The clinical features of migraine change in older age. Headache is less intense, and aura symptoms frequently become more prominent and sometimes even the only or the most bothersome feature of migraine (12). Depending on age, about 7–23% of migraine sufferers have migraine with aura (7). Patients with migraine develop co-morbidities such as cardiovascular or pulmonary diseases. Gastric, hepatic, and renal factors are more prominent in older people and play a role in the decision making for acute and preventative treatment (13).
This study aims to assess the prevalence of migraine with (MigA+) and without aura (MigA−) and non-migraine headache (nonMigHA) in women and men over 65 years of age in a population-based German cohort and to examine factors that might contribute to headache activity in older age.
Methods
Study design and study population
The analysis is based on data of the longitudinal population-based Heinz Nixdorf Recall (HNR) study. Details of the study design and cohort have been described previously (14). Briefly, participants for this study were randomly selected inhabitants of the Ruhr area living in Essen (589,676 residents), Bochum (371,582 residents), and Mülheim/Ruhr (172,759 residents). Baseline examination from 2000 to 2003 included 4814 men and women age 45–75 years with an overall recruitment efficacy proportion of 55.8% (15). Participants received annual questionnaires and follow-up examinations after 5 years and 10 years.
The current analysis included study participants investigated in the years 2011–2015 (10 years follow-up visit, third visit). Two thousand and thirty-eight subjects were 65 years or older then and provided information about their headaches. Figure 1 shows the flow chart of the study population. The interviewers collected information about general medical history and especially about headaches in the past and during the previous 3 months, smoking, alcohol, sports, sleep, and socio-economic status. The current use of medication was assessed. Participants were asked to bring all medication taken during the 7 days before the examination date. During the annual postal follow-up, participants reported events of stroke and coronary heart disease (CHD), which were validated by an external expert committee taking into account copies of medical records. Diagnostic headache questions were based on the criteria of the International Headache Society (16) (Supplemental Material 1).

Flow chart of the study population.
We defined the following headache groups:
Migraine with aura (MigA+) was defined as headache and one of the following accompanying symptoms immediately before headache: Visual impairments (like flickering, streaks, lines, scotoma, zigzag figures), spreading sensoric paresthesia or numbness, motoric dysfunction or disorders of speech or language. Migraine without aura (MigA−) was defined according to the criteria of the International Headache Society (16). Non-migraine headache (nonMigHA) was defined as any headache not fulfilling the criteria for MigA+ and MigA−.
MigA+, MigA− and nonMigHA were divided into active, if participants responded “yes” to the question ‘Do you still have this headache occasionally?’ and into inactive forms, if they responded “no”. Based on the reported headache frequency during the previous 3 months, we defined headache as episodic infrequent (0–8 headache days/month), episodic frequent (9–14 headache days/month), and chronic (≥15 days/month).
Definition of possible risk factors
Body mass index (BMI in kg/m2) was calculated from height and weight. “Obesity” was defined as BMI ≥ 30 kg/m2, otherwise “no”. Education was classified according to the International Standard Classification of Education (ISCED-97) as total years of formal education, combining school and vocational training. “Low education” was defined as <13 years of education, otherwise “no”. “Current smoking” was defined as a history of cigarette smoking during the past year. “Past smoking” was defined as quitting smoking more than a year ago, otherwise “no”. “Sports” was defined as “yes” when one or more sports had been practised in the last 4 weeks before the interview, otherwise “no”. Alcohol drinking behavior was assessed via a self-report questionnaire asking about the average consumption of different beverages (beer, red wine, white wine, spirits, and cocktails) within the last 4 weeks. The proportion of pure alcohol per beverage was then multiplied by the frequency of drinking. All beverages per person were summed up, resulting in the amount of total consumption of pure alcohol in g/month which was converted to g/day. “Alcohol abuse” among women was defined as average alcohol consumption of more than 20 g/day and among men of more than 30 g/day, otherwise “no”. Partnership status was defined as being with a partner or without. “Living alone” was defined when participants reported living alone. “Having children” was defined when participants reported having children. To assess sleep quality, participants were asked about length of sleep, subjective assessment of sleep quality, and the intake of sleeping pills. For “sleeping hours”, the following question was asked: “How many hours per night have you actually slept during the last 4 weeks? This does not have to be the same as the number of hours you have spent in bed.”. “Poor sleep” was defined as “yes” when participants answered the question “How would you judge the overall quality of your sleep during the last 4 weeks?” with “quite poor or very poor sleep quality”, otherwise “no”. “Sleeping pills” was defined as “yes” when participants answered the question “How often have you taken sleeping pills (prescribed by your doctor or over the counter) in the last 4 weeks?” with “once or more times”. In our study, a short version of the Center for Epidemiologic Studies Depression Scale (CES-D) (17) was applied. Answers were given on a 4-point Likert scale ranging from “less than one day” (0 points) to “5–7 days” (3 points). A sum score ranged from 0–45 points was calculated, with a higher score indicating more depressive symptoms. Positively formulated items were coded backwards and an average value was calculated over all 15 items. For up to three missing answers, the item value was replaced by the mean value of the answered questions. A cutoff point of ≥17 was defined as “depression” (18,19). Blood pressure categories were defined according to the Joint National Committee (JNC) 7 guidelines (20). “Hypertension” was defined as stage 1 or stage 2 hypertension, otherwise “no”. “Hypertensive medication” included the following classes of drugs or any combination of them: Diuretics, beta-blockers, angiotensin-converting enzyme inhibitors, angiotensin-receptor antagonists, calcium channel blockers, α-blockers, centrally active antihypertensive drugs, hydralazine. “Diabetes mellitus” was defined as present when participants reported a diagnosis of diabetes mellitus, or used antidiabetic medication, or had an elevated fasting serum glucose of ≥200 mg/dl. “Stroke” was defined as present when a history of stroke was noted from participants’ records or history. Events of strokes that occurred between the first and the third visit were validated by external experts. “CHD” was defined as present when a history of myocardial infarction, coronary revascularisation, and/or coronary artery disease was noted from participants’ records or history. Events of CHD that occurred between the first and the third visit were validated by external experts. “Polypharmacy” was defined as present when participants reported intake of five or more different medications within the last 7 days before examination. “Female hormone intake” (women only) was defined as present when women answered “yes” to the following question: “Have you taken hormone preparations (not the ‘pill’) for at least 1 month in the last 5 years?”.
Statistical analysis
We estimated the crude and sex-specific prevalence of all headaches, MigA+, MigA− and nonMigHA. Demographic data and possible risk factors were presented as mean ± standard deviation (SD), numbers, and percent rates. Multiple logistic regression was used to estimate the association between possible risk factors and headache activity (active vs. inactive) by estimating sex and age-adjusted odds ratios and 95% confidence intervals (OR [95%CI]) or sex-stratified and age-adjusted OR and 95% CI, respectively. If the 95% CI includes the one, the p-value is greater than 0.05 and the estimate is not significant. All analyses were performed using SAS 9.4 (Statistical Analysis System Corp., Cary, NC, USA).
Results
Table 1 presents the characteristics of all 2038 study participants (51% women, mean age: 72.9 ± 5.1 years). One thousand, four hundred and four (69%) of the participants reported ever having had a headache, with a higher percentage in women (79.0%/58.4%). More women than men reported ever having had migraines. The lifetime prevalence of MigA+ was 8.2% (n = 168, 76% women), of whom 71 (3.5%) were still active and 97 (4.8%) were in remission. The lifetime prevalence of MigA− was 20.4% (n = 416, 71% women), of whom 121 (5.9%) were still active and 295 (14.5%) were in remission. Eight hundred and twenty (40.2%) had nonMigHA, of whom 383 (18.8%) were still active and 433 (21.2%) were in remission; see also Figure 2, which shows the lifetime prevalence of active and inactive headache subtypes of all study participants. Further, Table 1 presents that 168 (8.2%) had episodic infrequent migraine, 10 (0.5%) had episodic frequent migraine and five (0.2%) had chronic migraine. Three hundred and fifty-one (17.2%) had episodic infrequent nonMigHA, 12 (0.6%) had episodic frequent nonMigHA and five (0.2%) had chronic nonMigHA. Overall, 10 (0.5%) had chronic headache. In Supplemental Tables 1 to 3, the characteristics of the study population are presented as sex specific. In Supplemental Table 4, the migraine and aura symptoms for MigA+ and MigA− are presented according to migraine activity.
Characteristics of the study population according to the headache subtype; mean ± SD, n (%).

MigA+: migraine with aura; MigA−: migraine without aura; nonMigHA: non-migraine headache; nHA: never headache; SD: standard deviation; CHD: coronary heart disease.
*missing headache activity, n = 4.

Prevalence of active and inactive headache subtypes of all participants aged 65 years or older.
The majority of participants with headache remitted at a mean age of about 53 years (women: 53.5 ± 12.7 years; men: 52.5 ± 15.7 years). Participants with migraine remitted at younger mean age than those with nonMigHA (MigA+: 52.4 ± 12.6 years; MigA−: 50.6 ± 12.8 years; nonMigHA: 55.4 ± 14.9 years).
Gender and age were the most important risk factors for headache activity in older people (see also Supplemental Tables 2 and 3). Active headache and active migraine were more common in women (all headache: n = 335 (32.3%); migraine: n = 135 (13.0%)) than in men (all headache: n = 240 (24.0%); migraine: n = 57 (5.7%)). Participants with active headaches were slightly younger than participants with inactive headache (72.0 ± 4.9 years vs. 72.7 ± 5.1 years, [crude OR: 0.97 (95% CI: 0.95; 0.99)]), reflecting the fact that the prevalence of headache decreases with age. Therefore, all other associations were given as sex- and age-adjusted, or stratified by sex and age adjusted, respectively.
Figure 3 shows the associations between the activity of all headaches, migraine, and nonMigHA, and possible associated risk factors compared to the respective inactive headache subtype. Headache activity in older people, in general, was associated with depression (OR: 1.62 [95% CI: 1.06; 2.47]), less sleeping time (0.96 [0.88; 1.05]), poor sleep (1.06 [1.00; 1.12]), and taking sleeping pills (1.06 [1.01; 1.12]). Activity in older migraineurs was associated with depression (2.55 [1.44; 4.50]), less sleeping time (0.92 [0.81; 1.05]), poor sleep (1.11 [1.03; 1.21]), and taking sleeping pills (1.06 [0.98; 1.15]). Participants with still active migraines were less likely to do sports (0.72 [0.51; 1.03]) compared to those with inactive migraines. Subjects with active headache were less likely to take antihypertensive medication (0.80 [0.64; 1.00]) compared to those with inactive headache.
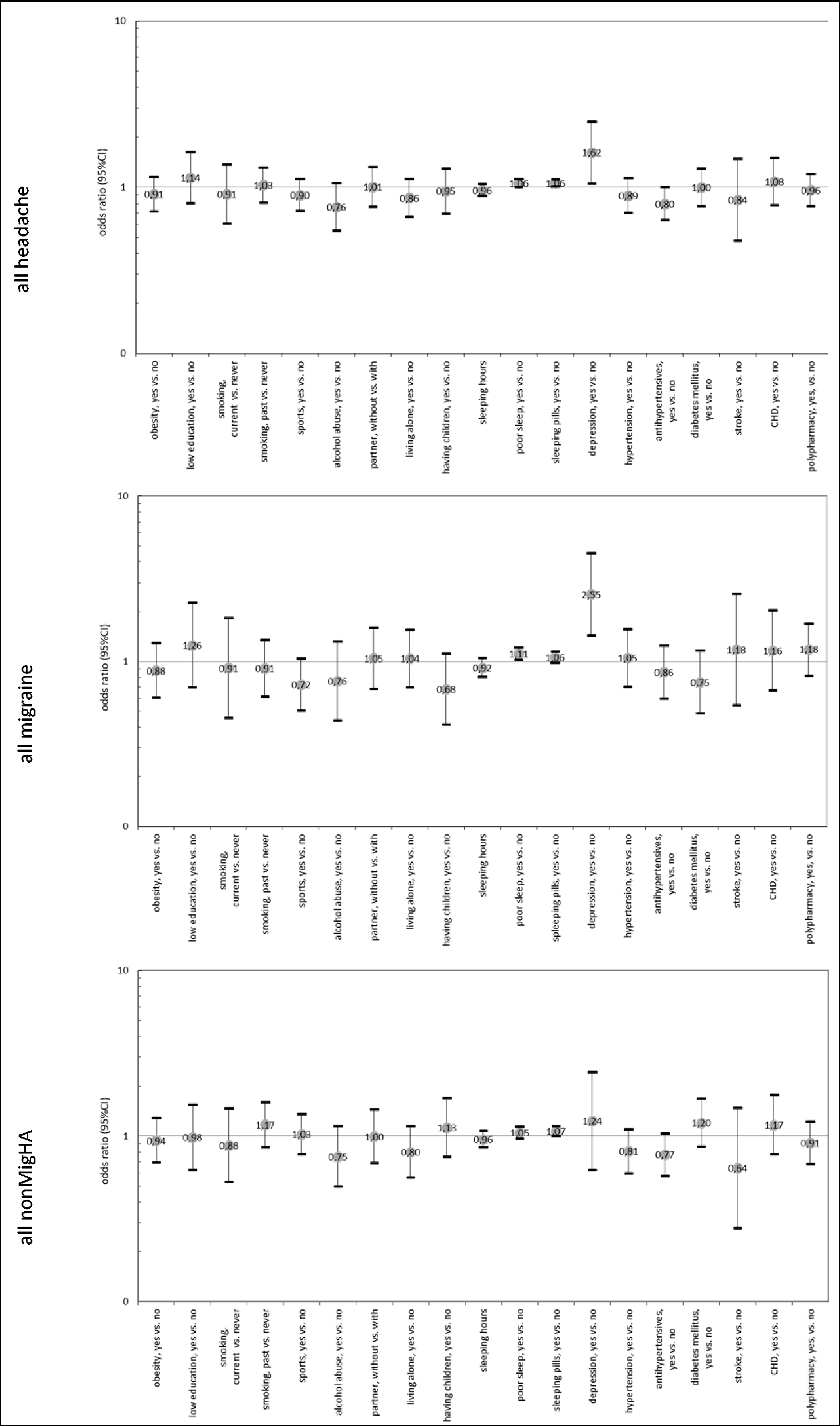
Association between possible risk factors and headache activity (age and sex adjusted OR and 95% CI, reference: inactive headache subtype).
Figures 4 and 5 show the sex-specific associations between the activity of headache, migraine, and nonMigHA and possible associated risk factors compared to the inactive headache subtypes. Active MigA+ in women was significantly associated with depression (3.67 [1.28; 10.49]). Women with active MigA+ were more likely to take female hormones (2.57 [0.88; 7.50]) and less likely to do sports (0.60 [0.30; 1.22]). Active MigA− in women was significantly associated with poor sleep (1.17 [1.04; 1.32]) and also associated with less sleeping time and intake of sleeping pills, with no significance. Women with active MigA− were also more likely to have depression (2.19 [0.98; 4.91]). There was no association between active MigA− and female hormone intake. Females with regular intake of several medications (polypharmacy) (0.72 [0.47; 1.10]) and especially of antihypertensive medication (0.66 [0.43; 1.00]) were less likely to have active nonMigHA (Figure 4). For men, the numbers for MigA+ were very small and did not allow meaningful analysis (Figure 5). Men with active MigA− also tended to sleep less (0.78 [0.59; 1.03]) and have poor sleep (1.11 [0.92; 1.35]) and depression (2.43 [0.31; 18.76]) compared to men with inactive MigA−, but there was no association with taking sleeping pills (0.97 [0.78; 1.21]). The associations between headache activity and CHD in women and men was controversial. Men with active nonMigHA tended to report CHD more often compared to those with inactive headache (1.55 [0.97; 2.47]). In women, it was the other way around (0.38 [0.15; 1.01]), but numbers in women were very small. We did not observe any associations between headache activity and stroke in both men and women.

Association between possible risk factors and headache activity in women (age adjusted OR and 95% CI, reference: inactive headache subtype).

Association between possible risk factors and headache activity in men (age adjusted OR and 95% CI, reference: inactive headache subtype).
The intake of possible acute and preventative medication according to the German guidelines of migraine therapy (21) is given in Table 2. Participants with active headaches more often used analgesics (22.1%) compared to participants with inactive headaches (17.6%) and those who never reported any headaches (16.9%). Very few used triptans (n = 2). Many study participants used beta-blockers, with a higher proportion in migraineurs (36.8%) compared to nonMigHA (30.3%) and people who never experienced headaches (32.2%). Also, the proportion of intake of antidepressants was higher in migraineurs (6.0%) compared to nonMigHA (4.3%) and those who never had headaches (“never headache”, 3.0%). The same was true with anticonvulsants (3.8% migraineurs, 1.6% nonMigHA, 3.0% never headache). We, however, were not able to evaluate the reasons for the medication intake (e.g. beta-blocker as antihypertensive or migraine preventative). No participant used ergotamins or botulinum toxin. Figure 6 shows the association between medication intake and headache activity, age and sex adjusted. Participants with active headaches were more likely to take analgesics (1.35 [1.03; 1.76]) and also tended to take antiemetics (1.95 [0.74; 5.17]) and anticonvulsants (1.22 [0.62; 2.39]) compared to participants with inactive headache. They were less likely to take antihypertensives (0.80 [0.64;1.00]). There was no association between intake of beta-blockers and headache activity (0.93 [0.74; 1.17]), but participants with active headache tended to report intake of ACE inhibitors (especially those with nonMigHA (0.67 [0.41; 1.09])) and angiotensin II receptor antagonists (0.83 [0.57; 1.19]) less frequently.
Intake of possible acute and preventative migraine medication, n (%).

MigA+: migraine with aura; MigA−: migraine without aura; nonMigHA: non-migraine headache; nHA: never headache.
*missing headache activity, n = 1 women and n = 3 men.

Association between medication intake and headache activity (age and sex adjusted OR and 95% CI, reference: inactive headache subtype).
Discussion
Our study provides current prevalence estimates of headache in older people in Germany. In general, older women had more headaches and migraines than older men. All kinds of headaches declined with age with a mean remission age of about 53 years. Depression and poor sleep quality contribute to persisting headache and migraine in older age and physical activity contributes to migraine remission.
In our study, the prevalence of active headache in participants aged 65 years and older was 28%, of whom 3% had active MigA+, 6% had active MigA− and 19% had active nonMigHA. Prencipe et al. reported comparable results from Italy: The one-year prevalence of migraine was 11%, for tension-type headache 42%, and for chronic headache 4.4% (22). An Austrian study estimated a one-year prevalence for migraine of about 6% and a one-year prevalence for tension-type headache of 36% (23). The numbers in an elderly Chinese population were lower: Migraine 3% and chronic daily headache 3.9% (24,25). The reported headache frequency in our study is based on the previous 3 months. The majority of study subjects had episodic infrequent headache/migraine, some had episodic frequent headache/migraine and a minority had chronic headache/migraine. This distribution is typical for migraine and tension-type headache in the majority of studies (8). A recent German study evaluated prevalence of primary headaches and headache frequency in a population-based sample aged 14–94 years but did not differentiate between migraine and tension-type headache. The 6-month prevalence rate of headache was 39%, it peaked at the age of 35–55 years and decreased with older age. Twenty-seven percent of participants older than 75 years suffered from headaches and reported a significant headache-related burden and frequent analgesic intake (26). Our data demonstrate that overall, about 1.5% of the general population 65 years of age and older suffer from frequent or daily headache and therefore requires preventative treatment.
In our study, the lifetime prevalence of headache was 69% (women 79%, men 58%). The prevalence is similar to the literature, where a global lifetime prevalence for all primary headaches of 64% was reported (4). The lifetime prevalence of migraine was 29% (women 12.2%, men 4.1%), of whom 21% had MigA− and 8% MigA+; these numbers are also in line with the international literature (8).
Further results of our study are that the majority of people, both men and women, with headaches remitted at the age of about 53 years. Factors associated with headache and migraine activity in the elderly were depression, poor sleep quality and, in migraine only, less sports activity. In general, women had more headaches and migraines than men and all kinds of headaches declined with age.
Higher headache prevalence in women is well documented in the literature (3,27,28). The relationship between age and headache in general, migraine, and tension-type headache is well known too. Migraine reaches a peak during the fourth decade of life and remains constant during the fifth decade, tension-type headache peaks between the ages of 30 and 39 years (2,29). The lowest prevalence of tension-type headache and migraine is at age 70 years and older (2,23,28,30,31).
The association between migraine and depression has been reported previously (32). Studies reported that depression and anxiety disorders are more prevalent in migraineurs than in the general population (33) and are related to poorer quality of life and increased suicide risk (34–36). In addition, psychiatric comorbidities are risk factors for the progression from episodic to chronic migraine (37). A strong association between migraine and depression was confirmed in a population-based cohort of elderly people as well (38).
Sleep disturbances and migraine share a complex relationship that is poorly understood (39). The relationship is complex because it involves several sleep disorders, including respiratory and non-respiratory conditions. Poor sleep is frequently present during the premonitory or postdrome migraine phases (40,41). Patients with migraines frequently report increased insomnia complaints interictally – including difficulty initiating sleep, staying asleep, poor sleep quality, self-reported decreased total sleep time, excessive daytime sleepiness, and lack of refreshment after sleep (42). This relationship is robust and persists when controlling for comorbid anxiety and depression (43). The relationship is bidirectional since migraine and nonMigHA are risk factors for later development of insomnia (44). Poor sleep is associated with chronic migraine (45) and may also be a risk factor for migraine progression (46). Our findings confirm the results of previous studies. We, however, cannot further elucidate details of this complex relationship between headache, migraine, and sleep.
In our study, being active in sports even in the elderly seems to correlate with remission of migraines. The relationship between migraine and physical activity has been studied extensively. Various large population-based studies demonstrated that a low level of physical activity is associated with a higher prevalence of migraine and other headaches as well (47–49). Vice versa, higher physical activity levels are associated with lower migraine frequency and with less migraine-related disability (50–52). Finally, the efficacy of exercise as a prophylactic treatment for migraine has been documented in several studies (53–55). The relationship with less physical activity and higher prevalence of migraines in our study is a result of a cross-sectional investigation and does not confirm but suggests that being active in sports in the elderly might be positive for the prevention of migraines.
We documented any intake of medication. Most study participants used analgesics for acute treatment and only two used triptans, which is not surprising because the use of triptans is not recommended in the elderly. We observed that many of the study participants received beta-blockers, ACE inhibitors, antidepressants, and anticonvulsants but we were not able to distinguish whether these medications were used as migraine preventatives or for comorbidities such as hypertension or depression. However, the proportions of beta-blockers, antidepressants and anticonvulsants were higher in migraineurs. Women with active MigA+ were more likely to take female hormones, but there was no association between active MigA− and female hormone intake. We observed a significant association between the use of antihypertensives and the remission of headaches. Stratified by classes, ACE inhibitors (especially in participants with nonMigHA) and angiotensin II receptor antagonists appeared to be responsible for the effect, but not beta-blockers. Calcium-channel blockers were taken too rarely. It is well known that some antihypertensive drugs are effective prophylactics for migraines, including beta-blockers, calcium-channel blockers, and angiotensin II receptor blockers (56), and the positive effect of antihypertensives on headache prevalence has been shown earlier (57,58). However, it remains unclear whether the association is causal (59). Meta-analyses have shown that antihypertensives reduced the incidence of non-specific headaches compared to placebo (57,58). The effect was particularly strong for beta-blockers. Law et al. concluded that lowering systemic blood pressure is probably the primary mechanism (58). The studies also demonstrated significant heterogeneity between drug classes in their effects on headache, which could not be explained by differences in mean blood pressure. Our study confirms the results for antihypertensives in general but not for beta-blockers. We cannot provide a conclusive explanation as to why beta-blockers were not associated with headache remission in our study. This could be because of many possible unspecific interactions. In our study, the positive effect of antihypertensive drugs on the occurrence of headaches in older people was most likely due to the use of ACE inhibitors and angiotensin-II receptor antagonists.
We did not find any associations with gender, obesity, education, and smoking, which were described as possible risk factors for chronic migraine (60). Obesity is associated with the frequency and severity of migraine attacks (61), but it remains unclear whether the relationship between obesity and migraine is causal. In our study, there was no clear association between obesity and headache activity in older people. Studies describe a link between migraine severity and lower educational status, but most have not been adjusted for painkiller abuse and may have been misinterpreted (62). Whether education has an impact on migraine or not remains unclear. In our study, there was no clear association between low education and headache activity in older people. The data on the influence of smoking on the severity of migraine are heterogeneous (62,63). Headache is also a known withdrawal syndrome in nicotine withdrawal (64). In our study, there was no clear association between smoking status and headache activity in older people. Migraine is associated with a significant burden on individuals but also on their social environment. Migraine affects family activities and relationships (65,66). Therefore, we have investigated whether partnership status, living alone, or having children had an influence on the activity of migraine in older people, but we couldn’t detect one. None of the above studies investigated the relationship between sex, obesity, education, smoking, partnership status, living alone, and having children and migraine activity in older people.
It has been shown that migraine, especially migraine with aura, is associated with a long-term risk for cerebrovascular and cardiovascular events (67). In the Northern Manhattan Study, migraine was associated with an increased risk of stroke among active smokers but not among non-smokers (68). In the Oxford Vascular Study, migraine was strongly associated with ischemic stroke, particularly at older ages (69). Another study in elderly Chinese has shown that the prevalence of migraine was significantly higher in elderly Chinese patients with acute cerebral infarction (70). Epidemiological and genetic studies also suggest an overlapping pathophysiological mechanism in migraine and CHD. Migraine has been shown to be associated with cardial events such as myocardial infarction, atrial fibrillation or flutter and heart failure (67,71). However, the extent of coronary calcification in patients with or without migraine does not appear to differ (72,73). This suggests that coronary artery calcification as a marker of coronary atherosclerosis is not the cause of increased cardiovascular risk in migraine patients. Genome-wide association (GWA) studies revealed a significant number of gene loci associated with migraine that code for various proteins of the blood vessels and smooth muscle tissue (74). Among these genes are those that specifically increase the risk of migraine and CHD, but also genes that are important for endothelial function. This suggests common biological processes for both diseases, such as endothelial dysfunction or vasospasms (72,75). In contrast to the previous studies, we investigated the likelihood of headache remission in participants with stroke or CHD. We did not detect any significant associations, probably due to the small number of participants with migraine and stroke or CHD. The sample size did not allow further stratification for other vascular risk factors such as smoking and diabetes.
Concepts for the physiology of the transformation of active migraine into an inactive form in older people are lacking. Migraine attacks become shorter and less typical with age (76–78). Aura without headache tends to occur later in life and is more common in older migraine sufferers (79,80). In our study, we unfortunately did not examine isolated aura without a headache. In older people, neurovascular inflammation, which is believed to be responsible for migraine pain, appears to be less pronounced (81). Other reasons for reduced pain may be age-related changes in blood vessels or the central pain-transmitting pathways (7). Stress appears to be less important in the headache etiology of older people. Unlike younger migraine sufferers, older people do not experience an increase in headache frequency as stress increases (82). Different hormone fluctuations in women and men over their entire life span were discussed as possible explanations for the gender-specific differences in migraine prevalence (83,84), but the higher prevalence in women, which is still present in older people, cannot be explained by circulating hormones alone (5). Menopause also seems to be an insufficient explanation, since the mean remission age of about 53 years in our study did not differ between women and men. A better understanding of the physiology of migraine remission in older people will help to improve clinical migraine management.
Our study has several strengths and limitations. Strengths are the large, randomly selected population-based sample and the high quality of data collection and processing, which was confirmed by external certification of the HNR study. Limitations of our study are that standardised interviews were only conducted by trained study nurses, but not by neurologists. Our questionnaire was a modified version of the validated questionnaire of the German Headache Consortium study (11). The original questionnaire was developed and validated for headaches and migraine symptoms within the last 12 months, but we asked for headache and migraine symptoms that had ever occurred in the lifetime. The headache questions were only asked at the third visit (10-year follow-up). We have pooled probable and definitive migraines because of the small numbers. The migraine diagnosis in our study is not as reliable as in a clinical setting. Misclassifications are possible. This is not unusual in epidemiological studies. We did not collect data on secondary headache prevalence, which increases with age (85). Since primary headache types are also the most common headache types in older people, we assume that the majority of migraine headaches are correctly classified, but misclassification cannot be ruled out. Overall, our study provides essential data on the elderly German population.
Conclusions
Headaches and migraines are not rare in people from the age of 65 years up. One-quarter of older people still have active headaches. One-third of all migraine sufferers still have an active migraine from the age of 65. The estimated prevalence of active migraine in older people is 13% in women and 5% in men. The majority have migraines on less than 9 days a month, but 1.2% of women and 0.3% of men still have migraines on 9 days a month or more. Overall, about 0.5% of older people have chronic headaches. Depression and poor sleep quality contribute to headache and migraine in general. Intake of antihypertensives contribute to headache remission and physical activity contributes to remission of migraine.
Clinical implications
More awareness of active migraine in older people is necessary, because it is not rare. The estimated prevalence of active migraine in older people is 13% in women and 5% in men. Although the majority of older migraineurs have migraines on less than 9 days a month, 1.2% of older women and 0.3% of older men still have migraines very often, 9 days a month or more, which requires specific acute and preventive medication. Depression and poor quality of sleep contribute to headaches and migraines in older people and taking antihypertensives reduces headaches. A modification of these factors could have a positive effect on the occurrence of headaches in older people. Physical activity is associated with remission of migraine. Therefore, it can be used as an additional argument in favor of regular physical activity in elderly migraineurs.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420977183 - Supplemental material for Prevalence and risk factors of migraine and non-migraine headache in older people – results of the Heinz Nixdorf Recall study
Supplemental material, sj-pdf-1-cep-10.1177_0333102420977183 for Prevalence and risk factors of migraine and non-migraine headache in older people – results of the Heinz Nixdorf Recall study by Sara Schramm, Isabell Tenhagen, Börge Schmidt, Dagny Holle-Lee, Steffen Naegel, Zaza Katsarava, Karl-Heinz Jöckel and Susanne Moebus in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420977183 - Supplemental material for Prevalence and risk factors of migraine and non-migraine headache in older people – results of the Heinz Nixdorf Recall study
Supplemental material, sj-pdf-2-cep-10.1177_0333102420977183 for Prevalence and risk factors of migraine and non-migraine headache in older people – results of the Heinz Nixdorf Recall study by Sara Schramm, Isabell Tenhagen, Börge Schmidt, Dagny Holle-Lee, Steffen Naegel, Zaza Katsarava, Karl-Heinz Jöckel and Susanne Moebus in Cephalalgia
Footnotes
Acknowledgments
The authors express their gratitude to all study participants of the HNR Study, the personnel of the HNR study centre and the EBT-scanner facilities, the investigative group and all former employees of the HNR study. The authors also thank the Advisory Board of the HNR Study: T Meinertz, Hamburg, Germany (Chair); C Bode, Freiburg, Germany; PJ de Feyter, Rotterdam, Netherlands; B Güntert, Hall iT, Austria; F Gutzwiller, Bern, Switzerland; H Heinen, Bonn, Germany; O Hess, Bern, Switzerland; B Klein, Essen, Germany; H Löwel, Neuherberg, Germany; M Reiser, Munich, Germany; G Schmidt, Essen, Germany; M Schwaiger, Munich, Germany; C Steinmüller, Bonn, Germany; T Theorell, Stockholm, Sweden; and SN Willich, Berlin, Germany.
Authors’ contributions
SS: Analysis and interpretation of data, drafting the manuscript. KHJ: Study concept and design of the HNR study, assistance with drafting the manuscript for intellectual content. All authors assisted with drafting the manuscript for intellectual content. All authors read and approved the final manuscript.
Availability of data and material
The corresponding author has full access to all data in the study and final responsibility for the submission of the article for publication. Due to data security reasons (i.e. data contain potentially participant identifying information), the HNR study does not allow sharing of data as a public use file. Data requests can also be addressed to recall@uk-essen.de.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS, IT, BS, KHJ, and SM have no competing interests. ZK received honoraria from Allergan, Novartis, Lilly, TEVA, Merck, and Bayer. DHL received honoraria from Allergan, Novartis, Lilly, Hormosan, Bayer, and Teva. SN received honoraria from Novartis, Lilly, and TEVA.
Ethics approval and consent to participate
This study was approved by the ethical commission of the Medical Faculty of the University Duisburg-Essen.
Consent for publication
All participants gave informed written consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This subproject of the HNR Study was not funded. The HNR study was supported by the Heinz Nixdorf Foundation (Chairman: Martin Nixdorf; Past Chairman: Dr jur. Gerhard Schmidt). Parts of the HNR study were also supported by the German Research Council (DFG) (DFG project: EI 969/2-3, ER 155/6-1;6-2, HO 3314/2-1;2-2;2-3;4-3, INST 58219/32-1, JO 170/8-1, KN 885/3-1, PE 2309/2-1, SI 236/8-1;9-1;10-1,), the German Ministry of Education and Science (BMBF project: 01EG0401, 01GI0856, 01GI0860, 01GS0820_WB2-C, 01ER1001D, 01GI0205), the Ministry of Innovation, Science, Research and Technology, North Rhine-Westphalia (MIWFT-NRW), the Else Kröner-Fresenius-Stiftung (project: 2015_A119) and the German Social Accident Insurance (DGUV project: FF-FP295). Furthermore, the HNR study was supported by the Competence Network for HIV/AIDS, the deanship of the University Hospital and IFORES of the University Duisburg-Essen, the European Union, the German Competence Network Heart Failure, Kulturstiftung Essen, the Protein Research Unit within Europe (PURE), the Dr. Werner-Jackstädt Stiftung and the following companies: Celgene GmbH München, Imatron/GE-Imatron, Janssen, Merck KG, Philips, ResMed Foundation, Roche Diagnostics, Sarstedt AG & Co, Siemens HealthCare Diagnostics, Volkswagen Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
