Abstract
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is caused by mutations in the NOTCH3 gene and is clinically characterized by recurrent stroke, cognitive decline, psychiatric disturbances and migraine. The prevalence of migraine in CADASIL is slightly higher than in the general population, and the proportion of migraine with aura is much higher. The pathophysiological mechanism that leads to increased aura prevalence in CADASIL is unknown. Possible mechanisms of the excess of migraine with aura are an increased susceptibility to cortical spreading depression (CSD) or a different expression of CSD. It is also possible that the brainstem migraine area is involved in CADASIL. Last, it is possible that the NOTCH3 mutation acts as a migraine aura susceptibility gene by itself.
In this narrative review we summarize the literature about migraine in CADASIL, with a special focus on what CADASIL might teach us about the pathophysiology of migraine.
Introduction
Migraine is a common, disabling, episodic headache disorder with a lifetime prevalence of around 13% in men and 33% in women (1). Aura symptoms may occur in around 31% of patients (1). The pathophysiology of migraine headache and aura symptoms is largely unknown (2). The classical vascular theory that migraine aura is primarily caused by vasoconstriction and migraine headache by vasodilatation is too simplistic (3). Part of the current theory is that migraine aura results from cortical spreading depression (CSD) (4,5), which is a short-lasting depolarization of neuronal and glial cell membranes spreading over the brain cortex. Activation of the trigeminal vascular system plays a role in migraine headache, but its exact mechanism is unknown. However, there are indications that blood vessels also play an important role in the migraine attacks. Some genetic and acquired vasculopathies have migraine as one of their main symptoms (6), and some animal studies show evidence for a strong vascular component in migraine pathophysiology (7,8).
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is a monogenic disease characterized by middle-age onset of cerebrovascular disease that often progresses to dementia (9). Around 30% of CADASIL patients get affected by migraine attacks—the majority with aura—often as the first symptom of the disease. CADASIL is a microvascular disease with partly known pathophysiology. It is caused by mutations in the NOTCH3 gene located on chromosome 19 (10). The NOTCH3 gene encodes for a transmembrane receptor which is solely expressed in vascular muscle cells in humans. Histopathological findings in CADASIL consist of degeneration of vascular smooth muscle cells with adjacent deposits of granular osmiophilic material (GOM) and fibrous thickening of the arterial walls. The microvascular changes are systemic, but the cerebral vasculature carries the brunt of the pathology (11). Cerebral MRI reveals characteristic white matter hyperintensities (WMHs) with or without lacunar infarctions and microbleeds in symptomatic, as well as asymptomatic adult mutation carriers (MCs) (12,13). The number, sites and type of lesions are age-dependent. Sites of predilection of WMHs are the anterior temporal lobe, the periventricular white matter, external and internal capsule and the pons (12,13). Lacunar infarcts are associated with cognitive dysfunction (14). In this narrative review we focus on migraine in CADASIL.
Search strategy
We electronically searched PubMed by using the search terms CADASIL and migraine. We checked reference lists of retrieved studies and reviews for additional reports of relevant studies. We also used unpublished data from patients in our Dutch CADASIL database (12). Retrieved articles were studied for migraine lifetime prevalence, subtype, associated risk factors and MRI characteristics. Studies were also included in our epidemiological survey if they included 10 or more patients or patients from more than five families, and if inclusion was independent from the presence of migraine. The overall prevalences of migraine and a 95% confidence interval (CI) were calculated after pooling all studies together. Gender risk ratios were calculated for studies with more than 10 patients with migraine. If multiple publications were retrieved from the same research center and it was not stated if the study populations overlapped, only the study with the largest number of patients was used.
Retrieved articles
We identified 27 articles that reported on migraine characteristics in CADASIL. Fifteen articles matched our inclusion criteria for the epidemiological analysis. Three studies were excluded because of possible overlap with other studies. Three studies were excluded because the inclusion was based on migraine diagnosis, and six studies were excluded because of insufficient sample size. In all remaining studies, diagnosis of migraine and of migraine with aura was based on a direct interview by a physician.
Epidemiology of migraine in CADASIL
Epidemiological surveys of migraine in CADASIL
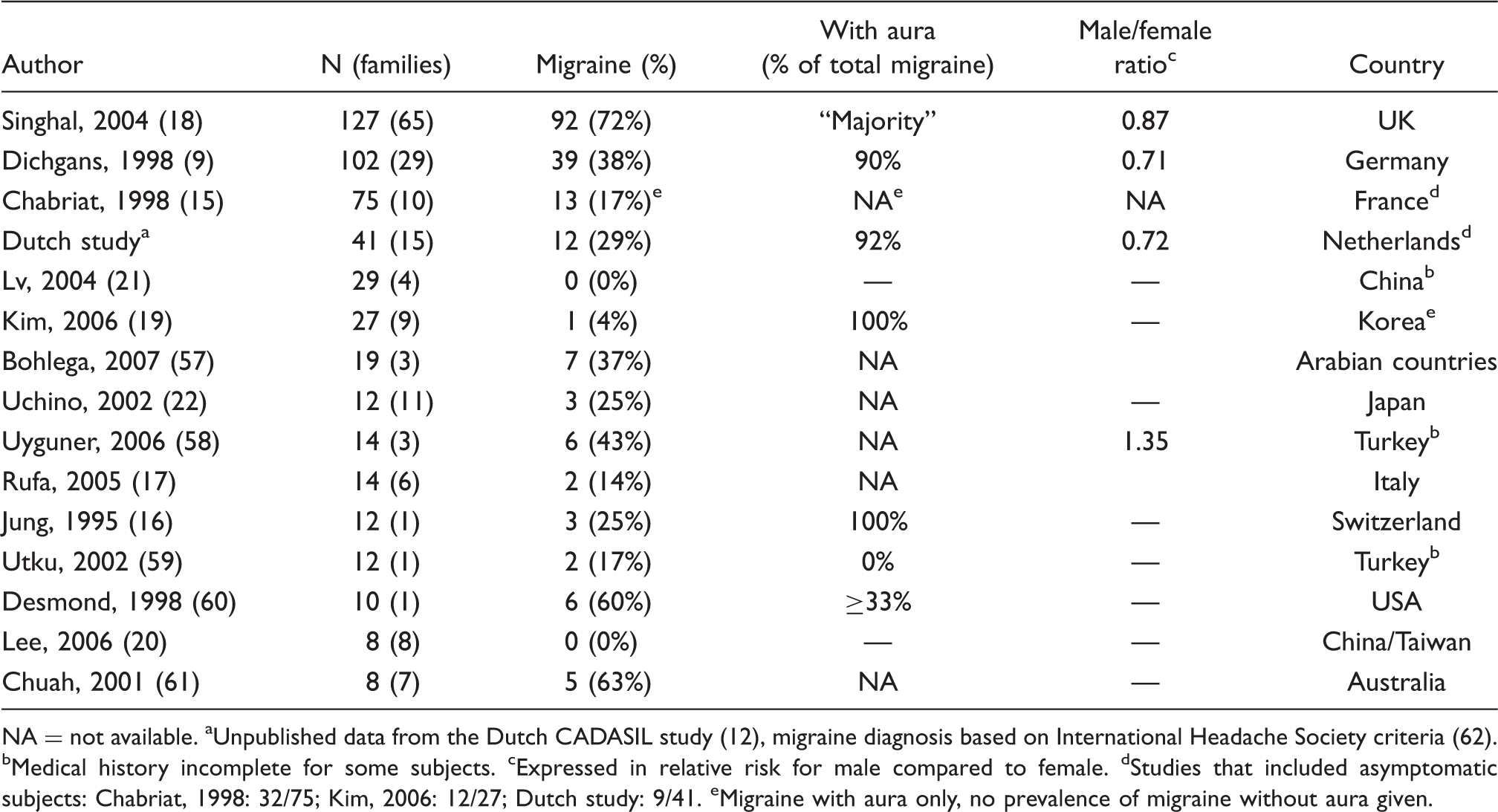
NA = not available. aUnpublished data from the Dutch CADASIL study (12), migraine diagnosis based on International Headache Society criteria (62). bMedical history incomplete for some subjects. cExpressed in relative risk for male compared to female. dStudies that included asymptomatic subjects: Chabriat, 1998: 32/75; Kim, 2006: 12/27; Dutch study: 9/41. eMigraine with aura only, no prevalence of migraine without aura given.
The migraine prevalence in CADASIL was higher in females than in males, but this difference did not reach statistical significance. One study found lower age of onset of migraine with aura in females (26.2 years) than in males (35.8 years) (23). Dichgans et al. (9) reported that 14% of patients had an increase in attack frequency before their first ischemic event, and that 54% reported a decrease or cease of migraine attacks after the first stroke.
The percentage of migraine with aura could only be inferred from two of the larger studies (Table 1) (9,12), which showed that the majority (80–90%) of migraineurs with CADASIL have migraine with aura.
Migraine aura subtype
Vahedi et al. (23) reported that the migraine aura in CADASIL is typical in 44% of cases, while the other 56% also experience atypical features: aura without headache, hemiplegic auras, basilar-type migraine symptoms, prolonged aura and acute-onset aura.
Associated factors
Some studies looked for factors that might possibly modulate migraine susceptibility in CADASIL. One study reported an association between hyperhomocysteinemia and younger age of onset of migraine in CADASIL (18). Hyperhomocysteinemia was not associated with the presence of stroke or with WMHs on MRI. No relation was found between other cardiovascular risk factors (hypertension, hypercholesterolaemia, diabetes, smoking, serum cholesterol, ApoE genotype) and migraine. Two case studies reported CADASIL families with a high prevalence of migraine, which suggests that the NOTCH3 genotype or other genetic or environmental factors play a role in susceptibility to migraine in CADASIL (24,25). However, larger studies failed to confirm an association between NOTCH3 genotype and migraine in CADASIL (9,18).
MRI abnormalities
Vahedi et al. (23) investigated possible associations between MRI abnormalities and migraine in CADASIL and found that MRI findings (WMHs and lacunar infarcts) did not differ between patients with migraine with aura and those without migraine. Also, no association was found between migraine and WMHs and lacunar infarcts in pons, occipital lobe and cerebellum.
Imaging studies of cerebral hemodynamics in CADASIL reported decreased total cerebral blood flow (26) and decreased cerebral perfusion in normal and abnormal appearing white matter (27). Also, decreased vasoreactivity was found in areas of white matter hyperintensities. It was not reported whether these flow changes are associated with migraine. In one case study a SPECT (single photon emission computed tomography) examination was performed during a prolonged aphasic aura attack in a CADASIL patient, showing hyperperfusion in the left hemisphere on day 5 of the aura (28).
Possible mechanisms of migraine in CADASIL
This narrative literature review shows that the prevalence of migraine in CADASIL is slightly higher than that of the general population. Females may be affected more frequently than males in CADASIL, but this difference was relatively small. Migraine prevalence varied by geography, showing higher prevalences in Europe and lower reported prevalences in Asian countries, which coincides with general epidemiological data (2,29). Aura symptoms occurred much more frequently in CADASIL than in the general population. The pathway through which the NOTCH3 genotype leads to an excess of migraine with aura is unknown. In our opinion, there are several possible theories for the increased aura prevalence in CADASIL.
First, as CADASIL is a vascular disease with decreased blood flow and cerebrovascular reactivity, migraine aura could be a result of episodic ischemia. This would be consistent with the classical vascular theory for migraine. However, animal experiments and in vivo functional MRI (fMRI) studies have demonstrated that migraine aura is not caused by ischemia, but by CSD (4,5), a short-lasting depolarization of neuronal and glial cell membranes, that spreads across the brain cortex. CSD is associated with cerebral blood flow (CBF) changes, which include phases of hyperperfusion and hypoperfusion (30,31). In normal conditions these flow changes, as observed during a CSD in cats, do not reach ischemia tresholds (30). However, an experiment in rats showed that when the brain is in a preexisting state of hypotension or hypoxia, the flow changes, especially the early hypoperfusion phase, become more pronounced and more prolonged (32). It is possible that a similar mechanism occurs in CADASIL patients since these patients have a decreased baseline level of cerebral blood flow (26,27). A more pronounced hypoperfusion phase, or a prolonged hyperperfusion phase, as has been observed in a CADASIL patient (28), could affect the type and intensity of the aura. Since hypoperfusion as well as prolonged hyperperfusion has been associated with prolonged duration of migraine aura (28,33), it is possible that these flow changes lead to more impairment of cortical function during the attack. For example, normal CSDs, which usually leads to typical visual aura in the general migraineurs, may cause atypical prolonged hemiplegic aura in CADASIL, because of the pronounced flow changes (23). Also, it has been suggested that some migraine patients have silent or subclinical CSDs that do not lead to aura symptoms (34–36). If these CSDs are accompanied by more pronounced flow changes in CADASIL patients, it is possible that these CSDs become symptomatic. This could explain the increased aura prevalence in CADASIL. If CSDs cause more intense flow disturbances, one might possibly expect that CADASIL patients with migraine have more WMHs and lacunar infarcts than CADASIL patients without migraine, but Vahedi et al. failed to demonstrate this (23). However, this might be hard to demonstrate as the contribution of migraine-related MRI abnormalities might be overshadowed by the full spectrum of MRI abnormalities in CADASIL.
A second theory is that in CADASIL there is an increased cortical susceptibility to CSD, which could be caused by chronic vascular brain damage or acute episodes of hypoperfusion. In the general migraine population, structural and functional MRI studies, magnetoencephalograpy and visual stimulation studies suggest that migraine is caused by a hyperexcitable brain state (37). It is possible that the cerebral cortex in CADASIL is more susceptible to CSD because of chronic vascular brain damage (infarcts, WMHs). A decreased threshold to CSD and to peri-infarct depolarizations that resemble CSD have already been demonstrated in mice with a deletion or mutation of the NOTCH3 gene (38–40). An additional mechanism leading to CSD may be that CSDs can be triggered by episodes of focal, mild and transient ischemia that do not cause requisite tissue injury. A close interaction between cerebral hemodynamics and neural activity has been suggested (41). This mechanism of vascular-induced CSD has been demonstrated in recent experimental data in mice (40,42). However, it should be realized that MRI abnormalities in the occipital lobe occur relatively late in CADASIL, while cortical hyperexcitability occurs mainly in the occipital cortex.
A third possibility is that the brainstem migraine area is involved in CADASIL. PET studies and functional MRI studies showed an area of activation located in the dorsal pons in migraine with and without aura (43–46). One MRI study reported that migraineurs with aura have an increased gray matter density in the periaqueductal gray (PAG) and dorsolateral pons (47). Also, some studies illustrated that migraine with and without aura can develop after a hemorrhage in a cavernoma in the PAG (48) or with a vascular malformation in the pons (49). Kruit et al. reported an increased prevalence of hyperintense lesions in the dorsal pons in migraineurs with aura and to a lesser extent in migraineurs without aura (50). Pontine WMHs are frequently observed in CADASIL (13,51). Therefore, it can be hypothesized that these pontine WMHs are directly or indirectly related to migraine with aura attacks in CADASIL.
A fourth possibility is that the microangiopathy and the migraine aura in CADASIL are not related. In the general population, family members of a migraine patient have a two-fold increased risk of having migraine (52). This risk is even higher for migraine with aura (53). Twin studies show that the heritability of migraine is 40–50% and that shared environmental factors are likely to have a minor effect on the susceptibility to migraine (54). Possibly, the NOTCH3 mutation is a genetic factor that acts in concert with other migraine aura susceptibility genes via pathways alternate to the vascular one. One study found a still unconfirmed association between the NOTCH3 polymorphism G684A and migraine (55). Another study however found no association between the NOTCH3 polymorphism T6746C and migraine (56).
Conclusion
The exact pathway through which the NOTCH3 mutation in CADASIL leads to an increased prevalence of migraine with aura is unknown. The monogenic disease CADASIL could serve as a model for the pathophysiology of migraine aura. Further research is needed, particularly into flow changes in CADASIL patients during auras and migraine attacks. Additional studies of structural MRI changes in relation to migraine in CADASIL are also needed.
