Abstract
Background and objective
The importance of neck pain and the trigeminocervical complex in migraine is of high pathophysiological interest since a block to the greater occipital nerve is more effective for some primary headaches than others. This observational study hypothesised that the response to manual palpation of the upper cervical spine predicts the efficacy of the greater occipital nerve-block.
Methods
We divided patients, scheduled by a neurologist to receive a greater occipital nerve-block to reduce their migraine symptoms, into three groups: Patients with no pain response to manual palpation of the neck, patients with local pain, and those with referred pain to the head. Primary outcome was the percentage change in headache frequency. Additionally, items from the quantitative sensory testing protocol were included.
Results
Eighty-seven chronic migraine patients were recruited consecutively from a specialised outpatient clinic and 71 were included for analyses and stratified into the three groups: No pain (n = 11), local pain (n = 28), and referred pain to the head (n = 32). Overall, patients experienced a reduction of 1.9 headache days per month (SD 3.4, p < 0,0001). The groups differed significantly in the percentage change of headache frequency (p = 0.041) with the “no pain” group showing the largest reduction. The pressure-pain-threshold over C2 and headache on the day of the intervention influenced the outcome significantly (R2 0,27, p = 0,00078). No serious adverse events occurred. Sixty-five percent of the patients had headaches during the examination. The groups did not differ regarding the distribution of patients with neck-pain in absence of migraine at baseline (p = 0.618).
Conclusion
Patients that were less sensitive to palpation in the cervical region and headache-free on the day of the intervention improved more after the greater occipital nerve-block.
Registration: Registered a priori at the German Clinical Trials Register (DRKS00015995).
Keywords
Introduction
Neck pain is prevalent in up to 76% of migraine patients (1,2) and therefore at least as frequently reported as nausea and photo/phonophobia. Photophobia has a prevalence of 74–78%, phonophobia of 67–76%, and nausea is reported by 36–55% of patients, depending on the data sources (3–5). Migraine patients show significantly more musculoskeletal findings in the cervical spine compared to non-headache controls (6) when using a standardised international consensus procedure (7).
Despite this obvious clinical significance, the (patho-) physiological implication of neck findings on migraine are not clear. A model for the connection between the trigeminal system and the greater occipital nerve has been suggested as the so-called trigeminocervical complex (TCC). Its foundation is the assumed convergence of trigeminal and cervical afferents (8,9). Afferent signals from the trigeminal nerve are led to and processed in the nuclei of the trigeminal nerve. Different modalities of stimuli are processed in different areas of the trigeminal nuclei: Pain afferents, which are of particular interest in headache research, synapse with second order neurons in the caudal part of the brainstem in the pars caudalis of the spinal trigeminal nucleus (10). In the upper cervical spine, cervical afferents innervate the suboccipital and upper posterior muscles of the neck and the spinal nerve C2, which is richly converged, forms the greater occipital nerve (GON) (11). Furthermore, collaterals from the trigeminal nerve synapse in the spinal dorsal horn with second order neurons that are also connected to afferents from C1-C3, and collaterals from C1-C3 synapse in nearby segments (8). This connection of the trigeminal and occipital/cervical system is the mechanistic justification for blocking the greater occipital nerve (GON-block) as a treatment option for headache patients. This has long been utilised in clinical practice for primary headaches such as cluster headache (12), but there is conflicting evidence for its efficacy in migraine (13). Open trials have reported some reduction in frequency and intensity of attacks (14–17). However, it does not seem to exert a convincing preventative effect on migraine frequency (18) and it has been proposed that the GON-block might be more effective in patients suffering from migraine with occipital pain focus (19). It is therefore possible that only subgroups of migraine patients have the potential to benefit from a GON-block. How these subgroups can be identified is currently unclear.
Subgrouping patients by physical examination to identify responders to treatment has been successfully applied for other diseases such as low-back pain (20) and neck pain (21). Manual cervical spine examination distinguished three subgroups of migraine patients (22): Those with no pain response, those with local pain, and those with pain referred to the head.
The aim of this study was therefore to investigate whether migraine patients with a higher sensitivity of the upper cervical spine will experience a greater relief of their migraine symptoms after a treatment that reduces the input from the cervical system. A secondary aim was to identify additional predictors for the reduction of the headache frequency after a GON-block.
Methods
This observational study was registered a priori at the German Clinical Trials Register (DRKS00015995). The study has been approved by the ethics review board of the Hamburg chamber of physicians (Ärztekammer Hamburg) (PV5757).
Study participants
Migraine patients were recruited consecutively from the outpatient headache clinic at the University Medical Center Hamburg-Eppendorf between January 2019 and January 2020. Adult patients with chronic migraine (CM) according to the International Classification of Headache Disorders (ICHD-3) (4) or high frequent episodic migraine (HFEM) (23), diagnosed by a headache specialist, with a disease duration of a minimum of 6 months, were eligible for study participation. All patients who were scheduled by a neurologist to receive a GON-block to reduce their migraine symptoms were asked to participate. Exclusion criteria were neurological or psychiatric diseases influencing pain perception, diagnosed trauma to the head or the cervical spine, other diagnosed acute and chronic pain conditions, allergy to lidocaine or fortecortin, misuse of alcohol or drugs, and inability to speak and understand German. As occipital neuralgia (ON) was shown to coexist with chronic migraine and could influence the results (24), no patients fulfilling the criteria of the ICHD-3 for ON were included (4). All patients were informed verbally and via a patient information leaflet about the intervention and the study objective and signed an informed consent sheet in accordance with the Declaration of Helsinki.
Manual joint testing/palpation
Migraine patients were stratified into three subgroups according to manual palpation of the upper cervical spine as described by Luedtke and May (22). To perform the manual joint palpation, the patient was positioned in prone position with the hands resting under the head. Manual pressure (ventral glide) was applied over the dorsal area of the joints C0/1 (lateral posterior arc of C1) and C1/2 (articular pillar of C2) bilaterally (Figure 1). This procedure was described by Maitland (25) to test joint dysfunction with a dynamic technique and by Watson and Drummond for the examination of headache patients (26) using a sustained pressure technique. Outcomes were pain during palpation and referred pain to the head during sustained pressure for 5 sec. According to the pain response, patients were stratified into the three groups: “No pain”, “local pain” and “referred pain to the head”. The examination was performed by a trained physical therapist with 15 years of clinical experience (AS). Since none of the patients suffered from any symptoms of ON, no occipital Tinel’s sign test was performed.
Figure1. Palpation of the upper cervical spine.

Quantitative sensory testing
Hyperalgesia and allodynia in the neck (laterally of the spinal process of C2) and in the receptive field of the first branch of the trigeminal nerve (V1) (above the eyebrow) were tested prior to the manual examination using elements of the quantitative sensory testing (QST) protocol to quantify the sensitivity and address potential differences in the groups due to the current migraine phase. The elements of the QST protocol included: Mechanical detection thresholds, mechanical pain thresholds, pressure pain thresholds, and a test for brush allodynia (27). For the mechanical detection threshold (MDT), standardised von Frey hairs were used (Optihair2-Set; Marstock Nervtest, Schriesheim, Germany, 0.25–512 mN). For the mechanical pain threshold (MPT), standardised weighted PinPrick stimulators were used (MRI Compatible Pinprick Stimulator Set; MRC Systems GmbH, Heidelberg, Germany, 8–512 mN). To test for brush allodynia, a soft brush from the Pin-Prick test set was applied that can exert a force of 200–400 mN. Pressure pain thresholds (PPT) were tested using a mechanical pressure algometer with a 1 cm2 probe measuring 1–10 kg/cm2 (Wagner Instruments, Greenwich, UK).
MDT and MPT were measured with the modified method of limits according to the test manual for QST, provided by the German Research Network on Neuropathic Pain (28). After five series of threshold determination with ascending and descending stimulus intensities, the final threshold was determined as the geometric mean of all repetitions. To determine the PPT, the algometer was placed on the skin and the pressure was increased by 0.5 kg/sec until the first pressure-induced pain was perceived. The threshold was the mean of three repetitions. Brush allodynia was tested with a light tactile stimulus on the skin and rated as painful or non-painful. In addition to testing the receptive field of V1 and C2, PPTs were measured over the masseter muscle and over the GON. Both were located by palpation. For the identification of the GON, palpation along a line between the landmarks of the external occipital protuberance and the mastoid process was performed (29,30). MDT was not feasible in the receptive field of C2 because of the patient’s hair. All measurements were conducted bilaterally.
Intervention
For the GON-block, the GON was located manually by palpation according to validated procedures (29,30). All patients were treated with bilateral injections (4 mg fortecortin and 4 ml lidocaine 1% per side) approximately 2 cm lateral to the occipital protuberance (31) on each side. The physicians performing the GON-blocks (SD and CZ) were equally experienced headache specialists trained in the treatment of migraine patients and blinded towards the group allocation (no pain, local pain, referred pain) and treated the same proportion of patients in all groups (p = 0.879).
Primary outcome
The effectiveness of the GON-block was measured as the percentage change in headache frequency comparing the 4 weeks prior to the 4 weeks after the intervention. Especially for chronic migraine, percentage change is a more meaningful marker than absolute numbers, since patients start with a high baseline frequency (32). A headache day was any day marked in the patients’ headache diary, independent of duration and quality of the headache. Patients kept a headache diary for 4 weeks prior to and for 12 weeks after the intervention.
Questionnaires and secondary outcomes
Questionnaires were used to document personal and migraine-associated patient characteristics such as age, gender, usual headache side, maximum and minimum headache intensity on a numerical rating scale from zero (no pain) to ten (maximum imaginable pain), last day of headache, associated symptoms chosen from a list of typically reported items and neck pain during/before/after attacks. The patients also answered if they had neck pain or headache on the day of examination. Additional headache-specific standardised questionnaires were used at baseline to evaluate the presence of allodynia (12-item Allodynia Symptom Checklist, ASC-12) (33), the level of migraine associated disability over 4 weeks (Migraine Disability Assessment, MIDAS) (34,35), disability due to neck pain (Neck Disability Index, NDI) (36,37), the degree of depression (Personal Health Questionnaire, PHQ-9) (38,39) and self-perceived change (Global Rating of Change, GROC) (40). In addition to the headache frequency, secondary outcomes of this study were the MIDAS and GROC to account for disability and self-perceived improvement at 4 weeks’ follow-up.
Study procedure and follow-up
All consecutive patients fulfilling the eligibility criteria were asked to participate in the study (Figure 2). Immediately prior to the GON-block, patients were examined and stratified into the three groups. After the GON-block, they filled out the questionnaires. Thirty minutes after the GON-block, the immediate effect and area of numbness was tested with the largest von Frey hair (512 mN). After 24 hours, patients were contacted by phone and asked about the duration of skin numbness, adverse events and the development of a migraine attack. Since migraine has been reported to improve mainly during the first month following a GON-block (41), the baseline assessment including manual joint testing and QST elements was repeated 4 weeks following the GON-block to identify changes in hypersensitivity or allodynia and in responses to manual joint testing.

Flow diagram of the study procedure.
Statistical analysis
Statistical analysis was performed with the IBM Statistical Package for Social Science (SPSS Version 25, Armonk, NY, USA), using a significance level of 5%. Demographic data is presented as mean values and standard deviations. The Kolmogorov-Smirnov test was used to test for normal distribution. QST data were transformed logarithmically before statistical analysis (42). Homoscedasticity was tested with Levene’s test. Differences between groups were tested with ANOVA for normally distributed data, Kruskal-Wallis-test for non-normally distributed data and χ2 test for categorical data. Bonferroni-correction was used to correct for multiple testing. Since the size of the groups differed, Fisher’s exact test was used for Kruskal-Wallis and χ2 test. Differences between baseline and follow-up within groups were tested with dependent t-test. Pre-post difference in headache days for the whole sample was tested with Wilcoxon test. Correlations between the outcomes were calculated using the Pearson’s correlation test. A linear backward regression was conducted with the response variable “percentage change in headache days” and the explorative variables “PPT over GON and C2”, “headache on the day of the GON-block”, “percentage of migraine attacks beginning in the neck”, “headache frequency at baseline” and baseline values of the questionnaires NDI, PHQ-9, ASC-12. The interaction of the significant variables from the backward regression was included in a linear regressions model. Simple imputation was used for three missing values of the variable “percentage of migraine attacks beginning in the neck”. A power analysis was performed a priori using G*power (43). Based on data from a GON-block study with a low risk of bias (16), a sample size of 20 patients per group was required to detect a statistically significant difference between groups using ANOVA (80% power, alpha = 0.05).
Results
Characteristics of the participants
Between January 2019 and January 2020, 87 eligible patients were recruited, of which six were excluded due to the presence of other disease such as systemic lupus erythematosus, psychiatric disease, trauma to the cervical spine or post-traumatic headache. Nine patients refused to participate due to fear of injections or lack of time. Seventy-one patients with CM (n = 56) and HFEM (n = 15) met the inclusion criteria and follow-up data was analysed (Figure 2). In total, 89% of the participants were female, 65% of the patients reported headache during the examination and intervention, 17% of the patients were examined interictally. At baseline, the three groups (no pain n = 11, local pain n = 28 and referred pain n = 32) did not significantly differ regarding demographic data, baseline questionnaires, neck pain or headache characteristics (p > 0.05, Table 1). The groups did not differ significantly regarding medication overuse. No significant differences were found for the QST parameters MDT and MPT (p > 0.05, Table 2). However, the three groups differed significantly regarding the PPTs at all test sites (p < 0.01), except for the masseter muscle (p = 0.085). The “no pain” group presented significantly higher PPTs than both the “local pain” group and the group with “referred pain to the head” (Table 2). None of the participants showed brush allodynia. For detailed information on the exact QST values, see Table 2. The groups did not differ significantly in the duration of numbness (p = 0.061, Table 3). Interestingly, there was a small negative correlation between the duration of numbness and the primary outcome “percentage change in headache days” (Pearson r = −0.239).
Mean values and standard deviations (SD) of sociodemographic data, questionnaires and headache characteristics at baseline for the three groups “no pain”, “local pain” and “pain referred to the head”.

Note: ANOVA was used for normally distributed data, Kruskal-Wallis test (H = Kruskal H) for non-normal distributed data and χ2 test for categorical variables (χ2 value). Fisher’s exact test was used because of the different group sizes.
MOH: medication overuse headache; CM: chronic migraine (the remaining percentage is high frequency episodic migraine); BMI: Body Mass Index; NDI: Neck Disability Index; PHQ-9: Personal Health Questionnaire; ASC-12: Allodynia Symptom Checklist; MIDAS: migraine disability assessment.
Mean values, standard deviations (SD) and post hoc for the results of physical examination and QST at baseline for the three groups “no pain”, “local pain” and “pain referred to the head”.

Note: ANOVA was used for normally distributed data, Kruskal-Wallis test for non-normal (H = Kruskal H) distributed data and χ2 test for categorical variables (χ2 value). Fisher’s exact test was used because of the different group sizes. Bonferroni corrected post-hoc p-value between the groups.
*No pain versus local pain.
**No pain versus referred pain.
***Local pain versus referred pain.
QST: quantitative sensory testing; MDT: mechanical detection threshold; MPT: mechanical pain threshold; PPT: pressure pain threshold (data for MDT, MPT, and PPT are shown after logarithmic transformation); V1: receptive field of the first trigeminal branch; GON: greater occipital nerve. Pain intensity measured on a numeric rating scale 0–100 in response to manual palpation. Significant p-values are marked in bold.
Mean values, standard deviations (SD) and post hoc of the headache characteristics at 4-week follow-up for the three groups “no pain”, “local pain” and “pain referred to the head”.
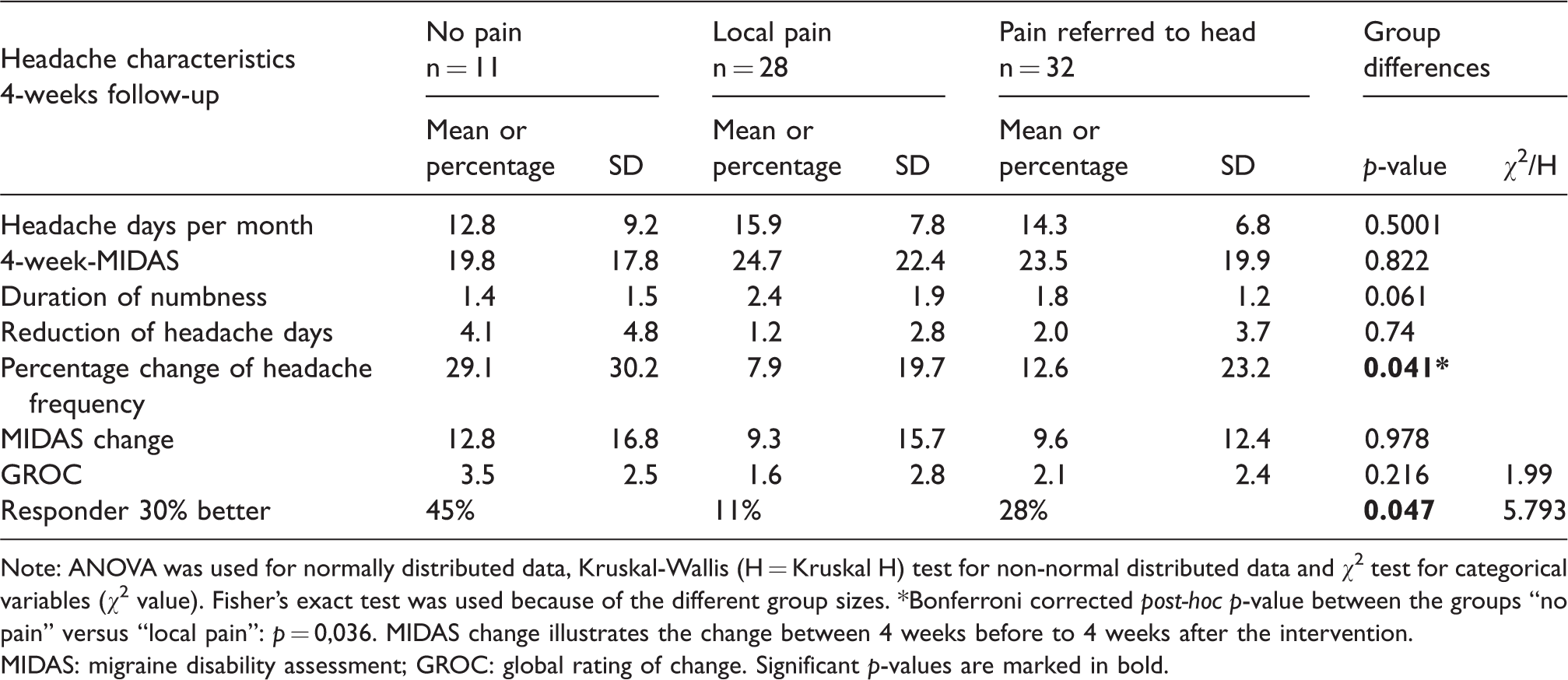
Note: ANOVA was used for normally distributed data, Kruskal-Wallis (H = Kruskal H) test for non-normal distributed data and χ2 test for categorical variables (χ2 value). Fisher’s exact test was used because of the different group sizes. *Bonferroni corrected post-hoc p-value between the groups “no pain” versus “local pain”: p = 0,036. MIDAS change illustrates the change between 4 weeks before to 4 weeks after the intervention.
MIDAS: migraine disability assessment; GROC: global rating of change. Significant p-values are marked in bold.
Differences between groups for percentage change of headache frequency
A significant between-group difference was found on the percentage change of headache frequency (p = 0.041) with a mean change of 29.1% (SD 30.2%) in the group with no pain, 7.9% (SD 19.7%) in the group with local pain and 12.6% (SD 23.2%) in the group with referred pain. Post hoc comparison showed a significant difference between the group with no pain compared to the group with local pain (Figure 3).

Between-group differences for percentage change of headache frequency for the three groups “no pain”, “local pain” and “pain referred to the head”. The p-value represents the Bonferroni-corrected difference between the groups no pain and local pain. Error bars show the standard error of the mean.
Change in migraine frequency and effect of the GON-block
Overall, migraine patients in all three groups showed a significant reduction in headache days after GON-block (p < 0.000034). This corresponds with a percentage change of 13.3% (SD 23.8). Twenty-four percent of the patients were responders to the GON-block, defined as a 30% reduction in headache frequency. The reduction of headache days was also significant within the groups. With a change of 4.1 days (SD 4.8, p = 0.018) in the group with no pain, 1.2 days (SD 2.6, p = 0.024) in the group with local pain and 2.0 days (SD 3.7, p = 0.005) in the group with pain referred to the head (Table 3).
Secondary outcomes
Overall, migraine patients showed a reduction in migraine disability after GON-block with a mean of 10 points reduction in 4-week MIDAS from baseline to follow-up (SD 14.3, p < 0.0001). This significant reduction is consistent within each of the three groups. The MIDAS score had a mean change of 12.8 (SD 16.8, p = 0.03) in the group with no pain, 9.1 (SD 15.4, p = 0.004) in the group with local pain and 9.7 (SD 12.6, p = 0.0001) in the group with referred pain to the head. This change in MIDAS was not significantly different between groups (p = 0.978).
The groups did not differ significantly for the GROC ratings (p = 0.216, H = 1.9). When considering two points on the GROC scale as a minimum clinically important change (MIC), 39 patients reported no change, three reported a deterioration and 29 reported an improvement (40). All patients reporting a deterioration were in the local pain group. The two secondary outcomes GROC and MIDAS correlated with the percentage change of headache frequency: GROC r = 0.435, p = 0.00015; 4-week MIDAS r = 0.369, p = 0.002. Detailed information on the headache characteristics at 4 weeks follow-up can be found in Table 3.
Exploratory analysis of predictors for percentage change in headache frequency
The following variables were entered into a linear backward regression model: PPT over GON and C2, having acute headache on the day of the GON-block, percentage of migraine attacks beginning in the neck, headache frequency at baseline and baseline values of NDI, PHQ-9, and ASC-12. Of these, headache on the day of the GON-block (p = 0.01) and the PPT on C2 (p = 0.002) showed a significant influence on the outcome percentage change of headache frequency. Both variables had a comparable impact on the regression model, influencing percentage change of headache frequency with a standardised beta of 0.35 (PPT C2) and −0.29 (headache on the day of GON-block) (R2 = 0.204, F = 8.72). All other predictors had no significant influence on the model. Taking the interaction of these two significant variables (headache on the day of the GON-block and the PPT on C2) into account in a linear regression model showed that patients who were pain free on the day of the examination had a greater benefit from the GON-block, the higher the PPT over C2 (Figure 4). This regression model explains 27% of the variability and is significant (p = 0.00078) with a standardised beta of −0.27 for the interaction (R2 = 0.274, adjusted R2 = 0.242, F = 8.44). Patients with headache on the day of the GON-block did not differ regarding the PPT over C2 from those without headache (p = 0.96). All three groups had the same percentage of patients reporting headaches on the day of the GON-block (p = 0.944). The post hoc calculated power of the regression with an alpha error of 0.05 was 0.995 with an effect size of 0.406.

Grouped scatterplot for the influence of headache on the day of the GON-block and the pressure pain threshold on C2 (kg/cm2) on the percentage change of headache frequency. This figure illustrates the interaction of PPT on C2 and having a headache on the day of the GON-block according to a linear regression model. The combined model explains 27% of the variability: R2 = 0.27, adjusted R2 = 0.24, F = 8.4, p = 0.000078. The combined model explains 27% of the variability: R2 = 0.27, adjusted R2 = 0.24, F = 8.4, p = 0.000078.
Adverse events
Eight participants reported adverse events. Six persons reported redness of the face on the following day, two reported local tenderness of the injections site and two reported dizziness.
Discussion
Our findings confirm the effect of a GON-block on migraine frequency. The main effect (i.e. for all three groups) was a significant reduction not only for the headache frequency but also for migraine disability and thus replicated the results of previous studies (15–17,44,45). However, although this was mathematically significant, it is questionable whether a 2-day reduction of headache days can be regarded as clinically important when taking into account that a GON-block is an invasive intervention. Invasive procedures may have more adverse events and a heightened placebo effect due to high expectations to injections, as known from studies on invasive prophylaxis (46) compared to less invasive treatments. Similar effect sizes were reported for less invasive interventions such as aerobic exercise (47,48) and relaxation techniques (49). Based on statistical results and on self-report, the GON-block remains of arguable benefit for the preventive treatment of migraine.
Our main question was whether subgroups of migraine patients would benefit more from a GON-block and to investigate whether a physiotherapeutic examination procedure can determine such subgroups of responders. Against our own hypothesis, we found no differences between the two groups who responded with pain during manual palpation of the upper cervical spine, whereas the “no pain” group responded with a larger reduction in headache frequency. These findings seem to be in contrast to a previous study, where tenderness around the GON region was observed as predictive for a positive response to the GON-block (14). The researchers assessed different types of chronic headaches with 54 out of 101 patients diagnosed as having migraine. It was not reported how tenderness was assessed and there was no differentiation between headache types. Furthermore, it is not mentioned whether the participants had headache on the day of the appointment. Therefore, a direct comparison of results is not justified. On the other hand, and in spite of a long recruitment phase, the group with no pain on cervical spine palpation remained small in this current study. The estimated sample size was reached with an unregular distribution across the groups, which resulted in a reduced power for the ANOVA calculation. The most likely reason is that patients who never complained of neck problems related to their migraine had no intuitive concept of a causative association between the neck and the headache, and consequently were less likely to accept a bilateral GON-block. Additionally, those who knew or suspected an influence of the cervical or occipital system due to their symptoms might have had a higher interest in taking part in the study. Another explanation would be that patients with chronic migraine are in general more sensitised (50), and since this study examined more affected patients with CM and HFEM, the “no pain” group remained the smallest in this population of migraineurs.
One could argue that the mere fact that our group of (neck-) pain free migraine patients was too small skews the data and explains the result. However, homogeneity within the group is given. The group is consistent, has no outliers, and does not differ from the other groups regarding variance. Furthermore, the exploratory analyses involving all data regardless of the group affiliation, confirmed the main findings: The less tender the neck of the patient was, and the less headache they showed, the better the benefit from the GON-block. The regression had a high statistical power and included 71 participants. The results of the regression suggested an interaction of three factors: PPT over C2 in interaction with headache on the day of the GON-block and the influence of these two variables on headache frequency. This interaction showed that especially those who had no pain on the day of the GON-block had a better benefit from the GON-block, the higher the PPT value over C2 was. Interestingly, when considering the overall sample, patients with headache on the day of the intervention did not differ from patients without headache on that day regarding PPTs, although across the three stratified groups PPTs were significantly different. Not only the PPT over C2 but also the PPT results over the GON and V1 indicated that patients in the “no pain” group were significantly less sensitive than both the local and the referred pain groups. There were no significant group differences for MPT, which is in line with the results of a recent meta-analysis, which did not show differences for MPT between migraine patients and healthy controls (51). In fact, the interaction of PPT over C2 and headache during the intervention was the combination of predictors best explaining the results.
Peripheral and central sensitisation are states of the nervous system that are frequently discussed in relation to chronic pain conditions including migraine (52–54) and altered sensory responses of migraine patients in the cervical musculoskeletal region have been reported (6,55,56). In the previous stratification study (22), a correlation was found between headache frequency and pain provocation on palpation indicating that patients with a pain response were more affected. Since migraine is a sensory threshold disease with varying thresholds throughout the different migraine phases (50,57,58), patients can be regarded as more prone to sensory neck dysfunction the higher the headache frequency and the closer they are to an attack. Indeed, patients in the local pain or referred pain groups were more tender to manual palpation, although the groups did not differ regarding headache frequency or the number of ictal or interictal patients at the baseline assessment. One could argue that the neck and innervation of the neck was somewhat more “sensitised” in those two groups compared to the “no pain” group, although only mild cutaneous hypersensitivity in the ASC-12 questionnaire (33) and no brush allodynia was found. Both are indicators for allodynia and therefore for central sensitisation (54). However, these indicators might not be sensitive to detect differences between different hyperalgesic states. Pain hypersensitivity from the trigeminal region might be extended via the TCC to the upper cervical system, expressing widespread pain and hyperalgesia as known for other chronic pain conditions (59). Our current results suggest that a GON-block may be less effective in a hypersensitive cervical system and thus may have a greater effect on migraine in a less altered system.
We note that the GON-block that we performed in this study is targeting the nerve. One could argue that the painful groups might have a stronger involvement of the C2 root. A peripheral nerve block will presumably not relieve the nociceptive input from the root, and this would explain why the GON-block is not efficient in the painful groups. Indeed, a recent double-blinded randomised controlled trial showed that the effect of a proximal GON-block is a little stronger and longer lasting than that of a distal GON-block (60).
A limitation of our study is that no placebo intervention was included. The primary aim was not to investigate the effectiveness of the GON-block but to predict a subgroup of responders. Otherwise, all participants were treated and examined equally by the same persons. Furthermore, despite all patients receiving the same intervention, they reacted differently in correlation to their physical alteration, which suggests an underlying effect independent of the placebo effect that might be caused by different pathophysiology of the patients having no pain on manual palpation. Due to the group of patients all coming from the same outpatient clinic, and the fact that patients who are suspected to have an involvement of the cervical region might be more likely to take part in the study, we note that results have to be viewed with caution in terms of external validity. The stratification was performed on patients with headache and without headache on the day of the examination. Arguably, the state of headache could influence the stratification. Since all three groups had the same proportion of patients with headache on the day of examination, we assume that combining ictal and interictal patients did not influence the results. The exploratory analysis addresses this point, and quantitative sensory testing was included to control for this effect.
Nevertheless, our data suggest that a GON-block is most effective in less affected patients, which are less sensitive to palpation of the cervical spine, and preferably do not have headaches on the day of the intervention. A recent study has shown a phenotypic difference of patients with ictal neck pain to those without ictal neck pain regarding interictal tenderness of the neck region (61). This distinction could be a useful addition to the current data, and it should be investigated whether patients with ictal neck pain benefit more from a GON-block than patients without.
Article highlights
Patients without pain on manual palpation of the upper cervical spine benefit significantly more from a GON-block than patients with local pain. Applicability of stratification into subgroups of migraine patients could be confirmed in a clinical context. Migraine patients with no pain on manual palpation and higher pressure-pain thresholds seem to benefit more from a GON-block, especially when headache-free during the intervention. Increased tenderness in the neck region is predictive for a less effective GON-block.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AS, SD, AM and KL declare no conflict of interest. CZ received speaker honoraria from Allergan, Lilly, Novartis and Teva and served on advisory boards for Lilly, Novartis and Teva.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Migraine Research Foundation (NY, USA) to AM and KL.
