Abstract
Background
The diagnostic criteria for headache attributable to cranial venous sinus stenting were first formalized in the recently published third edition of the International Classification of Headache Disorders (ICHD-3). However, the diagnostic criteria for headache caused by cranial venous sinus stenting are based on very few data and the condition is poorly characterized.
Objectives
To validate the diagnostic criteria for cranial venous sinus stenting headache by retrospectively studying the characteristics of headache in patients with isolated pulsatile tinnitus who underwent curative cranial venous sinus stenting and who had not previously complained of headache.
Patients and methods
We retrospectively studied clinical, radiological, and manometric data from patients with isolated venous pulsatile tinnitus who had not previously reported headache. All patients underwent lateral sinus stenting in our institution between October 2010 and February 2018.
Results
Forty eight patients, 47 females and one male, were enrolled. The mean age at symptom onset was 36.2 ± 8.7 years and the mean body mass index was 24.0 ± 3.2 kg/m2. Lateral sinus stenosis was evident in 47 patients and a sigmoid diverticulum in one. Fourteen patients experienced headaches after recovering from general anesthesia. All were female, with a mean age of 35.5 ± 9.6 years. Headache persisted for less than 3 days in six patients (42.8%); for 3 days to 3 months in four (28.6%); and for longer than 3 months in four (28.6%). The headaches were located on the same sides as the cranial venous sinus stents in 13 patients (92.9%) and were principally occipital, being oppressive in nine patients (64.3%) and of moderate intensity in seven (50%). Age at onset of pulsatile tinnitus and body mass index were significantly associated with headache (p < 0.05; t-test).
Conclusion
To the best of our knowledge, this is the first study to describe cranial venous sinus stenting headache in detail. We found that de novo headache developed after cranial venous sinus stenting, and was usually mild to moderate, unilateral, but oppressive; almost one-third of such headaches persisted for more than 3 months. Researchers and clinicians must become familiar with this headache spectrum; further prospective studies are required.
Keywords
Introduction
Idiopathic intracranial hypertension (IIH), giant arachnoid granulations, and venous sinus wall anomalies can trigger venous sinus stenosis (VSS), which in turn aggravates IH and/or triggers pulsatile tinnitus (PT) (1,2). The latter term was first used in 1977. Holgate et al. referred specifically to self-perception of a vascular somatosound (3) caused by murmurs in vascular flow. This tinnitus is objective and can severely compromise quality of life and social functioning. To date, cerebral venous anomalies are the most commonly identified abnormalities during PT work-up (4,5); it has been speculated that elevated flow velocity/volume rates adjacent to the anomalies may be causative of PT. Commencing in 2009, Li et al. found that some patients with PT caused by cranial venous sinus wall anomalies benefitted from stenting; the PT immediately disappeared (4). Over the past years, cranial venous sinus stenting (CVSS) has been extremely successful. However, some patients complain of debilitating headache after the procedure; this was formalized in the third edition of the International Classification of Headache Disorders (ICHD-3) (6). However, the diagnostic criteria for such headaches are based on few data from small case series; all patients had IIH, headache was the principal preoperative symptom, and we suggest that this affected headache assessment after stenting. Here, we retrospectively enrolled consecutive patients without IIH (thus without primary headache) but with PT caused by venous sinus wall anomalies. All consented to neuroendovascular treatment. We here evaluate the ICHD-3 diagnostic criteria for CVSS headache and we describe the headaches of our patients. This study was approved by the research ethics committee of the Chinese PLA General Hospital and all patients gave written informed consent.
Patients and methods
Patients: From October 2010 to February 2018, we screened 99 patients with isolated PT. Those who refused operations and patients with any type of primary headache, intracranial artery stenosis, intracranial aneurysm, cerebral arteriovenous malformation, dural arteriovenous fistula, carotid artery dissection, or clinical signs of IH (headache, vomiting, papilledema, dizziness, or visual loss), were excluded. Finally, 47 patients with lateral sinus stenoses and one with sigmoid diverticula were included. All complained of “whooshing” PT synchronized to the heartbeat and could identify the locations thereof. Loudness was reduced by head rotation (ipsilateral internal jugular vein compression by the ipsilateral sternocleidomastoid) or ipsilateral neck compression. Pure tone audiometry was used to approximate tinnitus pitch and loudness via sound frequency and level matching. The PT matched to frequencies between 125 and 1,000 Hz with loudness values of 30 to 89 decibel hearing level (dBHL).
Methods: We retrospectively studied clinical, radiological, and manometric data. The clinical files included ages at PT onset and stenting, gender, body mass index (BMI), operation time, headache timing, duration, and clinical characteristics (side [bilateral or unilateral], location [frontal, occipital, generalized, or half-sided], quality [pressing, throbbing, or stabbing], severity [mild, moderate, or severe]); and accompanying symptoms. During follow-up, all patients repeatedly reported the features of headache. We noted any PT recurrence and any drugs used.
All patients underwent brain magnetic resonance imaging (MRI) using a Siemens 1.5-T Avanto or a 3.00 T TIM Trio system (Erlangen, Germany) fitted with 12-channel head coils. The MRI protocol included a T2-weighted fluid-attenuated inversion-recovery sequence (axial sections), a T2* sequence (axial sections), a T2 sequence (coronal sections), a large three-dimensional time-of-flight (TOF), and intracranial arterial magnetic resonance angiography (MRA). The cerebral venous system was examined using magnetic resonance venography (MRV) (Figure 1) and digital subtraction angiography (DSA) (Figure 1). The type of stenosis, the affected side (unilateral or bilateral), and the location within the dominant sinus were recorded. We sought radiological signs of IIH, including an empty sella turcica, widening of optic nerve sheaths, and/or optic nerve tortuosity. If any of these features was evident, the patient was excluded. Prior to endovascular treatment, all patients underwent three-vessel catheter angiography via 4-F, arterial femoral access with injection into both internal carotid arteries and one vertebral artery; we used long runs to visualize the cerebral veins and intracranial venous sinuses. CVSS was performed under general anesthesia. During the procedure, all patients received intravenous heparin to maintain the activated clotting time at more than 150 s. All patients initially underwent conventional four-vessel cerebral angiography, and stenting was performed in a separate session using the sheath or guiding catheter as a road map or employing the overlay technique. Venous pressure measurements were acquired via jugular vein access using the introducer subsequently employed to perform stenting. Lateral sinus stenosis was defined as any focal area exhibiting a prominent change in vessel caliber (at least a 50% decrease in diameter). The dominant sinus was of diameter greater than 150% that of the non-dominant sinus. Pressure was measured using an invasive transducer connected to a microcatheter of 0.018 in internal diameter. All results are expressed in mmHg. Pressures were measured on both sides of each stenosis (or diverticulum), thus in the proximal part of the sigmoid sinus and the middle third of the superior longitudinal sinus. The trans-stenotic gradient was the difference in pressure between these two points. The stent diameter was that of the transverse sinus. The pressure was measured at the end of each procedure, using the same method, and an angiographic control was routinely performed to verify venous system permeability (Figure 1).
(a) and (b), Isolated PT of the right ear caused by an extrinsic stenosis of the right lateral sinus. The brain MRV and angiography reveal extrinsic stenoses of the right lateral sinuses (arrows); (c) a transtenotic gradient of 2 mm Hg; (d) stenting of the lateral sinus (arrow) allowed PT to disappear.
Antiplatelet treatment (aspirin, 100 mg/day) and anticoagulation therapy (warfarin, 6–9 mg/day) were prescribed before stenting; the plasma thrombinogen time was maintained at 20–25 s. Antiplatelet treatment continued for 3 months and warfarin for 6 months. All patients were followed up at 6 and 12 months after stenting. PT disappeared in all patients immediately after stenting and did not recur during follow-up. All patients were re-contacted by telephone in July 2018, all but one (see below) were well.
Statistical analyses
Data were analyzed with the aid of SPSS software v. 25.0 (IBM Corporation, Armonk, NY, USA). We evaluated age, sex, age at PT onset, sinus stenosis location, PT side, operative duration, and headache status and persistence if present. Continuous variables are expressed as means ± SDs, frequencies, or percentages. The t-test and Wilcoxon test were used to compare quantitative data, and the chi-squared test to compare qualitative data. We calculated two-tailed p-values and set the significance level at p < 0.05.
Results: The mean age at stent placement was 41.5 ± 9.7 years. The mean age at symptom onset was 36.2 ± 8.7 years. Females predominated (47, 97.9%, vs. one male, 2.1%). The mean BMI was 24.0 ± 3.2 kg/m2. The mean duration of symptoms prior to stenting was 5.1 years. PT was right-sided in 28 cases, left-sided in 16, and bilateral in four. Post-CVSS headache was observed in 14 patients; such patients were younger than those without headache (35.5 ± 9.6 vs. 43.9 ± 8.1 years of age, p = 0.005). Headache was ipsilateral, pressing in nature, mostly occipital (71.5%), and located on the side of stenting. Headache generally commenced within 1–2 h after recovery from general anesthesia, persisted for less than 3 days in six patients, between 3 days and 3 months in four, and for more than 3 months in four; in one patient with PT in the sigmoid diverticulum, the headache has not yet disappeared (1 year after stenting), and is aggravated by coughing and sneezing.
Demographics and clinical features in patients with or without headache after cranial venous sinus stenting.
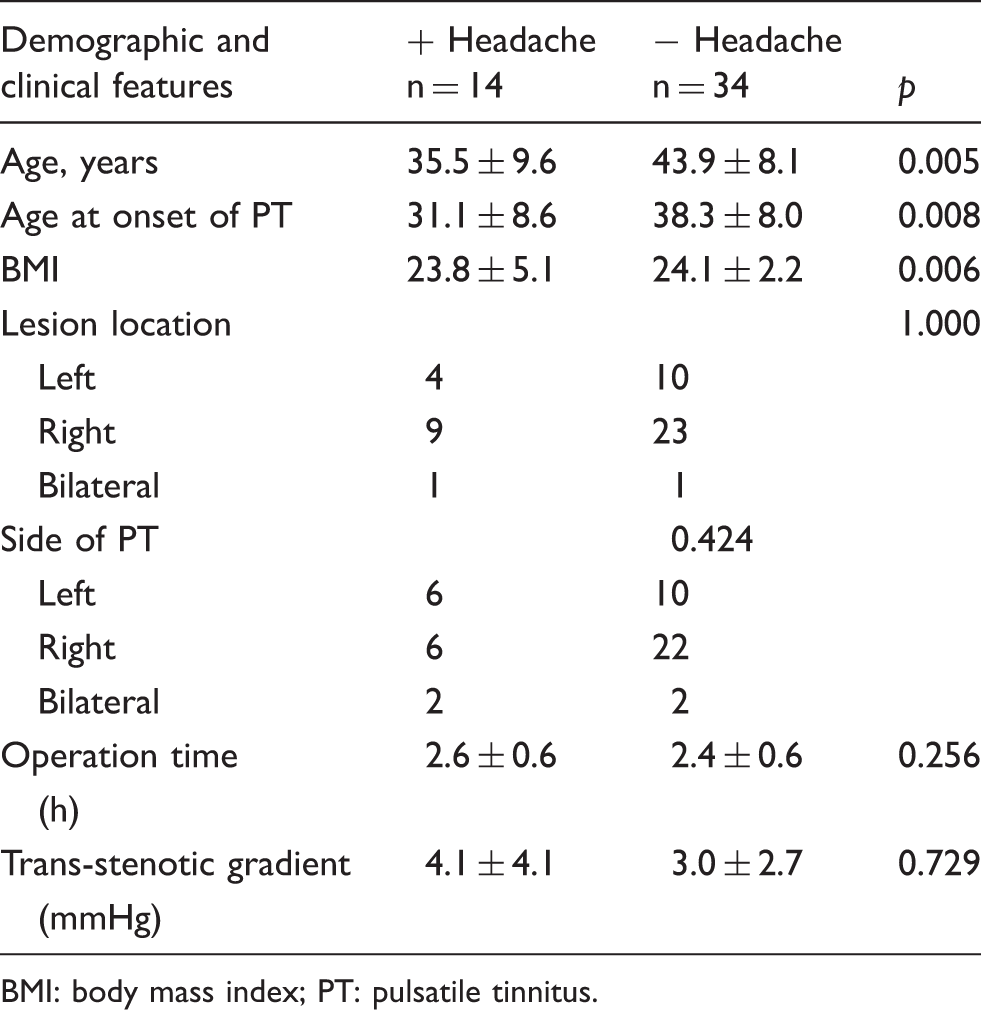
BMI: body mass index; PT: pulsatile tinnitus.
Characteristics of headache after cranial venous sinus stenting.

Headache after cranial venous sinus stenting (n = 14): According to the criteria of ICHD-3.

Discussion
Literature on headache (new or worse) after cranial venous sinus stenting.

NO: not mentioned in the study; new: new headache; worse: residual headache increased; IIH: idiopathic intracranial hypertension.
Modified diagnostic criteria for headache attributed to cranial venous sinus stenting.

The mechanism of CVSS headache remains unknown. Suggestions have included: a) mechanical stimulation of the venous sinus wall (stretching; distortion by balloons, catheters, or the stent; or local hemodynamic changes that trigger the trigeminovascular pathway via trigeminal C fiber stimulation); b) local toxicity, a chemical reaction to the dye, or inflammatory changes; c) a lower pain threshold; d) variation in the locations of the hypersensitive pressure receptors of venous vessel walls (20); and/or e) physical and psychological stress (pain or fear). Other studies reported a (statistically significant) increased risk of headache after intracranial neuroendovascular procedures in patients with psychiatric comorbidities (anxiety/depression). Anxiety sensitized patients to pain, anxiety and depression predicted postoperative pain, and depression and pain were correlated (21). These mechanisms resemble those causing headache after intracranial endovascular procedures (22). During follow-up, we detected stent migration in the single patient who had experienced postoperative headache for more than 1 year.
Do factors other than the above influence headache? Does primary headache play a role (23)? Are other factors in play, such as persistent mechanical stimulation of the venous sinus wall caused by stent stretching? If the stenotic angle is small, restenosis after stenting may increase intracranial pressure. Further research is necessary.
Limitations
Our study is limited by its retrospective nature and the small number of subjects. Also, most patients refused lumbar puncture; we have venous sinus pressure data only. Patients did not undergo psychological testing. Retrospective studies may underestimate headache incidence because of recall bias. Despite the power limitation, our data seem robust, but future prospective studies are needed.
Conclusion
CVSS headache criteria were first proposed in the International Classification of Headache Disorders (3rd edition), based on data from small numbers of IIH patients. Our data on patients with isolated PT caused by venous sinus wall anomalies clearly show that headaches are more frequent, and their locations and durations differ from those of the current diagnostic criteria, which thus require revision. We found that CVSS headache commonly arose soon after the procedure and resolved within 6 months. Additionally, headache was not always stent-ipsilateral. Most headache was mild to moderate, occipital in location, and pressing in quality. Larger studies are required.
Article highlights
With the remarkable developments in interventional therapy, CVSS headache has attracted increasing attention; we are the first to focus on the problem.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the study, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Early warning study of cognitive dysfunction in cerebral vascular disease by magnetic resonance (15BJZ49) and the Chinese people’s liberation army (PLA) college comprehensive program (The Diagnosis and Treatment Training Base of Acute Ischemic Stroke).
