Abstract
Objective
To develop a claims-based algorithm to identify undiagnosed chronic migraine among patients enrolled in a healthcare system.
Methods
An observational study using claims and patient survey data was conducted in a large medical group. Eligible patients had an International Classification of Diseases, Ninth/Tenth Revision (ICD-9/10) migraine diagnosis, without a chronic migraine diagnosis, in the 12 months before screening and did not have a migraine-related onabotulinumtoxinA claim in the 12 months before enrollment. Trained clinicians administered a semi-structured diagnostic interview, which served as the gold standard to diagnose chronic migraine, to enrolled patients. Potential claims-based predictors of chronic migraine that differentiated semi-structured diagnostic interview-positive (chronic migraine) and semi-structured diagnostic interview-negative (non-chronic migraine) patients were identified in bivariate analyses for inclusion in a logistic regression model.
Results
The final sample included 108 patients (chronic migraine = 64; non-chronic migraine = 44). Four significant predictors for chronic migraine were identified using claims in the 12 months before enrollment: ≥15 versus <15 claims for acute treatment of migraine, including opioids (odds ratio = 5.87 [95% confidence interval: 1.34–25.63]); ≥24 versus <24 healthcare visits (odds ratio = 2.80 [confidence interval: 1.08–7.25]); female versus male sex (odds ratio = 9.17 [confidence interval: 1.26–66.50); claims for ≥2 versus 0 unique migraine preventive classes (odds ratio = 4.39 [confidence interval: 1.19–16.22]). Model sensitivity was 78.1%; specificity was 72.7%.
Conclusions
The claims-based algorithm identified undiagnosed chronic migraine with sufficient sensitivity and specificity to have potential utility as a chronic migraine case-finding tool using health claims data. Research to further validate the algorithm is recommended.
Introduction
Chronic migraine (CM), defined as ≥15 headache days per month with ≥8 migraine days per month (1), is underdiagnosed and undertreated worldwide (2–4). In the American Migraine Prevalence and Prevention Study, only 20.2% of individuals with CM received a diagnosis of CM, transformed migraine, or chronic daily headache (4). In the more recent Chronic Migraine Epidemiology and Outcomes (CaMEO) Study, only 24.6% of individuals with CM consulting a healthcare professional received an accurate CM diagnosis (2). The global prevalence of CM is estimated to be 1.4% to 2.2% (5) and is comparable to other common neurologic disorders such as epilepsy (6). Compared with episodic migraine (EM; generally characterized by < 15 headache days per month (1)), individuals with CM also have higher healthcare resource utilization, lower productivity, lower health-related quality of life, greater limitations of daily activities, higher rates of comorbidities, and substantial unmet treatment needs (7–9). It is thus important for individuals with CM to be diagnosed and treated optimally with guideline-approved acute and preventive treatments to minimize the burden of the disease at both the individual and societal levels.
Once CM is accurately diagnosed, disease management should be aimed at reducing the frequency of migraine attacks and the level of migraine-related disability (10). Effective preventive treatment not only reduces headache day frequency but also improves health-related quality of life (11) and may relieve the distress associated with frequent migraine attacks (12). To avoid the overuse of acute treatments for migraine (e.g. triptans, nonsteroidal anti-inflammatory drugs, opioids) (13), CM should be managed with both behavioral and preventive treatments (10). People with CM should also be educated to use preventive treatment in combination with judicious use of acute treatments (10). OnabotulinumtoxinA is approved worldwide specifically for the prevention of CM (10); however, beta-blockers (e.g. propranolol), tricyclic antidepressants (e.g. amitriptyline), and antiepileptics (e.g. topiramate, divalproex), some of which are approved for the prevention of migraine, are also commonly used for prevention of CM (10,14), as is the recently approved erenumab (15).
Although a diagnostic interview by a headache specialist remains the gold standard for CM diagnosis, efforts to diagnose CM are often limited by the lack of availability of clinical experts and lack of access to effective diagnostic tools. Indeed, it has been demonstrated that only 43% of patients with CM (as determined by the “gold standard” diagnostic interview) have an ICD-9/10 code for CM (16), suggesting that more than 50% have not been formally diagnosed with CM. The diagnostic process can be improved by using traditional approaches to detect underdiagnosed conditions, which use screening or case-finding tools (17). The Identify Chronic Migraine (ID-CM) patient screener was developed in 2015 to identify undiagnosed CM patients (18). In a sample of 111 individuals, the ID-CM correctly classified most CM cases (sensitivity of 80.6%, specificity of 88.6%), making it a simple and accurate tool that can be used by a broad range of healthcare providers to identify CM. With the availability of a range of pharmacy and medical claims data in many healthcare settings, however, additional opportunities are now available to further enhance case finding based on administrative claims among patients enrolled with a healthcare system.
Claims-based algorithms can be used by health systems and payer organizations that are interested in efficiently identifying patients of interest at the healthcare system level (19). As a next step toward optimizing management of CM, a claims-based algorithm was developed for use in individuals with migraine to help identify potentially undiagnosed CM in healthcare settings.
Methods
Patient screening and enrollment
An observational study using medical and pharmacy claims data and patient survey data was conducted in a large regional medical group providing primary care, hospital-based, and specialist services to >600,000 members in Southern California, delivering care through a combination of an employed staff model and a contracted independent physician association model. A claims database query was run every two months from 1 October 2015 to 1 November 2016 to identify eligible patients. The date on which the query was run was the screening date for that group of patients. The date on which the patient provided informed consent and was enrolled in the study was considered the enrollment date. The study was approved by IRB Company, Inc. (Buena Park, CA) review board.
Selection criteria
Patients were eligible for inclusion in the study if they were ≥ 18 years old on the screening date; had a claim with an International Classification of Diseases, Ninth/Tenth Revision (ICD-9/10) diagnostic code for migraine (346.xx/G43.xxx), without a diagnosis code of CM (346.7x/G43.7xx), in the 12-month period from the screening date to 364 days before screening (i.e. the screening period); had continuous medical and pharmacy enrollment in the 12-month period from the enrollment date to 364 days before enrollment (i.e. the enrollment period); provided informed consent; and were willing to provide either an email address or physical mailing address for distribution of the study questionnaires. Patients with a head injury, head or neck surgery, or a history of illicit drug use during the 12-month screening period were excluded. Additionally, patients with a migraine-related onabotulinumtoxinA claim (i.e. ICD-9/10 code of 346.xx/G43.xxx and Healthcare Common Procedures Coding System code of J0585 on the same claim), without a diagnosis code on the same claim for another on-label onabotulinumtoxinA-treatable condition (e.g. cervical dystonia, spasticity), in the 12-month enrollment period were excluded. To determine eligibility for study inclusion, diagnosis codes were evaluated in the first, second, and third claim positions. No a priori sample size calculations were undertaken. However, a sample size of approximately 200 patients was targeted for enrollment to ensure an adequate sample size for statistical analyses after accounting for study dropouts as a result of applying additional selection criteria (previous claim-based diagnosis of CM or a migraine-related onabotulinumtoxinA claim).
Determination of CM status
All eligible patients were first administered the ID-CM screener, a validated case-finding tool that can be readily used by healthcare professionals to correctly identify the majority of individuals with migraine or CM (18). The ID-CM is a 12-item, patient-reported screener based on headache frequency (over the previous 30 and 90 days), headache symptoms, medication use, and effect of headache on activities and making plans (over the previous 30 days) that an individual completes independently and subsequently reviews with a clinician to arrive at the diagnosis of CM or non-CM. After administering the ID-CM to the enrolled patients, their CM status was confirmed in a convenience sample of 196 patients through physician administration of a semi-structured diagnostic interview (SSDI) over the phone. All physicians were trained by a headache expert before administering the SSDI, which served as the study gold standard for CM diagnosis. The SSDI was based on the International Classification of Headache Disorders, 3rd edition (beta version; ICHD-3b) criteria for CM (20) and modified Silberstein-Lipton criteria for CM (21); it included 31 questions on headache symptoms, headache day frequency, productivity, medication use, and other aspects of migraine based on 30-day and 90-day patient recall. Based on the results of the SSDI, the physician who conducted the interview used clinical judgment to assign a diagnosis of CM. Chronic migraine status was also assessed through computer analysis of data obtained from the SSDI by the physician. In the event of a lack of agreement between the physician-derived and computer-derived diagnosis (based on data obtained from the SSDI), an independent clinician reviewed all of the SSDI data (i.e. computer-derived and recorded physician interviews) and assigned a final diagnosis. The SSDI diagnosis was then used to classify patients as CM (i.e. SSDI-positive) or non-CM (i.e. SSDI-negative).
Analysis of potential predictors
Definitions of potential predictors of chronic migraine status.
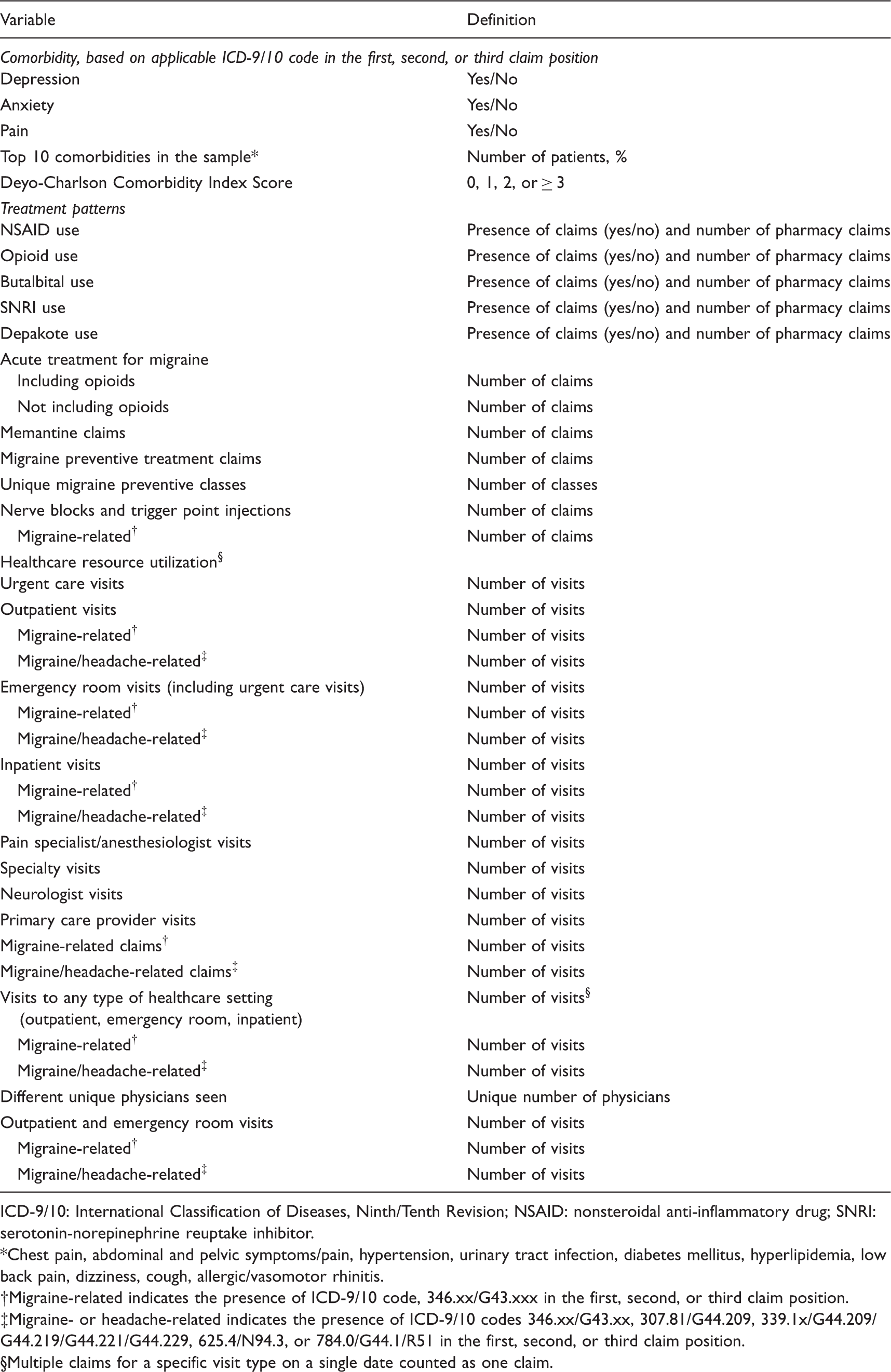
ICD-9/10: International Classification of Diseases, Ninth/Tenth Revision; NSAID: nonsteroidal anti-inflammatory drug; SNRI: serotonin-norepinephrine reuptake inhibitor.
Chest pain, abdominal and pelvic symptoms/pain, hypertension, urinary tract infection, diabetes mellitus, hyperlipidemia, low back pain, dizziness, cough, allergic/vasomotor rhinitis.
Migraine-related indicates the presence of ICD-9/10 code, 346.xx/G43.xxx in the first, second, or third claim position.
Migraine- or headache-related indicates the presence of ICD-9/10 codes 346.xx/G43.xx, 307.81/G44.209, 339.1x/G44.209/G44.219/G44.221/G44.229, 625.4/N94.3, or 784.0/G44.1/R51 in the first, second, or third claim position.
Multiple claims for a specific visit type on a single date counted as one claim.
Patient demographics and clinical characteristics included age, sex, and comorbidities (i.e. anxiety, depression, and pain, as suggested by clinical experts) and also the 10 most common comorbidities in the patient sample (i.e. chest pain, abdominal and pelvic symptoms/pain, hypertension, urinary tract infection, diabetes mellitus, hyperlipidemia, low back pain, dizziness, cough, and allergic/vasomotor rhinitis). The mean (SD) Deyo-Charlson Comorbidity Index score was determined for individuals; scores were categorized as 0, 1, 2 or ≥ 3 (22). Healthcare resource utilization included visits to neurologists, primary care physicians, pain specialists/anesthesiologists, emergency rooms (including any urgent care visits), outpatient settings, and inpatient settings. Emergency room, outpatient, and inpatient visits were further evaluated as migraine-related (i.e. ICD-9/10 code: 346.xx/G43.xxx) or migraine- or headache-related (migraine [346.xx/G43.xx], tension headache [307.81/G44.209], tension-type headache [339.1x/G44.209/G44.219/G44.221/G44.229], premenstrual tension syndromes [625.4/N94.3], or headache [784.0/G44.1/R51]) based on diagnosis codes in the first, second, or third position. Multiple claims on a single date for a specific visit type were counted as one visit. Additional potential predictors included the number of different unique physicians seen, number of healthcare visits of any type (i.e. combination of outpatient, emergency room, and inpatient visits, with multiple claims on a single date for a specific visit type counted as one visit), number of migraine-related healthcare visits of any type, and the number of migraine- or headache-related healthcare visits of any type. Acute and preventive treatments for migraine that were evaluated were selected based on medications with Level A or B evidence and clinical expert opinion (Supplemental Table 1) (13,23,24).
The total number of acute treatment claims, acute treatment claims including opioids filled at an outpatient pharmacy, preventive treatment claims, and claims for unique migraine preventive classes (i.e. antihypertensives, antidepressants, antiepileptics, and nonsteroidal anti-inflammatory drugs [NSAIDs]) were also evaluated as potential predictors. Additionally, the number of nerve blocks and trigger point injections (total and migraine-related) were assessed. Other medications that were evaluated included NSAIDs, opioids, butalbital, serotonin-norepinephrine reuptake inhibitors, and depakote; both the presence of claims (“yes/no”) and number of claims were evaluated. In the event that claims for combination medications were present, such claims were counted in each corresponding medication variable.
A multivariate logistic regression model was used to identify predictors of CM (i.e. SSDI-positive status) based on the claims data collected in the 12-month enrollment period. Only variables that significantly (p < 0.05) predicted SSDI-positive status in bivariate analyses and were considered by our clinical experts to be clinically relevant were included in the logistic regression model. To reduce multicollinearity, variables were excluded if they were redundant based on clinical expert opinion. For example, the “number of opioid claims” was excluded since it was included in “number of claims for acute treatment of migraine (including opioids).” The remaining variables were then categorized (e.g. ≥24 vs. <24 healthcare visits of any type) based on the distribution of data and clinical input. Clinical experts reviewed the data distribution models and determined the most appropriate cut-off point for each predictor value to optimize separation between EM and CM cohorts. The final model included all predictors that had a p < 0.05, after adjustment for all other predictors. Multicollinearity was assessed using the variance inflation factor (VIF).
Statistical analysis of the claims-based algorithm
Using the CM status assigned by the SSDI as the gold standard, the characteristics of the claims-based algorithm as a patient screener were evaluated, with a cutoff probability for CM set at a percentage above 0.50 and which would result in balanced sensitivity and specificity (e.g. patients with ≥55% predicted probability of CM based on the logistic regression model were classified as having CM). The key performance characteristics included the C-statistic, sensitivity, specificity, positive predictive value, and negative predictive value.
Definitions of performance characteristics.

CM: chronic migraine; FN: false-negative; FP: false-positive; TN: true-negative; TP: true-positive.
Sensitivity analyses
Sensitivity analyses were conducted in which all patients with a migraine-related onabotulinumtoxinA claim, with or without diagnosis of another on-label onabotulinumtoxinA-treatable condition (e.g. cervical dystonia, spasticity), in the 12-month enrollment period were excluded. For both the main analysis and the sensitivity analysis, further analysis was undertaken with a range of cutoff probabilities from 0.40 (i.e. patients with ≥40% predicted probability of CM based on the logistic regression model were classified as having CM) to 0.60.
Results
Patient disposition and demographics
A total of 536 patients met the inclusion criteria and completed the ID-CM screener. Among these patients, 196 completed the SSDI; 132 (67.3%) patients received a diagnosis of CM and were classified as SSDI-positive, and 64 (32.7%) patients did not receive a diagnosis of CM and were classified as SSDI-negative. After patients with a claims-based diagnosis of CM in the 12-month screening period (n = 76) and those with a migraine-related onabotulinumtoxinA claim without a diagnosis code on the same claim for another on-label onabotulinumtoxinA-treatable condition (n = 12) in the 12-month enrollment period were excluded, the final sample included 108 patients. Of this final sample, 64 patients (59.3%) were classified as CM and 44 patients (40.7%) were classified as non-CM (Figure 1, Table 3). Demographic characteristics of age and race were similar across the CM and non-CM groups, but the CM group had a higher percentage of female patients (96.9% vs. 84.1%, p = 0.018). Based on claims in the 12-month enrollment period, CM patients were also significantly more likely to have hypertension and slightly more likely to have depression, lower back pain, and general pain (Supplemental Table 2). Lastly, NSAID claims and opioid claims were significantly higher in patients who were classified as CM than in those classified as non-CM. There was no significant difference in the use of any other medications across the groups (Supplemental Table 3).
Patient selection diagram. Patient demographics and clinical characteristics of the final sample in which the claims-based algorithm was developed. CM: chronic migraine; Deyo-CCI: Deyo-Charlson Comorbidity Index; SSDI: semi-structured diagnostic interview. Based on claims data collected in the 12-month time period from enrollment date to 364 days before enrollment. Included the following comorbidities: Chest pain, abdominal and pelvic symptoms/pain, hypertension, urinary tract infection, diabetes mellitus, hyperlipidemia, low back pain, dizziness, cough, allergic/vasomotor rhinitis. Includes only treatments with Level A or B evidence for use (14,22,23) and those supported by clinical expert opinion. No p values were estimated, as there was a cell with a 0 count.

Predictors of CM status
Summary of logistic regression model for predicting SSDI-positive status (i.e. CM) based on claims data collected in the 12-month enrollment period.

CM: chronic migraine; SSDI: semi-structured diagnostic interview.
Performance characteristics of the claims-based algorithm
When the ability of the claims-based algorithm to detect CM was compared with that of the gold standard SSDI using a probability cutoff of 0.55 (i.e. probability ≥0.55 that a patient in the sample has CM based on the logistic regression model), the claims-based algorithm resulted in a C-statistic of 0.80 (Figure 2), a sensitivity of 78.1%, a specificity of 72.7%, a positive predictive value of 80.7%, and a negative predictive value of 69.7%.
Receiver operating characteristic curve for the claims-based algorithm model and SSDI data from 108 patients with migraine.
Sensitivity analyses
Performance characteristics of the claims-based algorithm.*

Values reported are mean (95% CI).
Discussion
In this study, we developed a claims-based algorithm to identify potentially undiagnosed CM patients among patients with migraine-related claims not otherwise recognized as having CM for implementation in electronic databases containing patient health data. Claims databases, which document healthcare visits, procedures performed, diagnoses assigned, and medications filled, have commonly been used to assess prevalence rates for medically diagnosed diseases (26) costs (27,28) healthcare resource utilization (29) and treatment patterns (30). Additionally, claims databases have been used to develop algorithms to identify at-risk patients to improve population health management (31–33). For example, internationally, claims databases have been developed with varying degrees of success to support patient management, including to estimate the prevalence of depression in healthcare users (31) and to identify undiagnosed chronic obstructive pulmonary disease (32) and acute episodes of gout (33).
It is recognized that <50% of people with migraine seek medical advice (34), with approximately 15% having a migraine-related inpatient or outpatient claim in a 24-month period (35). Despite the burden of disease and associated healthcare costs, CM also is often undiagnosed (2). The availability of a claims-based approach to detect individuals with undiagnosed CM among patients with migraine accessing medical care represents important progress in the management of CM in any large health system or payer population. Others have developed claims-based algorithms to detect CM but have not reported the sensitivity or specificity of their model (36). Using multivariate logistic regression and clinical expert input, we developed a claims-based algorithm that can be used to screen for CM in a healthcare system setting that included sex, number of claims for acute treatment of migraine (including opioids; <15, ≥15), number of healthcare visits of any type (emergency room, inpatient, and outpatient visits; <24, ≥24), and number of unique migraine preventive classes (0, 1, or ≥2) as predictors of CM. By using dichotomized predictor variables, the final model could potentially be developed as a paper-based screening tool; however, additional rigorous research would be required. These variables were all statistically significant and clinically relevant predictors of migraine, as confirmed by clinical expert opinion and prior studies demonstrating higher medication use (7,37), healthcare resource utilization (7,8), and female prevalence (38) in CM versus EM patients. Furthermore, the area under the receiver operating characteristic curve of 0.80 for our algorithm suggests that the model performed well in detecting patients with CM. Nonetheless, it must be recognized that as a screening tool, any patient identified should be fully assessed by a healthcare practitioner to confirm their CM status.
Despite the strengths of our approach (i.e. using real-world data from the healthcare database of a large medical group and verifying CM status using the SSDI administered by trained clinicians) and the acceptable performance measures of the final claims-based algorithm, this study has some limitations. First, the study cohort was primarily female, which may limit the generalizability of this claims-based algorithm for detecting CM in male subjects. However, it is well established that women are approximately threefold more likely to have migraine and CM than men (38,39), so this algorithm has been shown to have applicability in the population most at risk of migraine. Nonetheless, validation of this claims-based algorithm in the male population would further increase its applicability as a real-world tool. Secondly, the claims-based algorithm was limited to variables that were available in the medical and pharmacy claims database. Data such as the number of headache days per month or the use of over-the-counter medicines were not available in the claims database and could not be included as variables. Third, the lists of acute and preventive treatments for migraine were restricted to treatments with Level A or B evidence from the clinical practice guidelines and also informed by clinical expert opinion (13,23,24). Patients may have used other medications for acute or preventive treatment of migraine; these medications would not have contributed to the count of claims for acute treatment of migraine or the claims for the unique migraine preventive classes. Fourth, the enrolled sample had a substantively higher prevalence of CM (SSDI-positive, 59.3%) than would be expected based on population data, where 6.3% to 8.8% (38,39) of all people with migraine have CM. However, the higher prevalence of CM in a healthcare system sample than in the general population was to be expected, as not all people with migraine seek medical intervention (34,35), and of those who do seek medical attention not all are diagnosed with or receive treatment for migraine (34). Additionally, as the sample for our claims-based algorithm required all enrolled patients to have at least one claim with a migraine diagnosis in the 12-month screening period, it is likely that more severe migraine patients were enrolled in this study. Given that the study sample was from a large healthcare system, it is likely that the prevalence of CM among people with migraine-related claims is similarly high in other US healthcare settings. Nonetheless, the high CM prevalence in this sample may have inflated the odds ratios for this analysis (19). This could have led to an overestimate of the positive predictive value and an underestimate of the negative predictive value (19). In addition, the sample size of 108 patients was relatively small to predict CM status using medical and pharmacy claims. For this reason, the confidence intervals observed in the analysis of the claims-based algorithm were relatively wide. Although the development of the algorithm demonstrated good sensitivity and specificity, it is to be noted that a separate validation sample was not used to further evaluate the validity of this algorithm. Further research is recommended to assess the validity of the algorithm in samples of patients from different health plan populations and different healthcare systems.
Notwithstanding these limitations, the claim-based screening algorithm developed is a useful tool to support the identification of CM in a healthcare system. Any claims-based screening tool will have false-positive and false-negative screening errors. False-positive screening errors are not of concern, as those patients identified would have a high burden of migraine disease and would benefit from further clinical assessment.
Conclusions
Four key predictors, from more than 40 potential predictors of CM, were identified, including female sex, ≥15 claims for acute treatment of migraine (including opioids), ≥24 healthcare visits of any type, and claims for ≥2 unique migraine preventive classes. These variables were statistically significant predictors of CM status and were the most clinically relevant. Based on these key predictors, the claims-based algorithm demonstrated acceptable sensitivity (78.1%) and specificity (72.7%) in identifying undiagnosed CM.
This claims-based algorithm represents a novel predictive analytic approach to identify patients within a healthcare setting who may require better management of their migraines, given their high amount of acute medication use, preventive medication use, and healthcare resource utilization. This algorithm has the potential to be applied in real-world settings with access to medical and pharmacy claims data to identify patients with potentially undiagnosed CM, and to subsequently improve the diagnosis and management of those patients who may not otherwise be detected.
Clinical implications
The claims-based algorithm to identify potentially undiagnosed chronic migraine patients demonstrated acceptable sensitivity and specificity for use in real-world healthcare settings. Statistically significant predictors of chronic migraine diagnosis included female sex, ≥ 15 claims for acute treatment of migraine (including opioids) per year, ≥ 24 visits of any type to a healthcare setting per year, and claims for ≥ 2 unique migraine preventive classes per year. The use of this claims-based algorithm in real-world healthcare settings can help detect patients with chronic migraine, and subsequently improve the diagnosis and management of the disease in patients who may not otherwise be detected.
Supplemental Material
Supplemental material for Development of a claims-based algorithm to identify potentially undiagnosed chronic migraine patients
Supplemental Material for Development of a claims-based algorithm to identify potentially undiagnosed chronic migraine patients by Jelena M Pavlovic, Justin S Yu, Stephen D Silberstein, Michael L Reed, Steve H Kawahara, Robert P Cowan, Firas Dabbous, Karen L Campbell, Anand R Shewale, Riya Pulicharam, Jonathan W Kowalski, Hema N Viswanathan and Richard B Lipton in Cephalalgia
Footnotes
Acknowledgments
Editorial support for development of this manuscript was provided by Lee B Hohaia, at Complete Healthcare Communications, LLC (West Chester, PA), a CHC Group company, and funded by Allergan plc (Dublin, Ireland).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jelena M Pavlovic has served as a consultant and/or advisory panel member for Alder Biopharmaceuticals, Allergan, and Dr Reddy’s Laboratories, and has received travel support from the American Headache Society and North American Menopause Society. She has received research support from the NIH/NIA. Justin S Yu is a former employee of Allergan plc and owns stock in the company. Stephen D Silberstein has served as a consultant and/or advisory panel member for Alder Biopharmaceuticals, Allergan Inc, Amgen, Avanir Pharmaceuticals Inc, eNeura, ElectroCore Medical, LLC, Dr Reddy’s Laboratories, Medscape, LLC, NINDS, Teva Pharmaceuticals, Supernus Pharmaceuticals Inc, and Theranica. His employer receives research support from Allergan Inc, Amgen, Dr Reddy’s Laboratories, ElectroCore Medical Inc, Eli Lilly and Company, and Supernus Pharmaceuticals. Michael L Reed is Managing Director of Vedanta Research, which has received research funding from Allergan, Amgen, Dr Reddy’s Laboratories, Eli Lilly, GlaxoSmithKline, Merck & Co., Inc., and Promius, via grants to the National Headache Foundation. Steve Kawahara and Riya Pulicharam are full-time employees of DaVita Medical Group. Robert P Cowan and Firas Dabbous have no financial disclosures to report. Karen Campbell, Anand R Shewale and Jonathan Kowalski are full-time employees of Allergan plc and own stock in the company. Hema N Viswanathan is an employee of Allergan plc and holds stock or stock options, patents, or other intellectual property. Richard B Lipton serves on the editorial boards of Neurology and Cephalalgia and as senior advisor to Headache. He has received research support from the NIH. He also receives support from the Migraine Research Foundation and the National Headache Foundation. He has reviewed for the NIA and NINDS; serves as consultant, advisory board member, or has received honoraria from Alder, Allergan, Amgen, Autonomic Technologies, Avanir, Biohaven, Biovision, Boston Scientific, Dr Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Pernix, Pfizer, Supernus, Teva, Vector, and Vedanta. He receives royalties from Wolff’s Headache (8th Edition, Oxford University Press), Informa, and Wiley. He holds stock options in eNeura Therapeutics and Biohaven.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Allergan plc (Dublin, Ireland).
*Current affiliation: Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
