Abstract
Background
The relationship between high sensitivity C-reactive protein and migraine is unclear. The aim of this cross-sectional population-based study was to investigate the association between high sensitivity C-reactive protein and types of headache, and to evaluate the impact of insomnia on this association.
Methods
A total of 20,486 (63%) out of 32,591 invited, aged ≥40 years or older, participated in the seventh wave of the Tromsø study conducted in 2015–2016 and had valid information on headache, insomnia and high sensitivity C-reactive protein. The influence of insomnia on the association between questionnaire-based diagnoses of headache and elevated high sensitivity C-reactive protein defined as >3.0 mg/L was assessed using multiple logistic regression, estimating prevalence odds ratio with 95% confidence intervals.
Results
A total of 6290 participants (30.7%) suffered from headache during the last year. Among these, 1736 (8.5%) fulfilled the criteria of migraine, 991 (4.8%) had migraine with aura, 746 (3.6%) migraine without aura (3.8%), and 4554 (22.2%) had non-migrainous headache. In the final multi-adjusted analysis, elevated high sensitivity C-reactive protein was associated with headache (odds ratio 1.10, 95% confidence interval 1.01–1.20), migraine (odds ratio 1.17, 95% confidence interval 1.01–1.35), and migraine with aura (odds ratio 1.23, 95% confidence interval 1.01–1.53). No association was found between elevated high sensitivity C-reactive protein and migraine without aura or non-migrainous headache. The association between high sensitivity C-reactive protein and migraine was strongly dependent on insomnia status. Among individuals with insomnia, elevated high sensitivity C-reactive protein was associated with migraine (odds ratio 1.49, 95% confidence interval 1.02–2.17), and migraine with aura (odds ratio 1.59, 95% confidence interval 1.03–2.45), whereas no such relationship was found among those without insomnia.
Conclusions
In this cross-sectional study, participants with migraine, in particular migraine with aura, were more likely to have elevated high sensitivity C-reactive protein, evident only among those with insomnia.
Introduction
Migraine is a chronic disorder with a global prevalence of approximately 15% (1) and ranked as the second leading cause of years lived with disability (2).
Migraine, particularly migraine with aura (MA), is associated with ischemic stroke (3). The precise mechanisms remain unclear, but a neurovascular inflammation as a part of the complex pathophysiological process has been suggested (4). High sensitivity C-reactive protein (hs-CRP), a non-specific marker of inflammation, is associated with risk of cardiovascular events (5). This relationship has most likely been contributing to the increasing focus on CRP and migraine for the last decade (6–30).
Most previous studies evaluating the association between CRP and migraine have been case-control studies with small sample sizes reporting inconsistent results. Some have reported significantly higher CRP values in migraine patients (8,12,13,19,23–27), whereas others have not (7,10,15,16,20,21). Various results have also been found in population-based studies (9,11,14,29). CRP levels were not increased in migraineurs compared to non-migraineurs in a study from Iceland (11) or in adjusted analyses in a Dutch population-based case-control study (29). In contrast, an association was found between increased CRP and migraine in a large cross-sectional study on women (9). An association between CRP and headache has also been reported in children and adolescents (14). A review of studies published between 2006 and 2014 indicated higher CRP levels in individuals with migraine than in controls (31). However, relatively few previous studies have performed analyses with adjustments for relevant potential confounders.
Insomnia may potentially modify the relationship between hs-CRP and migraine. The relationship between headache and insomnia is well known (32), and shorter sleep has been associated with a higher level of CRP in a prospective study (33). However, to the best of our knowledge, no previous studies have explored the impact of insomnia on the relationship between CRP and migraine. Such an approach may be of value for better understanding the mechanism behind a potential association between CRP and migraine.
The aim of the present large-scale population-based study was to evaluate the cross-sectional association between hs-CRP and types of headache, and to investigate the impact of insomnia on this association. Based on previous knowledge, we hypothesized that migraineurs would be more likely to have elevated hs-CRP.
Methods
The Tromsø Study
The Tromsø Study is a population-based cohort study initiated in the municipality of Tromsø, Northern Norway. The study includes seven surveys (Tromsø 1–7) conducted between 1974 and 2016, to which total birth cohorts and representative random samples have been invited. The surveys included questionnaires and interview, biological sampling and clinical examination.
Study population
In Tromsø 7 (2015–2016), all inhabitants aged ≥40 years living in Tromsø (n = 32,591) were invited, of which 21,083 (65%) women and men aged 40–99 years participated. Information about headache and insomnia were collected via questionnaires and hs-CRP was analyzed from non-fasting samples. The present analyses are based on data from all 20,486 participants with valid measures of hs-CRP, headache and insomnia (Figure 1).
Flow chart of participation in Tromsø Study 2015–2016.
C-reactive protein
Hs-CRP was analyzed at the Department of Laboratory Medicine, University Hospital of North Norway, Tromsø, Norway, and assessed by a particle-enhanced immunoturbidimetric assay on a Modular P autoanalyzer (Roche Diagnostics, Mannheim, Germany) with reagents from the manufacturer with a detection limit of 0.12 mg/L (34). In recent population studies from the US, approximately two-thirds of Americans had serum CRP concentrations less than 3 mg/L, suggesting that 3 mg/L is a reasonable cut point between normal and elevated hs- CRP (35,36). Thus, in the present study, we replicated the strategy used in a previous published study from Tromsø 6 evaluating hs-CRP and cold-pressure tolerance (34), defining normal CRP as 0–3.00 mg/L, and elevated CRP as 3.01–20.00 mg/L. Participants with hs-CRP values >20.00 mg/L, which may indicate acute or chronic disease (38), were excluded (n = 116) (Figure 1). The study did not include other exclusion criteria than lack of response on questions about headache and insomnia.
Headache
A questionnaire screening question (“Have you suffered from headache during the last 12 months?”) was used to identify participants with and without headache (reference group).
Participants who answered “yes” to the screening question completed a set of additional questions adapted from the third Nord-Trøndelag Health Study (HUNT3) (39), mainly to diagnose active migraine (i.e. during the last 12 months) according to the second version of the International Classification of Headache Disorders (ICHD-II) (40). Headache sufferers were classified as having active migraine if they fulfilled the following three criteria: a) Headache attacks lasting 4–72 hours (<4 hours were accepted for those who reported visual disturbance before headache); b) headache with at least two of the following characteristics: Pulsating quality, unilateral location, moderate or severe pain intensity, and aggravation of physical activity; and c) during headache, at least one of the following: Nausea and/or vomiting, or photophobia and phonophobia. Participants who fulfilled the migraine diagnosis and reported visual disturbance prior to headache were classified as MA, whereas the remaining participants with migraine had migraine without aura (MO). The remaining participants with headache were classified as having “non-migrainous headache”. The merged group of “any headache” consisted of participants with migraine or non-migrainous headache.
A question about number of headache days per months, with the following options: <1 day, 1–6 days, 7–14 days, and more than 14 days, was also available.
The validity of the questionnaire-based headache diagnoses has been evaluated in HUNT3 (39): For any headache, the sensitivity was 88%, and specificity 86% (kappa value at 0.70, 95% CI 0.61–0.79); for migraine the sensitivity was 51%, and specificity 95% (kappa value 0.50, 95% CI 0.32–0.68), and for MA the sensitivity was 50%, and specificity 95% (kappa value 0.44, 95% CI 0.38–0.50) (39).
Insomnia
The sleep-related questions were adapted from the validated Berge Insomnia scale (41) and the questionnaire-based definition of insomnia was given in accordance with DSM-V (Diagnostic and Statistical Manual of Mental Disorders, 5th edn.). Insomnia was defined as a) Sleep disturbance for at least 3 months during the last year (“If sleep problem, how long time?); b) at least three times per week with sleep disturbance that reduce performance at school, job and/or social life at least three times per week (“How many days per week have you felt so tired that it has affected your work, school or private life?”); c) dissatisfied with sleep at least three times per week (“How many days per have you been dissatisfied with sleep?”); and d) at least three times per week with a duration of a minimum of 30 minutes having difficulty initiating (“How many nights per week do you usually use more than 30 minutes to fall asleep?”) and/or maintaining (“How many nights per week do you usually wake up for more than 30 minutes in the middle of the night?”) and/or waking up too early (“How many nights per week do you usually wake up 30 minutes earlier than you wished, without being able to go back to sleep again?”).
Covariates
The following variables were examined as potential confounders based on previous literature (35,37,42): age (continuous); sex, years of education (≤9, 10–12 and ≥13 years); body mass index (BMI) (<25, 25.0–29.9, and ≥30 kg/m2) (28,32); smoking (current, previous, and never), physical activity (never, ≤1 time per week, ≥2 times per week), alcohol consumption (never, <2 times/week, ≥2times/week), self-reported diabetes (yes/no), self-reported stroke and/or heart infarction (yes/no), and self-reported hypertension (yes/no). Symptoms of anxiety and depression were measured by the 10-item questionnaire of the Hopkins symptom checklist (HSCL-10), using mean scores of ≥1.85 as cut-off for emotional distress (43).
Ethics
This study was approved by the Regional Committee for Ethics in Medical Research (#2016/1997/Rek sør-øst C and #2014/940/Rek Nord), and the Norwegian Data Protection Authority. The participants have given written informed consent.
Statistical analysis
In the multivariate analyses, using logistic regression, we estimated the prevalence odds ratio (OR) with 95% confidence interval (CI) for the association between two categories of hs-CRP (dependent variable) and types of headache. Participants without headache were used as reference.
We initially adjusted for age and sex (model 1), and subsequently for predefined confounding factors. To determine which factor contributed most to the effect on the adjustment, analyses were carried out with adjustment for each separate factor in addition to age and sex. In model 2, we adjusted for age, sex and BMI. In addition, multiple factors were also tested together. We excluded factors from model 3 if they did not change OR at all or just changed marginally (≤0.01) when evaluating each factor separately or by including several factors grouped together (i.e. self-reported diabetes, stroke/heart infarction, and hypertension). Potential interaction between two variables was evaluated by including the product of the variable in the logistic regression analyses, and the interaction was tested using Wald χ2 statistics. Subjects with missing data on confounding factors were included in the analysis to reduce the impact of possible bias.
To evaluate insomnia as a modifying factor on the association between CRP and types of headache, we repeated the multi-adjusted analyses in model 3, separating by insomnia status.
Analyses were performed with the IBM SPSS version 25 (SPSS, Chicago, IL, USA).
Results
Characteristics of participants (n = 20,486) in the Tromsø Study 2015–2016 according to types of headache.
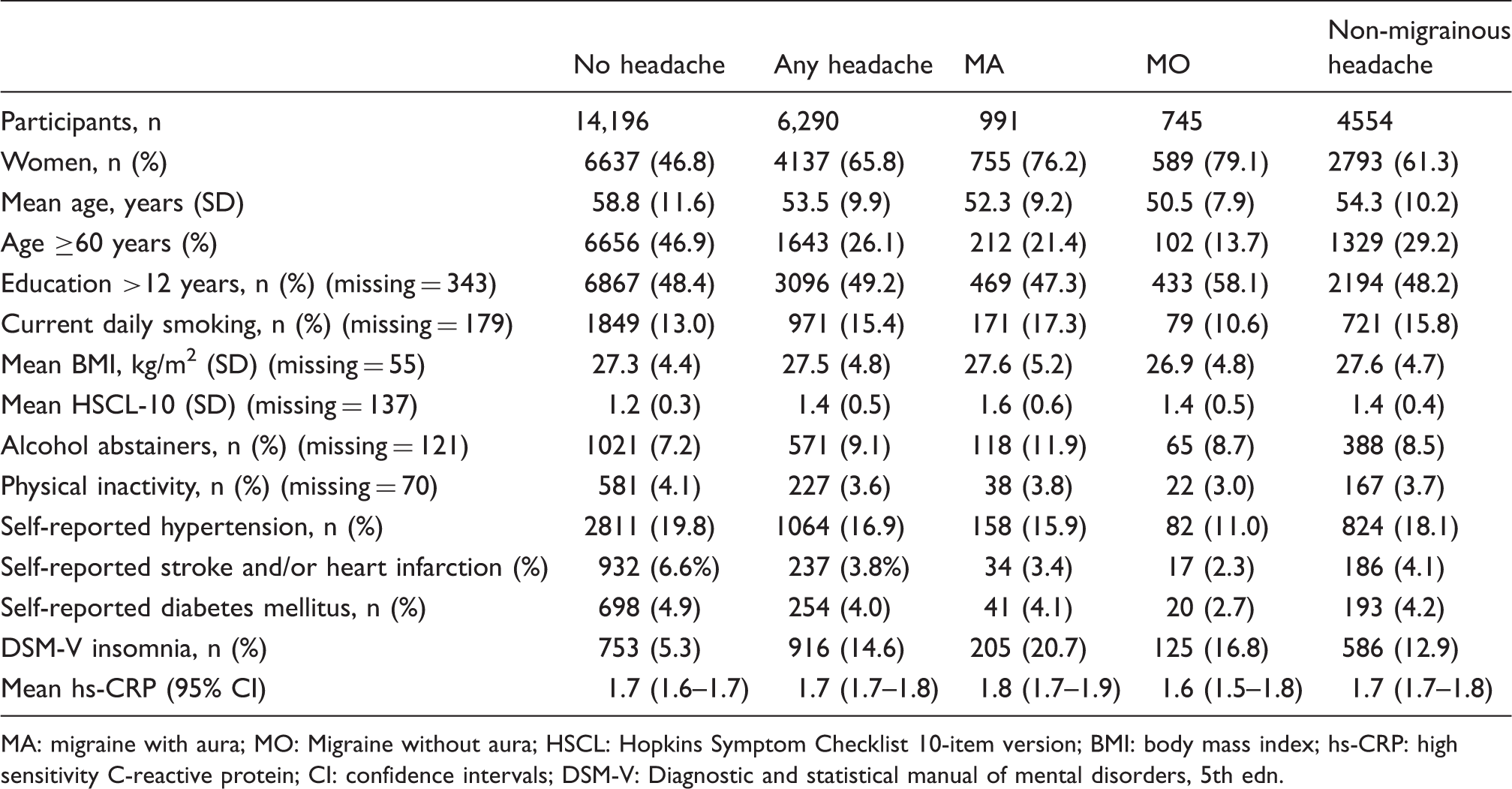
MA: migraine with aura; MO: Migraine without aura; HSCL: Hopkins Symptom Checklist 10-item version; BMI: body mass index; hs-CRP: high sensitivity C-reactive protein; CI: confidence intervals; DSM-V: Diagnostic and statistical manual of mental disorders, 5th edn.
Prevalence of headache
A total of 6290 (30.7%) participants suffered from headache during the last year, 1736 (8.5%) fulfilled the criteria for migraine, 991 (4.8%) had migraine with aura (MA), 746 (3.6%) migraine without aura (MO) (3.8%), and 4554 (22.2%) had non-migrainous headache.
The association between elevated hs-CRP and headache
Odds ratio (OR) with 95% confidence interval (CI) of C-reactive protein (CRP) defined as >3.0-20.0 mg/l according type of headache.

MA: migraine with aura; MO: Migraine without aura. Model 1: Adjusted for age, and sex. Model 2: Adjusted for age, sex and body mass index. Model 3: Adjusted for age, sex, body mass index, smoking, education level, alcohol use, physical activity, and anxiety and depression measured by the Hopkins Symptom Checklist.
Although no interaction was observed, the impact of headache frequency was evaluated. In age- and sex-adjusted analyses, somewhat stronger association with elevated hs-CRP was found for individuals with headache ≥7 days/month than for those with headache less than 7 days/month (Table 2). In the final multi-adjusted analyses, elevated hs-CRP was associated with migraine ≥7 days/month (OR 1.21, 95% CI 1.01–1.46), but not migraine less than 7 days/month (OR 1.14; 95% CI 0.96–1.35).
The impact of insomnia
Multi-adjusted* odds ratio (OR) with 95% confidence interval (CI) of elevated C-reactive protein (CRP) defined as >3.0–20.0 mg/l according to type of headache separated by insomnia status.

Adjusted for age, sex, body mass index, smoking, education level, alcohol use, physical activity and anxiety and depression measured by the Hopkins Symptom Checklist.
MA: migraine with aura; MO: Migraine without aura.
Discussion
In this population-based cross-sectional study, participants with migraine, in particular MA, were more likely to have elevated hs-CRP (between >3–20 mg/L), evident only among those with insomnia.
Comparison with other population-based studies
In accordance with our results, the Women's Health study showed that women with migraine were more likely to have CRP of 4.2 mg/l or above (OR 1.13, 95% CI 1.05–1.22) than women without migraine (9). Furthermore, among children and adolescents aged 4–19 years, those in the highest CRP quintile were associated with headache (OR 1.23, 95% CI 1.00–1.51) (14). In the Dutch case-control study (29), greater than median value was defined as elevated hs-CRP. In the unadjusted analyses, MA participants were more likely to have elevated hs-CRP (1.74, 95% CI 1.09–2.78), and borderline elevated in the multi-adjusted model (OR 1.60, 95% CI 0.97–2.64). In the study from Iceland, CRP levels were not increased in migraineurs compared to non-migraineurs (11). It should be noted that the Reykjavik study excluded subjects with CRP values above 10 mg/L (11), whereas the exclusion cut-off was above 20 mg/L in the present study.
In accordance with the Dutch study (29), MA was most consistently associated with elevated hs-CRP in the present study. We did not find any association with MO. By reviewing previous studies, a consistently stronger association in MA than in MO has not been demonstrated (31). However, in the Dutch study, the average number of aura attacks and total number of years of aura were both found to be associated with hs-CRP (29). Correspondingly, in the present study, the highest OR for having elevated hs-CRP was found for individuals with migraine at least 7 days/month.
To the best of our knowledge, no previous study has evaluated insomnia as a modifying factor on the association between CRP and migraine or included insomnia as one of the confounding factors. Interestingly, we found that the significant association between elevated hs-CRP and migraine, in particular MA, was only evident among individuals with insomnia.
Interpretation
If there is a causal link between hs-CRP, ischemic cardiovascular disease, and migraine, a stronger relationship between elevated hs-CRP and MA should be expected, because a relationship with ischemic cardiovascular disease is most evident for MA (3). On the other hand, the present results could represent an epiphenomenon, not specific for migraine (31). Based on our results, one may speculate that insomnia may be of major importance in the relationship between CRP and migraine. Possibly, migraine, insomnia and elevated hs-CRP may share a common underlying factor; for example, related to endothelial function. However, this explanation seems less likely because no consistent relationship has been found between endothelial function measured by flow-mediated dilatation and insomnia (44) or migraine (45) in population-based studies. In future studies evaluating the association between CRP and migraine, the influence of insomnia should be considered.
Strengths and limitations of the study
The major strengths of this study are the population-based design and the possibility of performing adjustments for important potential confounding factors. However, the possibility of residual confounding by an unrecognized factor cannot be ruled out. The participation rate (65%) was good, but generalization of results should be performed with caution. Finally, the diagnosis of insomnia in this study was in accordance with the current DSM-V criteria for insomnia.
Several study limitations should also be considered. Firstly, the cross-sectional design does not permit any conclusions about causality. Secondly, the diagnoses of headache and insomnia were questionnaire-based, which may have led to misclassification. However, the questionnaire-based diagnosis of migraine has previously shown acceptable agreement with corresponding diagnosis in a clinical interview (39). MA diagnosis was based on one question about visual disturbance before headache, with high specificity but low sensitivity (39). Ideally, more details about the aura symptoms would have been informative; for example, the proportion of migraine attacks that were accompanied by aura. The sleep-related questions were adapted from the Bergen Insomnia Scale, which has previously been validated against the DSM-IV criteria for insomnia (41). The DSM-V criteria for insomnia are somewhat different from other recent population-based studies that have either used the older DSM-IV definition of insomnia (46) or studies that have operationalized the ICDS-3 criteria for insomnia, considering dissatisfaction with sleep quality or sleep length as not mandatory for the diagnosis of insomnia (47). The definition of insomnia used in the present study is stricter, demanding dissatisfaction with sleep as a mandatory criterion for the definition of insomnia.
Conclusions
In this population-based cross-sectional study, participants with migraine, in particular MA, were more likely to have elevated hs-CRP, evident only among those with insomnia.
Footnotes
Clinical implications
Participants with migraine, in particular migraine with aura, were more likely to have elevated hs-CRP.
The association between elevated hs-CRP and migraine was only found among participants with insomnia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
