Abstract
Cluster headache (CH) is a primary headache with a close relation to sleep. CH presents a circa-annual rhythmicity; attacks occur preferably during the night, in rapid eye movement (REM) sleep, and they are associated with autonomic and neuroendocrine modifications. The posterior hypothalamus is the key structure for the biological phenomenon of CH. Our aim is to describe a 55-year-old man presenting a typical episodic CH, in whom we performed a prolonged sleep study, consisting of a 9-week actigraphic recording and repeated polysomnography, with evaluation of both sleep macrostructure and microstructure. During the acute bout of the cluster we observed an irregular sleep-wake pattern and abnormalities of REM sleep. After the cluster phase these alterations remitted. We conclude that CH was associated, in this patient, with sleep dysregulation involving the biological clock and the arousal mechanisms, particularly in REM. All these abnormalities are consistent with posterior hypothalamic dysfunction.
Introduction
Cluster headache (CH) is a primary headache (1) of neurovascular origin (2). Clinically, episodic CH is characterized by a very severe pain, always unilateral and usually retro-orbital, associated with restlessness or agitation and autonomic symptoms (2). The attacks of CH occur typically in bouts (clusters) of a few months. The rhythmic course, the autonomic activation, the circadian biological rhythmic changes and the neuroendocrine disturbances (3), in particular involving melatonin (4), have suggested a pivotal role for the hypothalamus which has been demonstrated by functional neuroimaging with positron emission tomography (5) and by anatomical imaging with voxel-based morphometry (6).
Clusters have a half-yearly, yearly or even biennial periodicity (7). Within a CH cluster about 50% of attacks occur at night, and in the same individual they often occur at the same hour each day (7). CH is closely related to sleep (8). Wolff (9) observed that the majority of cluster headaches occur during sleep, with pain that startles the patient from bed. Dexter and Weitzmann (10) first reported that episodic attacks of CH tend to occur during rapid eye movement (REM) sleep; these data have been confirmed (11) and recently (12, 13) CH attacks have been observed during REM sleep, following haemoglobin desaturations.
The aim of the present study was to investigate the sleep pattern of a patient with typical episodic CH during a cluster and in the pain-free interval. We used wrist actigraphy to investigate circadian sleep–wake cycles, polysomnography (PSG) to analyse sleep structure and to observe in which sleep stage pain occurred; also, an analysis of sleep microstructure was made to investigate the relation between CH and arousal.
Case report
A 55-year-old man presented with episodic CH since the age of 50 years. The typical attacks consisted of a very severe (10/10) pricking pain in the right retro-orbital and right temporal areas. The pain was associated with photophobia. Cluster periods occurred once per year and lasted 3–4 weeks. During the cluster, attacks occurred twice per day, each attack lasting 45–90 min, and most of them (more than one half) awakened the patient from night sleep. During the attacks he had tearing, ipsilateral conjunctival injection and eyelid oedema; the right nostril was congested. At the moment of our first observation the patient was in the cluster period, which had begun 12 days before. His physical and neurological examinations were normal. Brain magnetic resonance imaging was normal. Prophylactic treatment was started with gabapentin up to 900 mg/day; gabapentin was chosen because EKG documented a sinusal bradicardia (45–50 beats/min), which contraindicated the administration of verapamil. Subcutaneous sumatriptan injections (6 mg) for the treatment of pain attacks were administered on two occasions. Prophylaxis was immediately effective in reducing the frequency, duration and severity of attacks. On a follow-up visit the patient reported that these episodes ceased after a period of 3 weeks. As concerns the sleep–wake habits, the patient did not complain of any sleep disorder before, during or after the cluster episode. The patient gave his written informed consent to enter the study.
Methods
Sleep study included: (i) cardiorespiratory monitoring; (ii) actigraphy; and (iii) PSG.
Cardiorespiratory monitoring
A one-night polygraphic recording was performed with MESAM 4 (14) in order to rule out obstructive sleep apnoea syndrome (12, 13).
Actigraphy
Actigraphy was performed to evaluate the circadian rhythms of sleep and wake. Three separate recordings were performed, each lasting three consecutive weeks. The first recording started at the moment of our first observation, 12 days after the onset of the cluster period; the second was performed during pharmacological treatment (7 weeks after the onset); the third was performed during follow-up, 3.5 months after the last attack and 2.5 months after drug withdrawal. Actigraphic monitoring was performed in agreement with guidelines reported in the literature (15–17). Data from the single nights in the two conditions (cluster period and remission) were compared by two-tailed unpaired Student's t-test followed by Bonferroni correction for multiple comparisons, with an adjusted significance level of ≤0.00625.
Polysomnography
Three full-night ambulatory polygraphic recordings (EEG, EOG, EMG) were performed, each lasting 48 consecutive hours, in order to allow the patient to sleep in his home setting and to keep his natural sleep–wake schedule. The first study was performed before starting the treatment (nights 1 and 2), the second during the treatment (nights 3 and 4), the third during remission, after drug withdrawal (nights 5 and 6). Sleep analysis was performed according to the criteria of Rechtschaffen and Kales (18), and, for the arousals, to the rules of the American Sleep Disorders Association (ASDA) (19).
Results
Cardiorespiratory monitoring
MESAM 4 ruled out the presence of any significant oxygen desaturation during sleep.
Actigraphy
Figure 1 shows three actograms, two referring to the cluster period (Fig. 1A,B) and one referring to the remission period (Fig. 1C). Several attacks of CH occurred during the recordings. Actigraphic recordings revealed that the patient's sleep–wake schedule was irregular during the CH period and progressively modified, becoming more regular, after remission. In particular, the interdaily variability index (19) was 0.513 and 0.545 in the first two recordings, and rose to 0.681 in the recording after remission.

Actograms from the three recordings, each lasting three consecutive weeks. (A, B) Cluster period. (C) Remission period. The upper horizontal bar indicates time of day. Arrows indicate the days in which cluster headache attacks occurred. Days when polygraphic (PSG) recordings were performed are specified on the left of the actogram. GP, Gabapentin. Start of treatment and dose modification are specified on the right side of the actogram.
Several episodes of diurnal sleep (naps) were observed in the cluster periods; napping was almost completely absent in the remission period. As concerns the statistical comparison among conditions (C = cluster period, R = remission period), we observed a significant decrease of actual sleep time (AST) (average AST C = 376 ± 74 min; R = 350 ± 61 min; P = 0.0040) and sleep efficiency index (SEI) (average SEI C = 73.1 ± 6.8%; R = 67.6 ± 7.8%; P = 0.0051). By contrast, we observed a significant increase in mean activity score (MAS) (average MAS C = 20.32 ± 4.22; R = 24.66 ± 6.81; P = 0.0027) and fragmentation index (FI) (average FI C = 53.30 ± 22.77; R = 73.15 ± 28.71; P = 0.0002). No significant difference between the two conditions was found in time in bed (P = 0.0558), sleep latency (P = 0.6017), assumed sleep duration (P = 0.0134) and wake time (P = 0.8529).
Polysomnography
The main results of polygraphic recordings are listed in Table 1, and hypnograms are shown in Fig. 2. Headache attacks occurred during recordings 1 and 2; both occurred during REM. Both attacks were treated successfully with sumatriptan and the patient returned to sleep. PSGs performed in the cluster period confirmed the great variability in sleep duration observed with actigraphy. PSGs 5 and 6, after remission, showed slightly shorter sleep duration, and a reduction of the total amount of REM (in minutes) and of REM percentages. No modification of the arousal parameters across the recordings was observed, except for arousal index in REM. This index was much lower in the first two nights, those with attacks (index = 5.56 and 3.58 events/h of REM sleep), than in the remaining four (12.45, 13.02, 15.00, 12.79 events/h of REM sleep).
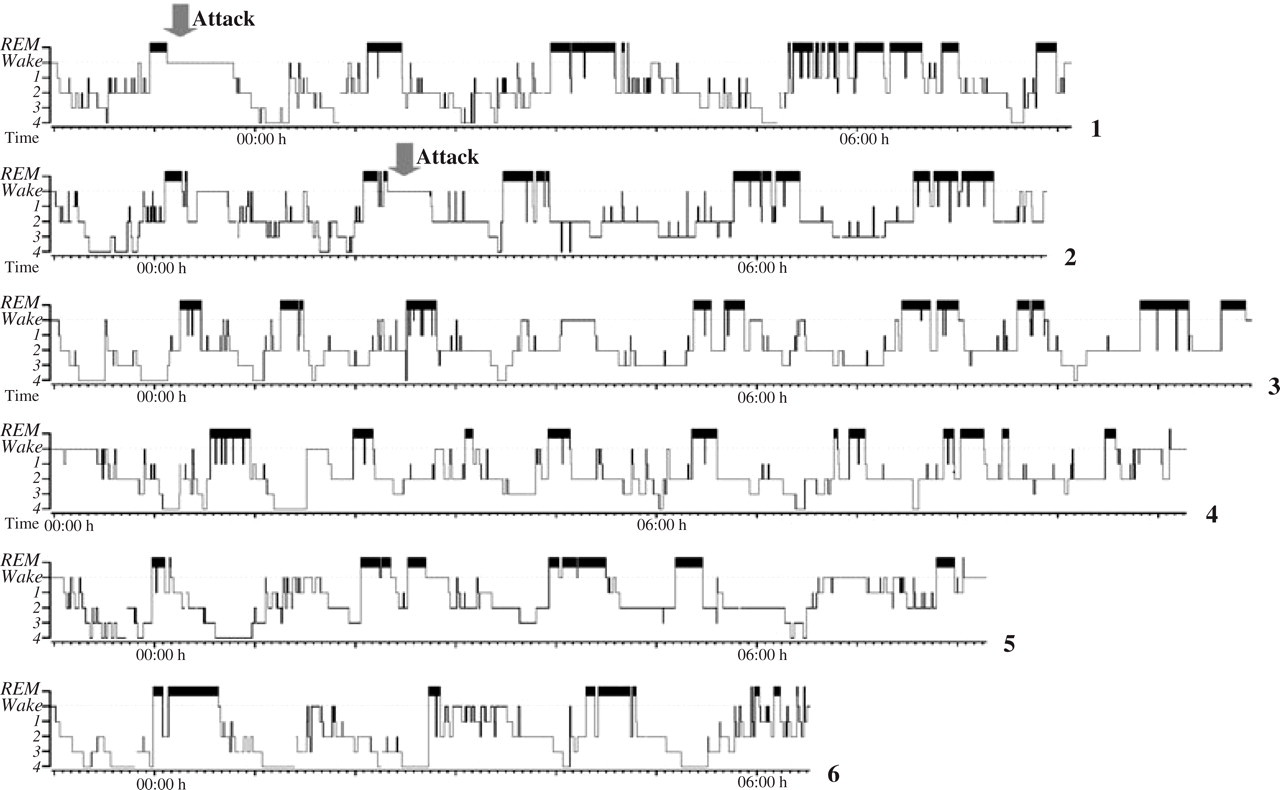
Sleep stage histograms (hypnograms) in the six nights of polysomnographic recording. On the left of the hypnograms sleep stages are indicated (REM, rapid eye movement sleep; 1, stage 1 NREM; 2, stage 2 NREM; 3, stage 3 NREM; 4, stage 4 NREM); the lower horizontal bar indicates time. Grey arrows mark headache attack onset.
Polysomnography (PSG) parameters

Sleep period time, interval between sleep start and sleep end; Sleep efficiency, percentage of time spent asleep whilst in bed. Sleep stages percentages are calculated on sleep period time.
Discussion
CH induced relevant modifications of the sleep–wake pattern of our patient. During the cluster period, actigraphy showed an irregular sleep–wake cycle, characterized by high interdaily variability, inconstant sleep duration, repeated nocturnal awakenings and frequent daytime napping (Fig. 1A,B). When the cluster period ended and gabapentin was withdrawn (Fig. 1C), the sleep–wake pattern appeared fully normalized. This indicates that the cluster period was associated with a transient dysfunction of the biological clock. Alternatively, the irregular sleep–wake cycle could be a consequence of pain (20). The latter interpretation is not consistent with the observation that a marked interdaily variability of the sleep–wake rhythm still persisted even when headache attacks were controlled by gabapentin. Therefore, we believe that the sleep–wake pattern dysregulation observed in our patient is a specific clinical feature of the CH. This is consistent with previous data in literature, which documented that CH is a chronobiological disorder (7), due to hyperactivation of the posterior hypothalamus (5, 6, 21). PSGs confirmed the variability in sleep–wake schedule observed with actigraphy.
As regards the PSGs, the most relevant findings concern REM sleep. Two severe attacks occurred in REM at the beginning of the night. Besides, during the cluster, REM episodes were fragmented, and interspersed by periods of NREM sleep. After remission, REM sleep time and REM percentages and REM fragmentation were reduced (Fig. 2). Therefore, PSG findings suggest some dysregulation of the ultradian NREM/REM cyclicity within sleep in this patient.
At a microstructural level, the arousal index calculated in REM was much lower in the first two nights, in which attacks occurred. An effect of sumatriptan cannot be ruled out, but it seems unlikely, since arousal modifications involved REM sleep. Alternatively, we may speculate that REM sleep in the acute bout of the cluster is associated with a condition of hypoarousability. Also, the reduced motor activity during sleep shown by actigraphy in the cluster period is consistent with hypoarousability. Hypoarousal is a pathogenic mechanism in migraine (22–24); impaired arousal activity during CH has been suggested (25, 26). The posterior hypothalamus has a role in the arousal mechanisms, since it projects directly to the cerebral cortex, and its stimulation elicits a cascade of arousal responses, including cortical activation, motor activity and sympathetic responses (27). The posterior hypothalamus also plays a crucial role in the inhibitory control of the executive mechanisms of paradoxical sleep (28, 29).
In conclusion, we believe that in our patient the CH induced an acute, transient, global modification of sleep regulation, at several levels. These levels included the circadian sleep–wake rhythmicity, the macrostructural pattern of NREM/REM ultradian cycle, and also the sleep microstructure, that is to say the dynamics of arousal; this latter abnormality selectively involved REM sleep. All the observed modifications are consistent with an abnormal function of the posterior hypothalamus.
