Abstract
Aim
To address whether, in patients with chronic migraine and medication overuse headache, mindfulness-based treatment is associated with changes in plasma levels of catecholamines and elusive amines that are similar to those observed in patients undergoing pharmacological prophylaxis.
Methods
In this non-randomized, clinic-based effectiveness study, patients aged 18–65, with a history of chronic migraine ≥ 10 years and overuse of triptans or non-steroidal anti-inflammatory drugs ≥ 5 years, were enrolled. Upon completion of a structured withdrawal program, patients received either pharmacological prophylaxis or six weekly sessions of mindfulness-based treatment and were followed for 12 months. Daily headache diaries were used to record headache frequency and medication intake; catecholamines (noradrenaline, epinephrine and dopamine) and levels of elusive amines were assayed from poor platelet plasma.
Results
Complete follow-up data were available for 15 patients in the pharmacological prophylaxis-group (14 females, average age 44.1) and 14 in the mindfulness treatment-group (all females, average age 46.4), and all variables were comparable between groups at baseline. At 12 months, significant improvement (p < .001) was found in the pharmacological prophylaxis group for headache frequency and medication intake (by 51% and 48.7%, respectively), noradrenaline, epinephrine and dopamine (by 98.7%, 120.8% and 501.9%, respectively); patients in the mindfulness treatment-group performed similarly. For elusive amines, no longitudinal changes were found.
Conclusions
The similar improvement trends observed in the two groups of patients further support the utility of mindfulness-based treatment in migraine care, and reinforce the hypothesis that alteration and normalization of tyrosine metabolism are implicated in migraine chronification and in remission of chronic migraine.
Keywords
Introduction
Chronic migraine (CM) is often accompanied by medication overuse headache (MOH): It usually develops from episodic migraine and results in a marked impact on patients in terms of disability and reduced quality of life, and on societies in terms of cost (1–3). Abnormalities connected to genetic and metabolic factors, the presence of comorbidities, lifestyle and the overuse of symptomatic medications have been implicated in migraine chronification (4–6). Abnormalities of tyrosine metabolism have specifically been found in patients with episodic and chronic migraine (7). Tyrosine is the amino-acid precursor of dopamine and noradrenaline and of elusive amines (EAs); for example, octopamine, synephrine and tyramine. EAs are present in the hypothalamus, the limbic system and some nuclei of the brain stem (8). Catecholamines, EAs and trace amine associated receptors (TAARs), the function of which is regulated by dopamine and EAs, are deemed to play a role in pain threshold regulation as well (9).
Treatment of CM-MOH requires appropriate prophylaxis and withdrawal of the overused drugs (4,5,10). Pharmacological treatments are often supplemented by behavioral approaches: Among them, mindfulness is an emerging treatment for migraine headache (11), and its effectiveness was similar to that of pharmacological prophylaxis in reducing headache frequency, medication intake, depression, anxiety, headache impact, and levels of interleukin-6 and neutrophils in patients with CM-MOH (12,13).
In this study, we assessed whether treatment with pharmacological prophylaxis and mindfulness alter plasma levels of catecholamines and EA, in addition to clinical improvement. Our exploratory hypothesis is that any such outcomes might be similar in response to both treatment options, pointing to similar mechanisms of action.
Methods
This was a prospective pilot exploratory study in which subsequent analyses were performed on additional data collected in a prior published study (12). In this manuscript, the focus is on the course of tyrosine metabolism and is based on a reduced number of patients. The study was approved by the Institute’s Ethical Committee and written informed consent was obtained from each patient prior to enrollment.
Patients
Details on procedures and selection criteria are reported in Grazzi and colleagues (12). In brief, eligible patients had a diagnosis of CM-MOH; that is, code 1.3 – Chronic Migraine with associated 8.2 – Medication Overuse Headache as defined by the International Classification of Headache Disorders III edition (ICHD-3 beta) (1). Patients were enrolled between February 2014 and June 2015 at the Headache Centre of the Neurological Institute C. Besta in Milan, Italy. Patients ranged in age from 18–65 years and had a history of CM lasting for at least 10 years and overuse of triptans or non-steroidal anti-inflammatory drugs (NSAIDs) for at least 5 years. Pregnancy, psychotic disorders and personality disorders, determined on the basis of clinical history and psychiatric evaluation, were exclusion criteria.
Procedure
All patients underwent a 5-day structured withdrawal program in a day-hospital setting, which consisted of intravenous steroids and ademetionine (14). Prior to discharge, they were informed of the possibility of participating in a new clinical trial, in which they could receive pharmacological prophylaxis (MED group) only, chosen on the basis of clinical history and medical comorbidities such as is provided in routine care (15,16), or mindfulness training only; that is, absent any specific pharmacological prophylaxis (MT group).
Mindfulness training was provided in small groups (5–6 patients) that met weekly for six sessions, each of approximately 45 minutes’ duration, guided by an experienced neurologist trained in mindfulness practice on the basis of the original model proposed by Kabat Zinn (17). These patients were additionally encouraged to supplement their training with regular home self-practice, of 7–10 minutes per day.
Patients in both groups were encouraged to engage in moderate physical activity (i.e. 45 minutes twice per week of aerobic exercise), remain well hydrated, consume three meals per day, and maintain a regular sleep/wake pattern with at least 7–8 hours of sleep per night.
Our limited resources, in terms of personnel and space, combined with the fact that the majority of patients attending our center resided outside our district, rendered us unable to implement random assignment. Participation was thus voluntary, with patients self-selecting their preferred treatment condition. Those choosing MT were informed of the importance of being available to attend weekly sessions on a consistent basis upon discharge. Those that were unable or unwilling to commit to the stated schedule were invited to enter the MED group. The neurologist who guided the mindfulness sessions was one of the authors (LG) and underwent extensive training in mindfulness, and certain aspects were not blinded: In particular, the investigators knew who attended mindfulness sessions and who did not. To limit possible source of bias with clinical outcomes, all follow-up evaluations were carried out by another neurologist (DD). For all the above-mentioned considerations, our study should be considered as an effectiveness trial (18).
Patients were followed up for 12 months and clinical assessments are herein presented for baseline, 6 and 12 months. During the study period, patients were instructed to continue with the treatment they preferred (i.e. prescribed pharmacological prophylaxis or 7–10 minutes/day of mindfulness practice) and were encouraged to restrict use of acute medications to headaches with “severe pain”; that is, those rated as 8 or greater on a 0–10 scale (0 = no pain to 10 = pain as bad as it could be). In these instances, patients were instructed to take Eletriptan (40 mg) and/or Almotriptan (12.5 mg) as the first-line treatment, indomethacin (50 mg) as the second line, and were specifically urged to avoid opioids.
Patients recorded headache frequency and consumption of acute medications (NSAIDS as well as triptans for severe headaches) in daily headache diaries, in which they reported the number of pills used in each headache episode. The total amount of single intakes was recorded, irrespective of the kind of medication.
Blood samples
Blood samples were taken at baseline, 6 and 12 months for all patients. Peripheral venous blood (20 ml) was drawn from the antecubital vein at 09:00 from participants after 10 minutes of resting in the supine position. For the 6 - and 12-month follow-ups, patients were required to be headache free for the prior 24 hours; those not meeting this requirement or developing a headache at the time of the follow-up visit in the hospital were rescheduled to be seen at a later date.
Blood was collected into tubes with citrate as an anticoagulant and centrifuged at 3500 rpm for 15 minutes to obtain poor platelet plasma. One ml of plasma was used to measure the levels of catecholamines (noradrenaline, epinephrine and dopamine) and of EAs (octopamine, synephrine and tyramine), expressed as pg/ml. All analyses were performed using high-performance liquid chromatography as previously described (19).
Statistical analysis
Data are expressed as means and standard deviations for continuous variables, with frequencies and percentages being used for categorical variables.
Attrition was handled through complete cases analysis as indicated by the European Medicine Agency Guidelines for exploratory studies (20). Following the suggestions by Gupta (21) and Hollis and Campbell (22), we opted not to perform any intention to treat analyses because of the lack of randomization. Dumville and colleagues have similarly recommended that researchers avoid statistical testing in designs such as ours because the imbalance of confounding variables might bias the results (23). Consequently, we provide only descriptive statistics for attrition rate.
The association between treatments and potential adjustment variables were tested by odds ratios and Fisher’s exact test for overused medications and gender, and unpaired t-test for age and CM duration. A stratified analysis for the two treatment groups was performed. Generalized estimating equation (GEE) models (24,25) for repeated measures were applied in order to assess the differences in clinical and biological markers among individuals across time and treatment. For each outcome, we fitted a GEE model where time – considered as a categorical three-level factor, i.e. baseline (t0) as reference, at 6 (t1) and 12 months (t2) – treatment (i.e. MT or MED, with MED as the reference), and their interaction terms were “focus” predictors. For headache frequency and medication intake outcomes, Poisson GEE models were fitted to manage their counting nature and minimize over-dispersion. For other outcomes, Gaussian GEE models were fitted by using “log” link, an exchangeable correlation structure, and sandwich robust estimation to consider model misspecifications. GEE models were chosen for their robustness to missing values and because they allow management of the intra-subject variability produced by three separate measurements carried out on the same patients (n = 29 × 3 = 87 observations, but only 29 independents) (26).
The models’ time parameters (βT) were exponentially transformed by achieving a more interpretable average variation rate (vrT) (as the multiplicative factor) of the outcome across time (from baseline) in the MED group. In addition, exp(βT)-1 allowed us to obtain the outcome expected percent variation (Δ%T) until the time point (from baseline) in the MED group. The exponential of the parameters associated to treatment (βTR) were interpreted as a baseline average variation rate (vrTR) between the two treatments (the MED group was the reference); analogously, exp(βTR)-1 allowed us to obtain the outcome average percent variation (Δ%TR) between MT and MED groups at baseline. Finally, the interaction parameters (βTxTR) transformed with exponential minus one (i.e. exp(βTxTR) -1) were interpretable as average percent variation of the MT group rate compared to the MED group (Δ%TxTR) after 6 months or 12 months. Wald tests and 95% confidence intervals were carried out in order to evaluate statistical significance on model parameters. Two-tailed p-values < 0.05 were considered significant. Calculations were carried out using the statistical software R, version 3.3.2 and its package geepack (27).
Results
Fifty patients met the inclusion criteria during the study period, but six declined to participate; thus, 44 patients were enrolled, with 22 in each group.
Figure 1 shows the flow of patients in each of the two groups: Three left the study, providing no reason, two left for concomitant health conditions and, in addition to these patients, another 10 patients arrived too late to have blood samples collected (after 09:00 at our headache center). Therefore, we had complete 12-month follow-up data (diary plus blood samples) available for 15 patients in the MED group and for 14 in the MT group (but reduced to 13 for octopamine, synephrine and tyramine, due to corruption of the blood sample during its preparation for EAs analysis).
Flowchart of the study.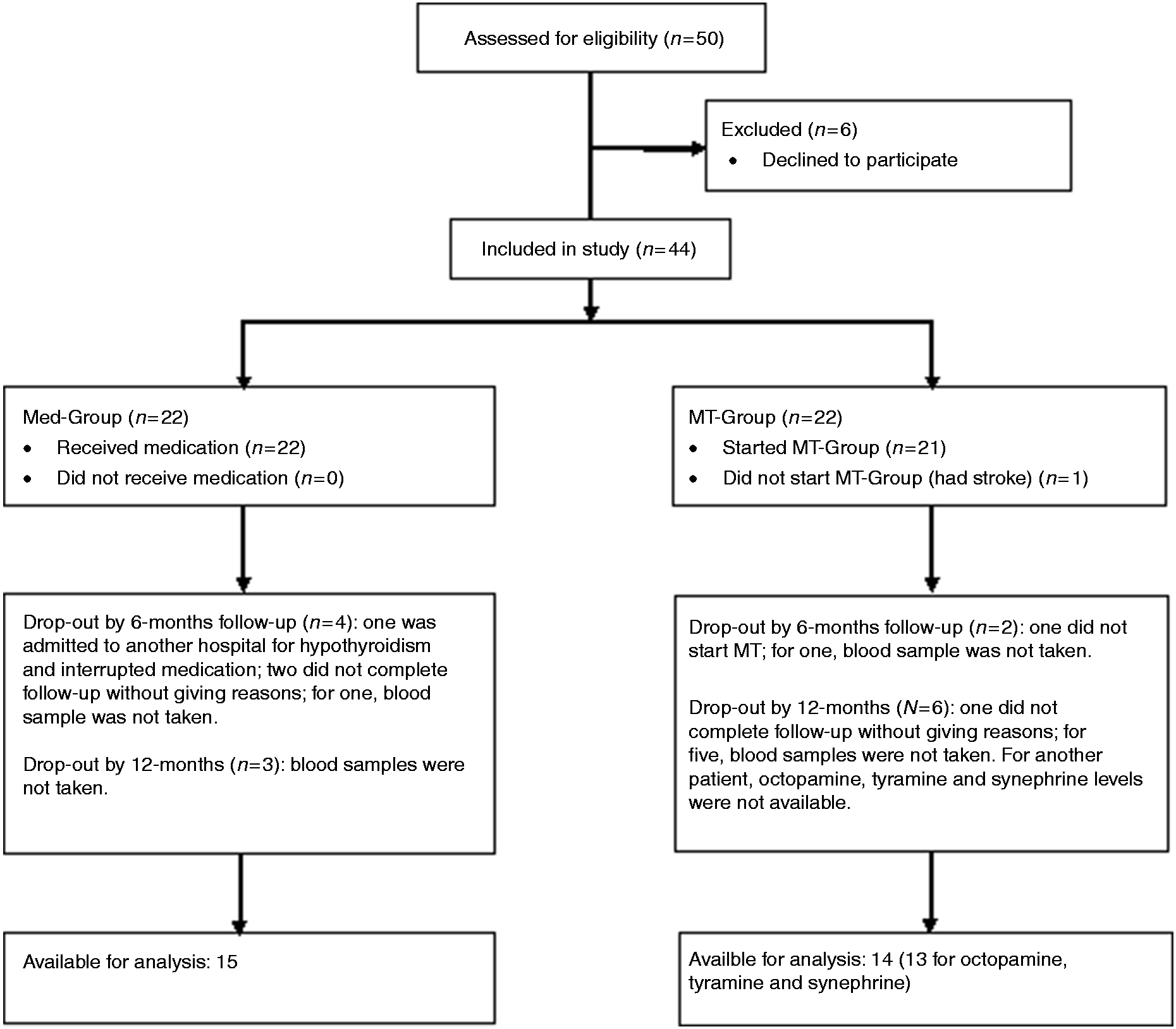
Key descriptive statistics for the two treatment conditions at baseline.

Notes: Categorical variables are expressed as frequencies and percentages; continuous as means and standard deviations; catecholamines and EAs are expressed as pg/ml. Octopamine, synephrine and tyramine levels were available for only 13 patients in the MT group.
Patients in the MED group were prescribed the following: Magnesium valproate to three patients (in one case combined with amitriptyline); onabotulinumtoxin-A to six patients; amitriptyline chlorhydrate to three patients (in one case combined with propranolol chlorhydrate); and pizotifen to three patients. When examining the relation between treatment and potential adjustment variables, only NSAID overuse was found to be significantly related (OR = 13.3, p = .042), but we did not include it in the final GEE models because the results were robust.
Averages comparison and modelling
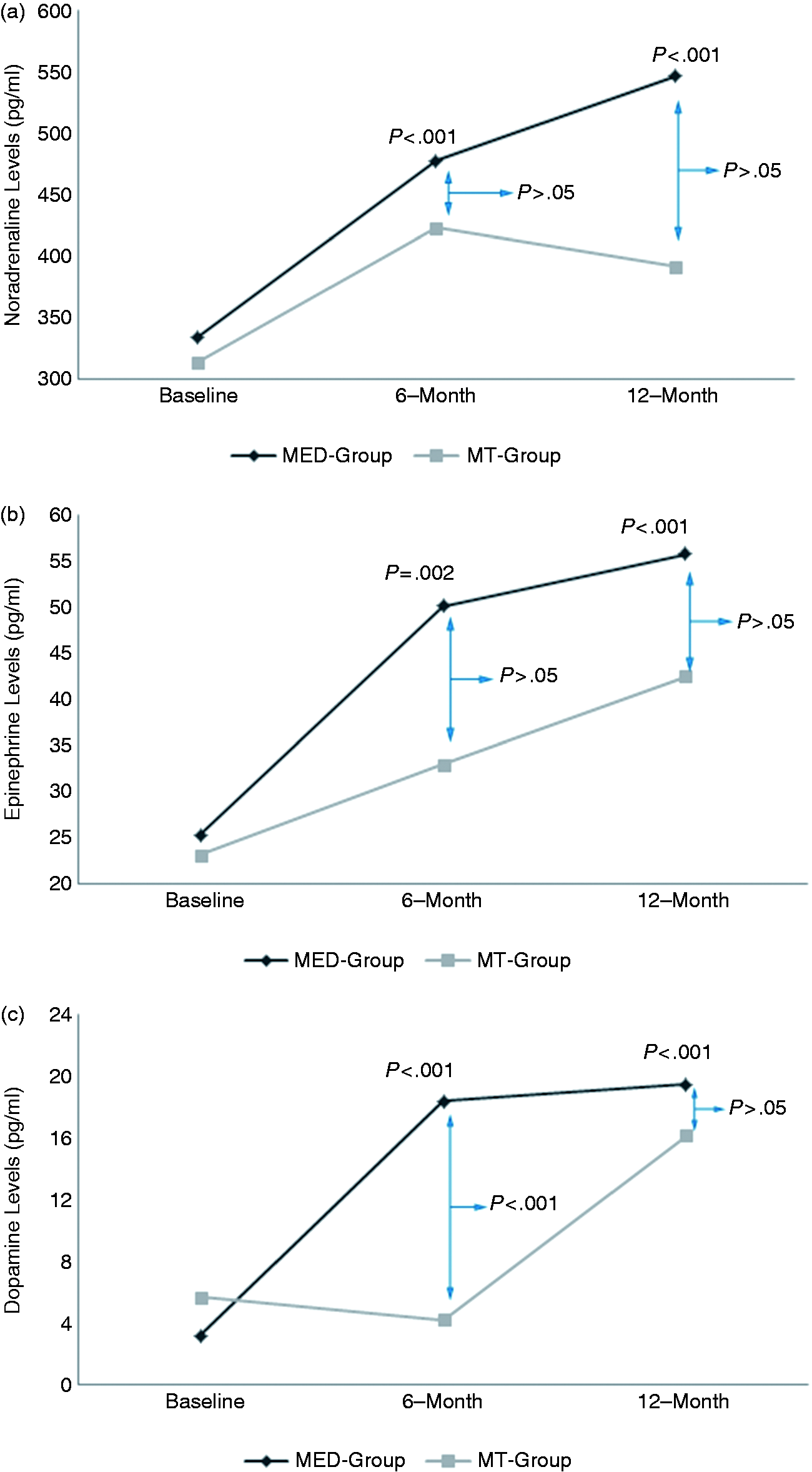
No baseline differences were found between the two treatments for any variable. Figure 2 reports the interaction mean plots for headache frequency and medication intake across time and treatments, and Figure 3 reports the same information with reference to noradrenaline, epinephrine and dopamine.
Interaction mean plots of headache frequency and medication intake across time and treatment. (a) Headache frequency; (b) medication intake. n = 15 in the MED group; n = 14 in the MT group. Interaction mean plots of catecholamines across time and treatment. (a) Noradrenaline; (b) epinephrine; (c) dopamine. n = 15 in the MED group; n = 14 in the MT group.
Headache frequency (Figure 2(a)) decreased significantly in the MED group, by 42% at 6 months and 51% at 12 months (both p < .001), and by 33% and 21%, respectively, in the MT group. Medication intake (Figure 2(b)) decreased significantly in the MED group by 36% at 6 months and 49% at 12 months (both p < .001), and by 40% and 31%, respectively, in the MT group. Although for both these clinical outcomes a lower level of improvement was observed at the 12-month follow-up among patients in the MT group, no significant differences were found at each time point against patients from the MED group at appropriate statistical evaluation.
Noradrenaline and epinephrine levels (Figures 3(a) and 3(b)) significantly increased by 43% and 99%, and by 63% and 121% (all p < .001), at 6 and 12 months, respectively, in the MED group. At the same follow-up evaluations, these levels among patients in the MT group increased by 35% and 25% (noradrenaline) and by 42% and 81% (epinephrine), with no differences being found between patients in the MED group and MT group at either follow-up period. With regard to dopamine (Figure 3(c)), a significant increase was observed for patients in the MED group at 6 and 12 months (by 468% and 502%, respectively, p < 0.001). In contrast, for patients in the MT group, dopamine levels decreased by 26% at 6 months, and increased by 175% at 12 months. At 6 months, levels of dopamine were different between the two groups (p < .001), while at the 12-month follow-up the two groups were not different (p = .19).
With regard to EAs levels, none of the observed fluctuations were significant at either follow-up period nor between the two treatment conditions. See Supplementary Files for detailed GEE results.
Clinical variables, catecholamines and elusive amines across time by treatment.
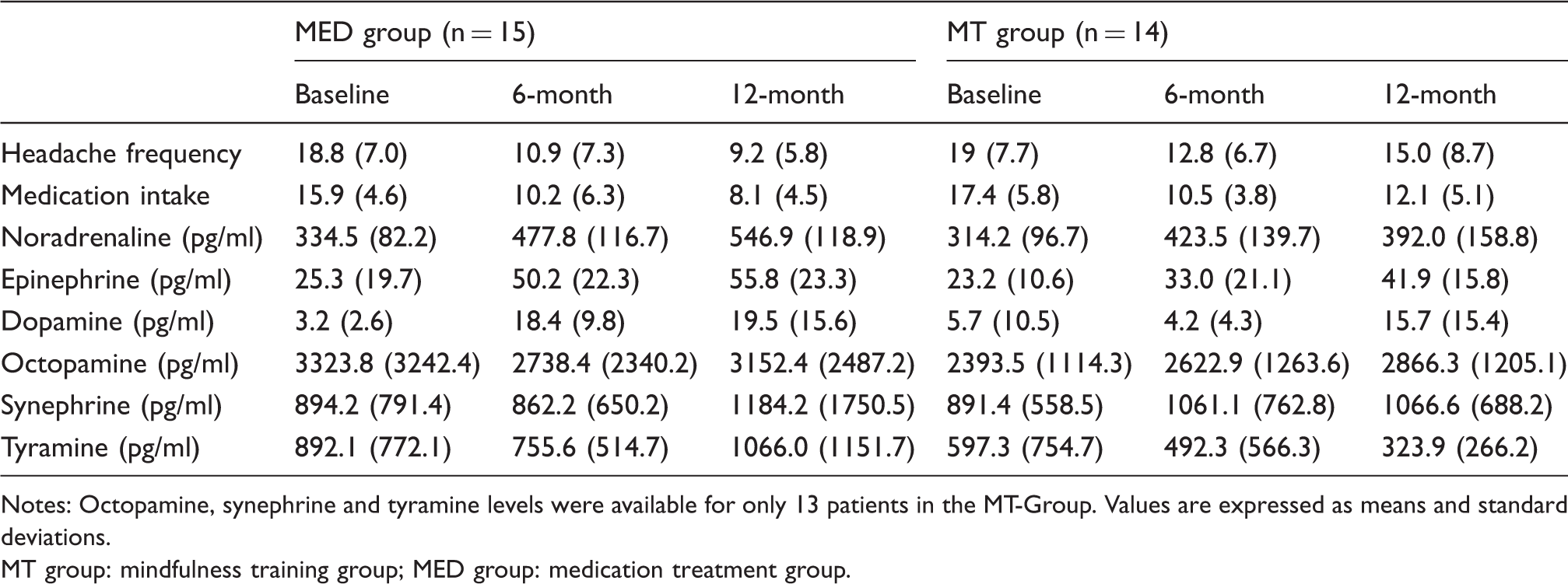
Notes: Octopamine, synephrine and tyramine levels were available for only 13 patients in the MT-Group. Values are expressed as means and standard deviations.
MT group: mindfulness training group; MED group: medication treatment group.
Discussion
This study showed that catecholamine plasma levels significantly increased in patients with CM-MOH who underwent a withdrawal protocol and were then treated with pharmacological prophylaxis, and that showed a significant reduction of headache frequency and intake of medications. Moreover, this study shows that the trend at 12 months was similar in those receiving mindfulness-based training only, both for clinical variables and for catecholamine plasma levels. The number of patients receiving each pharmacological treatment was too small to address the potential different effects on catecholamine levels exerted by each different drug. What we note is that the changes in catecholamine levels at 12 months in patients receiving MT were similar to those in the MED group as a whole, which suggests that these changes may be independent from the therapeutic approach. Thus, our results point out a novel observation, namely that the improvement of patients with CM-MOH, maintained over 12 months, is also accompanied by significant changes in key biomarkers of disease activity; that is, plasma levels of catecholamines, which are known to be abnormal in such patients (7,28).
Although the two groups were not significantly different at both follow-up time-points at appropriate statistical evaluation, we observed a lower level of improvement among those in the MT group at the 12-month follow-up with regard to headache frequency and in medication intake. Our hypothesis is that, in the long course, some patients might not have followed the indication of regular self-practice; this, however, was not assessed, thus highlighting a possible problem of persistence with the treatment that has also been reported with oral preventive medications (29).
The neuromodulators (EA) and the neurotransmitters (catecholamines) play an important role in the physiology of CNS and in the pain matrix, specifically in the pathogenesis of migraine and its chronification (9). In fact, it has been shown that patients with CM are characterized by an overactivity of the tyrosine decarboxylase enzyme with abnormal synthesis of EA and of catecholamines (7,28,30). Our pilot study showed that within 12 months of day-hospital withdrawal from overused medication (and a brief trial of treatment), the synthesis of catecholamines increased in patients receiving pharmacological prophylaxis, and that the trend was similar in those receiving mindfulness-based training only, while the levels of EAs remained stable. This suggests that both pharmacological and behavioral approaches have a similar effect on the regulation of tyrosine metabolism that, as previously mentioned, is deemed to play a role in the pathogenesis of migraine and in its chronification (7,28,31).
Catecholamine synthesis depends on the activity of tyrosine hydroxylase. The increase of plasma levels of noradrenaline, epinephrine and dopamine found in our patients may be due to an increase in the activity of this enzyme. In turn, this may reflect what happens in CNS pathways involved in migraine mechanisms, given that dopamine and noradrenaline in plasma derive from the spillover of the autonomic endings around the pial vessels and are considered markers of catecholamine synthesis in the hypothalamus. Of note, tyrosine hydroxylase requires an efficient energy supply, which is reduced in migraine patients who are known to have impaired mitochondrial functioning (32). We postulate that the studied interventions may stimulate a restoration in energy supply in the CNS areas involved in the complex processes leading to migraine chronification, with an activation of the tyrosine hydroxylase activity, and consequently of catecholamine levels. Assuming this is so, it is important to note that the metabolic changes found in our study were evident not only after pharmacological prophylaxis but also after a behavioral treatment; that is, mindfulness.
Tyramine levels at 6 months appeared slightly reduced, indicating a possible increased action of tyrosine hydroxylase after both treatments, consistent with the increased values of catecholamines. However, the distribution of these variables was too dispersed and prevents us from drawing any strong conclusion. So, we can hypothesize that the modest fluctuations of octopamine and synephrine levels in the plasma of our patients might be due to the normal fluctuation of enzyme activity, dopamine β-hydroxylase (DBH) and phenylethanolamine –N-methyltransferase (PMNT) respectively, that regulate the synthesis of the elusive amines. It has, however, to be remembered that this was an exploratory study and the results obtained for elusive amines cannot explain the role of mindfulness in their metabolism. Further investigation is needed to ascertain it.
The reason why dopamine plasma levels did not increase in the MT treated group at 6 months is unclear, and we can only make some hypotheses. We know that migraine patients have a reduction in the metabolism of cellular energy in different tissues, show impaired mitochondrial functioning and reduced activity of tyrosine hydroxylase (31). Research on the biological effects of mindfulness is relatively new and no studies have yet looked at its effect on catecholamines and biogenic amines or relative enzyme functions. To explain the different dopamine response in the MT group, we hypothesized that mindfulness therapy could act to increase mitochondrial energy in a slower way than pharmacological treatment, and consequently it needs more time to modify the activity of the tyrosine hydroxylase enzyme that transforms tyrosine into dopamine.
The mechanisms explaining the efficacy of mindfulness in migraine headaches are not fully clear. Neuroimaging data have shown that mindfulness induces a reduction in the activation of the amygdala, hippocampus, emotional/evaluative regions of the prefrontal cortex and the contralateral primary somatosensory cortex, as well as an increased activation of the midcingulate and anterior cingulate cortex, thalamus, and insula (33–35). Our results provide further evidence of the possible biological effects of mindfulness, as they show a change in those biomarkers of pain processing that, in the human brain, are located in the same subcortical structures where catecholamines, EAs and TAARs are commonly found (7,8,36). Moreover, recent studies provided new insights into how mind-body approaches influence biological responses at the molecular level (37): The authors found enhanced expression of genes associated with energy metabolism and mitochondrial function, and differences in adaptive responses in short-term and long-term practitioners. More research is needed to specify the biological target of the intervention of mindfulness and to confirm our hypothesis.
Certain limitations need to be taken into account when interpreting our results. First, our sample size was small and somewhat selective. Although our patients attended a specialty center and, as expected, had quite a long disease course, we excluded from participation those with major psychiatric comorbidities, and did not find any common comorbidity of CM, such as depression or hypertension. Further, no a priori computation for power was performed. Second, this was a non-randomized study, with patients deciding whether to participate or not in mindfulness training: Thus, the motivation to commit to the 6-week training period might have played a confounding role. Third, we were unable to assess the level of adherence and persistence to either pharmacological treatment or mindfulness home self-practice during the follow-up period, or to fully monitor use of rescue medications (i.e. no counts of pills from a pre-defined box). Consequently, we relied only on self-reported information on intake of medications used for pharmacological prophylaxis, as well as on the use of acute/abortive medications. Fourth, for practical as well as ethical reasons, we opted not to include a no-treatment or placebo control group and recognize this may limit the degree to which we are able to fully evaluate the clinical relevance of changes in catecholamine levels. Fifth, catecholamines and EAs were measured in peripheral blood, and the possibility exists that the observed changes may not fully reflect actual changes at the CNS level: This remains an open hypothesis.
In conclusion, the results of our prospective exploratory study pointed out a novel observation: 12 months after withdrawal from medication overuse and a brief course of outpatient treatment, the patients treated with pharmacological prophylaxis alone and mindfulness-based training alone not only experienced similar improvement in relevant clinical outcomes; that is, headache frequency and acute medication intake, but also showed a similar change in the plasma levels of noradrenaline, epinephrine and dopamine. Overall, these results add to the growing body of literature supporting the utility of mindfulness for treating the serious and disabling condition of CM-MOH. They also shed light on the possible biological mechanisms of action of pharmacological prophylaxis and, in turn, indicate the importance of pursing further research on the possible biological effects of behavioral approaches. Furthermore, our findings serve to reinforce the hypothesis of a major role of tyrosine metabolism alterations in migraine and in its chronification process, as postulated based on previous research findings.
Clinical implications
Altered plasma levels of catecholamines and elusive amines have been observed in patients with CM-MOH. Our study provides support for patients treated with pharmacological prophylaxis or mindfulness-based treatments alone (i.e. absent any prophylaxis) improving with regard to levels of catecholamines, headache frequency and medication intake by 12 months in a comparable way. Our results reinforce the hypothesis of a major role of tyrosine metabolism alterations in migraine and in its chronification process and shed light on the possible biological mechanisms of action of pharmacological prophylaxis as well as of behavioral approaches.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
