Abstract
Background
The use of frontal infrared thermography in the diagnosis of primary headaches provided scattering results due to measurement fluctuations and different types of headaches or research protocols.
Objective
This study aims to assess the reliability of frontal infrared thermography in healthy individuals and provide a preliminary evaluation in chronic migraine patients using a commercial infrared thermal camera.
Methods
Thermographic images were acquired in 20 controls and 15 patients at three consecutive time-points in two daily sessions. The Side Difference and Asymmetry Index parameters were defined. The reproducibility of the measurements, the correlation of Asymmetry Index and Side Difference with clinical evaluations and patient perceptions, and the ability of the parameters to discriminate between patients and controls were investigated.
Results
We reported a good reproducibility of the measurements (Inter-class Correlation Coefficient > 0.75 and Coefficient of Variation < 13.4%), independent from external factors. The Side Difference was significantly different between patients and controls ( p < 0.001). The Asymmetry Index showed good correlation with the side of unilateral pain ( p = 0.0056).
Conclusions
Frontal infrared thermography can be used to quantify the difference between the right and the left side of frontal vascular changes in chronic migraine patients, provided that standardized conditions are satisfied.
Abbreviations
Asymmetry Index
Analysis of variance
Between-subjects mean square
Coefficient of Variation
Residual mean square
Frontal Infrared Thermography
Intra-class correlation coefficient
Within-subjects mean square
Transcranial direct current stimulation
Visual Analogue Scale
Introduction
Infrared thermography detects infrared light emitted by the human body to visualize changes in heat due to abnormalities in the skin surface blood flow of diseased areas. This non-invasive and non-radiative imaging technique has different clinical applications including the detection of circulatory and/or inflammatory disorders such as rheumatoid arthritis, Raynaud's disease or osteoarthritis of the knee (1). It has also been demonstrated that thermography is able to capture thermal gradient in facial areas characterizing healthy individuals (2).
However, the literature investigating the use of external carotid region (forehead) thermal imaging for the characterization of vascular headaches has not reached consistent results due to the different types of headache evaluated, different timing of patients' evaluation (during headache attack or in the headache-free interval), different technologies used for image acquisition, and different methods for imaging or statistical analysis.
In 1986, Swerdlow and Dieter (3), comparing electronic thermography between 275 headache patients and 45 controls, defined the “cold patch” as a region of the face where the temperature is cooler (less than 0.5 ℃) than the surrounding areas (3,4). The presence of the cold patch is more frequent in vascular headaches than in healthy subjects or patients with tension type headache, psychogenic headaches or post-traumatic headaches.
There is an open question of whether the cold patch is a “fixed” entity (5) or whether it decreases with therapy (6,7). In fact, whereas according to Swerdlow and Dieter “the vascular cold patch is independent of prognosis and is most likely a permanent element of a vascular headache sufferer's facial thermal pattern” (5), in 1991, Dalla Volta et al. (7) suggested that the “cold patch” in vascular headache patients is ipsilateral to the prevailing side of pain and that the cooler area decreased after 6 months of prophylactic therapy. The differences observed by the two groups may be explained in terms of experimental protocol and patient selection, which introduced a higher variability in the cold patch response (7).
Anyway, there is large consensus about the fact that the cold patch represents an asymmetry in the forehead of migraine patients. Unfortunately, the location of the cold patch is seldom related to the side of the pain (5), probably due to the variation of temperature during headache attack or due to the lateralization of the headache (unilateral or bilateral) as measured by Drummond and Lance (8,9).
Finally, FIT, in conjunction with nitroglycerine administration, was suggested as a novel non-invasive approach to study vascular processes underlying headaches (10).
Taken together, all these studies demonstrate that FIT can be used as a diagnostic tool in migraine with and without aura, in cluster headache, and in other headache types.
In the light of previous observations and of new possible therapeutic applications, the present study has two primary end points: (a) to evaluate the reliability of FIT measurements in controls and chronic migraine patients using a modern commercial infrared thermal camera; (b) to verify whether FIT-based parameters correlate both with the visual evaluation of FIT by an expert clinician and with the patient's perception of pain side (at least in the case of unilateral pain).
Materials and methods
Subjects
Thirty-five right handed volunteers (26 females and nine males) with a mean age of 35 ± 11.6 years (range: 20–55) were enrolled at the Headache centre of the Fondazione IRCCS Istituto Neurologico Nazionale Casimiro Mondino. All the subjects were not medicated at the time of testing.
Fifteen of the 35 volunteers (three males) suffered from chronic migraine with medication overuse, as assessed according to the IHS classification (11). They were examined while hospitalized and during a washout period from analgesics or other symptomatic treatment, including non-headache medication. Patients were not on dietary/smoking restrictions ahead of measurements. Patients did not use preventive treatment and underwent daily parenteral detoxification (saline, cyanocobalamin, folic acid, nicotinamide, glutathione, delorazepam and metoclopramide on demand). Patients were assessed on the second or third day after symptomatic medication withdrawal that started on the day of hospital admission. No symptomatic medication was allowed during the evaluation period except a local ice bag (at least one day apart from the examination). Hence, patients were generally with headache during the examination. Pain severity was evaluated before each examination on a 0–3 score Visual Analogue Scale (0: No pain; 1: Mild pain; 2: Moderate pain; 3: Severe pain) with a mean value of (mean ± SE) 1.88 ± 0.078.
The remaining 20 of the 35 volunteers were used as controls since they suffered from migraine or tension type headache no more than 1–2 times a year.
The study was approved by the institutional review board (Ethical Committee of the Fondazione IRCCS Istituto Neurologico Nazionale Casimiro Mondino, date of approval: 29 July 2013) and conformed with the Declaration of Helsinki. All patients signed written informed consent before participating in the study.
Frontal infrared thermography (FIT)
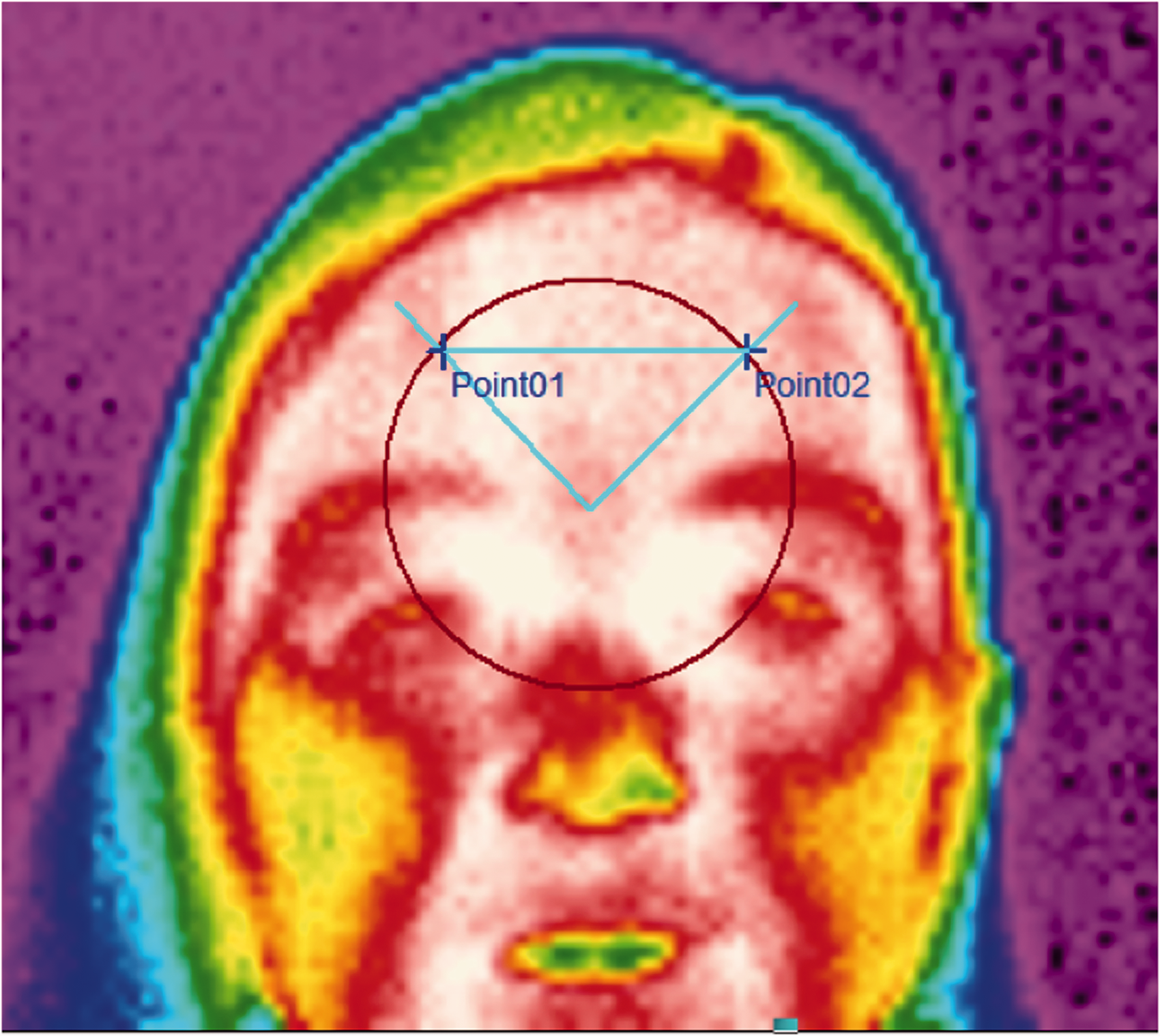
FIT was assessed with a modern infrared thermal camera (model LT3, produced by Zhejiang Dali Technology Co. Ltd) characterized by a thermal sensitivity below 0.08 ℃ at 30 ℃, according to suggested guidelines (12,13). FIT measured the spatial distribution of the heat over the human face (object emissivity 0.98): The camera was placed at distance of 1 m from the subject in a room with stable temperature (23.6 ± 1.57 ℃). To ensure comparability between patients and controls, since controls do not have a “cold area”, the temperature was evaluated in two target points (left and right side) in the frontal polar sites. In patients, we identified the coolest point in the side showing the cold patch, and then we identified its symmetric on the other side of the head (equidistant from the nasion, Figure 1). In controls there is no cold patch, so we used two symmetric points equidistant from the nasion on a radius of 2 cm (Figure 2). This protocol allowed repeatability and comparability among subjects.
Measurement point identification in patients. The radius of the circle was calculated as the distance between the nasion and the coolest point in the cold patch. Once the radius was defined, the second point was the point equidistant from the nasion and with the same vertical coordinate as the first point of measurement (the symmetric point). Measurement point identification in controls. The radius of the circle was constant.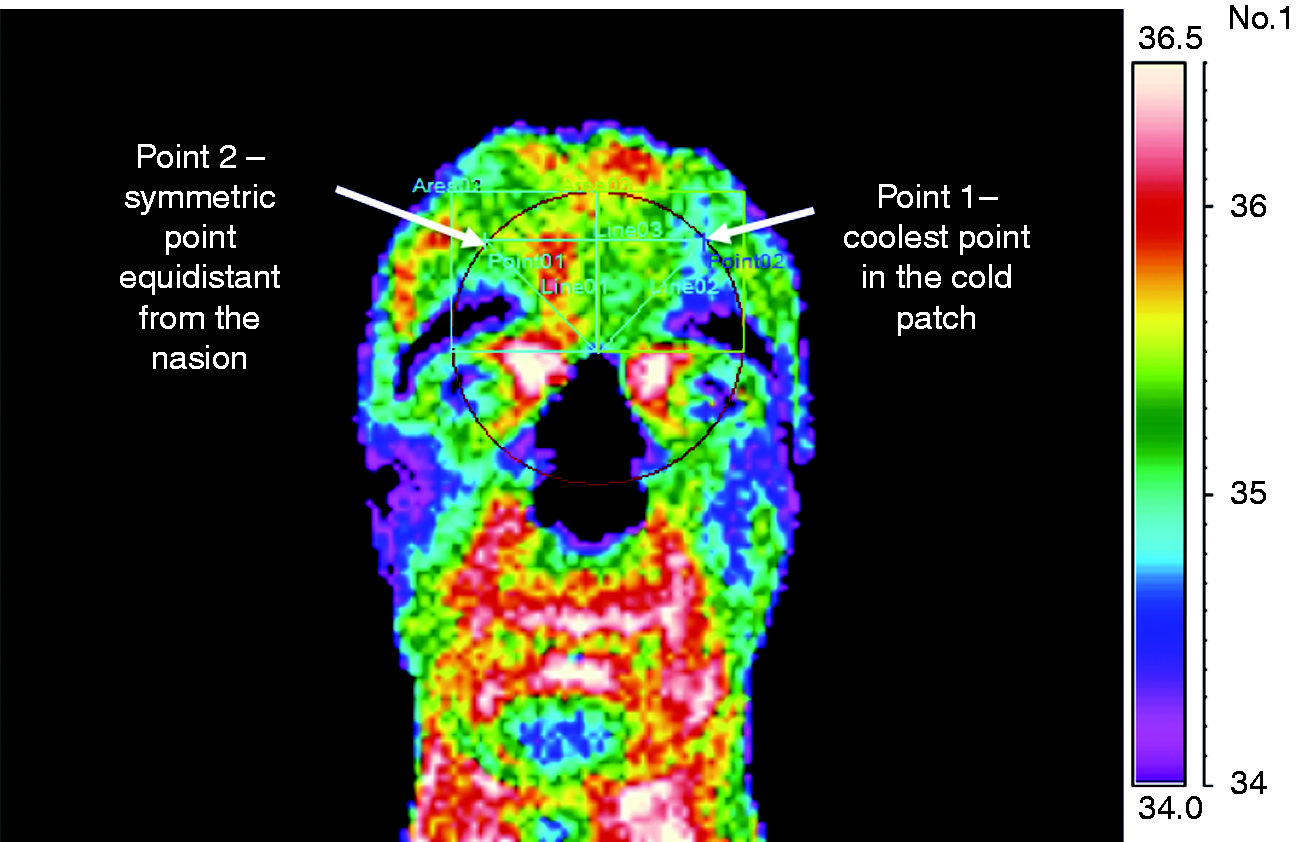
Study design
All measurements were taken in two test sessions (T1 and T2) for all subjects. The second session (T2) was run at least one day apart in order to evaluate the intra-subject variations in FIT. During each session (T1 and T2), the measurements were repeated three times (m1, m2, m3) by the same experimenter after 10 minutes of rest between each measurement. Room temperature was recorded during each session. Images were taken approximately at the same time of day in each patient (10–12 am or 2–4 pm).
Data analysis
Reliability of FIT
In order to verify whether measurements were influenced by external factors, correlation analysis was computed with respect to room temperature, sex and age of subjects, both at T1 and at T2, using m1, m2, and m3 as dependent variables.
Student's paired t test was used to determine whether three measurements belonging to the same session (m1 vs. m2, m2 vs. m3, m1 vs. m3) had the same mean. The Bonferroni correction for repeated measures was applied (p < 0.016).
Reliability of measurements was investigated using the intra-class correlation coefficient (ICC), defined as the fraction of variance that is caused by the variation between subjects. Thus, if the variance between tests is smaller than the variance between subjects then ICC is close to 1. According to Fleiss, ICC values above 0.75 generally mean “excellent” reliability (14). The intra-class correlation coefficient (ICC) was calculated for FIT at each point: Data during a single session (T1 and T2) were analysed using a two factor analysis of variance (ANOVA) without replication (factors: Subjects and FIT readings mi) and the values from the ANOVA table were substituted into equation (1):
The reproducibility of the method was tested by calculating the Coefficient of Variation (CV) during the first test session (16,17). For each comparison between measurements (m1 vs. m2, m2 vs. m3, m1 vs. m3) the CV (equation 2) was calculated as the absolute value of:
In order to evaluate the intra-subject variations in FIT, a two-way analysis of variance with replication (2-way ANOVA) was performed for a statistical comparison between two different test sessions (T1 and T2).
Characterization of images comparing controls and patients
In order to verify the difference between migraine FIT measurement and healthy subject FIT measurements, we defined two parameters:
The Asymmetry Index (14) (equation 3)
where Tleft is the temperature of the left forehead and Tright is the temperature of the right forehead. AI was calculated in order to assess the lateralization of FIT. If the temperature measurement on the two sides is the same, then the asymmetry index is equal to 0. A positive AI value means that the cold patch is located on the right side of the forehead. Conversely, a negative asymmetry index means lateralization on the left side.
The absolute value of percentage difference between left and right side
Side Difference (equation 4) is the difference between left and right side as a percentage of the average temperature of the forehead.
Both parameters were calculated from all the measurements of each patient (15 × 6 = 90 observations) and controls (20 × 6 = 120 observations). A comparison between the two groups was conducted in order to test whether there was a significant difference of AI or Side Difference. In headache patients, the AI was compared with FIT visual inspection by the doctor and with declarations of the patient before each session (the side of pain and intensity of pain using VAS scores).
Results
Reliability of frontal infrared thermography
FIT readings did not correlate with external factors during both during T1 and T2 sessions as far as sex (Tright r = 0.20, p = 0.24; Tleft r = 0.23, p = 0.17), room temperature (Tright r = 0.22, p = 0.20; Tleft r = 0.19, p = 0.27) and age (Tright r = −0.017, p = 0.92; Tleft r = 0.035, p = 0.83) is concerned.
During each session, paired t test analysis revealed no significant difference between consecutive FIT measurements during the same session ( p > 0.016) for all the three comparisons (m1 vs. m2, m2 vs. m3, m1 vs. m3) in both the right and left measurement side (Figure 3).
Mean FIT measured over 20 controls (left side) and 15 patients (right side) in three consecutive readings (m1, m2, m3 – x axis) with 10 minutes of rest between readings during T1 and T2. Error bars represent standard errors.
The reliability analysis demonstrated “excellent” (14) results during T1 (ICC values: Right side, 0.73; left side, 0.81). The same result was confirmed during T2 (ICC values: Right side, 0.80; left side, 0.74).
Regarding the reproducibility, mean CV between consecutive FIT measurements in the right and left side were similar in controls (T1: Left side: 1.63% ± 1.79; right side: 1.69% ± 2.09; T2: Left side: 2.71% ± 3.26; right side: 2.67% ± 3.22) and in patients (T1: Left side: 3.77% ± 3.57; right side: 3.87% ± 3.62; T2: Left side: 2.71% ± 3.26; right side: 2.67% ± 3.22). However, in patients, the CV range was slightly greater than in controls both in T1 and T2 session (patients: 0.00–13.40%; controls: 0.00–10.04%). In addition, the average smallest CV was observed among m2m3 measurements (m2m3: 1.56% ± 0.62 vs. m1m2: 3.13% ± 1.29 and m1m3: 3.54% ± 1.25). Despite this, the overall reproducibility of the measurements was very good because the maximum CV was less than 13.40%.
When examining intra-subject variations between two different days (inter-test), CV represents the percentage difference between two daily sessions (T1 and T2). The maximum CVs between test sessions (T1 and T2) were very low (patients range: 0.19–8.24%, controls range: 0.00–7.56%) thus suggesting a good reproducibility of the measurements over different days.
The range of temperature was not different between days or between the two groups of subjects (controls: T1: 33.10–36.73 ℃; T2: 34.13–36.70 ℃; patients: T1: 32.30–37.60 ℃; T2: 32.30–36.17 ℃). Patients were characterized by a larger standard deviation of temperature than controls (Figure 3). The two-way ANOVA with replication over 35 subjects demonstrated that there were no significant differences between FIT measurements on different days.
Characterization of FIT comparing patients and controls
In controls, the temperature measurements during T1 varied from a minimum of 32.60 ℃ to a maximum value of 38.90 ℃ (mean on the two sides: 35.05 ℃, standard deviation: 1 ℃, corresponding to 2.8% of the mean value). In patients with chronic migraine, the temperature measurements during T1 varied from a minimum of 31.90 ℃ to a maximum value of 38.30 ℃ with a larger standard deviation from the mean than controls (mean on the two sides 35.47 ℃, standard deviation 1.59 ℃, corresponding 4.5% of the mean value).
Whereas average AI was not different ( p > 0.05) between patients (−0.00002 ± 0.0164) and controls (−0.00118 ± 0.0091), the Side Difference, representing the absolute value of the difference between temperature measured in the left and right side, significantly discriminated controls (mean ± Standard Deviation (SD): 0.73% ± 0.55) from patients (mean ± SD: 1.37% ± 0.87, p-value one tail < 0.0001) (Figure 4).
Difference between left and right side as a percentage of the average temperature on the forehead (Side Difference) in patients (n = 20) and controls (n = 15).
Conversely, AI can be used to locate the cold patch in patients. In fact, AI polarity (positive or negative, where positive AI value means that the cold patch is located on the right side of the forehead) was in consort with the visual inspection of the FIT by the examiner (correlation, r2 = 0.7, p = 0.00072). In addition, when the patient referred to a bilateral pain before the test session, both the visual inspection and the AI index revealed an asymmetry in the forehead, thus suggesting that the AI can be a reliable index for the localization of the cold patch also when the patient's perception is not reliable.
Finally, in patients with unilateral pain, the correlation between AI and the patient-referred pain side before FIT was good (r2 = 0.6, p = 0.0056). Conversely, even though patients were examined mainly during attacks with different pain severities, as measured by VAS, the correlation between AI and pain severity was very low (r2 = 0.2, p = 0.37).
Discussion
In this study, we examined the usability and reproducibility of FIT measurement in patients with headache and in healthy subjects, in a research scenario in which previous experiences using FIT were not conclusive (1,3,4,6,7,18).
In order to obtain comparable results between patients and controls, we used a thermal punctual evaluation instead of an evaluation by area. In fact, whereas patients showed a cooler area (the cold patch), controls did not, making evaluation by area unreliable and not reproducible. We therefore chose to evaluate the temperature in two symmetric points equidistant from the nasion. In patients, one point was the coolest point in the cold patch, and the second was its symmetric; in controls, we took two symmetric points located on a circle of fixed radius. With this setup, our data showed that FIT is a reproducible tool provided that standard location and standard measurement procedures are carried out. In fact, we found good reproducibility of the measurements within the same session and between the two sessions, as measured by CV. However, we also found the smallest CV in the comparison between the second reading and the third reading during each test session, while the first reading, even though statistically not different from the others, cannot always be reliable during thermography. Since the first reading was taken immediately when the patient entered the room, it is likely that, after some minutes, there is some stabilization of the subject at the room temperature that affects the absolute measurements. This observation suggests that a stabilization period of the subject at the room temperature is recommended before the thermograph exam in order to guarantee an effective measurement. This is in line with available guidelines suggesting that thermography measures should be taken after a stabilization period of 15–20 minutes to allow equilibrating with the environment (12,13).
Our data also suggest that FIT is a reliable procedure in detecting the cold patch location in headache patients and that the AI parameter was able to localize the cold patch even when the patient referred to a bilateral pain before the image was recorded. In previous studies, no correlation was found between FIT measurements and pain side or VAS score in patients (5,18). Instead, our results demonstrate that the AI defined by Kurth et al. (19) well describes the lateralization of the cold patch in patients. Moreover, the AI correlates with the doctor's visual inspection of FIT, and in the case of unilateral pain there is a good correlation between AI and pain side. Even if the correlation between AI and VAS score is low, the AI could be a useful tool to localize the pathological side (where the cold patch is located) in each patient. Our results further confirm that, regardless of the patient's perception, the cold patch can be viewed as a “unilateral” entity, representing the neurochemical imbalance between the two sides in terms of microcirculation in the facial district. This hypothesis is in line with neurophysiological studies on Visual Evoked Potentials showing a prominent laterality of neurophysiological signatures in headache (20–24). Therefore, the AI estimated by FIT can have a relevant prognostic role going beyond a patient's perception.
Finally, the Side Difference parameter was able to discriminate headache patients from controls, even if the FIT image was taken with patients experiencing different pain intensities.
These observations suggest that FIT can be a useful neurophysiological aid in the diagnosis for pain medicine. An add-on marker in diagnosis may be useful in difficult cases or when there is a risk of diagnostic mistakes concerning unilateral or bilateral headache form (26). We foresee a time when FIT could be used in evaluating pharmacological and non-pharmacological treatment in headache and other facial neuralgias: FIT with a commercially available camera is a relatively inexpensive procedure that can be used in headache and pain centre settings.
However, even though promising, our results were obtained in a relatively small number of subjects, and acquisitions followed a shared protocol that guaranteed consistency across subjects. Therefore, for future applications, it will be important to create a consensus procedure for FIT acquisition, able to support diagnosis.
From a practical viewpoint, in the current therapeutic scenario where non-invasive neuromodulation techniques are increasingly used for the treatment of migraine (21–23,25–27), these results can reach an even more interesting meaning. In fact, transcranial direct current stimulation (tDCS) has been proposed for the treatment of migraine (27,28,30,31). tDCS is a non-invasive technique that acts sub-threshold ( <1 V/m vs. 100 V/m produced by other supra-threshold techniques (32)), producing an excitability change in the area below the electrode. Even though the electric field induced by tDCS is spread over neighbouring areas, the electrode montage is a crucial element in the optimization of tDCS therapy (29). In migraine, electrodes are usually placed in the frontal area, according to patient's perception and subjective analysis. Conversely, the cold patch, representing a vasoconstriction area characterizing the migraine, may be an effective target for neuromodulation intervention. In migraine patients, the cold patch corresponds to a vasoconstriction within the external carotid territory, which represents the end result of the haemodynamic changes due to the activity of the autonomic and the trigeminovascular systems. In this case, cathodal tDCS over this area may reduce the pathological frontal asymmetry. For this reason, the automatic localization of the cold patch using FIT based parameters, as proposed in this study, can be applied for the optimization of tDCS treatment in migraine patients.
Taken together, our results suggest that FIT could be useful as a diagnostic tool to localize the cold patch and to study unilateral headache without side shift like cluster headache, paroxysmal hemicrania, SUNCT or cranial neuralgias (trigeminal neuralgia). However, the case series is relatively small in order to evaluate the sensitivity and specificity of FIT in discriminating between different headache entities. In addition, even though at present there is no strong evidence supporting the change of FIT after a pharmacological or non- pharmacological treatment, the use of tDCS or prophylactic agents (beta blockers, calcium antagonists) shows a tendency to reducing or disappearing FIT asymmetry, thus suggesting the use of FIT also as a prognostic tool.
Conclusions
In conclusion, in respect to previous studies, this work introduces the possibility of using a modern commercial infrared thermal camera for the analysis of frontal thermography in migraine patients. Moreover, two new parameters (AI and Side Difference) provided meaningful results, one being able to localize the cold patch and the other able to distinguish patients from controls.
Footnotes
Article highlights
The use of Frontal Infrared Thermography (FIT) in the diagnosis of primary headaches is still debated.
In this study, we showed that FIT measurements have good reproducibility, even when using commercial cameras, in both patients and controls.
We defined a new parameter, the Side Difference, which is able to discriminate patients with headache from controls.
We defined another parameter, the Asymmetry Index, which correlates with the side of pain.
FIT can be used to characterize vascular changes in chronic migraine patients, but standardized recording conditions are needed to guarantee reliability.
Institutional Review Board Approval
The study was approved by the institutional review board (Ethical Committee of the Fondazione IRCCS Istituto Neurologico Nazionale Casimiro Mondino, date of approval: July 29th 2013) and conformed with the Declaration of Helsinki. All patients signed written informed consent before participating to the study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Elena Rossi, at the time of the work, was employed by Newronika Srl, a spin-off company of the Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico and of the University of Milan. Sara Marceglia is founder and shareholder of Newronika Srl. The other authors declare no conflict.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.




