Abstract
Background
On-demand stimulation of the sphenopalatine ganglion (SPG) by means of an implantable neurostimulation system has been shown to be a safe and effective therapy for treatment-refractory cluster headache patients. Our objective was to estimate changes in cluster headache medication cost observed in SPG-treated chronic patients.
Methods
Detailed patient-level data of 71 chronic patients treated with the Pulsante® SPG Microstimulator System were available from the Pathway R-1 Registry through 12 months’ follow-up. We used utilization data of preventive and acute medications reported at baseline, 3, 6, 9, and 12 months to estimate annualized drug costs for SPG-treated patients and compared it to baseline. Cost estimates for all drug/dosage combinations were developed based on German medication prices for 2016.
Results
In the base case analysis, mean annual acute and preventive medication costs decreased from €14,178 to €6924 (−€7254; −51%), and €559 to €328 (−€231; −41%), respectively, leading to total estimated annual drug cost savings of €7484, 97% of which were attributable to acute medications.
Conclusions
Our analysis suggests that SPG stimulation for the treatment of chronic cluster headache is associated with pronounced reductions in cluster headache medication usage that might lead to sizable annual savings in medication costs.
Keywords
Introduction
Cluster headache (CH) is a severe primary headache disorder, known to be highly disabling and severely compromising patients’ quality of life (1,2). It is associated with intense unilateral pain attacks up to eight times a day, each lasting between 15 and 180 minutes (3). Usually, CH occurs in episodes, but in a minority of about 15% of patients (4), CH attacks do not remit for a minimum of 4 weeks within one year and are then defined as chronic (3,5).
Current treatment of chronic cluster headache (cCH) follows two objectives: (a) to effectively treat an attack in a short timeframe, and (b) to prevent the onset of future attacks. While medical management for acute cCH attacks typically includes the use of oxygen inhalation, subcutaneous or intranasal application of triptans, or intranasal lidocaine, preventive medication strategies include high dosages of verapamil as first choice, and alternatively or concurrently lithium, topiramate, melatonin, and long-acting triptans. Occipital nerve blocks or steroids are also commonly used as transitional prophylactic treatments (6,7).
The cost of these medical treatment strategies is substantial and further contributes to an already high economic burden of the disease, stemming from disability, increased absence from work and frequent outpatient visits (1,8). Medical treatment costs alone were estimated to amount to more than €12,000 per year based on analysis of a German cluster headache cohort (8).
Stimulation of the sphenopalatine ganglion (SPG) by means of an implantable neurostimulation system (Pulsante® SPG Microstimulator System, Autonomic Technologies Inc., Mountain View, CA, USA) has been introduced in recent years as a novel minimally invasive treatment option that has been shown to be a safe and effective approach for the treatment of cluster headaches (9–12). In case of a CH attack, the patient can switch the device on via remote control, and in many cases can stop the attack effectively. For example, Jürgens et al. (13) report significant acute pain reductions as early as 15 minutes after switching on the device, a result that was achieved in 67.1% of all treated attacks. In that study, 68% of patients experienced a clinically significant improvement through achieving significant pain relief in case of an acute attack, while also experiencing a ≥50% reduction in attack frequency. Over 24 months follow-up, 65% of acute attacks were successfully treated with SPG stimulation, leading to alleviation or absence of pain, with the latter accomplished in 50% of all attacks (13).
The long-term cost-effectiveness of implantable SPG stimulation has been studied previously by means of a model-based health-economic analysis based on the Pathway CH-1 study (14). That study found SPG stimulation to add meaningful gains in health-related quality of life at a cost profile that rendered SPG stimulation a cost-effective treatment for analysis horizons longer than three years, and cost saving and therefore dominant for analysis horizons longer than 5 years.
The objectives of the current study were to analyze changes in acute and preventive medication use observed in subjects treated with SPG stimulation in the real-world Pathway R-1 Registry, and to estimate from these changes the overall difference in annual medication cost, using German medication cost data as the basis for the analysis.
Materials and methods
Cost calculations were based on patient-level drug utilization data from patients followed in the Pathway R-1 Registry, and published medication cost data from the German healthcare system for the year 2016. Annualized acute and preventive medication costs were computed based on drug utilization reported at baseline, and 3, 6, 9, and 12-month follow-up.
Clinical and drug utilization data
Clinical and utilization data were collected in May 2016 from data collected in the Pathway R1 Registry. That study is a post-market registry of the Pulsante® SPG Microstimulator System conducted to confirm the therapy’s safety and long-term effectiveness in a larger population. The registry enrolled patients who met CH criteria per the device’s CE regulatory labeling – clusters lasting a minimum of 16 weeks. The exclusion criterion was changes in preventive medication in the month prior to enrollment. A total of 12 European sites (10 in Germany, one in Denmark, one in Austria) enrolled patients. All patients were required to give written consent, and local ethics committee approval was obtained as required.
In addition to safety and effectiveness data, the registry collected detailed usage data of acute and preventive medications. These were collected retrospectively at each follow-up clinic visit, with patients asked to report their average weekly attack frequency and acute and preventive medication usage for the past 4 weeks.
Cohort characteristics of included subjects.

Drug cost data
For each of the study-reported medications, drug cost information was sourced from the official published year 2016 pharmaceutical prices for Germany (Rote Liste – German Red Book). Where information was not included in the official record, online searches were conducted to determine the respective drug’s current sales price in German online retail (searches conducted in June 2016). Oxygen prices per application dose were determined based on registry-reported flow in L/min and an assumed flow duration of 10 minutes per attack (in line with current guideline). Throughout, the lowest identifiable cost was assumed for each drug and dosage, relying on the least expensive medication where several products existed and choosing the largest and therefore most economic package size as the basis for computations.
Cost assessment
Changes in medication cost under SPG stimulation were determined based on assessed medication utilization recorded during follow-up, and subsequently comparing the corresponding cost to the baseline medication cost. All costs were computed on an annualized basis. For the baseline comparator, this was accomplished by multiplying the determined weekly cost by 52 weeks per year, assuming that the drug utilization collected at baseline is a representative sample that would be maintained in a cohort that would not choose to pursue SPG stimulation treatment.
For patients receiving SPG stimulation, four data points on drug utilization were available – weekly drug utilization reported at 3, 6, 9, and 12 months follow-up. Where data for select follow-up visits were not available, we imputed these values assuming the average of the prior and following reported drug usage. This was the case for one follow-up visit in 10 of the patients, and for two follow-up visits in one patient.
In order to compute an annual cost estimate from these data points, several analytical approaches were developed and implemented, as follows:
Approach 1: Using the weekly utilization reported at 12 months as the basis for computation of an annual medication cost. Approach 2: Computing estimated weekly cost for all 52 weeks of the year, by using a linear approximation between baseline, 3, 6, 9, and 12 months. These 52 weekly cost estimates were added to obtain an annual cost estimate. Approach 3: The same as Approach 2 but only using the period between 6 and 12 months as the basis for computation (i.e. 26 weekly inputs based on linear approximation, multiplied by two). Approach 4: The same as Approach 2 but only using the period between 9 and 12 months as the basis for computation (i.e. 13 weekly inputs based on linear approximation, multiplied by four). Approach 5: Computing the annualized cost under the assumption that each of the reported weekly utilizations at 3, 6, 9, and 12 months is maintained for a total of 13 weeks.
For the base case analysis, we opted to choose the most granular analysis approach, which relies on the full set of collected data points (Approach 2), with results of the other approaches shown in comparison.
For reference, the baseline annualized mean medication costs for those subjects excluded from the analysis were also computed and compared to the respective baseline costs of the 71 patients included in our study.
Statistical analyses and computations
All statistical analyses and calculations were performed using Excel 2016 (Microsoft Corporation, Redmond, WA, USA) and Stata 14 (Stata Corporation, College Station, TX, USA) software packages.
We performed paired t-test evaluations to assess the statistical significance of the changes in acute and preventive drug costs between baseline and the annualized estimate under SPG stimulation treatment.
In addition, we computed the weekly average attack frequency reported for the cohort at baseline, 3, 6, 9, and 12 months, and the percent changes in attack frequency, acute medication cost, and preventive medication costs, respectively, compared to their baseline values. Also, we analyzed whether annualized cost savings correlated with the baseline attack frequency.
Further, annualized acute medication costs at baseline and 12 months were compared by type of acute medication to provide perspective on the relative contribution of each medication category toward the total savings.
Finally, the annual estimated medication cost under SPG stimulation treatment was taken as the basis for approximation of multi-year savings that might materialize. This additional analysis provides useful insight to compare our findings to prior cost-effectiveness evaluations of SPG therapy and to estimate the possible amortization timeframe required for healthcare payers to recover the upfront cost for an SPG neurostimulation device and its implantation procedure of approximately €32,000, per the earlier cost-effectiveness study (14). For these multi-year scenarios, we computed discounted (at 3% p.a.) and undiscounted medication cost savings under the assumption that the computed 1-year results would be maintained in subsequent years. In addition to this steady-state assumption, the effects of a reduction and of an increase in annual savings were explored in sensitivity analyses.
In line with guidelines for health-economic assessment, all base case calculations considered mean costs. In addition, median costs were calculated for reference.
Results
Average baseline annualized medication costs were €14,737 ± 18,918, with a median cost of €6061. As indicated by the difference between mean and median and the sample’s standard deviation, annualized costs – based on patient-reported drug utilization – varied substantively, with costs ranging from €0 in three patients reporting neither preventive nor acute medication usage to €97,904 in one patient reporting extensive use of acute medications (see baseline of patient-specific annualized costs in Figure 1(a)). Corresponding calculations for the excluded subjects yielded baseline mean medication costs of $15,702, approximately 7% higher than the computed mean of the study population.
Individual computed annual medication costs (acute and preventive) at baseline, 3, 6, 9, and 12 months, based on reported weekly utilization reported at baseline and follow-up visits (a). Corresponding percent changes in mean attack frequency, acute medication cost, and preventive medication costs, as compared to baseline, for the full cohort.
At 3, 6, 9, and 12 months, annualized mean medication costs were €5928 ± 12,668, €5229 ± 12,442, €4098 ± 8151, and €6662 ± 13,711. The median annualized drug costs were €1745, €713, €353, and €1092. Figure 1(b) shows the relative change in attack frequency, acute medications, and preventive medications, compared to baseline.
Computational approaches explored to obtain estimate of total cost difference in annual medication use. Approach 2 uses the most granular computational approach and was therefore chosen as the base case.

The mean change in annual estimated total drug cost amounted to €7484 ± 14,574, with median drug cost reduction of €3002. Figure 2(a) shows the individual patients’ changes in total drug cost on SPG stimulation compared to baseline, and the resulting distributional box plot (Figure 2(b)). Correlation analysis did not suggest a relationship between baseline attack frequency and amount of medication cost savings (linear regression R2 = 0.03; see Supplementary Materials for detail).
Computed changes in total annual medication cost in the studied 71 patients. (a) Individual per-patient results. (b) Boxplot representation for total cohort. Negative values denote savings compared to baseline.
Both baseline drug costs and drug costs on SPG stimulation treatment were heavily driven by acute medications. In total, a drug cost reduction of 51% was estimated, based on estimated 51% reduction in acute medication cost and 41% reduction in preventive medication cost. Ninety seven percent of total savings resulted from reductions in acute medications (see Figure 3). Within acute medications, sumatriptan, zolmitriptan, and oxygen contributed the highest costs, with 75.0%, 17.2%, and 7.4% of the total cost, respectively, at baseline, and 72.3%, 13.8%, and 10.0%, respectively, at 12 months (see Supplementary Materials for further detail).
Computed total annual medication cost at baseline and on SPG therapy (using most conservative computational approach) and resulting percent changes.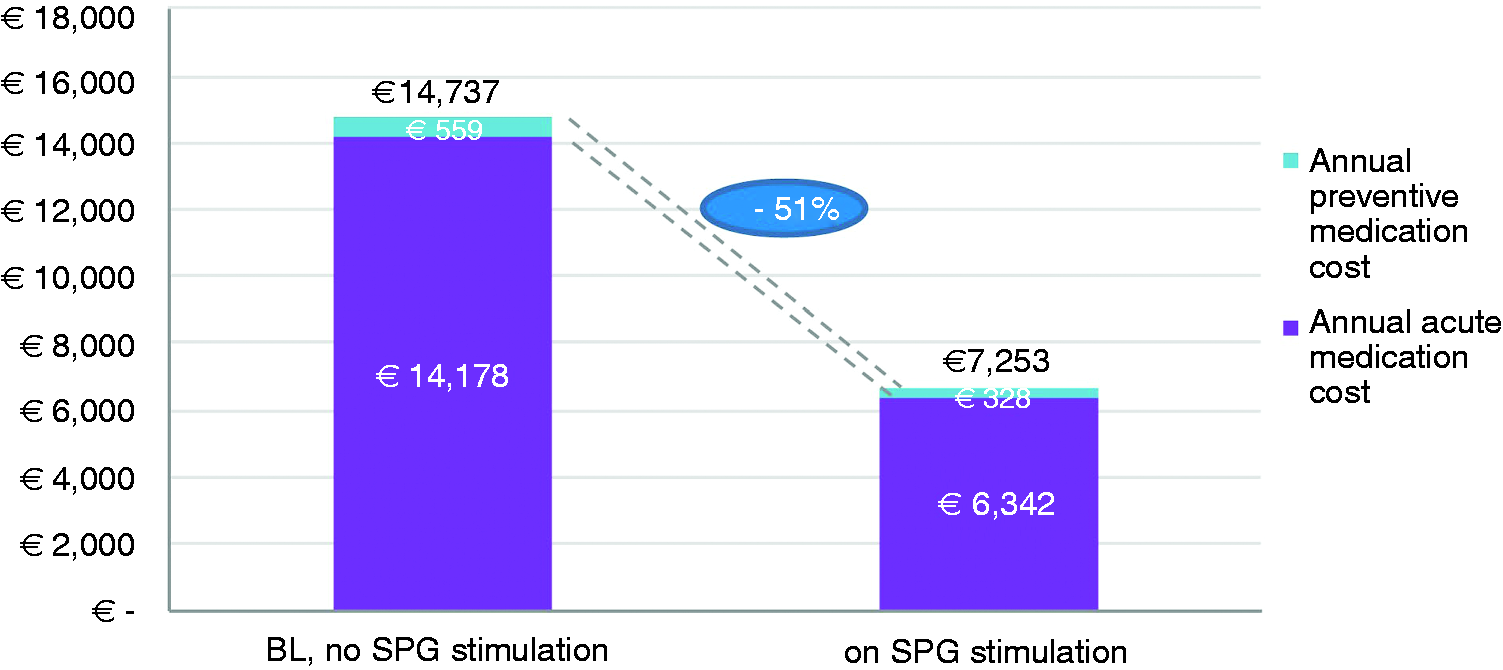
Under the assumption of maintained drug utilization levels in subsequent years on SPG stimulation treatment, 5-year undiscounted mean medication cost savings were projected to be €37,422 (discounted: €35,305). If, following the first – observed – year, drug cost savings were reduced to 75% of these initial year annual savings, 5-year undiscounted cost savings would amount to €29,937. If year 2 and following year drug cost savings were 25% higher than first-year drug-cost savings, the 5-year undiscounted cost savings, conversely, would amount to €44,906.
Discussion
Our study, based on real-world data from the Pathway R-1 Registry, provides, for the first time, detailed insight into medication patterns and adjustments that might be expected over time in patients treated with SPG stimulation. We found drug utilization in stimulation-treated patients to be substantially reduced as compared to baseline, with statistically significant annual reductions in acute and preventive medication costs averaging around €7500 per patient, or more, depending on the analysis approach.
These findings compare relatively closely with the results of a prior costing study of chronic cluster headache patients in Germany, which found attack-aborting drug costs to amount to approximately €12,400 per year, for a cohort with similar attack frequency of 3.8 attacks per day (8). Of note, that prior study focused solely on sumatriptan, zolmitriptan, and oxygen costs to estimate acute cost.
From a clinical perspective, our analysis revealed pronounced subject-to-subject variation in medication use in a subset of the cohort. Despite these sometimes extreme variations in a subset of patients, which are not fully unexpected given the nature of cluster headache disease, the large majority of patients observed a predictable reduction in the need for medication, both in acute and preventive medications. These findings are in line with data collected in the 24-month follow-up of the CH-1 study (13), which reported that 79% of acute attacks were treated without acute medications, and 64% of patients reported a positive development of preventive medication use at 24 months.
Prior clinical studies have reported on the effectiveness of SPG stimulation. These prior findings are in line with the observations in our study, namely a reduction in attack frequency of more than a third, a reduction in acute cost by about half, and of slightly less in preventive medications. As expected, our data suggest a close relationship exists between therapy effectiveness and achievable savings in medication costs.
The current analysis confirms and further corroborates the findings of the earlier cost-effectiveness analysis of SPG, which found SPG stimulation to be a cost-effective treatment strategy in the German healthcare system, as compared to current drug-based therapy (14). Of note, that earlier study found SPG stimulation to be cost-effective for analysis horizons of more than three years, and cost saving for analysis horizons just above 5 years post implantation. The findings of our current study, when adopting the same SPG implantation cost of approximately €32,000, suggest overall cost savings for the healthcare system might materialize in an even shorter period of less than 5 years, after which the cost of SPG implantation would be fully amortized (in this simplified calculation, not considering potential costs for SPG stimulation-related physician visits for programming, though).
Our study is subject to several limitations. First, our analysis was limited to inclusion of registry patients who reported medication usage data through 12 months follow-up. This criterion was chosen to ensure the longest follow-up possible was considered at the time of analysis, and to attenuate the effect of any shorter-term reductions that might or might not be representative of the stable long-term treatment effect. Especially for estimation of potential long-term savings, this broad data basis is desirable. At the same time, some patients might have opted out of the Pathway R-1 Registry or have been lost to follow-up, while inclusion of their early follow-up data might have led to some measured changes in our findings. However, imputation of their missing data would have introduced additional bias. Further, the fact that these excluded subjects had mean BL medication costs that were around 7% higher than the costs of included patients suggests our projected savings might err on the conservative side. On a related note, exclusion of the three included patients for whom no drug usage data were reported at baseline would have increased the projected savings, again suggesting conservatism in the current analysis. As such, our inclusion of these patients and assumption of no costs at baseline is conservative. Second, seasonal differences in disease severity, which are common in cluster headache (15) patients and might explain variation in attack frequency and drug usage, could possibly affect the analysis results. While this is true at the individual patient level, the aggregate costs for the cohort can be expected to represent a realistic annual average, as implantation dates were reasonably well distributed over the calendar year, and each patient included in the analysis was followed for a full year. Third, patients in the Pathway R1 registry were enrolled at sites across three European countries. Variation in clinical practice and prescription patterns might vary between these different healthcare systems. Fourth, some of the patients using oxygen used demand valve oxygen (DVO), where reported oxygen flow was only determined based on an estimated amount. Fifth, we maintained the registry-reported separation into acute versus chronic medications. In a few, very limited instances, individual subjects reported some prednisone, GON-block and frovatriptan use as part of their acute regimen, while these medications are more appropriately classified as preventive or transitional preventive medications. However, this does not affect the total medication cost calculations, and would have no meaningful impact on the estimated distribution of acute versus preventive medications. Fifth, because the Pathway R1 registry provides only single-arm data, we needed to rely on baseline medication use data to calculate savings, as opposed to using medication cost data from a control population. Finally, the analysis was conducted taking the perspective of the German healthcare system. Use of other countries’ drug prices will likely lead to different annual baseline medication costs and consequently also to other absolute drug savings amounts. However, the relative reductions in medication cost under SPG stimulation treatment can be expected to closely follow the results of the current analysis.
In summary, our analysis suggests that SPG stimulation for the treatment of chronic cluster headache is associated with pronounced reductions in cluster headache medication usage that might lead to sizable annual savings in medication costs in the German healthcare system. Further studies with longer-term follow-up would be desirable to confirm these findings.
Supplemental Material
Supplemental material for Changes in medication cost observed in chronic cluster headache patients treated with sphenopalatine ganglion (SPG) stimulation: Analysis based on 1-year data from the Pathway R-1 Registry
Supplemental material for Changes in medication cost observed in chronic cluster headache patients treated with sphenopalatine ganglion (SPG) stimulation: Analysis based on 1-year data from the Pathway R-1 Registry by Jan B Pietzsch, Simon A Weber, Nunu Lund and Charly Gaul in Cephalalgia
Footnotes
Article highlights
SPG stimulation therapy is associated with reductions in acute and preventive cluster headache medications.
In the German healthcare system, these reductions lead to cost reductions of more than half, averaging €7484 in savings per year.
More than 95% of these total savings are attributable to reductions in acute medications.
Decreased expenditures for acute and preventive medications stem from effective treatment of acute attacks and a reduction in attack frequency under SPG stimulation treatment.
While absolute monetary savings will differ from country to country based on respective drug prices, our study findings can be expected to directionally apply across healthcare systems.
Acknowledgments
The authors would like to thank all investigators of the Pathway R-1 Registry and the study sponsor Autonomic Technologies Inc. for providing access to the drug utilization data collected in the Pathway R-1 Registry. Further, the authors would acknowledge Dr. Benjamin Geisler of Wing Tech Inc. for reviewing and providing comment on statistical analyses.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JBP is president, CEO, and shareholder of Wing Tech Inc., a health economics consultancy. Wing Tech Inc. received consulting fees from Autonomic Technologies Inc. to conduct the health-economic analyses and model development underlying this study. NL has been a sub-investigator in the register studies on SPG neurostimulation. She has received financial support for her doctoral studies from the Danish Tryg Foundation, and travel grants from the Candy Foundation. SAW worked as a consultant to Wing Tech Inc. on this project, and through Wing Tech Inc., received consulting fees and travel support from Autonomic Technologies, Inc. CG has received honoraria for consulting and lectures within the past three years from Allergan Pharma, Ratiopharm, Boehringer Ingelheim Pharma, Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer vital, Hormosan Pharma, electroCore, Grünenthal, Reckitt Benckiser, and TEVA. He does not hold any stocks of pharmaceutical companies or medical device companies.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Autonomic Technologies Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
