Abstract
Background
The relationship between subcortical nuclei and headache is unclear. Most previous studies were conducted in small clinical migraine samples. In the present population-based MRI study, we hypothesized that headache sufferers exhibit reduced volume and deformation of the nucleus accumbens compared to non-sufferers. In addition, volume and deformation of the amygdala, caudate, hippocampus, pallidum, putamen and thalamus were examined.
Methods
In all, 1006 participants (50–66 years) from the third Nord-Trøndelag Health Survey, were randomly selected to undergo a brain MRI at 1.5 T. Volume and shape of the subcortical nuclei from T1 weighted 3D scans were obtained in FreeSurfer and FSL. The association with questionnaire-based headache categories (migraine and tension-type headache included) was evaluated using analysis of covariance. Individuals not suffering from headache were used as controls. Age, sex, intracranial volume and Hospital Anxiety and Depression Scale were used as covariates.
Results
No effect of headache status on accumbens volume and shape was present. Exploratory analyses showed significant but small differences in volume of caudate and putamen and in putamen shape between those with non-migrainous headache and the controls. A post hoc analysis showed that caudate volume was strongly associated with white matter hyperintensities.
Conclusion
We did not confirm our hypothesis that headache sufferers have smaller volume and different shape of the accumbens compared to non-sufferers. No or only small differences in volume and shape of subcortical nuclei between headache sufferers and non-sufferers appear to exist in the general population.
Background
Much is still unclear regarding the etiology and pathophysiology of headache and its subtypes. Migraine, for instance, was formerly believed to mostly be a vascular disease, but newer evidence points to a considerable neuronal component in the pathophysiology (1). Thus, it is of great interest to investigate the brain’s morphology among those suffering from headache.
The majority of the structural imaging studies of people with headache have investigated the brain’s cortex in migraineurs. A few studies have looked at subcortical grey matter and headache, mostly in small clinic-based samples of migraine patients. These studies have reported somewhat inconsistent findings with reduced volume of the nucleus accumbens (2), hippocampus (3,4) and different subnuclei of the thalamus (5), both increased and decreased volume of the caudate (2,6) and increased volume of the putamen (7). Further, the shape of thalamus, striatum and pallidum was reported to be similar in the only study of subcortical shape differences between a migraine and a control group (5).
In chronic pain conditions, sharing clinical characteristics with headache, meta-analyses have revealed somewhat similar results with altered structure of the accumbens, putamen and thalamus (8–10). Lately, increasing attention has been given to the nucleus accumbens as an important modulator of pain (10,11). One study recently found chronic pain patients to have a reduction in reward responsiveness and volume of the accumbens (12). Furthermore, a PET scan of a 36-year-old female with migraine showed reductions in µ-opioid receptor availability in the accumbens during the ictal phase (13).
The aim of the present population-based study was to investigate the association between headache (migraine and TTH included) and the morphology of the main subcortical grey matter structures (accumbens, amygdala, caudate, hippocampus, pallidum, putamen and thalamus). Firstly, analyses on the volumes of the nuclei were conducted. Secondly, to investigate whether there were regional differences in the morphology of the nuclei, shape analyses were performed. Based on previous knowledge, we hypothesized that headache sufferers, regardless of subtype, would show smaller volume and difference in shape of the nucleus accumbens compared to those not suffering from headache.
Material and methods
Ethical approval
This study was approved by the Regional Committee for Ethics in Medical Research. The HUNT study was in addition approved by the Norwegian Data Inspectorate. All participants gave their informed, written consent.
The HUNT cohort
Participants were recruited from the Nord-Trøndelag Health Surveys (HUNT), which is a general population survey of the entire population aged ≥20 years in Nord-Trøndelag county, Norway. Surveys collecting a wide range of health-related data from questionnaires and other investigations (e.g. blood samples, blood pressure) were conducted in 1984–1986 (HUNT1), 1995–1997 (HUNT2) and 2006–2008 (HUNT3).As part of HUNT3, a group of 1006 individuals (530 women), all between 50 and 65 years at the time of consent, were sampled for brain imaging with a standardized MRI protocol (HUNT-MRI). Participants that had previously participated in HUNT1, 2 and 3, and lived maximally 45 minutes away by car or public transport from Levanger hospital where the scanning was performed, were eligible for inclusion. Individuals exhibiting standard safety contraindications to MRI, that is, having a pacemaker, severe claustrophobia or body weight above 150 kg, were excluded. Details about the recruitment of participants to the HUNT-MRI study and the imaging procedure have been published previously (14,15) and a comparison of the non-invited, the non-participants and the participants of the HUNT-MRI study revealed that they were not widely different from the general population, with the possible exception of somewhat reduced cardiovascular risk factors (14).
MRI scanning
All imaging was performed on the same 1.5 T General Electric Signa HDx 1.5 T MRI scanner equipped with an eight-channel head coil and software version pre-14.0M (GE Healthcare). Scans included a T1 weighted volume, transverse T2, T2* and FLAIR sequences, a time of flight 3D angio sequence through the base of the brain and diffusion tensor imaging (DTI).
MRI analysis
The T1 weighted volumes were analysed using FreeSurfer 5.3 (http://surfer.nmr.mgh.harvard.edu/) and measurements of the volumes of the subcortical nuclei accumbens, amygdala, caudate, hippocampus, pallidum, putamen and thalamus were obtained using an automated procedure described previously (16) and quality assessed visually. The data on volume were imported into SPSS, where the statistical analyses were performed. The volumes of the different nuclei were combined for the right and left hemisphere to constitute the total bilateral volume of the nucleus.
Local shape differences in the nuclei were compared between groups using a vertex-by-vertex analysis based on FMRIB’s Integrated Registration and Segmentation Tool (FIRST) 1.2 (17). First, a surface mesh of the different nuclei in each subject was created using a deformable mesh model composed of a set of vertices. The number of vertices was then fixed so that corresponding vertices could be compared between groups (18). The surface for the right and left nuclei of each participant were then separately aligned to an average model provided by FIRST using a 6-degrees of freedom transformation. Group differences in the surface displacement maps were analyzed in FMRIB Software Library using randomize for nonparametric permutation-based inference (n = 5000) with a threshold at p < 0.05 and corrected using threshold-free cluster enhancement (19).
Intracranial volume (ICV) estimation was performed in Statistical Parametric Mapping 8 (SPM8) (rel. 5236) (http://www.fil.ion.ucl.ac.uk/spm/) using an automated version of the reverse brain mask method (RBM) (20, 21). ICV corrections using the residuals method were applied in all volume analyses (22).
Headache diagnoses
Participants of the HUNT study had answered headache questionnaires as part of both the HUNT2 and HUNT3 surveys. In both surveys, the headache questionnaires started with a screening question “Have you suffered from headache during the last 12 months?” and participants answering “yes” were classified as headache sufferers. The accuracy of being a headache sufferer was evaluated with face-to-face interviews by neurologists with special training in headache, showing a sensitivity of 85% and a specificity of 83% in HUNT2 and a sensitivity of 88% and a specificity of 86% in HUNT3(23,24).
In the HUNT3 survey, the headache sufferers were further categorized into three mutually exclusive headache categories: Migraine, TTH ≥1 day per month, and unclassified headache. The migraine and TTH diagnoses were based on the criteria of the 2nd edition of the International Classification of Headache Disorders (ICHD-II). The classification and accuracy of the questionnaire-based diagnoses have been described previously (23). For migraine, the sensitivity was 51% and specificity 95%, whereas for TTH the respective values were 96% and 69%. Headache sufferers not fulfilling the criteria of either migraine or TTH were categorized as having unclassified headache. In the present study, no specific analyses were conducted based on this group. In addition, the headache sufferers in HUNT3 were categorized into two different groups based on frequency of headache attacks (<7 days/month and ≥7 days/month).
The HUNT2 questionnaire lacked information regarding the strength of the headache and thus the diagnoses were not strictly according to the ICHD criteria. The liberal migraine diagnosis from the validation study (24) was applied, showing a sensitivity of 49% and a specificity of 96%. Individuals not fulfilling the criteria for this diagnosis were classified as non-migraineurs. The fact that 780 (78%) participants had answered the headache questionnaire in both HUNT2 and HUNT3 made it possible to classify individuals in four groups according to when they did or did not suffer from headache: Headache HUNT2 alone, headache HUNT3 alone, headache in both HUNT2 and 3, and no headache suffered in either HUNT2 or 3. The last group consisted of those answering “no” to the screening question in both surveys and was used as controls in all analyses.
Statistics
Volume and shape of the subcortical nuclei for the different headache groups were compared one-on-one to the control group, which consisted of those without headache in both HUNT2 and HUNT3. When analysing the hypothesis that headache sufferers have smaller volume and different shape of the accumbens, individuals suffering from headache in HUNT3, regardless of subtype, were compared to the control group. All other analyses were considered as being exploratory. In order to examine a dose-response relationship, analyses on the frequency of headache attacks in HUNT3 (headache <7 days/month or headache ≥7 days/month) and volume of the subcortical nuclei were performed. The analyses were corrected for age at the time of scanning (continuous), sex, and ICV. The volume analyses were in addition rerun after the Hospital Anxiety and Depression Scale (HADS) score (continuous) was added as a covariate. Data on HADS were collected from the HUNT3 survey. Further, to elucidate the possible contribution of outliers, the volume analyses corrected for age, sex and ICV were rerun after the removal of individuals deviating >1.5 × interquartile range above or below the third and first quartile respectively.
Volumes were compared using analysis of covariance (ANCOVA) with a two-tailed significance threshold set to p < 0.05. This threshold was used both when testing the hypothesis and in the exploratory analyses to minimize the risk of type II error. Effect sizes for the volume comparisons were calculated using Cohen’s d. Group differences in the surface displacement maps were thresholded at p < 0.05, two-tailed, and corrected using threshold-free cluster enhancement. This was true for both the hypothesis and the exploratory analyses. SPSS version 21 (SPSS IBM, New York, USA) was used for the volume analyses and the shape data were analysed using the FMRIB software.
Post hoc analyses
The most significant findings were increased caudate and putamen volumes in the non-migrainous headache groups. We have previously shown, in the present population, that white matter hyperintensities are more common in TTH (15). Since white matter hyperintensities alter image contrast and are often located around the caudate and putamen (25), post hoc analyses on the association between white matter hyperintensity load, as described by Fazeka’s score (15), and the volume of the caudate and the putamen were performed. In the ANOVA analyses, volume of the caudate and the putamen were used as the continuous dependent variable and the Fazeka’s score was used as the ordinal independent variable. Age, sex and ICV were implemented as covariates. In addition, the analyses were rerun with correction for having headache or not in HUNT3.
Results
Of the 1006 participants in HUNT-MRI, 21 individuals were excluded from the present study because of subcortical pathology. Individuals who exhibited pathology (ischemic lesions, traumatic lesions, vessel malformations) of the cerebral cortical mantle alone (n = 23) were not excluded. Furthermore, MRI data from 67 individuals were not included in the analyses owing to poor image quality (mostly motion artefacts) or other errors in the image data acquisition incompatible with the FreeSurfer algorithm. Of the remaining 918 individuals, 814 had answered the headache questionnaire in HUNT2, 786 in HUNT3 and 709 had answered the headache questionnaires in both HUNT2 and HUNT3. In addition, some individuals were excluded because of lack of data on ICV and HADS. Figure 1 and Table 1 show the participation of the present study and the basic characteristics of the population. Headache sufferers had significantly higher HADS scores than non-sufferers. This was true for all the headache diagnoses except for those with headache in HUNT2 alone.
Flowchart of participation in the present study. Basic characteristics of the present headache population. p < 0.05 (compared to those with no headache in HUNT2 and HUNT3). p < 0.01 (compared to those with no headache in HUNT2 and HUNT3. p < 0.001 (compared to those with no headache in HUNT2 and HUNT3). n: Number of individuals; SD = standard deviation; ICV: intracranial volume; HADS: Hospital Anxiety and Depression Scale. Not included in the analyses. Controls in all analyses. Headache non-sufferers HUNT2+3: Those who reported not suffering from headache in the HUNT2 and HUNT3 studies; Headache sufferers HUNT2 alone: Those who reported suffering from headache in the HUNT2 study but not in the follow-up HUNT3 study; Headache sufferers HUNT3 alone: Those who reported suffering from headache in the HUNT3 study but not in the previous HUNT2 study; Headache sufferers both HUNT2+3: Those who reported suffering from headache in the HUNT2 and HUNT3 studies.

Volume (mm3) of subcortical grey matter nuclei and headache status in HUNT2 and HUNT3 corrected for age, sex and intracranial volume.

Note: Significant results shown in bold.
Volumes (mm3) of subcortical grey matter nuclei and frequency of headache attacks corrected for age, sex and intracranial volume.
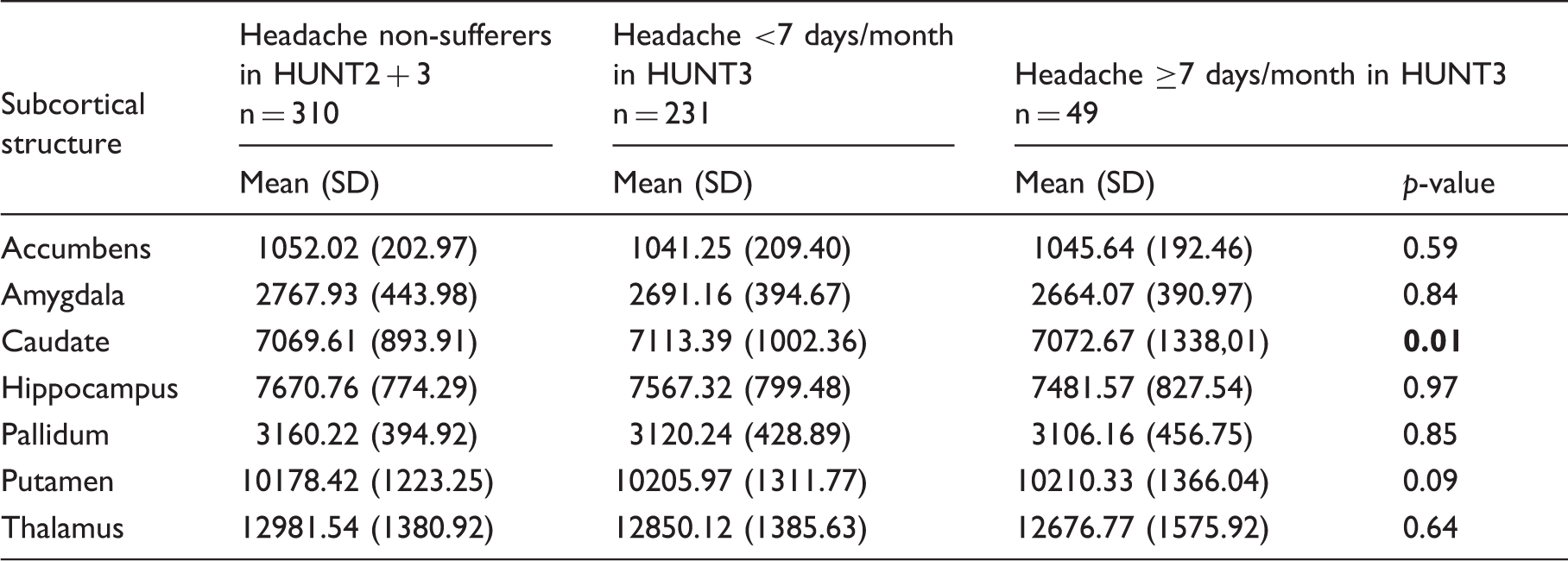
Note: Significant results shown in bold.
In the exploratory analyses corrected for age, sex and ICV (Table 2), the caudate was significantly larger in those suffering from headache in HUNT3 (p = 0.004), both HUNT2 and HUNT3 (p = 0.01) and HUNT3 alone (p = 0.002), but significantly smaller in headache sufferers in HUNT2 (p = 0.03). When examining the headache diagnoses, those with TTH in HUNT3 (p < 0.001) and those with non-migrainous headache in HUNT2 (p = 0.03) had larger volume of the caudate. In addition, a significantly larger putamen was found in those with headache in both HUNT2 and HUNT3 (p = 0.02) and TTH in HUNT3 (p = 0.03). Most effect sizes were very small to small (mean = 0.13, median = 0.13 and standard deviation = 0.095).
With regard to frequency of headache attacks (Table 3), those with headache <7 days/month had larger volume of the caudate than those with headache ≥7 days/month, who again had larger volume of the caudate than those not suffering from headache (p = 0.01).
When HADS was added as a covariate (Supplementary Tables 1 and 2), headache sufferers in HUNT3 showed significantly larger volume of the putamen than the controls (p = 0.03). Otherwise, these analyses gave similar results to the analyses corrected only for age, sex and ICV. The analyses where outliers were removed (Supplementary Table 3) gave higher p-values for all previously significant comparisons leading to non-significant results for those with headache in HUNT2 (caudate, p = 0.06), non-migrainous headache in HUNT2 (caudate, p = 0.05) and TTH in HUNT3 (putamen, p = 0.05). Further, after removing outliers, headache sufferers in HUNT3 (p = 0.01) and in both HUNT2 and HUNT3 (p = 0.01) were found to have smaller volume of the caudate, instead of larger.
Concurring with the volume analyses, between-group comparisons of vertex-wise nucleus shape demonstrated regional deformation of the putamen in those with headache in both HUNT2 and HUNT3. In addition, shape difference without volume difference of the putamen was present in the following groups: Headache in HUNT3, headache in HUNT2, migraine in HUNT2 and non-migrainous headache in HUNT2. More specifically, the headache sufferers displayed an expansion (outward vertex displacement) located mainly laterally and present in both the left and right putamen (Figure 2). Those suffering from migraine in HUNT2 had very limited shape deformation in two areas (6 and 15 vertices) in the accumbens compared to headache non-sufferers.
Putaminal shape differences between different headache groups and the controls (headache non-sufferers in HUNT2 and HUNT3) corrected for age, sex and intracranial volume. Areas in yellow indicate significant difference in shape (P < 0.05, threshold-free cluster enhancement) which in this case are all areas of expansion (outward vertex displacement) in the headache groups relative to the controls.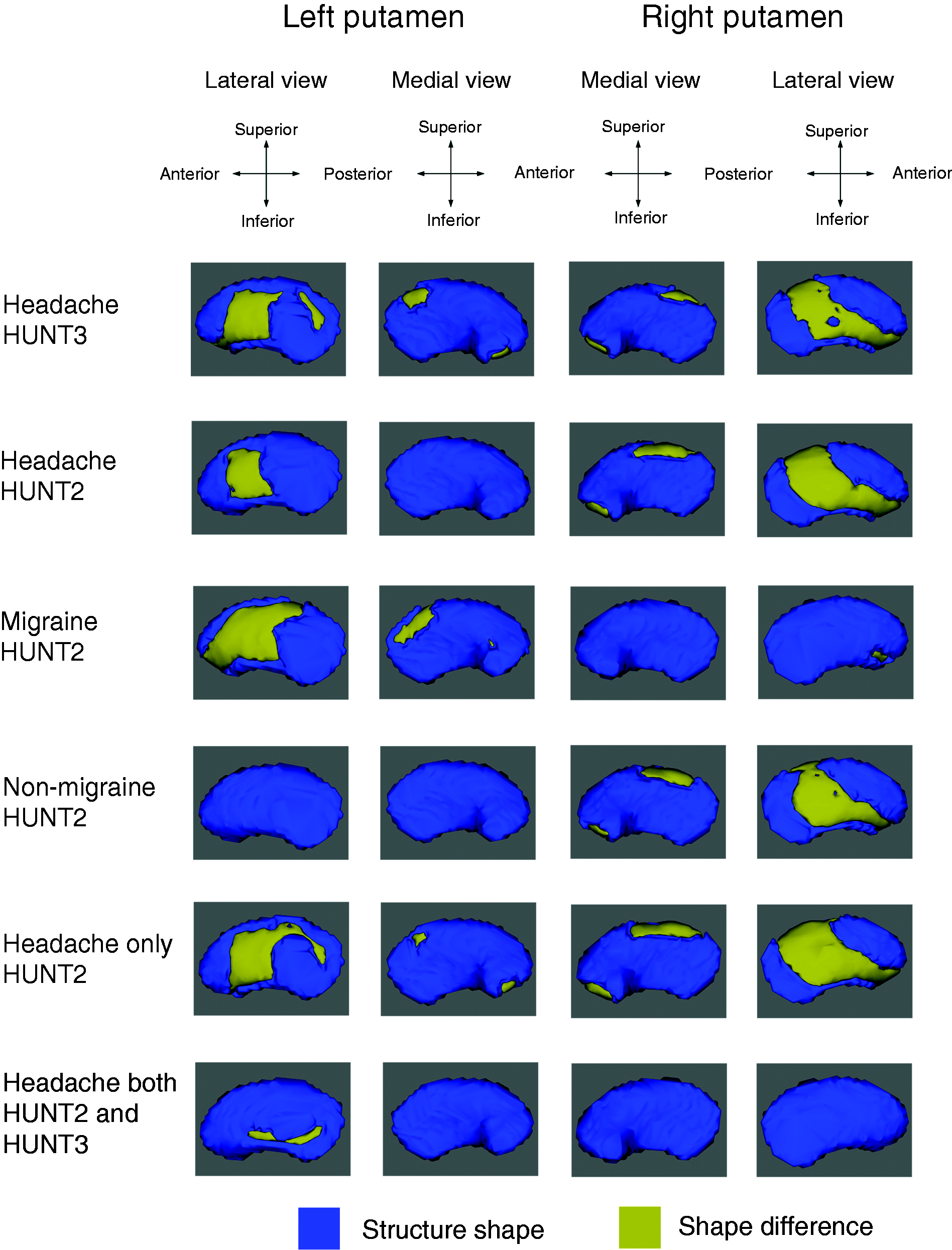
In total, 276 exploratory analyses on volume and shape of the subcortical nuclei were performed. No correction for multiple comparisons were performed.
Post hoc analyses
The post hoc analyses showed that the Fazeka’s score was positively associated to the volume of the caudate (F (3,906) = 29.81 and p < 0.001) (Figure 3), but not to the volume of the putamen (F (3,906) = 2.18 and p = 0.09). Similar results were obtained (caudate: F(3,906) = 20.23 and p < 0.001; putamen: F (3,906) = 0.78 and p = 0.51) when headache status was added as a covariate.
A positive association between the volume of the caudate (mm3) and the Fazeka s score (F (3,906) = 29.81 and P < 0.001).
Discussion
The present study failed to confirm our hypothesis that headache sufferers have smaller volume and deformation of the accumbens compared to headache non-sufferers. The exploratory analyses gave some significant, albeit inconsistent, results that may be useful for generating hypotheses in future studies. These results showed that headache sufferers have different volume of the dorsal striatum (caudate and putamen) compared to headache non-sufferers. Compared to headache non-sufferers, significantly different volume of the caudate was present among all headache groups except for those with migraine and those suffering from headache in HUNT2 alone. The effect sizes for the volume analyses were very small to small and correction for HADS did not affect the results. Furthermore, when removing outliers, all previously significant p-values became larger and the results more inconsistent. The shape analysis showed expansion on the lateral side of the right and left putamen in several of the headache groups but had limited correlation to the volume analyses. The two areas in the accumbens showing shape deformation among those with migraine in HUNT2 were extremely small and the present authors consider them to be of no relevance.
There are several strengths of the present study. Firstly, to our knowledge this is the first population-based imaging study relating subcortical structures to headache. The participants were randomly drawn among individuals attending a large longitudinal epidemiological study (HUNT), thereby avoiding potential selection bias of clinic-based studies. Secondly, headache sufferers were categorized into different headache categories, allowing for investigation of associations between different types of headache and subcortical volumes. The headache criteria used in this study have been validated, showing acceptable accuracy (23). The migraine diagnoses were highly specific but had lower sensitivity. This relationship was the opposite for the non-migrainous headache diagnoses; hence, we have probably classified some true migraineurs as non-migraineurs. Such misclassification will diminish rather than increase differences between the headache groups. Thirdly, the brain morphology was determined with a fully automated method, in which there is no risk of measurement bias related to interpretation of images by humans. Fourthly, before running the analyses, we postulated a precise hypothesis in order to enable definite conclusions. In addition, exploratory analyses with the purpose of generating hypotheses for later studies were performed. Fifthly, data on headache status in HUNT2 and HUNT3 enabled investigations of associations between subcortical nuclei morphology and evolution of headache. Further, it allowed selection of individuals with very few to no documented headache complaints over several years as control group. Last but not least, compared to the previous clinic-based studies, this population-based study was superior in terms of number of both headache sufferers and controls.
An important limitation in this study is the relatively long time interval from when the participants answered the headache questionnaire (1995–1997 in HUNT 2 and 2006–2008 in HUNT 3) to when they were scanned (2007–2009). Morphological changes can both arise and recede within a year (4,26). Although this effect cannot be ruled out, it seems unlikely that the headache had improved or increased dramatically in the majority during the time from the HUNT3 questionnaire to the MRI scanning (mean 1.2 years). Furthermore, longitudinal data existed only on headache status, and therefore the present study does not allow conclusions as to whether differences in subcortical morphology may be the cause or consequence of headache. Even though it is fair to assume individuals with consistent answers in the two surveys to be long-term headache sufferers and long-term non-sufferers, caution in interpretation of these results has to be taken because of the lack of information on headache status between the two time points. Also, estimating the headache status of individuals with a questionnaire is inferior to a clinical interview.
In contrast to previous studies (2,6,7), the present results indicate that non-migrainous types of headache, such as TTH, are more strongly associated to volumetric deviation of the striatum than migraine. Indeed, our data showed that migraineurs tended to have smaller volume of caudate and putamen and, considering the effect sizes, the lack of significance regarding the migraine groups could be caused by lack of power. However, the present migraine group was larger than those in previous studies. Based on the sensitivity and specificity of the present diagnoses, the migraine group should be well suited for investigating characteristics associated with migraine. The TTH group, on the other hand, probably included some individuals with migraine and thus larger volume of the caudate and putamen may in reality be even more strongly associated with TTH. The present analyses indicated that temporal proximity could be important for the association between headache and volume of caudate, since those who had headache a relatively short time before the MRI scanning (HUNT3) had a larger caudate than those who had headache only in the past (HUNT2). Furthermore, as correction for HADS had no impact on the results, anxiety and depression probably do not influence the association between headache and volume of the subcortical nuclei in the general population.
Before concluding that headache sufferers exhibit structural variations of the caudate and putamen, one should remember that the results obtained in the present sample were partially due to outliers and somewhat inconsistent, showing headache sufferers to have both larger and smaller volume of caudate. Furthermore, the effect sizes were small for all the significant comparisons, indicating that if the results are true the volume of the caudate and the putamen are almost indistinguishable between headache sufferers and non-sufferers in the general population.
The shape analyses showed lateral parts of the bilateral putamen to be expanded in headache sufferers, but not in the two migraine groups or the TTH group. No difference in shape was detected between headache sufferers and non-sufferers with regard to the caudate. The shape analyses showed limited correlation to the volume analyses. It might be that the present differences in volume of the caudate and putamen are attributed to an evenly distributed volume increase in headache sufferers and thus not detectable in a shape analysis.
The caudate and putamen, constituting the dorsal striatum, receive input from the cerebral cortex and thalamus and activation here enables execution of motor and cognitive cerebral programs (27,28). Hence, one may speculate that this region is larger in those with non-migrainous headache due to an increase in afferent/efferent signalling as part of headache pathophysiology. On the other hand, the post hoc analyses showing a highly significant dose-response relationship between Fazeka’s score and caudate volume may indicate that the dorsal striatum volume increase in headache sufferers represents an MRI artefact caused by white matter hyperintensities. The relationship between white matter hyperintensities and caudate volume was not confounded by headache status. Conceivably, changes in the nearby white matter alter the grey/white matter contrast and subsequently apparent grey matter volume size in T1 weighted MRI scans. It has previously been demonstrated, both in this population and in others, that white matter hyperintensities are more common in headache sufferers (15,29).
The exact interpretation of differences in deep grey matter nucleus volume seen on MRI remains to be fully understood, but the hypothesis that it may arise from white matter changes should be further explored. As in headache MRI studies, studies of other pain-related conditions have showed volumes of putamen and caudate to be both larger (30–32) and smaller (33,34) than in controls, but since chronic pain has also been associated with white matter hyperintensities (35) these findings too could be caused by changes in the white matter. The current findings in this headache population could therefore be of relevance for other pain conditions as well.
In conclusion, the present large population-based imaging study failed to confirm our hypothesis that headache sufferers exhibit smaller volume and different shape of accumbens. Instead, we found non-migrainous headache sufferers to have somewhat different volume of the dorsal striatum, especially caudate, compared to non-sufferers. A post hoc analysis showed that this could be explained by white matter hyperintensities. Overall, the effect sizes were very small, and the results were somewhat inconsistent. Hence, in the general population, there are probably no or only very small differences in the morphology of subcortical nuclei between headache sufferers and non-sufferers.
Public health relevance
In this large population-based imaging study, both volume and shape analyses were applied in order to investigate the relationship between headache and subcortical brain structures. Several headache diagnoses (migraine and tension-type headache included) were investigated and individuals with few documented headache complaints over several years were chosen as controls. We failed to confirm our hypotheses that headache sufferers (regardless of subtype) would have smaller volume and different shape of the nucleus accumbens compared to those not suffering from headache. The exploratory analyses found non-migrainous headache sufferers to have somewhat different volume of the dorsal striatum compared to the controls, but post hoc analyses showed that this could be explained by white matter hyperintensities. In the general population, there is probably no or only a very small difference in subcortical brain morphology between those who do and those who do not suffer from headache.
Supplemental Material
Supplementary Table 1 -Supplemental material for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study
Supplemental material, Supplementary Table 1 for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study by Andreas Kattem Husøy, Carl Pintzka, Live Eikenes, Asta K Håberg, Knut Hagen, Mattias Linde and Lars Jacob Stovner in Cephalalgia
Supplemental Material
Supplementary Table 2 -Supplemental material for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study
Supplemental material, Supplementary Table 2 for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study by Andreas Kattem Husøy, Carl Pintzka, Live Eikenes, Asta K Håberg, Knut Hagen, Mattias Linde and Lars Jacob Stovner in Cephalalgia
Supplemental Material
Supplementary Table 3 -Supplemental material for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study
Supplemental material, Supplementary Table 3 for Volume and shape of subcortical grey matter structures related to headache: A cross-sectional population-based imaging study in the Nord-Trøndelag Health Study by Andreas Kattem Husøy, Carl Pintzka, Live Eikenes, Asta K Håberg, Knut Hagen, Mattias Linde and Lars Jacob Stovner in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The Nord-Trøndelag Health Study (the HUNT Study) is a collaboration between HUNT Research Centre (Faculty of Medicine and Health Sceince, Norwegian University of Science and Technology (NTNU)), Nord-Trøndelag County Council, Central Norway Regional Health Authority, and the Norwegian Institute of Public Health.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
