Abstract
Background
According to the neurovascular theory of migraine, activation of the trigeminovascular system contributes to the development of migraine. This study examined the effects of chronic intraperitoneal ghrelin (150 µg/kg) treatment on the development of chronic migraine induced by intermittent injection of nitroglycerin 10 mg/kg.
Methods
Baseline and post-drug (2 h following nitroglycerin injection) mechanical and thermal sensitivity were assessed by von Frey hair and tail immersion tests, respectively on days 1, 3, 5, 7, 9 and 11. Moreover, we investigated the effect of ghrelin treatment on nitroglycerin-induced aversive behavior by using a two-chamber conditioned place aversion paradigm. At the end of behavioral testing, on day 11, animals were sacrificed and plasma concentration of calcitonin gene-related peptide was measured using a rat-specific enzyme-linked immunosorbent assay kit. Also, real time polymerase chain reaction was used to quantify mRNA expression of calcitonin gene-related peptide and transient receptor potential vanilloid 1 in the trigeminal ganglion.
Results
Our results indicated that nitroglycerin activated the trigeminovascular system, which was reflected by mechanical and thermal hypersensitivity and elevation of mRNA expression of calcitonin gene-related peptide and transient receptor potential vanilloid-1, as migraine markers, and plasma calcitonin gene-related peptide levels. Moreover, chronic nitroglycerin injection induced conditioned place aversion and body weight loss. Nevertheless, ghrelin modulated nitroglycerin-triggered changes in transient receptor potential vanilloid-1 and calcitonin gene-related peptide expression, and mitigated nitroglycerin-induced hyperalgesia.
Conclusion
These results provide the first convincing evidence that ghrelin has a modulating effect on central sensitization induced by chronic intermittent nitroglycerin, and its antinociceptive effect may be related to a reduction of these factors in the trigeminal ganglion.
Keywords
Introduction
Migraine is a common chronic neurovascular disorder affecting both the patients and society. The World Health Organization ranks migraine as the third most costly neurological disorder (1,2). Since there is no specific test to diagnose migraine, diagnosis is based on clinical history (3). This pain syndrome is associated with nausea and/or vomiting, photophobia and phonophobia (4). According to the International Headache Society (IHS), chronic migraine is recognized as more than 15 headache days per month over a three month period (5). Some evidence suggests that frequent episodes of migraine may lead to chronic sensitization of central pain pathways in chronic migraine. Therefore, transition of acute migraine into chronic migraine is associated with allodynia (6,7).
Migraine is a complex disorder with a variety of triggers whose exact mechanism is now being better understood. Activation of the trigeminovascular system (TS) plays a pivotal role in the pathophysiology of migraine pain and its associated symptoms (8). Extensive evidence shows that chemical activation of TS by administration of nitroglycerin (NTG), a nitric oxide (NO) donor, is a reliable and standard model for studying migraine (9–12). The mechanism of NTG-induced migraine pain is supposed to be the result of vasodilation of cranial blood vessels and activation of pathways involved in nociception (13).
Calcitonin gene related peptide (CGRP) is a vasodilatory neuropeptide that is widely expressed in migraine-related structures such as the trigeminal ganglion (TG) and trigeminal nucleus caudalis (TNC). CGRP has a causative role in the pathophysiology of migraine headache, and is considered as a biomarker of migraine headache (14–16). Previous studies reported that concentrations of CGRP in the plasma, saliva, and cerebrospinal fluid are elevated during migraine headache and normalized by triptans (17–19). It has also been shown that activation of the trigeminal system by administration of NTG increases CGRP levels in the brainstem and TG (20). Moreover, intravenous infusion of CGRP evokes a migraine-like headache in migraineurs (21). In another study, Bellamy et al. reported that CGRP intensifies nociceptive activity during migraine through vasodilation and release of pro-inflammatory cytokines from satellite glial cells (SGCs), which in turn leads to sensitization of peripheral and central neurons within the trigeminal system (1). In contrast, several CGRP receptor antagonists have been proven to be effective in acute treatment of migraine headache (19,22,23).
Transient receptor potential vanilloid-1 (TRPV1), a capsaicin receptor, is activated by inflammation, noxious heat (activated at 42℃) and painful chemical stimulus (24). Existing researches havehighlighted the critical role played by the TRPV1 ion channel in the pathophysiology of migraine (25). Activation of TRPV1, expressed on trigeminal nociceptors, leads to release of CGRP from trigeminal nerve terminals resulting in neurogenic inflammation within the meninges (26,27).
Ghrelin, an orexigenic neuropeptide, is chiefly derived from the stomach (28). Ghrelin affects a broad range of biological processes such as food consumption and energy hemostasis (29–32). Ghrelin has also been shown to have antinociceptive effects in a variety of experimental models of pain (33–36). In 2006, Sibilia et al. for the first time demonstrated that ghrelin exhibits an inhibitory role on inflammatory pain through interaction with hypothalamic opioid-containing neurons (37). Guneli et al. have also reported that administration of ghrelin prevented chronic constriction injury of the sciatic nerve through inhibition of pro-inflammatory cytokines in the spinal cord (38). Lately, our group showed that a single dose of ghrelin could block acute NTG-evoked hyperalgesia (34).
Recent evidence in management of migraine pain has heightened the need for new drugs. No previous study has investigated the effect of chronic administration of ghrelin in a chronic model of migraine. In the present study, the effects of NTG and ghrelin treatment on hyperalgesia and expression of CGRP and TRPV1 receptors in the TG were investigated in a chronic migraine model in rats.
Material and methods
Animals
Forty male Wistar rats weighing 220 to 250 g, about 90 days old, were obtained from the animal center of Tabriz University of Medical Sciences. Animals were kept three per cage at a 12 h light–dark cycle, 20–22℃; 45–55% humidity, and had free access to food and water in the laboratory animal house of the Neurosciences Research Center. All efforts were made to minimize animal suffering and the number of animals used. All experimental procedures were performed in accordance with the health guide for the care and use of laboratory animals of Tabriz University of Medical Sciences and approved by the Ethics Committee of Animal Research of Tabriz University of Medical Sciences (license No.: TBZmed.REC.1394.924). The sample size was determined with regard to previous studies (11,39,40) and based on the ethical principles of laboratory animals of Tabriz University of Medical Sciences. Furthermore, sample size was estimated using STATA software (alpha = 0.05 (two-sided), power = 80%, sd1 = 9, sd2 = 1, M1 = 67, and M2 = 56). This study adhered to the ARRIVE guidelines for animal research.
Experimental design
NTG (Caspian Tamin Pharmaceutical Company, Iran) was prepared from a stock solution of 5.0 mg/ml nitroglycerin in 30% alcohol, 30% propylene glycol, and water. NTG was freshly diluted each day in 0.9% saline to a dose of 10 mg/kg. In this study, saline 0.9% was used as vehicle. No significant differences in mechanical and thermal thresholds were observed between vehicle and NTG solvent received groups.
After one week of habituation, rats were randomly divided into four groups, 10 animals in each group, and treated according to the following experimental design: Vehicle group received intraperitoneal (i.p.) injection of saline (0.9%) every day for 11 days; the ghrelin group received i.p. injection of 150 µg/kg/day ghrelin (Innovagen, Sweden) for 11 days; the NTG group was treated every second day (on days 3, 5, 7, and 9) with i.p injections of NTG (10 mg/kg) for 9 days (41); the NTG + ghrelin group was injected with ghrelin (150 µg/kg, i.p.) every day for 11 days in addition to NTG (39). All injections were administered as a maximum volume of 10 ml/kg.
Measurement of food intake and body weight
Every day, food was weighed and provided in standard stainless steel hoppers. The amount of remaining food, either on the hopper or any that had spilled on the bottom of the cage was recorded for 11 days. The food intake was calculated as the difference between the weight of the food provided and the remaining food after 24 h. Changes in body weight (or weight gain) were also recorded for each animal every second day for 11 days.
Behavioral assays
Behavioral tests were performed between 8:00 a.m. and 2:00 p.m. in a quiet room. All animals were transferred to the test room at least 30 minutes before the experiments to habituate, and a person blinded to the experimental treatments performed all tests. Baseline mechanical and thermal responses were determined on days 1, 3, 5, 7, 9, and 11 before injection of NTG or vehicle, and post-drug sensitivity tests were performed two hours after NTG injection on days 3, 5, 7, 9, and 11. Our experimental design is summarized in Figure 1.
The experimental design. NTG: nitroglycerin (10 mg/kg); D: days.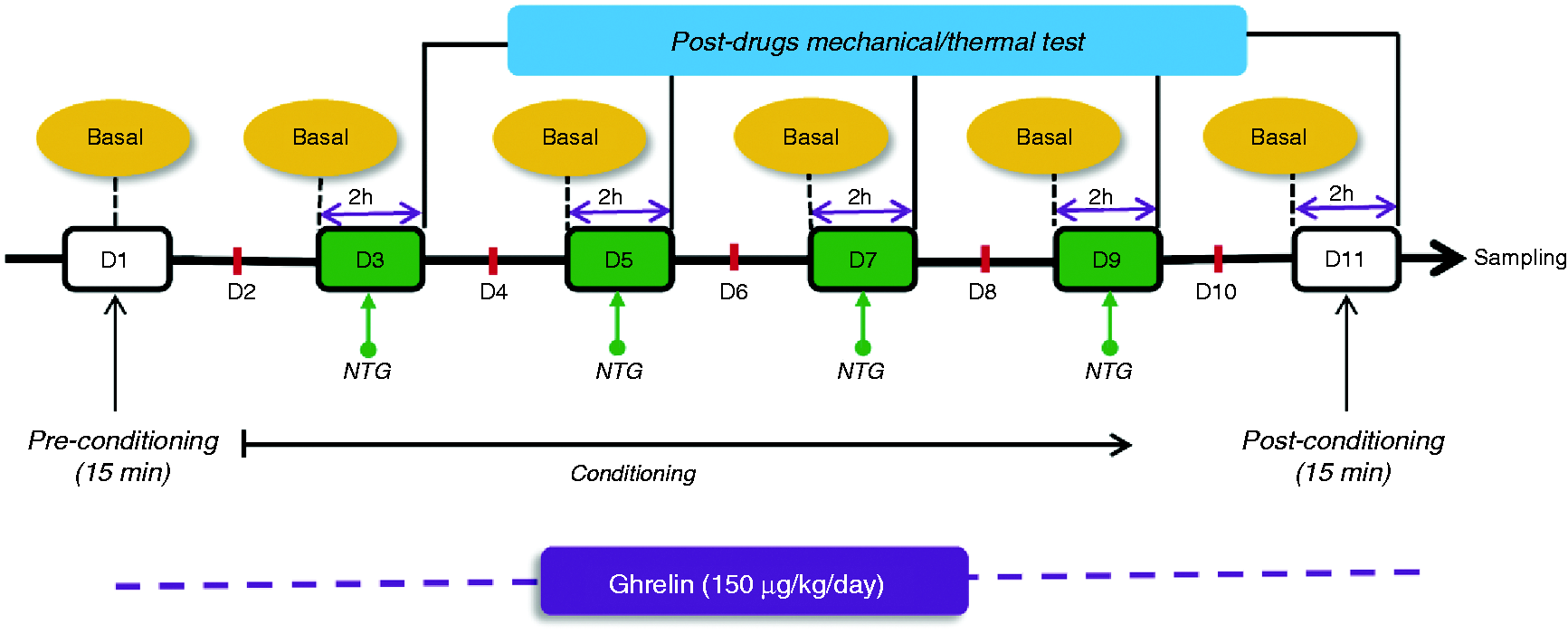
Mechanical and thermal sensitivity
Mechanical sensitivity was determined with a series of calibrated von Frey hairs (Touch Test Sensory Evaluator Kit, USA) with a bending force range from 0.002 to 300 g according to the up-and-down method as previously described (42). In brief, rats were placed on wire mesh platforms in clear cylindrical plastic cages and allowed to acclimate for 30 min. A series of eight von Frey filaments were applied to the mid-plantar region of the hind paw of the animals three times with each filament. The first filament tested was 2 g. A response was defined as a shaking or lifting of the paw upon stimulation. If there was a lack of response, a heavier filament (up) was applied, and in the presence of a response a lighter filament (down) was tested. The mechanical threshold was defined as the minimum force that caused at least two positive responses out of three trials.
To determine thermal sensitivity, rats were gently restrained and 5 cm from the tip of the tail was immersed in the hot water bath (48 ± 0.5°c), and the latency to tail withdrawal was recorded. Tail withdrawal latency was defined as an obvious flick or bend of the tail out of the water. A cut-off of 40 s was established to avoid damage to the tail (43).
Conditioned place aversion
We used a two-chamber conditioned place aversion (CPA) paradigm, as previously described in detail (44). Briefly, the apparatus consisted of two identical Plexiglas boxes (40 × 40 × 30 cm), divided into two individual compartments, each with a different wall pattern (horizontal or vertical white stripes) and floor texture (stainless steel perforated mesh or round holes). A plastic guillotine door (15 × 15 cm) separated the two compartments. CPA consisted of three phases:
Pre-conditioning phase: On the first day, the guillotine door was raised 12 cm above the floor and animals were allowed to freely explore both compartments for 15 min, and the time spent in each compartment for 900 s was recorded. Based on the results of the pre-conditioning, the drug-paired compartments were allocated in an unbiased manner to the saline and NTG groups.
Conditioning phase: On days 2, 4, 6 and 8, animals were injected with saline and restricted to one of the conditioning chambers for between 1.5–2 h post-injection. On days 3, 5, 7 and 9, rats were injected with saline (vehicle group) or 10 mg/kg i.p. NTG (NTG group), and were restricted to the other conditioning chamber for 1.5–2 h post-injection. Animals in the vehicle or NTG + ghrelin groups received saline 0.9% or ghrelin (150 µg/kg i.p) respectively, 30 min prior to the conditioning session.
Post-conditioning phase: On day 10, animals were allowed free access to both chambers with no aversive stimulus present and the time spent in each compartment was recorded. On day 11, animals received their treatments and had free access to the entire apparatus as a test of state-dependent aversion. The difference in the time spent in the NTG-paired and vehicle-paired sides on the test day served as an index of CPA magnitude (10).
Blood and trigeminal ganglion tissue sampling
At the end of behavioral testing, all animals were deeply anaesthetized with a mixture of ketamine (60 mg/kg) and xylazine (10 mg/kg). Blood samples (2 ml) were collected from the heart into tubes containing ethylenediaminetetraacetic acid (EDTA). Then the plasma was separated by centrifugation at 5000 g for 10 min at 4℃ and aliquots for the determination of CGRP levels were stored at −20℃ until analysis. Animals were immediately decapitated, the TG removed and stored at −80℃ for further analysis.
Determination of plasma levels of CGRP
The concentrations of CGRP in the plasma were determined as duplicates by a rat-specific CGRP enzyme-linked immunosorbent assay (ELISA) kit (Zellbio, Germany) according to the manufacturer's instructions.
Quantitative real-time PCR
Total RNA was extracted from the homogenized TG by using Trizol reagent (Invitrogen) and an alcohol-chloroform protocol according to the manufacturer’s recommendations. The mRNA concentration was quantified using a Nano Drop Spectrophotometer (Thermo Scientific, USA). Reverse transcription into cDNA was carried out with the RT-PCR kit (EURx, Poland) according to the manufacturer’s recommendations. Quantitative real-time PCR was performed using SYBR Green PCR Master Mix (Takara Bio, Shiga, Japan) in a final volume of 10 μl. Primer sequences for TRPV1, CGRP, and GAPDH, an internal control, were as follows: TRPV1 (Forward 5′-GTGTGCCTGCACCTAGCTGGTT-3′, Reverse 5′-GGGTCCAGGCAGGAGTTCTCTT-3), CGRP (Forward 5′-GGCCTCCAGGCAGTTCCTTT-3′, Reverse 5′-GTGCAGCCAGTAGGCGAGCTT-3′), GAPDH (Forward 5′-CCTGTGACTTCAACAGCAACTC-3′, Reverse 5′-CAAGTTCAACGGCACAGTCAAG-3′). The CT values given by a Light Cycler 96 (Roche, Germany) real-time PCR instrument were analyzed by the relative quantitative method (efficiency = 100%). The mRNA expressions of CGRP and TRPV1 were normalized to that of GAPDH. The following formula was used to calculate the relative expression of the target genes: 2–ΔΔCT is ΔCt = Ct(sample) – Ct(GAPDH) and ΔΔCt = (ΔCt(sample)–ΔCt(control)).
Statistical analysis
Data are expressed as mean ± standard error of mean (SEM). All statistical analyses were performed by the SPSS version 16 statistics software package. A two-way repeated measure ANOVA followed by the Holm-Sidak post-hoc test were used to determine the significance of differences in the behaviors of animals in the von Frey and tail immersion tests. For time spent in the paired sides, a paired sample t-test was used. For real time PCR results, the Kruskal-Wallis test followed by the Mann-Whitney post-hoc test were used. Unless otherwise noted, all experiments were statistically analyzed using one way ANOVA followed by post-hoc Tukey’s test. In all cases, p < 0.05 was considered as statistically significant.
Results
Ghrelin increased food intake and reversed body weight loss induced by chronic NTG
The effects of chronic administration of ghrelin on food intake (a) and body weight changes (b) are summarized in Figure 2. The NTG-treated rats in this experiment consumed less food than the vehicle- and ghrelin-treated rats, indicating the presence of nausea. Consequently, weight gain in the animals that received multiple NTG injections was significantly less than vehicle animals. Nevertheless, chronic ghrelin administration increased food intake and weight gain in the ghrelin group compared to the NTG group in a time-dependent manner. Moreover, animals in the NTG + ghrelin group that received ghrelin treatment for 11 days had significantly higher food intake and weight gain than the NTG group receiving saline.
Chronic nitroglycerin (NTG) administration suppressed food consumption (a), and body-weight gain (b) compared to the vehicle animals. Chronic (11 days) ghrelin treatment improved food intake and weight gain in the NTG-received rats. Data are shown as mean ± SEM for 10 animals in each group. Repeated measures two-way ANOVA was followed by the Holm-Sidak modified t-test, *p < 0.05, **p < 0.01, ***p < 0.001.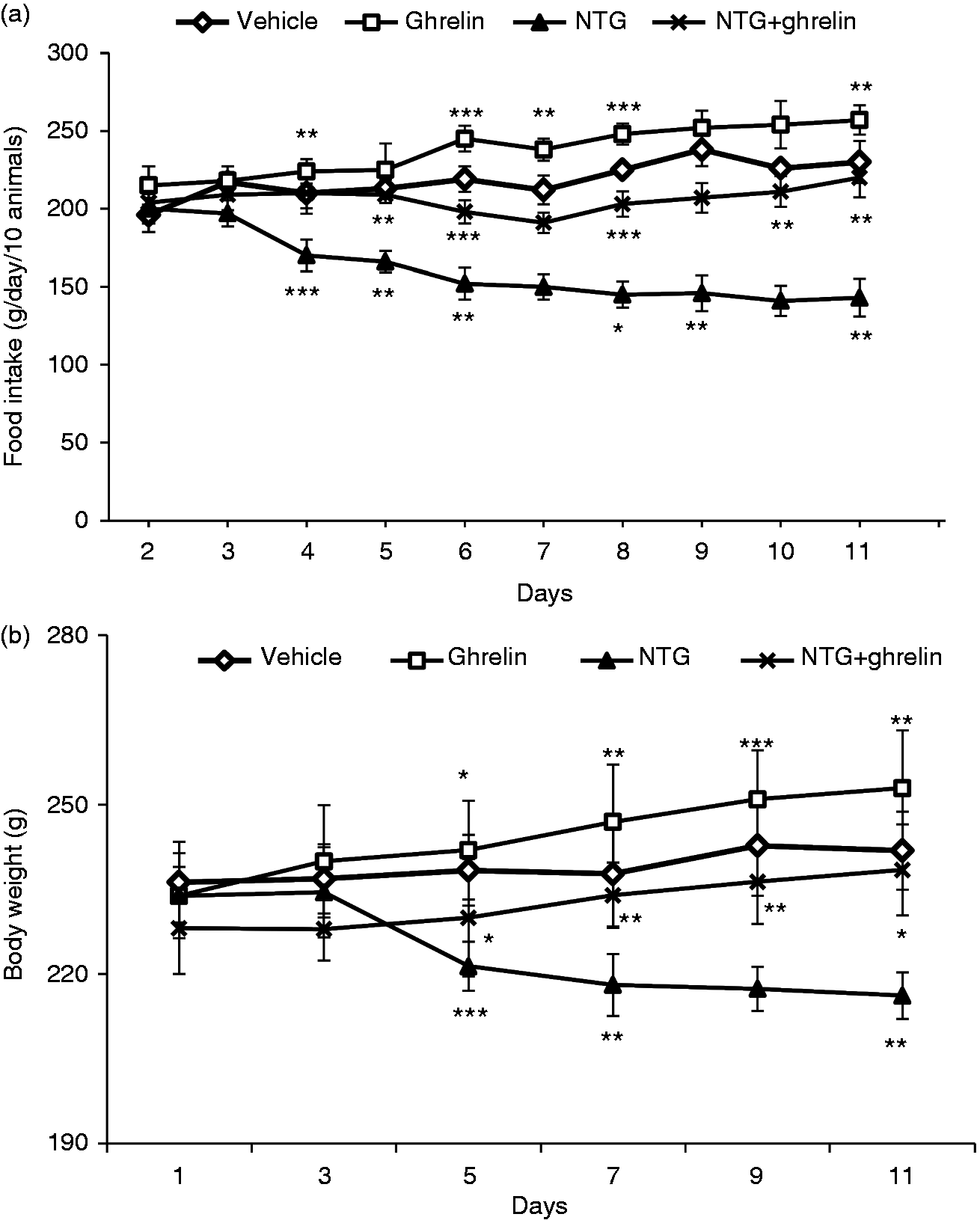
Chronic ghrelin treatment attenuated mechanical and thermal hyperalgesia induced by chronic NTG
Repeated intermittent injection of 10 mg/kg NTG gradually produced basal mechanical and thermal hypersensitivity (Figure 3(a) and (c), respectively). Moreover, post-drug results showed that NTG induced significant acute mechanical and thermal hyperalgesia on each test day (on days 3, 5, 7 and 9) (Figure 3(b) and (d)), which continued over the 11 days of the study period compared with the vehicle group. At all other time points, the nociceptive scores of the NTG group were significantly higher than those of the vehicle rats. On the other hand, chronic treatment with ghrelin for 11 days significantly increased basal mechanical and thermal thresholds, and reversed post-drug mechanical and thermal hyperalgesia following administration of each dose of NTG (Figure 3(b) and (d)), which started from day 5 and gradually continued throughout the 11 days. In the ghrelin group, repeated administration of ghrelin (150 µ/kg) did not change the basal and post-drug mechanical and thermal responses compared to the vehicle group.
Chronic nitroglycerin (NTG) evoked hyperalgesia, which was significantly attenuated by ghrelin treatment. Basal mechanical (a) and thermal (c) responses, as tested before vehicle or NTG administration, significantly decreased in the NTG group. Post-drug mechanical (b) and thermal (d) responses, as tested 2 h after NTG or vehicle administration, showed that hyperalgesia, induced by chronic NTG, was reversed by ghrelin (150 µg/kg/day) treatment for 11 days; n = 10/group, repeated measures two-way ANOVA was followed by the Holm-Sidak modified t-test, *p < 0.05 **p < 0.01, ***p < 0.001.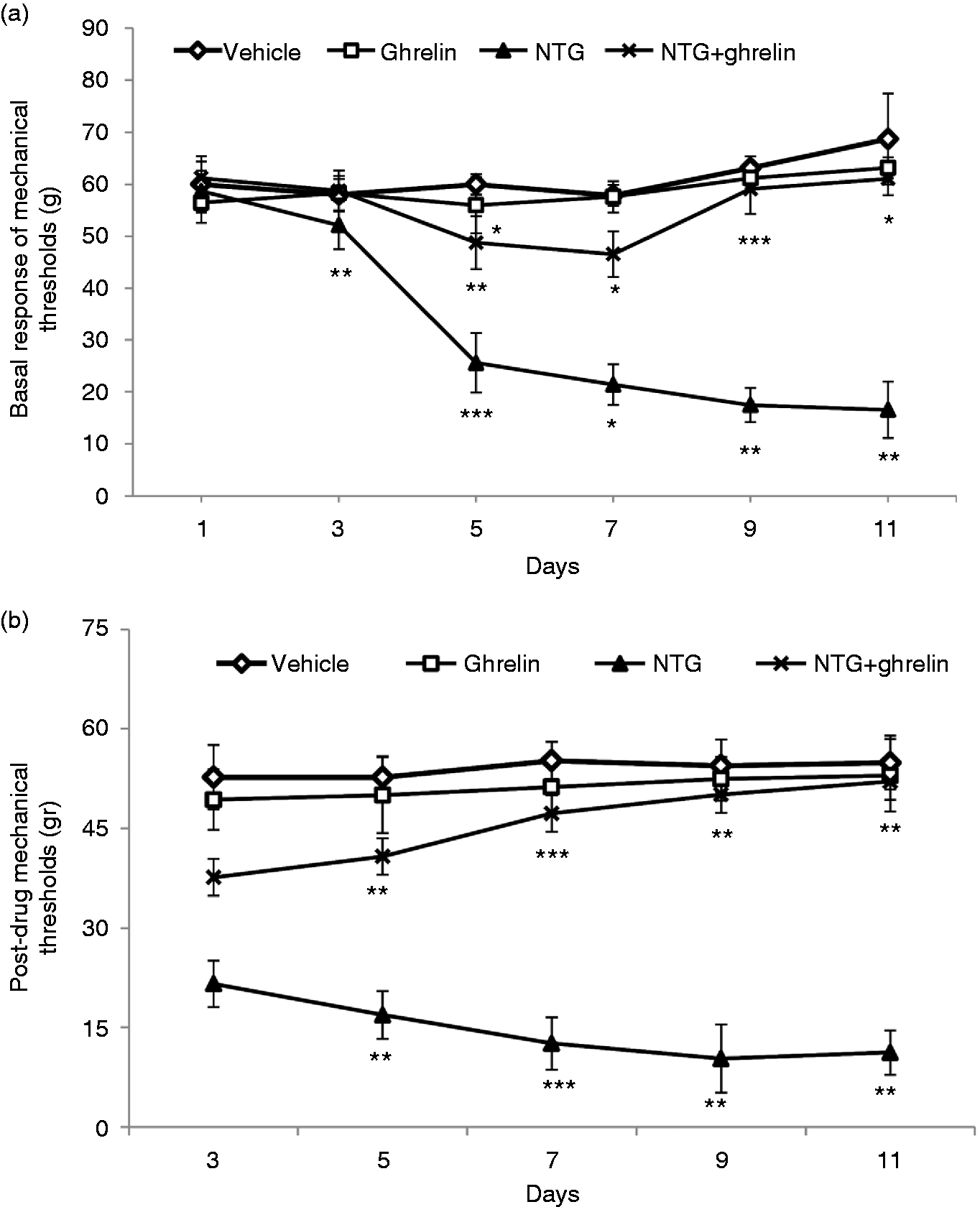
Ghrelin reversed conditioned place aversion induced by chronic intermittent NTG
The results of the CPA test showed that there was no initial preference for either chamber during the pre-conditioning test among the four groups (Figure 4(a)). On day 10, in a treatment-free state, administration of NTG or vehicle did not produce aversion or preference (data are not shown). However, in the post-conditioning phase in a state-dependent test, animals in the NTG group spent significantly less time (p = 0.001) in the chamber where they had previously experienced NTG-induced headache compared with the pre-conditioning phase. Moreover, NTG-receiving rats displayed significantly greater magnitude of CPA scores than vehicle rats (Figure 4(b)). However, administration of ghrelin in the NTG + ghrelin group reversed CPA induced by NTG and decreased its magnitude. By contrast, chronic administration of ghrelin in the ghrelin group caused neither production of aversion nor preference in rats.
Conditioned place aversion (CPA) produced by chronic intermittent nitroglycerin (NTG) reversed by ghrelin treatment. (a) Percentage of time spent in paired side. On the pre-conditioning phase, no significant preference for either chamber was seen. However, on the test day, animals in the NTG group spent significantly less time on the NTG-paired side. Paired sample t-test, ***p < 0.001, ##p < 0.01. (b) The difference in the time spent in the NTG-paired and vehicle-paired sides on the test day served as an index of CPA magnitude. On day 11, in a state-dependent test, NTG evoked a significant conditioned place aversion, which was reversed by chronic ghrelin treatment. A negative score represents aversion, whereas a positive score represents preference. Data are shown as mean ± SEM for 8–10 animals per group, one-way ANOVA, Tukey’s post-hoc test, ***p < 0.001 compared with the vehicle group, ##p < 0.01 compared with the NTG group.
Chronic ghrelin treatment attenuated plasma concentration of CGRP
We examined the effect of ghrelin treatment on the plasma levels of CGRP using the ELISA method. Our study clearly demonstrated that ghrelin treatment reduces plasma levels of CGRP (Figure 5). Although a single dose injection of NTG, within 3 h, significantly (p < 0.05) increased the plasma concentration of CGRP, those found after the chronic intermittent NTG injection, in the chronic NTG-receiving animals, were more prominent (p < 0.001) compared to the vehicle group. On the other hand, acute treatment with a single dose of ghrelin significantly (p < 0.05) decreased CGRP concentration compared to the acute NTG group. Furthermore, chronic ghrelin treatment markedly (p < 0.01) reduced CGRP concentration in the NTG + ghrelin group compared to the chronic NTG group. However, administration of ghrelin in the ghrelin group did not significantly change plasma CGRP levels.
Elevation of CGRP plasma levels induced by nitroglycerin (NTG) administrations and its reduction by ghrelin treatment. Data are shown as mean ± SEM for 10 animals per group. One-way ANOVA followed by Tukey post hoc test, *p < 0.05, ***p < 0.001 compared with the vehicle control group; $p < 0.05 compared with the Acute NTG group; ##p < 0.01 compared with the chronic NTG group.
Chronic ghrelin treatment down-regulated CGRP and TRPV1 mRNA expression in the TG
To investigate the changes in mRNA expression of CGRP and TRPV1, trigeminal ganglia were removed 3 h after a single injection of NTG (acute NTG) and 11 days after chronic intermittent injection of NTG (chronic NTG). Relative expressions of CGRP mRNA and TRPV1 mRNA were analyzed with real time PCR. Figure 6 shows that acute NTG significantly (p = 0.02) increased CGRP and slightly increased TRPV1 mRNA expression in the TG. Moreover, a single dose treatment with ghrelin insignificantly decreased CGRP and TRPV1 mRNA in the TG. Nevertheless, chronic intermittent injection of NTG markedly (p = 0.001) increased mRNA expressions of both CGRP and TRPV1 in the TG. On the other hand, chronic ghrelin treatment in the NTG + ghrelin group significantly (p = 0.01) decreased mRNA expressions of both CGRP and TRPV1 compared to the chronic NTG group. Besides, chronic ghrelin administration insignificantly decreased mRNA expression of CGRP and TRPV1 in the TG compared to the vehicle group.
Relative expression CGRP and TRPV1 mRNA in TG in different groups; n = 6, Kruskal-Wallis test was followed by the Mann-Whitney post-hoc test, *p < 0.05, ***p < 0.001 compared with vehicle group; ##p < 0.01 compared with chronic NTG; $p < 0.05, $$p < 0.01 compared with acute NTG group.
Discussion
The principal new findings of the present study were that ghrelin treatment attenuated chronic intermittent NTG-induced mechanical and thermal hypersensitivity, indicating that chronic ghrelin treatments inhibited the development of hypersensitivity. Moreover, hypersensitivity was accompanied with elevated plasma levels of CGRP and up-regulation of CGRP and TRPV1 mRNA expressions in the TG, which were both attenuated by ghrelin treatment. In addition, our results showed that ghrelin reversed NTG-induced place aversion, as well as improving the food intake and body weight in the NTG-treated group.
Migraine has a considerable impact on both patients and society by reducing quality of life and work productivity (45,46). Despite the high prevalence and burden of migraine on the patient and society, available treatments are surprisingly limited, which may reflect the absence of a clinically relevant animal model. Evidence suggests that stimulation of TS is the most important factor for inducing migraine (8). Administration of NTG, as a reliable experimental model, demonstrates several behavioral similarities to human migraine (7). Numerous studies demonstrated that systemic administration of NTG induced a delayed spontaneous headache attack via stimulation of the TS system, vasodilation, and central sensitization (47–50). In this study, we found that chronic intermittent injection of NTG evoked acute hyperalgesia following each NTG injection, and a baseline hypersensitivity that progressively developed over time. Similar effects were reported by Pradhan et al., who showed that chronic NTG is able to evoke mechanical and thermal hypersensitivity (10,41). It seems that NTG produces a hyperalgesic condition, possibly through vasodilatation and activation of the TS.
CGRP, as an endogenous pain mediator, plays a pivotal role in the development of hyperalgesia and is categorized as the most important neuropeptide involved in the pathophysiology of migraine and other primary headaches (14,21). Cell bodies of trigeminal ganglia neurons are the main source of CGRP (51). It is now well established that increased CGRP levels are a biological marker of activation of the TS, which in turn leads to vasodilation and neurogenic inflammation, and decreases threshold activation of trigeminal neurons (52,53). In contrast, CGRP receptor antagonists effectively reduce trigeminal activity in rodents and migraineurs (22,40,54). There is accumulating evidence demonstrating that migraine treatment with triptans efficiently relieves pain and returns the levels of CGRP to normal (1,55,56). Previous studies have also found that NTG increases the release of CGRP and administration of CGRP receptor antagonists reverses NTG-induced hyperalgesia (20,23). Previous researches have also shown that chemical activation of TS by NTG increased CGRP levels (1,20). We consistently found that administration of NTG increased the plasma concentration of CGRP. We suggest that CGRP may be involved in the development of NTG-induced hyperalgesia.
Recently, several studies have focused on the TRPV1 receptor as a target for the development of new therapeutic approaches in pain management (57–59). It has been shown that TRPV1 contributes to peripheral sensitization, allodynia and hyperalgesia, and blockade of these receptors attenuates central sensitization (58,60). Moreover, various studies have shown that activation of TRPV1 receptors initiates migraine attacks, possibly through release of CGRP from trigeminal nerve terminals, which is capable of triggering trigeminal firing and neurogenic inflammation within the meninges (26,27,61). Experimental studies also showed that NO donors increased TRPV1 expression in an inflammatory pain model (62,63). Furthermore, systemic administration of CGRP increases the expression of TRPV1 in the TG of rats (64). Moreover, previous studies showed that NTG administration results in the release of pro-inflammatory cytokines and neurogenic inflammation that subsequently contribute to activation of TRPV1 channels (11,65). In this study, chronic NTG injection increased the mRNA expression of CGRP and TRPV1 in the TG. We suggest that NTG increased TRPV1 levels indirectly through activation of inflammatory mediators and directly through elevation of CGRP levels. Therefore, it seems that fluctuation of CGRP and TRPV1 are involved in the NTG-triggered trigeminal sensitization, and ghrelin treatment inhibits the nociceptive process through modulation of these markers. Taken together, these results highlight the importance of the interplay of CGRP and TRPV1 in the pathophysiology of migraine-related hyperalgesia.
We also demonstrated that a single i.p injection of NTG, within 3 h, resulted in a significant rise in plasma levels and mRNA expression of CGRP in the TG. However, a single dose NTG injection did not significantly change TRPV1 mRNA expression in the TG. In contrast to our findings, Nagy-Grocz et al. found that NTG 10 mg/kg after 4 hours increased TRPV1 expression in the C1–C2 segments in rats (11). The lack of change in TRPV1 mRNA expression in the TG in the acute NTG-received rats might be related to the fact that latency was not sufficient to cause significant expression changes. Although chronic administration of ghrelin (150 µg/kg/day for 11 days) significantly decreased mRNA expression of TRPV1 and CGRP levels in the TG, mRNA expression of TRPV1 and CGRP did not significantly change in the TG after a single dose of ghrelin following NTG. These results provide the first convincing evidence that the antinociceptive effect of ghrelin may be related to the reduced levels of these factors in the TG.
There is accumulating evidence supporting ghrelin and its receptors, growth hormone secretagogue receptor (GHS-R1a), being found in the brain regions involved in the field of pain control (33,36,37,43). Ghrelin exhibits antinociceptive effects in a variety of experimental models of pain, possibly through interaction with the GHS-R1a receptor and endogenous opioid system and suppression of pro-inflammatory cytokines (37,38). Moreover, administration of the ghrelin receptor agonist, GHRP-2, produces an antinociceptive effect via activation of GHSR-1α (66). In addition, Wei et al. demonstrated that co-administration of [d-Lys3]-GHRP-6, a selective GHS-R1a antagonist, and ghrelin, antagonized the antinociceptive effect of ghrelin (67). A previous study showed that activation of the mitogen-activated protein kinase (MAPK) pathway can increase the synthesis and release of CGRP in the TG neurons (68). Likewise, administration of triptan blocks the MAPK-mediated activation of CGRP gene expression (69). Zhou et al. demonstrated that ghrelin alleviates neuropathic pain via suppression of the p38 MAPK/NF-κB pathway in a chronic constriction injury model in rats (36). Furthermore, Jansen-Olesen et al. found that infusion of PACAP-38 induces the release of CGRP in the TNC (70). In addition, we have previously shown that chronic ghrelin treatment decreased the plasma concentration of PACAP (39). Meanwhile, in this study, treatment with ghrelin reduced CGRP and TRPV1 expression in an NTG-induced migraine model. Therefore, it is likely that ghrelin decreases plasma CGRP and mRNA expression via inhibition of the MAPK signaling pathway and attenuation of PACAP levels. All results suggested that the antinociceptive effect of ghrelin might be related to inhibition of CGRP and TRPV1 expression and reduction of plasma levels of CGRP.
Pain is an unpleasant experience composed of sensory discriminative and negative affective components (71). The unpleasantness of pain is frustrating and influences future decisions about selection of action (72). The emotional dysfunction and affective-motivational components of chronic pain are assessed by CPA (73,74). In the current study, the sensory component of migraine was investigated by assessment of mechanical and thermal thresholds, and the CPA paradigm was used to assess the negative affective component. Consistent with a previous report, our results indicated that NTG induced place avoidance behaviors, in which animals learned to associate one chamber with an aversive stimulus and spent less time in the chamber that was associated with the pain experience (10). Nevertheless, animals in the NTG + ghrelin group showed a preference for the chamber that had been previously paired with ghrelin treatment, suggesting that ghrelin might alleviate the migraine-induced negative emotional state. Increasing evidence has demonstrated that alleviation of the painful state is rewarding and associated with activation of the mesolimbic dopaminergic circuit, which is involved in reward and motivation-related behaviors (75–77). Moreover, previous studies have shown that ghrelin interacts with brain reward pathways and modulates the activity of the mesolimbic dopaminergic system (78,79). Therefore, ghrelin may prevent conditioned place aversion via alleviation of NTG-induced hyperalgesia or by stimulating reward pathways.
Another finding of the present study that provides additional support for the NTG model of migraine is the body weight change. In the vehicle group, food intake and body weight gradually increased with time. However, animals in the NTG group demonstrated unusual patterns of food intake and body weight, and showed lower food intake and body weight compared to the vehicle group. On the other hand, chronic administration of ghrelin in intact animals (the ghrelin group) remarkably increased food intake and body weight during the experiment. In addition, chronic ghrelin treatment in the NTG-receiving animals improved food intake and consequently reversed body weight loss. These results are parallels to the clinical picture of migraine, and may support the validity of this model. Consistent with the present study, Sufka et al. showed that NTG produced a significant lower body weight gain than control animals over two weeks (80). Nausea and reduced appetite are common symptoms of migraine, which lead to less food consumption and weight loss in migraineurs (5). As a result, it seems that failure to gain weight in the NTG group is possibly due to NTG-induced nausea, which modifies food intake and inhibits weight gain in rats. Conversely, chronic ghrelin treatment amended food intake and body weight gain in the NTG + ghrelin group. In line with our results, previous studies demonstrated that peripheral or central administration of ghrelin to rodents increases food intake and body weight (81–83). To our knowledge, this is the first report that ghrelin improves food intake and body weight in the chronic migraine model.
Conclusion
Our results provide the first evidence that ghrelin can reduce both plasma CGRP levels and inhibit CGRP and TRPV1 mRNA expression in the TG. These results suggest that ghrelin may have beneficial effects against migraine pain sensory and negative affective components. We suggest that the effectiveness of ghrelin in treatment of chronic migraine may be due to its ability to decrease migraine biomarkers. Further experiments are necessary to clarify the underlying mechanisms.
Footnotes
Article highlights
Chronic intermittent NTG injection activates the trigeminal system, which is reflected by up-regulation of mRNA expression of migraine biomarkers (CGRP and TRPV1) and hyperalgesia.
Ghrelin improved food intake and body weight gain in chronic migraine induced by NTG.
Chronic ghrelin treatment attenuated mechanical and thermal hypersensitivity, and reversed CPA induced by chronic NTG.
Chronic ghrelin treatment down-regulated CGRP and TRPV1 mRNA expression in the trigeminal ganglion and attenuated the plasma concentration of CGRP.
Acknowledgement
We are especially grateful to Dr. Javad Mahmoudi for his technical assistance.
