Abstract
Background and objective
The present study aimed to investigate the effects of selective calcitonin gene related peptide (CGRP) receptor antagonist (MK-8825) on cortical spreading depression (CSD) induced pain behavior and anxiety in freely-moving rats, and neuronal activation in the correlated anatomical regions.
Methods
CSD was induced while keeping all meningeal layers and BBB intact and MK-8825 was administered in two different doses. Regional cerebral blood flow (rCBF), arterial pressure and DC shift were recorded. Behavioral studies were conducted in freely-moving rats. Spontaneous behavior, mechanical allodynia, ultrasonic vocalization, and anxiety were evaluated. Immunohistochemistry of c-fos, CGRP, calcitonin receptor like-receptor (CLR) and receptor activity modifying protein 1 (RAMP1) were studied.
Results
MK-8825 did not block DC shifts in the cerebral cortex and accompanied hemodynamic response. CSD significantly induced freezing and grooming behavior in freely-moving rats. MK-8825 reversed increased episodes of freezing, grooming, wet dog shake and head shake behavior. MK-8825 increased CSD-induced reductions in von Frey thresholds, but did not change elevated plus maze results. MK-8825 blocked c-fos induction by CSD in the brainstem trigeminal nucleus caudalis (TNC) and reticular nucleus of thalamus (TRN) but not in the amygdala. Immunofluorescence analysis showed no co-localization of CGRP, CLR or RAMP1 with c-fos positive cells.
Conclusion
CGRP receptor antagonist MK-8825 dose dependently attenuated CSD-induced trigeminal nerve mediated pain response without altering CSD waves and accompanied rCBF response. While blocking TNC activation, MK-8825 did not exert any effect on amygdala and anxiety behavior. CGRP receptor antagonists may also modulate thalamo-cortical gating.
Introduction
Migraine is the most prevalent neurovascular disorder in the population worldwide. Migraine affects many regions in the nervous system; the activation of the trigeminal pathway is involved in headache symptoms. Several aspects of migraine still warrant clarification, such as the involvement of subcortical structures in the generation and/or abortion of an attack (1). Cortical spreading depression (CSD) waves are considered as neurobiological mechanisms underlying migraine attacks. CSD is a cortical phenomenon that involves a spreading wave of depolarization in neurons and glial cells, with massive release of glutamate and other nociceptive and vasoactive substances resulting in prominent changes in local cerebral blood flow and metabolism (2). CSD is a counterpart of migraine aura, and is able to induce lateralized headache as evidenced by activation of both perivascular trigeminal nerve endings and trigeminal second order neurons in the brain stem (3). Though behavioral changes associated with head pain are not obvious following a single CSD, functional responses of CSD waves in freely-moving rodents are consistent with pain behavior and accompanied by activation of the trigeminal pain nucleus, amygdala, and thalamic reticular nuclei among other structures (4,5). Involvement of the thalamic reticular nucleus (TRN) by CSD is critical because of its gatekeeper role for sensory flow to the cerebral cortex (6).
Calcitonin gene-related peptide (CGRP) is an essential molecule in the development of migraine headache. CGRP is a very strong vasodilator neuropeptide in the cranial vasculature and plays a part in nociception, motor function, secretion, and olfaction (7,8). CGRP is abundantly expressed in the trigeminal nerve, trigeminal brainstem nucleus, and other CNS structures such as the cerebellum, amygdala and striatum. Targeting CGRP receptor antagonism is a breakthrough in migraine treatment (7,9).
CGRP receptor antagonist drugs effectively abort migraine attacks in clinical trials (10,11). The site of action for CGRP receptor antagonism is still debated, though multiple levels of localizations have been demonstrated for CGRP receptors in the CNS (12,13). While the gepant class of CGRP receptor antagonist consists of small molecules with limited access to the blood-brain barrier, humanized monoclonal antibodies against CGRP receptors are large molecules without any penetration to the CNS (7).
Studying CSD in awake animals, as a relevant model of migraine, is crucial for translational migraine research, because it is known that sedatives and anesthetic agents depress CNS activities (14). CSD waves, which are an intrinsic mammalian brain response to noxious stimuli, have definitive peripheral and central effects leading to the trigeminal pathway activation. CSD-induced neuronal and vascular reactions are mediated by various molecules including CGRP. To understand the role of CGRP receptor blockade in a CSD model of migraine, MK-8825, a highly selective and potent antagonist at rat CGRP receptors, is employed. MK-8825 is capable of reversing pain behavior and central sensitization elicited by various models such as nitroglycerin infusion, temporomandibular dysfunction, and constriction injury to the trigeminal nerve (15–17). MK-8825 on CSD has only been studied before in brain slices (18). Therefore, we aimed to examine the consequences of MK-8825 on CSD-induced neuronal, vascular responses and pain behavior in rats. Our goal was particularly to test the potency of MK-8825 in reversing the neuronal activation in the trigeminal pain nucleus in the brain stem (TNC), amygdala, and reticular nucleus of the thalamus (TRN) in freely-moving rats.
Methods
Animals
Experiments were approved by Gazi University Animal Studies Ethical Committee (G.U. ET-09.097/ET-11.061) and conducted according to the National Guidelines for the Care and Use of Laboratory Animals. All surgical procedures were performed under anesthesia.
Male Wistar rats, apparently healthy, weighing 200–250 g, were provided from the Laboratory Animal Breeding and Experimental Research Facility of Gazi University. Rats were housed where the temperature of the environment was kept at 22 ± 2℃ and were subject to a 12 h daylight and 12 h dark cycle with ad libitum access to food and water.
Surgical procedure and CSD induction
Deep anesthesia was induced with a mixture of intraperitoneal (i.p.) ketamine (Ketalar®, Pfizer, 60 mg/kg) and xylazine (RompunTM, Bayer, 8 mg/kg). Anesthesia depth was titrated based on the heart rate and animals hind leg reaction to painful pinch. Body temperature was stabilized at 37 ± 0.5℃ during surgical procedure. Animals were placed in a stereotaxic instrument (Stoelting Wood Dale, IL, USA). A burr hole was drilled on the right side (bregma −7 mm, lateral 3 mm), and the dura mater was kept intact following an incision in the scalp (4,5). A plastic tube was sealed over the burr hole and the opening of the tube was kept closed with a rubber cap. All behavioral experiments were conducted at the same time of day, following 6 days of a recovery period after the surgery. Two rats were excluded after the surgery because of problems with the tubing. Two more animals were excluded from the study because of failure to obtain whole sections from TNC + TRN + Amygdala sections to analyze along with behavior. Topical KCl over the intact dura mater was chosen to induce multiple CSD, as single CSD by topical NMDA did not induce prominent pain behavior (4). In the preliminary experiments CSD was not consistently induced by 1 M KCl application through the tubing (n = 6). When the cap was removed, the applied KCl solution was diluted due to the fluid accumulated in the tubing. Therefore, 10 µL solution of 3 M KCl was used to fill the tubing without handling rats, as described before (5).
Regional cerebral blood flow (rCBF) measurement
Male Wistar rats weighing 200–250 g were anesthetized with urethane (Sigma-Aldrich, 1.2 g/kg i.p.), and the femoral artery was cannulated for continuous blood pressure (BP) measurement throughout all experiments. The temperature of the rat was maintained at 37℃ during all surgical procedures using a homoeothermic blanket (Harvard Apparatus, Holliston, MA, US). For electrophysiological and blood flow recordings, rats were placed in a stereotaxic frame (Stoelting Co. Wood Dale, IL, USA) Under saline cooling, a burr hole (∼1 mm diameter) was drilled over the right hemisphere at the following coordinates from bregma: 5.6 mm posterior and 3.5 mm lateral for CSD induction. The dura was carefully kept intact to minimize trauma. Two separate skull regions, approximately 1 mm posterior to the burr hole, were thinned using an abrasive drill bit under saline cooling to place the laser Doppler flowmeter probe (OxyLab LDF; Oxford Optronix, Abingdon Oxfordshire, UK) and the metal electrode for electrocorticogram (ECoG) recordings (Batiray EX-1, Kaldiray EX-2C; YSED, Ankara, Turkey). A thin layer of mineral oil was spread over the skull to prevent drying. In a group of animals, rCBF and BP were recorded for 30 minutes before and after the i.p. injection of MK-8825 (30 mg/kg and 100 mg/kg, n = 5). In a second group of animals, a single CSD was elicited using a pinprick, following the baseline recordings of rCBF. Two further CSDs were precipitated by the pinprick method 30 minutes following 30 and 100 mg/kg i.p. injection of MK-8825 in the same animal (n = 5). The area under the curve of CSD-induced blood flow changes before and after the injection of MK-8825 were measured and compared.
Behavior
Behavior was tested 6 days following recovery from surgery. All behavioral experiments were carried out at a pre-defined time period in a given day, in cages where rats were free to move. Following KCl application, behavioral parameters were obtained for 20 min. Rat behaviors of rearing, sleeping, grooming, freezing, wet dog shake, and head shake, besides food and water consumption, were automatically recorded via a behavioral analysis system (Laboras; Metris, Hoofddorp, The Netherlands). The behavioral analysis system used was a standard cage, equipped with the system, which is able to detect and classify behavioral variables based on vibrations produced by the movements of the rats. Video recordings were captured to investigate the details of immobility periods (see details in 4,5). The output of the system and the video recordings were assessed to differentiate freezing behavior from resting or sleep. In three separate groups, the behavioral experiments were carried out: i) CSD + saline group (n = 9), ii) CSD + MK-8825, 30 mg/kg group (n = 9), iii) CSD + MK-8825, 100 mg/kg group (n = 8). MK-8825 was shown not to change basal behavioral and pain responses in rodent orofacial pain models (16,17).
Anxiety responses in rats were assessed by elevated plus maze (EPM) (19), which is a reliable method in rodents (20). Each rat was placed in the center, facing the same closed arm, and spontaneous behavior was documented for 5 minutes. Total entry numbers and total time spent in both arms were evaluated. The elevated plus maze was cleaned meticulously with alcohol after experiments. Rats were subjected to EPM test only once.
Allodynia was examined by using von Frey filaments. Rats were acclimatized to platforms 40 cm high, and von Frey filaments were applied in the periorbital region to assess tactile allodynia. Von Frey filaments were tested by using the “up and down method” (21), starting with a force of 4 g. Avoidance of rats from von Frey filament and ipsilateral head grooming was considered as a positive response. Baseline values were obtained a day before CSD, and test values were obtained 30 minutes after CSD.
Ultrasonic vocalization (USV) calls
Rats can communicate in the ultrasonic range and (>20 kHz) (22). The 15–100 kHz band of ultrasonic sounds was recorded during experiments and analyzed by a USV detector system (Sonotrack, Metris, The Netherlands). The 22–27 kHz range of vocalizations of rats was considered as a response to pain or a distressing event, as described before (4).
Drugs
CGRP receptor antagonist MK-8825 (2-[(8R)-8-(3,5-difluorophenyl)-8-methyl-10-oxo-6,9-diazaspiro[4.5]dec-9-yl]-N-[(6S)-2-oxo-1,2,5,7-tetrahydrospiro [cyclopenta [b] pyridine-6,3-pyrrolo [2,3-b]pyridin]-3-yl] acetamide), was kindly provided from Merck Sharp Dome. CGRP receptor antagonist MK-8825 (30 mg/kg or 100 mg/kg) or its vehicle alone (0.9% NaCl) was given intraperitoneally, 30 min before the induction of CSD.
Immunohistochemistry and immunofluorescence analysis
At the end of each experiment, rats were sacrificed 2 hours following CSD induction. Coronal brain sections and axial brainstem and upper cervical sections were obtained. Tissue sections 50 µm thick were taken every 150 µm, and processed for c-fos immunostaining by the free-floating method (for details, see (3–5)). For detecting and counting the neuronal activation, the avidin-biotin-peroxidase method was employed for c-fos immunoreactivity. A blinded observer confirmed the existence of CSD in the cerebral cortex and then counted c-Fos immunoreactive neurons in different brain regions. Each region was sampled according to rat atlas (23); TNC (obex to −2.5 mm), amygdala (−2.0 mm to −3.3 mm), reticular nucleus thalamus (bregma, −2.2 mm to −4.6 mm). At least eight to nine tissue sections for each region were evaluated for c-fos, under bright-field microscopy. In the TNC region, c-fos positive cells were counted only in lamina I and II.
Detailed description of primary and secondary antibodies.

Data analysis
Data was analyzed by SPSS® (Statistical Package for Social Sciences) for Windows Version 11.5. Statistical significance was tested with one-way ANOVA and post hoc Tukey test. The results were presented as mean ± standard error of the mean. p < 0.05 was considered as significant. Regional CBF measurements were tested and presented as percentage changes with respect to baseline values.
Results
rCBF and electrophysiology
Baseline mean blood pressure and basal rCBF recording were not altered by the infusion of MK-8825 30 mg/kg or MK-8825 100 mg/kg. Mean arterial blood pressure values of baseline, MK-8825 (30 mg/kg) and MK-8825 (100 mg/kg) groups were (73.1 ± 5.5 mmHg), (74.8 ± 4.8 mmHg), and (78.1 ± 4.7 mmHg), respectively. MK-8825 infusion did not significantly alter the rCBF (Figure 1(a) –(c)). CSD induction elicited a typical rCBF increase (Figure 1(c)). MK-8825 30 mg or 100 mg/kg did not inhibit CSD. MK-8825 also did not alter CSD-induced rCBF response (Figure 1(b)). CSD-induced rCBF increase was measured as the area under the curve and compared with the first CSD-elicited rCBF response (Figure 1(b), (c)).
The effect of MK-8825 on basal regional CBF in anesthetized rats. (a) 30 and 100 mg/kg MK-8825 did not significantly increase the basal rCBF in rats. (b) Comparison of rCBF responses (AUC) to CSD. MK-8825 did not change the rCBF response to CSD at either dose. Median (horizontal line), minimum and maximum data points (whiskers) are shown. (c) Representative tracings show recordings of arterial blood pressure, electrocorticogram, and laser Doppler flowmetry measurements following the induction of CSD (arrowhead) by pinprick, after i.p. injection of MK-8825 (30 mg/kg). Electrophysiology and rCBF data are digitally filtered (Low pass: 5 Hz).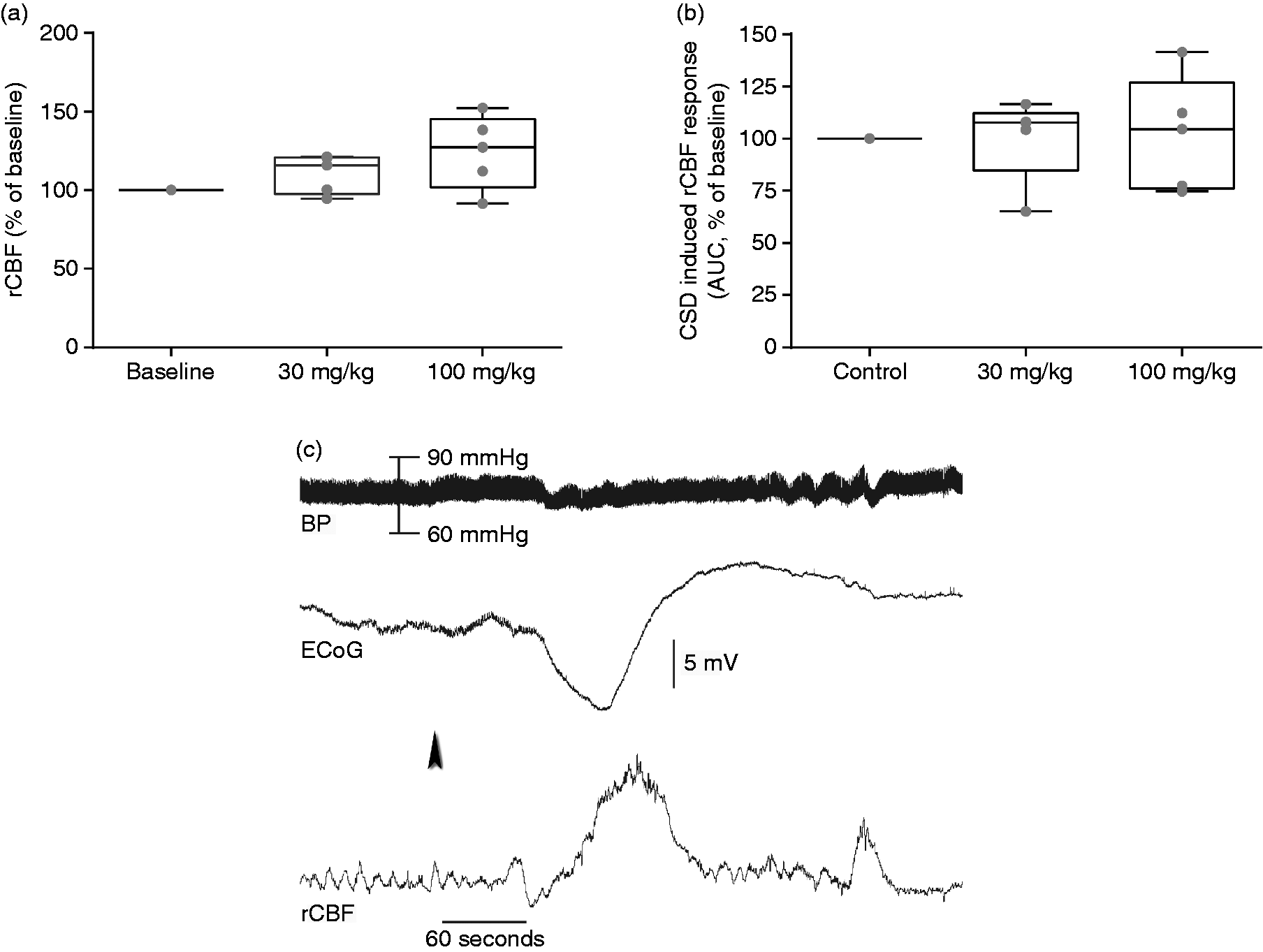
CSD and behavior
CSD-induced pronounced freezing episodes in freely-moving rats, consistent with previous reports (4,5). Freezing was the most noticeable behavioral component of CSD. Freezing was defined when the rat abruptly stopped moving, walking, rearing, grooming, or eating and stared at an uncertain spot. During the freezing, eyes were open and blinking. Episodes of freezing behavior were reproducible and clustered within 10 minutes following CSD. Administration of MK-8825 in 30 mg/kg and 100 mg/kg doses significantly reduced the total duration of CSD-induced freezing episodes (Figure 2(a)).
Behavioral alterations: (a) Cortical spreading depression (CSD) increased episodes of freezing behavior. Total freezing time enhanced by CSD was significantly reversed by MK-8825 in a dose-dependent manner. (b) CSD-induced grooming behavior was reduced by MK-8825 administration. (c) The total number of wet dog shake (WDS) behaviors induced by CSD was significantly reduced by MK-8825. (d) The total number of head shake (HS) behaviors induced by CSD was significantly reduced by MK-8825. (e) CSD significantly lowered the thresholds for VF filaments, which were reversed by MK-8825. (f) Total duration spent in the closed arms of the elevated plus maze was not significantly altered by MK-8825 administration. Mean + standard error of the mean, *p < 0.05, **p < 0.01.
Grooming behavior in rats could indicate a pain perception and/or anxiety (25). The total duration of both body and head grooming induced by CSD was significantly reduced in both MK-8825 groups (Figure 2(b)). Additionally, wet dog shakes, and head shakes, were significantly reduced in the MK-8825 groups (Figure 2(c), (d)). Other behavioral parameters, such as total eating/drinking, rearing and clockwise or counter clockwise turning were not changed by administration of either dose of MK-8825.
In the CSD + saline group, von Frey thresholds 30 minutes following CSD were significantly reduced compared to baseline values. Administration of MK-8825, 30 mg/kg and MK-8825, 100 mg/kg statistically significantly reversed mechanical allodynia (Von Frey) thresholds lowered by CSD (Figure 2(e)). USV recordings revealed that pain calls in the 23–27 KHz band were emitted only in three rats from each group during mechanical allodynia examination.
EPM test results revealed that the total duration spent in the closed arms was higher in all groups. Either dose of MK-8825 reversed the duration spent in the closed arms (Figure 2(f)). The ratio of total entrance to closed arms/total entrance to all arms was comparable. The differences were not statistically significant.
c-fos immunohistostaining
Induction of pronounced c-fos immunoreactive neurons widely distributed in all cortical layers of one hemisphere was detected as a hallmark of CSD. Such a c-fos staining pattern was never observed on the contralateral cerebral cortex in any group. c-fos expression was scarce on the contralateral side and c-fos positive cells were mainly found in all layers of the cerebral cortex. c-fos activation in the cerebral cortex was massive, and always unilateral.
c-fos expression was significantly induced by CSD in the ipsilateral brainstem trigeminal pain nuclei (TNC) (Figure 3(a)). CSD induced c-fos activation was statistically significantly decreased by MK-8825 pretreatment in a dose dependent manner (Figure 3(a)). Number of c-fos positive cells were significantly lower on ipsilateral (CSD side) lamina I-II in both CGRP receptor antagonist groups compared to saline group.
c-fos activation induced by cortical spreading depression (CSD): (a) CSD significantly increased the c-fos positive cell number in lamina I and II of the trigeminal nucleus caudalis (TNC), ipsilaterally. MK-8825 dose dependently blocked CSD-induced c-fos expression in the nociceptive laminae. (b) the c-fos positive cell number in the amygdala significantly induced by ipsilateral CSD. MK-8825 30 mg/kg dose reduced total stained cell number, though the difference did not reach statistical significance. No reduction in the total number of c-fos was observed following MK-8825 100 mg/kg dose administration. (c) The total number of c-fos immunoreactivity in the thalamic reticular nucleus was increased by ipsilateral CSD. The statistically-significant decrease in the total c-fos positive cell number obtained by 30 mg/kg CGRP receptor antagonist MK-8825 administration was reversed when the higher dose of 100 mg/kg was used. Mean ± standard error of the mean, *p < 0.05, **p < 0.01, ***p < 0.001.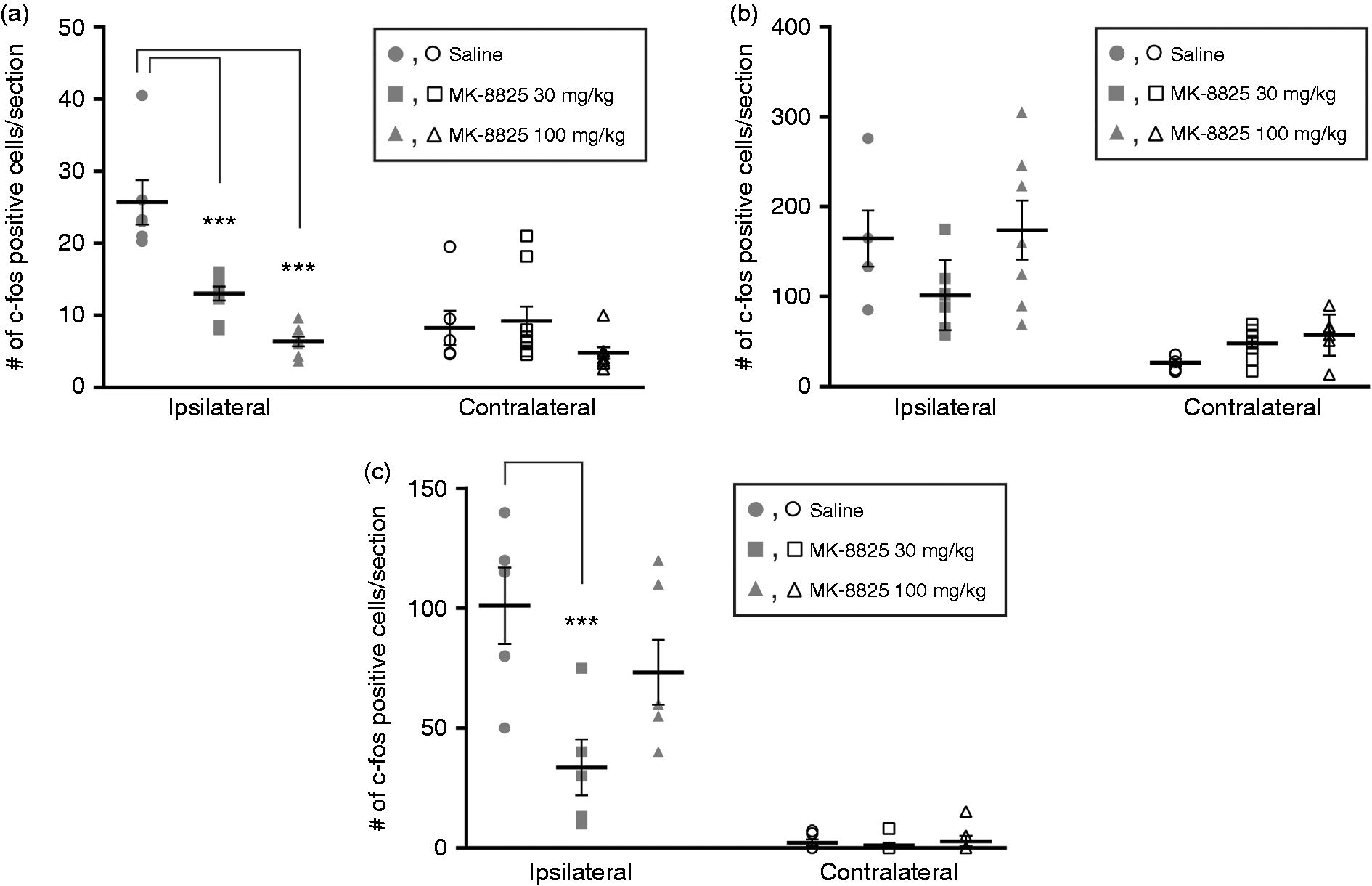
CSD induced pronounced c-fos expression in the amygdala ipsilateral to CSD (Figure 3(b)). Activation of amygdaloid nuclei by CSD was not influenced by either MK-8825 30 mg/kg or MK-8825 100 mg/kg pre- treatment (Figure 3(b)). Both doses of MK-8825 were effective in suppressing TNC activation, though none of them reversed amygdala activation (Figure 3(b)).
c-fos induction was detected prominently in the TRN ipsilateral to CSD in freely-moving rats. The topographic distribution of c-fos positive neurons in TRN was in dorsal TRN including visual sector and not observed in the ventral TRN. This pattern was consistent with the previous report (5).
MK-8825 administration abrogated CSD induced c-fos activation in the TRN only in the 30 mg/kg dose. MK-8825 100 mg/kg did not revert increased the c-fos positivity in TRN (p > 0.05).
CGRP, CLR and RAMP1 immunohistochemistry
CGRP expression was detected in nerve fibers in indusium griseum (IG), lateral septal nucleus (LSI) and lateral parts of the caudate putamen (CPu). The immunoreactive fibers appeared to reach the central and lateral amygdaloid nucleus, where some fibers extended to the cortex. No c-fos positive neurons were observed in areas holding CGRP expression. Interestingly, CGRP immunoreactive fibers were often observed close to the c-fos immunoreactive cortex (Figure 4). CLR was frequently found in fiber structures and capillaries. RAMP1, however, showed less frequent immunoreactivity.
CGRP and receptor components in cortex. (A) c-fos and CGRP/CLR/RAMP1 in cortex.
Discussion
The effect of MK-8825, a selective CGRP receptor antagonist given systemically, on CSD-induced alterations was studied in awake rats. The behavioral responses of freezing, grooming, and decreased locomotor activity to CSD in freely-moving rodents were consistent with previous reports (4,5). CGRP receptor antagonist MK-8825 reversed CSD-induced behavioral alterations associated with pain perception such as grooming, freezing episodes, rapid shaking behavior of the body and reduced thresholds of tactile allodynia. In support of behavioral alterations associated with pain, MK-8825 dose dependently blocked c-fos expression by CSD in the ipsilateral trigeminal pain nucleus. Additionally, MK-8825 was effective in reducing thalamic reticular nucleus (TRN) activation induced by CSD in fully-awake, freely-moving rats. On the other hand, MK-8825 did not alter CSD-induced anxiety behavior and amygdala activation. MK-8825 did not block CSD waves in the cortex. MK-8825 administration per se did not change basal hemodynamic responses or rCBF responses associated with CSD. The presented findings provided the evidence that MK-8825 could inhibit the trigeminal pain induced by CSD without blocking CSD waves and altering hemodynamic responses. CGRP receptor antagonist MK-8825 can abort nociceptive transmission centrally at the TNC, and also modulate at the thalamic level.
Despite the claims that MK-8825 may inhibit CSD in in vitro brain slice preparations (18), it did not block CSD waves elicited by either KCl or pin prick application in our study on awake animals. It is important to note that MK-8825 may block CSD waves when BBB is lacking, as shown in in vitro brain slices. However, when we used the CSD induction method while keeping all meningeal layers and BBB intact, MK-8825 did not influence CSD waves. Moreover, MK-8825 alone did not change the basal rCBF in a statistically-significant manner, nor did it change the CSD-induced rCBF increases in anesthetized animals. These findings are in accordance with previous studies reporting that the gepant class of CGRP receptor antagonists do not change the hemodynamic properties in humans (26,27). In our experiments, MK-8825 efficiently blocked CSD-induced trigeminal pain (Figures 1, 2) without blocking CSD waves and altering vascular dynamics, as revealed by direct measurement of mean arterial pressure and laser Doppler flowmeter recordings of rCBF.
Activation of the trigeminovascular system during a migraine attack in humans leads to throbbing headache triggered by routine physical activity. Therefore, freezing episodes in rodents might reflect a defense mechanism to restrict head and body movements. Freezing occurred especially within the first 20 minutes after induction of CSD. Early start of freezing, usually within the first five minutes, suggests its association with the propagation of CSD waves. Significantly shortened freezing duration along with reduced grooming, body shakes, allodynia and c-fos positivity in the TNC by MK-8825 verifies that freezing was related to the sensation of trigeminal-mediated pain. Additionally, activation of the amygdala could contribute to freezing behavior due to fear and/or anxiety response. Failure of MK-8825 to reverse c-fos expression in the amygdala and EPM results suggests that the role of the amygdala in freezing behavior is not secondary to pain perception and its contribution to freezing behavior is negligible. Alternatively, freezing episodes in rats may manifest as absence seizures subsequent to TRN involvement. However, that possibility was investigated in detail and epileptic origin was excluded in CSD-induced freezing behavior. Cataleptic freezing from basal ganglia involvement is unlikely due to normal muscular tonus and interruption of freezing by sensorial stimulus. The brain regions involved in grooming and shaking behavior of the body and head are not known, though they are related to perception of pain.
Grooming is considered a behavioral indicator of pain sensation in rats, linked to various stressors. Increased grooming behavior was observed in rats with development of sensitization to chronic pain patterns. Our study demonstrated that CSD increased the grooming behavior, which was significantly decreased by CGRP receptor antagonists. Increased grooming behavior following intraventricular administration of CGRP (28) supports the observed anti-nociceptive effect. Though head and body shakes are not directly related to CSD, they are manifested in response to physical stress and associated with the activation of the 5-HT2A receptors (29). The basal ganglia has been implicated in grooming behavior, and the locus coeruleus in shakes. In our study, head shake was observed in the CSD-induced rats and was significantly decreased in the rats receiving the CGRP receptor antagonist. Therefore, it may be inferred that head shake and wet dog shake behavior may occur during a headache induced by CSD.
CGRP receptor antagonist MK-8825 significantly suppressed the CSD-related allodynia in the periorbital region. Cutaneous allodynia develops during migraine attacks, and mechanical and thermal thresholds are lowered. Development of cutaneous allodynia 30 minutes after CSD was also reported. The presented findings are consistent with the allodynia during headache in migraine patients. Lowered mechanical thresholds in the periorbital region following CSD were reversed by either dose of MK-8825. Behavioral results were consistent with the reports that MK-8825 effectively relieved trigeminal-mediated pain behavior in rodents following nerve constriction injury, temporomandibular inflammation or nitroglycerin infusion (15–17).
Mechanical or chemical stimulation of intra- or extracranial structures innervated by the trigeminal nerve can induce the c-fos protein in the brain stem trigeminal neurons. Following induction of CSD, c-fos induction occurs in the ventrolateral section of lamina I and II, where ophthalmic branches terminate (3). Following administration of CGRP receptor antagonists, a dose-dependent decrease in the c-fos induction was observed in the trigeminal pain nuclei, the TNC. In conclusion, the present findings clearly demonstrated the antinociceptive action of MK-8825, as the CGRP receptor antagonist effectively reversed the CSD-induced behavioral parameters associated with pain, increased sensorial thresholds, and blocked neuronal activation in the brain stem trigeminal nucleus.
The involvement of the amygdala during CSD is significant, since it interacts with other cerebral regions related to vision, pain, the senses, and autonomic functions. The amygdala is conventionally associated with fear/anxiety behavior. Therefore, involvement of the amygdala may account for anxiety and freezing behavior induced by CSD. The amygdala is an important nociceptive relay station, considered as a part of the medial pain pathway (30). However, while MK-8825 dose-dependently blocked TNC activation, freezing, grooming behavior and allodynia, none of the doses reversed amygdala activation and the anxiety response. Therefore, our results demonstrate that amygdala activation by CSD was not downstream to trigeminal mediated pain perception. The latter notion is concordant with previous reports that CSD-induced amygdala activation was not reversed by anti-migraine drugs such as sumatriptan or valproic acid (4,5).
Amygdala activation may reflect a direct invasion of CSD waves. Although the data is limited for penetrability of MK 8825 through BBB, the drug was effective centrally, as reflected in the TNC activation and pain thresholds. Even though CGRP was associated with nociception and its receptors were abundant in the amygdala (31), its antagonism by MK-8825 did not influence CSD-induced amygdala activation. The functional effects of CGRP receptor antagonists on the amygdala regarding anxiety and/or nociception, however, are not fully explored yet.
Involvement of TRN by CSD is very critical to understanding the sensorial disturbances seen in migraine. The known functions of TRN includes a “gatekeeper” role for sensory information flow to the cortex, an “attentional searchlight” to focus on a certain stimulus, lateral inhibition, and maintenance of sleep (6). TRN has been linked to pain conditions (32,33). Recently, TRN activation during CSD in freely-moving rats was demonstrated, and CSD waves were able to propagate into TRN (5). Activation of dorsal TRN predominantly in the hemisphere ipsilateral to the CSD was concordant with the previous report. The CSD-elicited TRN activation was totally blocked by sodium valproate while alleviating pain responses (5). MK-8825 in the present study significantly reduced TRN activation in the 30 mg/kg dose in parallel to its effects on trigeminally-mediated pain parameters. TRN c-fos reduction was parallel to reduced TNC activation and pain response in the 30 mg dose of MK-8825. Burst firing of the thalamus generating spindles plays a crucial role in gating sensory inputs, and is linked to nociception and neuropathic pain (32,33). Oscillations in the 7–15 Hz spindle range detected during CSD-induced freezing behavior suggested perception of pain in awake rats (5). Significant reduction in freezing episodes as well as inhibition of c-fos activation in both the brain stem trigeminal pain nucleus and thalamic reticular nucleus by MK-8825 administration supports the above notion. The latter effect on TRN was disinhibited by MK-8825 in the 100 mg/kg dose, which very efficiently blocked the TNC response and pain behavior. The dual effect of MK-8825 in two doses on TRN must be investigated with further studies. It is also possible that the discrepancy may also arise from the amygdala activation, as direct projections from the amygdala to TRN were recently revealed (34). C-fos induction in the amygdala, un-affected by MK-8825 administration, could drive dorsal TRN activation, and may reflect an attention to emotional response in freely-moving rats.
Chronic blockade of CGRP receptors may reduce the activation of the amygdala and other central structures particularly in the medial pain pathway; even though the humanized antibodies do not cross the BBB, their peripheral effect via the trigeminal system may silence central sensitization. Functions of CGRP are mainly mediated by either CGRP receptors (composed of RAMP1 + CRL/calcitonin receptor like receptors) or AMY1 receptors (formed by RAMP1 + CTR/calcitonin receptors) (35). Both receptors are abundantly found in the TNC, and mediate antinociceptive actions as evidenced by significantly blocked c-fos expression in the nociceptive laminae. However, in the cerebral cortex and thalamus, CSD-activated c-fos protein expression did not occur in the RAMP1/CRL positive neurons. Likewise, the amygdala’s unresponsiveness to MK-8825 may occur due to activations of discrete cells. A lack of CGRP receptor component investigation in the amygdala, TRN and TNC is one of the limitations of the presented study. The strength of the study is the induction of CSD while keeping the meninges and BBB intact, and the avoidance of experiment-related passage of systemic MK-8825 to central structures via brain damage.
In conclusion, CGRP receptor antagonist MK-8825 dose dependently attenuated CSD-induced trigeminal nerve mediated pain response without altering CSD waves and the accompanying rCBF response. MK-8825 did not directly block c-fos induction by CSD, as c-fos positive cells were not co-localized with CGRP receptor elements of CRL or RAMP1. CSD-induced TRN involvement in consciously-behaving rats was also modulated by MK-8825, suggesting that thalamocortical gating could be a novel target for CGRP receptor antagonists. While blocking TNC activation, MK-8825 did not exert any effect on the amygdala and anxiety behavior. The differential effect of MK-8825 on central structures needs further studies, with particular interest in their possible anti-nociceptive role.
Article highlights
CGRP receptor antagonist MK-8825 attenuated CSD-induced trigeminal nerve mediated pain response without blocking SD waves in the cortex and the accompanying rCBF response. While blocking TNC activation, MK-8825 did not exert any effect on the amygdala and anxiety behavior. The differential effect of MK-8825 on central structures needs further studies with particular interest in their possible anti-nociceptive role. MK-8825 did not directly block c-fos induction by CSD, as c-fos positive cells were not co-localized with CGRP receptor elements of CRL or RAMP1. Thalamo-cortical gating could be a novel target for CGRP receptor antagonists.
Footnotes
Acknowledgement
The study was supported by the research grant GU-01-2009-41 from Gazi University. The authors are thankful to Merck & Co. Inc for kindly providing MK-8825 and Tony Ho and Christopher Salvatore for critical discussions about MK-8825. The authors also gratefully acknowledge the valuable critiques of Didem Akcali, Doga Vuralli and Yildirim Sara. Part of the study was presented at the EHMTIC 2014.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
