Abstract
Objective
To systematically review the association between migraine and rosacea.
Background
Migraine is a complex disorder with episodes of headache, nausea, photo- and phonophobia. Rosacea is an inflammatory skin condition with flushing, erythema, telangiectasia, papules, and pustules. Both are chronic disorders with exacerbations of symptoms almost exclusively in areas innervated by the trigeminal nerve. Previous studies found an association between these disorders. We review these findings, provide a meta-analysis, and discuss possible pathophysiological commonalities.
Methods
A search through PubMed and EMBASE was undertaken for studies investigating the association between all forms of migraine and rosacea published until November 2016, and meta-analysis of eligible studies.
Results
Nine studies on eight populations were identified. Studies differed in methodology and diagnostic process, but all investigated co-occurrence of migraine and rosacea. Four studies were eligible for meta-analysis, resulting in a pooled odds ratio of 1.96 (95% confidence interval 1.41–2.72) for migraine in a rosacea population compared to a non-rosacea population.
Conclusion
Our meta-analysis confirmed an association in occurrence of migraine and rosacea. Future studies should specifically investigate possible shared pathophysiological mechanisms between the two disorders.
Introduction
Migraine is a complex neurovascular disorder characterised by recurrent episodes of headache accompanied by nausea, photo- and phonophobia (1). Rosacea is an inflammatory facial skin condition characterised by episodes of flushing, intermittent or chronic centrofacial erythema, telangiectasia, papules, and pustules (2). In subgroups, patients may suffer from phymatous changes or ocular complaints including xerophthalmia and conjunctivitis. Cutaneous erythema can be accompanied by secondary xerosis, as well as burning and stinging pain (3,4).
Migraine and rosacea are both chronic relapsing disorders with paroxysmal and highly disabling symptoms arising in the trigeminal innervated area (1,2,5,6). A number of shared features between migraine and rosacea include similarities in disease prevalence reaching 15% for migraine and 2–20% for rosacea, increased risk among white-skinned individuals and in young to middle-aged women (6–9). However, migraine prevalence is highest between the third and fifth decennium, often followed by diminishment of symptoms, whereas rosacea might worsen or even progress between subtypes into old age (7,10,11). To date, no biological markers exist for the diagnosis of either disorder and both are diagnosed according to internationally-agreed guidelines based on clinical signs and symptoms (Figure 1). Importantly, some clinical manifestations, along with epidemiological data, suggest a rosacea/migraine co-occurrence (12,13), possibly pointing towards the existence of a pathophysiological and clinical overlap between the two disorders.
Diagnostic criteria for rosacea.
In this systematic review article, we reviewed data on the association between migraine and rosacea, focusing on epidemiology, clinical aspects, and pathophysiology. Additionally, we conducted a meta-analysis on the association between rosacea and migraine.
Methods
Search strategy and selection criteria
We searched PubMed and EMBASE for articles on migraine and rosacea with information on etiology, pathophysiology, and treatment options for the two diseases. Keywords used in the PubMed search were “rosacea” AND “migraine” combined with “epidemiology”, “pathophysiology”, “treatment”, “neurogenic”, “inflammation”, “vascular”, “sensitization”, “dysregulation”, “augmented” or “immune”. The EMBASE search was conducted using a series of seven search strings as follows: 1) ”exp migraine/”; 2) ”exp rosacea/”; 3) ”(review or meta-analysis or metaanalysis).m_titl.”; 4) ”(animal/or nonhuman/) not human/”; 5) ”1 and 2”; 6) ”5 not 3”; 7) ”6 not 4”. This series was used to identify all articles on migraine and rosacea, leaving out reviews, meta-analyses, and animal studies.
The search was completed on 15 November 2016. Articles were restricted to English language and reviewed first by title and abstract and then full text to confirm eligibility for this review. Only publications dealing with etiology, pathophysiology, treatment and/or epidemiology of both rosacea and migraine were included. Furthermore, a review of reference lists from the included articles was performed to identify supplementary works not included through the initial search protocol. Two reviewers (CEC and FSA) assessed the eligibility of the records found, and disagreements were resolved through discussion. A search flow chart is depicted in Figure 2.
Flow chart of search protocol.
Statistical analysis
Statistical analyses were performed using StatsDirect version 3.0 (StatsDirect Ltd., Cheshire, UK). Odds ratios (OR) with 95% confidence intervals (CI) of migraine in individuals with rosacea compared to controls were estimated. Heterogeneity of included studies was assessed using I2 statistics, and a forest plot was constructed. I2 statistic describes the percentage of variation across studies that arises due to heterogeneity rather than chance. Random effects models with DerSimonian-Laird methods were utilised as between-studies heterogeneity was found with I2 = 98%. Funnel plot progression of logarithmic OR and standard errors was used to assess for publication bias. All statistical tests were 2-sided, with a significance level of <0.05.
Results
Summary of included studies.

One study reported a 16% prevalence of migraine amongst Saudi women with rosacea but no control population was included (17). One reported that previously identified co-occurrence between migraine and rosacea in 809 office employees (16) was driven by a subgroup of women between ages 50 and 59 (15). A study of 201 patients referred to a specialist due to skin problems after working with visual display terminals found 46% suffering from rosacea and 40% suffering from migraine, but did not provide data on overlap (18). In another study, 44% of 27 female rosacea patients in a dermatology clinic reported a history of migraine compared to 10% of 21 male rosacea patients (20). The only study included in this review that did not find an association between migraine and rosacea was a case-control study of 65 rosacea patients and matched controls, examining associations between various systemic diseases and rosacea (14). Significant associations were found with allergies, gastro-intestinal diseases, respiratory diseases and so on, but not with migraine (14).
Four studies (12,13,16,21) contained quantitative data (103,620 cases and 4,367,029 controls) and were eligible for meta-analysis according to our selection criteria (Figure 3) . We calculated a pooled OR of 1.96 (95% CI 1.41–2.72) for migraine in the rosacea population compared to the non-rosacea control population. A nationwide study from Denmark reported a migraine prevalence among women with rosacea of 16%, whereas prevalence was only 4% among men compared to 11.4% and 3.2%, respectively, in the control population (12). A large population-based study from the UK investigated cases of incident rosacea through first-time medical codes for the disease and found a higher rosacea incidence among migraine patients than controls (OR 1.18) (21). In a cross-sectional analysis of 809 office employees, 27% of rosacea patients also suffered from migraine (13% in control population) (16). Another case-control study found that 44% of rosacea patients had migraine compared with 13.1% in the control group (13). Furthermore, 29 volunteers in the control group suffered from flushing, but did not meet the criteria used for rosacea – 55% of these had migraine. Excluding these volunteers from the control population further increased the significance of the primary finding of higher migraine prevalence in the rosacea population (13).
Forest plot depicting results from meta-analysis. Results are odds ratios followed by 95% confidence intervals.
Discussion
This meta-analysis showed a significant association between migraine and rosacea. Although the underlying cause for the observed association remains unclear, pathophysiological, molecular, and therapeutic aspects may be considered as explanatory factors.
The finding that 16% of dark-skinned women with rosacea from Saudi Arabia also suffer from migraine should be seen in context with the background migraine prevalence from that region (17). Population-based studies from the same area report that migraine affects 6.8% of the general population (22,23). Therefore, 16% suggests an overrepresentation of migraine in rosacea patients. However, no firm conclusions can be made without a control population. The two largest studies included in this review found an association between rosacea and migraine through database capturing (12,21). Both studies report an overlap, but as cases were incident in one (21) and migraine prevalence increases during follow-up in the other (12), conclusions should be drawn carefully so as not to underestimate overall prevalence and co-occurrence.
Four studies showed that the association between rosacea and migraine was largely or solely driven by female subgroups, and even suggest that the association was stronger around menopause (12,15,20,21).
Heredity
Migraine is believed to occur due to a combination of environmental and genetic factors, with a heritability of almost 60% (24). Several single nucleotide polymorphisms have been shown to increase susceptibility to migraine, and a stronger correlation is seen for migraine with aura than migraine without aura (25–28). However, these genetic variants have modest effect sizes and cannot alone explain the observed heritability of the most prevalent migraine subtypes (29).
Rosacea prevalence is highest among people of northern European descent, and having a family member with rosacea increases one’s risk of developing the disease fourfold, leading to a theoretical basis for a genetic rosacea etiology (5,30). A genome-wide association study identified two single nucleotide polymorphisms correlated with rosacea along with three human leukocyte antigen associations also linked to the disease (30). These data, together with findings of alterations in genes protecting the skin from reactive oxygen species, suggest that certain genetic factors may be associated with rosacea development (5,31).
Phenotype
A migraine attack is a multiphasic condition that can be triggered by various triggers of endogenous (e.g. hormonal changes) and exogenous (e.g. stress, sleep disturbances, alcohol, heat, certain foods, exercise etc.) origin (32). Migraine attacks often begin with a premonitory phase, where patients may display a wide range of symptoms that may include tiredness, fatigue, mood change, food cravings, and neck stiffness, although these may display great variability from patient to patient and attack to attack (33). Roughly one third of migraine patients experience transient neurological symptoms (aura) followed by a headache phase lasting between 4–72 hours. After this headache, patients may experience a postmonitory phase with symptoms such as tiredness, head soreness, cognitive difficulties, and mood changes (34–36).
As it is the case with migraine, rosacea is a chronic condition where exacerbations or attacks can occur. In particular, burning and stinging pain as well as flushing can be triggered by various stimuli such as UV light, spicy food, stress, exercise, heat, and alcohol consumption (5,37). An exacerbation of rosacea often starts with facial flushing that evolves into a more persistent erythema, which is the most common feature of rosacea (5). Four specific grouping patterns of symptoms divide rosacea into four distinct subtypes (i.e. erythematotelangiectatic, papulopustular, phymatous and ocular rosacea) (2). Recently, a fifth subtype termed neurogenic rosacea has been proposed (4). One case series investigated 14 patients with neurogenic rosacea, who all suffered from burning and/or stinging pain in addition to previously described rosacea symptoms. Moreover, an unusually high percentage of these patients (71%) suffered from unspecified headaches in addition to rosacea (4).
Neurovascular mechanisms
Migraine and rosacea may share common neurovascular pathophysiological features including alterations in facial skin blood flow and perivascular neuropeptide involvement.
The facial circulation undergoes an increase in cutaneous blood flow in the frontotemporal region of the face during migraine headache (38,39). Blood flow increase is more prominent on the pain side than non-pain side and can be abolished along with the headache using the anti-migraine drug ergotamine tartrate (38). One study reported asymmetrical heat loss from the facial skin in migraine attacks, but with a lower temperature in the skin on the headache side, contrary to what was expected based on previous findings regarding flow changes (38,40). Increased pulsation amplitude is seen in the superficial temporal artery (STA) during attacks of migraine, and pain can be relieved by compression of either the STA or the common carotid artery (41,42). Interestingly, pulsation increase was correlated with increased heat loss from the facial skin on the headache pain side, especially in the subgroup of migraine patients who experienced pain relief with STA compression (42). This suggests that a subgroup of migraine patients exhibit either changes in facial vasculature during attacks (39) or a diminished sympathetic regulation of facial microcirculation due to release of vasoactive neuropeptides in the perivascular space as a response to various trigger stimuli (43,44). During a migraine attack several peptides (vasoactive intestinal peptide (VIP), calcitonin gene-related peptide (CGRP), pituitary adenylate cyclase activating polypeptide (PACAP)) and molecules (nitric oxide (NO)) can mediate vasodilatation and neurogenic inflammation, as well as efferent and afferent nerve activation (45–48). Intravenous infusion of vasoactive neuropeptides induces migraine attacks (49–52) and immediate rosacea-like facial flushing in 60–100% of participants (53–56).
In rosacea skin, precapillary arterioles and postcapillary venules are dilated and their walls disrupted, causing the initial transient flushing, persistent erythema and extravasation of proteins that characterise the disease (9,57,58). Dilated vessels in the skin are still susceptible to further stimulation by vasoactive agents, suggesting a vicious cycle of dilatation, extravasation and peptide release, which causes irreversible damage to the microstructures in the skin (9,59,60). Vascular hyperreactivity is reflected by increased facial blood flow in lesional skin and the diminishment in flushing seen using alpha-adrenergic receptor agonists (61–64). The neuropeptides involved in abnormal vascular tone in rosacea include CGRP, VIP and PACAP (5,59,65,66). Interestingly, both physical and mental stress, well-known migraine triggers (32), induce an augmented sympathetic outflow in the facial skin of rosacea patients compared to controls, a mechanism like the one described for migraine above (67).
Neuroinflammatory mechanisms
The transient receptor potential vanilloid type 1 (TRPV1) receptor mediates proinflammatory substance release and is often co-localised with CGRP receptors in the human trigeminal ganglion, and could be involved in migraine pathophysiology (68,69). TRPV receptors are more abundant specifically in scalp arteries in patients with migraine compared to controls (70). Activation of TRPV receptors induces CGRP release and can, via this mechanism, induce both vasodilatation and mast cell degranulation followed by perivascular inflammation (70–72). Novel therapeutics targeting the TRPV1 receptor have been studied in migraine, but have not proved effective so far (73).
TRPV receptors also play a key part in rosacea pathophysiology and are more abundant in the skin of patients compared to controls (74). TRPV channels are activated by various stimuli such as heat or capsaicin (the pungent constituent of chili peppers), leaving a clear trail from known triggers to pathophysiological theory in rosacea. Activation of TRPV1 and transient receptor potential cation channel 1 (TRPA1), another member of the TRP family, can mediate release of neuropeptides and induce flushing in human skin (66,75). Furthermore, capsaicin-induced increase in dermal blood flow can be blocked by a CGRP antagonist, suggesting that vasodilatation and inflammation can be mediated, in part, by a TRPV-controlled CGRP release (76). PACAP and VIP levels are increased in the skin in patients with all subtypes of rosacea, and may contribute both to vascular as well as inflammatory features of rosacea pathophysiology (65), whereas receptors for PACAP and VIP are less abundant in skin biopsies of rosacea patients, suggesting that the neuropeptide increase is rather chronic, leading to a downregulation of receptor availability (59). Mast cells are more abundant in the skin of rosacea patients than healthy controls (59), and rosacea features cannot be provoked in mast cell-deficient mice (77), which emphasises the importance of these key inflammatory cells in the pathophysiology of rosacea. Inflammatory reactions in rosacea have also been linked to skin colonisation of demodex folliculorum mites and several bacterial microbes are also hypothesised to be implicated, which has led to different antibiotic treatment strategies that fall outside the scope of this review (5).
Treatment options
Medications used for migraine can be divided into two categories: Acute and preventive treatment. Preventive treatment for migraine comprise beta blockers, calcium channel blockers, angiotensin converting enzyme inhibitors, angiotensin receptor antagonists, anticonvulsants, non-steroidal anti-inflammatory drugs, antidepressants, and others. Efficacy is quite individual, and a high rate of adverse effects poses a major issue in terms of treatment compliance (78,79). Beta blockers are also used in the treatment of rosacea with efficacy for flushing and erythematous reactions (80). Calcium channel blockers have been suspected of worsening or inducing rosacea, but a recent study concluded no increased risk of incident rosacea with calcium channel blocker prescriptions; the study design did not, however, allow for evaluation of worsening of pre-existing rosacea (81). In so-called neurogenic rosacea, it was found to be beneficial to treat patients with channel modulators (gabapentin, pregabalin) or tricyclic anti-depressants (2). Clonidine, an alpha-adrenergic agonist, has been tried for migraine prophylaxis and for flushing reactions in rosacea, but its efficacy in either is disputed (78,82,83). Botulinum toxin injections are used in severe cases of migraine with some effect, and are also effective in treating rosacea flushing (84,85).
5-HT1B/1D-agonists, triptans, are migraine-specific acute medications that are marketed in a number of formulations and brands with only minor differences in efficacy (78,86). As triptans elicit vascular responses, one could speculate that flushing and erythematous symptoms in rosacea could be helped with these agents. Furthermore, triptans diminish neurogenic inflammation, block release of CGRP, and inhibit TRPV1 channels, which could benefit rosacea patients, possibly as abortive medication during exacerbations (87–89). Facial flushing is a known side effect of sumatriptan in some patients, possibly due to arteriovenous anastomose constriction (90). Thus, sumatriptan could worsen the erythematous symptoms in rosacea. One study reported an increase in rosacea risk with triptan use (21), but whether this was due to an increase in underlying migraine severity or an actual drug effect is not known.
5-HT1F receptor agonists, so-called “ditans”, as well as CGRP receptor antagonists, are currently showing promise in acute treatment clinical trials and might represent the next phase of abortive migraine treatment (79). Monoclonal antibodies against CGRP and the CGRP receptor are under development and are showing promise in several phase II and phase III studies, thus posing a new wave of opportunities in migraine prevention (79,91–94). Novel abortive and preventive medications for migraine elicit effects in cascades that are implemented in flushing and erythematous reactions of rosacea, which leaves room to consider whether they could be tried for subtypes of this condition as well.
Collectively, these data suggest several overlapping neurovascular mechanisms linking migraine and rosacea (summarised in figure 4).
Potential mechanisms in migraine and rosacea pathophysiology. Trigger factors stimulate activation of trigeminal sensory nerves in blood vessels of either facial skin or the brain, resulting in release of calcitonin gene-related (CGRP), pituitary adenylate cyclase-activating polypeptide (PACAP), substance P and neuropeptide Y in the perivascular space. Here, the neuropeptides induce vasodilation, increased vascular permeability and neurogenic inflammation. Depending on the patient’s susceptibility, this chain of reactions will lead to either migraine headache or rosacea.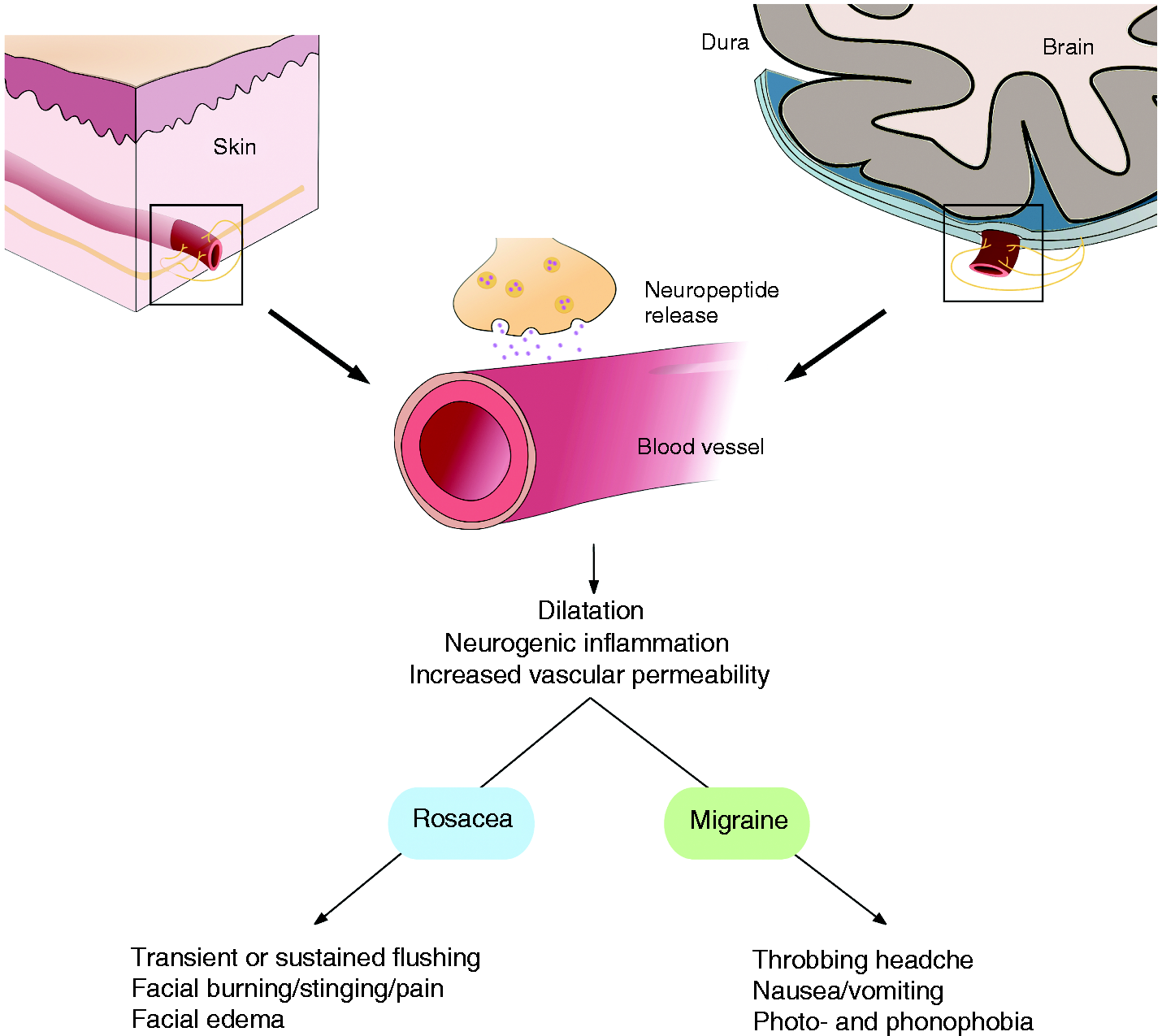
Limitations and strengths
A common limitation is that the reviewed studies did not diagnose migraine according to the international classification criteria (1). Three studies reported migraine as unilateral, pulsating headache accompanied by nausea and vomiting or visual aura phenomena (13,16,20), and two population-based studies included migraine cases based on international classification of disease codes or pharmacotherapy used almost exclusively for treatment of migraine (12,21), whereby the true migraine risk may be underestimated. The same limitation applies to rosacea diagnoses, as the standard classification was not published until 2002 and was not applied to the reviewed studies (2). Furthermore, the studies included show quite high heterogeneity in terms of selection criteria, population demography and population sizes, rendering direct comparisons difficult.
Conclusion
Migraine and rosacea show clear co-occurrence and we suggest a pathophysiological overlap, which leads to the assumption that the two diseases are somehow linked. However, the link has yet to be identified. Rosacea studies using the same provocation designs as seen in migraine research could increase understanding of the pathophysiological link and supply a platform for acute and preventive treatment trials. Furthermore, the pharmacology of triptans (vasoconstriction (95) and inhibition of pro-inflammatory neuropeptide release (96)) used to treat migraine, as well as novel anti-migraine drugs targeting CGRP or PACAP, prompts investigation into whether they are also effective in treating rosacea, specifically the erythematous and flushing symptoms. Deep phenotyping studies of clinical cohorts of rosacea patients are important for sub-categorisation of patients, as well as randomised clinical trials to determine the efficacy of migraine medications in rosacea.
Clinical implications
There is an association in occurrence of migraine and rosacea. Migraine and rosacea may share neurovascular and neuroinflammatory pathophysiological mechanisms. Future studies should investigate shared mechanisms possibly explaining co-occurrence. Studies should investigate whether patients might benefit from treatment targeting migraine and alleviating rosacea as well.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Christensen, Ms Andersen and Dr Wienholtz report no disclosures relevant to the manuscript. Dr Egeberg has received research funding from Pfizer and Eli Lilly, and speaker and/or consultancy honoraria from Pfizer, Eli Lilly, Galderma, Novartis, and Janssen Pharmaceuticals. Dr Thyssen is supported by an unrestricted grant from the Lundbeck Foundation and has received speaker honoraria from Galderma and MEDA, and has attended an advisory board for Roche. Dr Ashina is a consultant or scientific advisor for Allergan, Amgen, Alder, ATI and Eli Lilly, primary investigator for Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3) and GM-11 gamma-Core-R trials, and reports grants from Lundbeck Foundation (R155-2014-171), Research Foundation of the Capital Region of Copenhagen, Danish Council for Independent Research-Medical Sciences.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by the Lundbeck Foundation (R155-2014-171).
