Abstract
Background
The migraine brain is believed to have altered cortical excitability compared to controls and between migraine cycle phases. Our aim was to evaluate post-activation excitability through post-movement beta event related synchronization (PMBS) in sensorimotor cortices with and without sensory discrimination.
Subjects and methods
We recorded EEG of 41 migraine patients and 31 healthy controls on three different days with classification of days in relation to migraine phases. During each recording, subjects performed one motor and one sensorimotor task with the right wrist. Controls and migraine patients in the interictal phase were compared with repeated measures (R-) ANOVA and two sample Student’s t-test. Migraine phases were compared to the interictal phase with R-ANOVA and paired Student’s t-test.
Results
The difference between PMBS at the contralateral and ipsilateral sensorimotor cortex was altered throughout the migraine cycle. Compared to the interictal phase, we found decreased PMBS at the ipsilateral sensorimotor cortex in the ictal phase and increased PMBS in the preictal phase. Lower ictal PMBS was found in bilateral sensorimotor cortices in patients with right side headache predominance.
Conclusion
The cyclic changes of PMBS in migraine patients may indicate that a dysfunction in deactivation and interhemispheric inhibition of the sensorimotor cortex is involved in the migraine attack cascade.
Introduction
Studies of migraine neurophysiology have found contradicting results regarding cortical excitability in migraine. Both a cortical hypo- and hyperexcitability have been suggested compared to healthy controls (1–5). Several studies have also found variations in cortical excitability in relation to the migraine cycle (4,6–8). One recent cross-sectional study with repetitive transcranial magnetic stimulation found preictal facilitation of motor evoked potentials interpreted as an increased preictal threshold for activating inhibitory homeostatic mechanisms followed by an ictally decreased threshold for inhibitory activation (6). Another magnetoencephalographic study reported a mainly ipsilateral cortical hyperexcitability to finger movements during the migraine attack (5). It has also been reported that thalamocortical high-frequency oscillations are abnormal in migraine measured by somatosensory evoked potentials (SSEP) (9,10). However, many of the other formerly used techniques only measure the rhythmicity generated by the thalamocortical circuits to a limited extent. Another useful complimentary technique that reflects various aspects of thalamocortical rhythmic activation patterns is called “event-related desynchronization and synchronization” (ERD/ERS) (11).
ERD/ERS encompass a set of frequency-specific response types characterized by either a decrease or an increase in rhythmic EEG activity. ERD/ERS represents an induced, time-locked, non-phase-locked response to an event, reflecting either decreased or increased neuronal synchrony. More specifically, the ERD/ERS represents changes in the activity of local interactions between the thalamocortical projection neurons and the cortical interneurons that control important aspects of cerebral rhythmicity (11). Post-movement beta ERS (PMBS) recorded from the sensorimotor cortex is a robust ERD/ERS-subtype with good signal to noise ratio (11). The PMBS-maximum coincides with a reduced excitability of motor cortex neurons, and PMBS may accordingly reflect a deactivated cortical state (11–13). Furthermore, PMBS may also reflect active inhibition of the sensorimotor cortex executed by somatosensory afferents from joint receptors, muscle spindles and cutaneous receptors, reaching precentral motor cortex neurons via either direct connections from the thalamus or cortico-cortical connections from the somatosensory cortex (14–16). PMBS has been used to study cortical excitability in other neurological conditions such as neuropathic pain and ALS (17,18), and it may also be useful in the study of cortical excitability in migraine, describing the relative excitability in a post-activation state of the sensorimotor cortex.
The variations in neurophysiology between migraine phases suggest that longitudinal studies are advantageous compared to cross-sectional studies to detect differences between phases (19), but such studies have seldom been performed. The purpose of the present controlled longitudinal blinded study was accordingly to evaluate cortical inhibition and post-activation excitability patterns from the sensorimotor cortex by PMBS, related to the migraine cycle in migraine patients. To our knowledge, PMBS has not been studied previously in migraine. One specific aim was to compare PMBS between healthy controls and interictal migraine patients. Secondly, we aimed to evaluate the intraindividual PMBS changes before, during and after the migraine attack in comparison to an interictal baseline. Our third aim was to explore whether clinical migraine features correlated with PMBS throughout the migraine cycle.
Subjects and methods
Subjects
We included 41 migraine patients and 31 healthy controls. Migraine patients were recruited by a newspaper advertisement and subsequently screened via telephone by nurses trained in headache research. Thereafter, a neurologist evaluated 52 migraine patients for inclusion according to the International Headache Society classification of headache disorders, 2nd edition (19). The study group consisted of men and women aged 18–65 years with episodic migraine consisting of two to six migraine attacks each month for the last three months, who did not use prophylactic medication. Thirteen had mainly headache without side predominance while 15 had right and 13 left-sided predominance. Healthy controls were recruited among blood donors. Before inclusion, the controls underwent a semi-structured interview by an experienced nurse. All but four patients and three controls were right handed. Exclusion criteria for migraine patients and controls were frequent episodic or chronic tension-type headache, acute or chronic neurological disease, connective tissue disorder or other painful conditions, malignancy, previous craniotomy or cervical spine surgery, cardiopulmonary or cerebrovascular disease, hypertension, pregnancy, medication for acute or chronic pain, neuroleptics, alcohol or drug abuse, ferromagnetic implants, and use of neuroactive substances such as anti-depressive, anti-epileptic, or migraine prophylactic drugs within the four weeks before the test.
Demographic and clinical data on groups used in interictal analysis and subgroups used in paired analysis.

MA: migraine with aura; MwoA: migraine without aura. Mean (SD) or numbers. Subgroups with both an interictal EEG recording (>36 h from attack) and a preictal (<36 h before attack), ictal or postictal (<36 h after attack) EEG recording.
Each subject had three EEG recordings except for one migraine patient who abstained from undergoing all recordings because headache worsened after the tests. Hence this subject only had two tests.
Staff involved in data reduction and analysis were blinded regarding the diagnostic status of the subjects. The subjects received NOK 1000 (about EUR 108 with current exchange rates) as compensation to cover expenses after completing all three recordings. The compensation was not mentioned in the advertisement. Written consent was obtained by all subjects. The study was approved by the regional committee for medical and health research ethics.
EEG recordings and experimental setup
EEG recordings were performed at the same time of day at 3–10 day intervals. An approximately 30 min EEG was recorded with eyes closed. The first five minutes were undisturbed relaxed wakefulness, followed by a motor/sensorimotor test and thereafter photo stimulation trains. Based on the first and last recording periods, resting state quantitative EEG and steady state visual evoked potential results have been reported previously by Bjork et al. (4,19,21,22). The present paper evaluates PMBS data from the motor and sensorimotor tests, which have not previously been analysed.
Twenty four scalp electrodes were attached according to the 10/20 international system (24), with channels for lateral anterior temporal electrodes, horizontal and vertical eye movements, and ECG. EEG was recorded digitally in Nervus 3.0 with M40 amplifier (Natus Medical Inc, Pleasanton, CA 94566, USA) and common reference with a 256 Hz sampling rate. An average reference montage was used with low and high-pass filters of 0.5 and 70 Hz in addition to a notch filter (50 Hz). To avoid drowsiness, the subjects were asked to open their eyes every minute as well as being talked to by the technician if drowsiness occurred during the first five minutes of the EEG recording.
Each subject performed both a motor test (M) and a sensorimotor test (SM) with approximately 30 repeated movements in each test. The order of tests was randomized for each subject, and fixed for each day of recording for the same subject. The instructions given to all subjects were the following. Each test would last for about eight minutes with a light blink indicating when to start each movement. For the motor test, subjects were to first flex their wrist for two seconds, then extend their wrist for two seconds, followed by about 15 seconds of relaxation. For the sensorimotor test an identical flexion-extension movement was performed. However, a bowl of different material spheres (wood or metal) was placed about 5 cm below the neutrally positioned fingers, so that the fingers were in contact with the spheres in the flexed position. The task was to use the two seconds in the flexed position to scan the spheres lightly with their fingertips to detect if a sphere of wood was present in the bowl or not. The right arm was used in both sequences. Two EMG-channels for flexion and extension were included in the EEG recording for determination of movement epochs (Figure 1).
Example from one single movement with EEG, EMG (ecR: radial wrist extensors, fR: forearm wrist flexors), and ECG channels. The photic channel marker represents a light blink as a sign for the subject to prepare for executing the task. “MovStart” and “MovEnd” markers were placed manually at the start and the end of EMG movement.
ERD/ERS analysis
IIR-filtered data in the 12–19 Hz beta frequency band (25,26) from sensorimotor cortices electrodes C3 (left side, contralateral) and C4 (right side, ipsilateral), were exported in 256 Hz resolution from each test and used for the PMBS analysis. Previous PMBS studies have found maximal responses close to the central sulcus (11,27,28). The amplitude was squared to obtain the power and then averaged across all movements within the same test (11). Movement onset and offset were marked (Figure 1) and used to determine epochs for analysis. Time-power graphs for all subjects were visually inspected by a blinded researcher to determine the response timings used to select epochs for analysis. Selections from −3 to −1 second prior to start of movement were used as the baseline. This choice was considered advantageous over a baseline prior to the end of movement, as the movement period was of varying duration (29).
PMBS has mostly been reported to peak within the first second after termination of voluntary movement (11,14,30). However, the post-movement period was defined from one to three seconds after movement offset based on blinded, visual inspection of power-time graphs for every subject. The natural logarithm of average power in the post-movement period divided by average power in baseline was then calculated as the PMBS response variable.
Statistical analysis
For each migraine patient, one test was selected for each cyclic phase that was available (interictal, preictal, ictal and postictal). If several tests for the same cyclic phase were available, the second was chosen. Control EEGs were chosen to have a similar test-order distribution as the interictal migraine group.
We conducted repeated measures ANOVA (R-ANOVA) with within-subject factors “side” (C3 vs C4) and “SM/M” (sensorimotor vs. motor test), and between-subjects factor “group” (CO vs. MIG). This was done to evaluate differences between the groups in the interictal phase. Post-hoc two sample Student’s t-test was used to further evaluate significant factors regarding the detailed nature of the differences between diagnostic groups.
Three R-ANOVAs within the migraine group were carried out with the factors “phase” (separate R-ANOVAs with preictal-interictal, ictal-interictal and postictal-interictal), “side” (C3 vs. C4) and “SM/M” (sensorimotor vs. motor test). We also conducted post-hoc paired t-tests for each variable to evaluate the detailed nature of the significant R-ANOVA factors. Two-sided p-values < 0.05 were regarded as significant and p-values < 0.10 were regarded as trends. A paired analysis with 11 (9) pairs has approximately 77% (65%) power to detect an effect = 90% of group SD (19).
Variables with significant group or phase differences were examined visually for possible outliers. A few (n = 6) tests with outlying data points and artefactual response-peaks were replaced with a test from another day in the same category if available (ictal, pre-, post-, or inter-ictal) from the same subject, or else the artefactual test was excluded from the analysis (control n = 1; preictal migraine n = 1); leaving 30 controls for the final comparison with interictal migraine and 11 patients for the paired within-subject preictal-interictal analysis.
Exploratory Spearman rho correlations were calculated for headache history duration, usual attack duration, individual scores on headache frequency (0–4), usual headache intensity (0–4), photophobia (0–2) and phonophobia (0–2), and PMBS. Correlation coefficients were calculated separately from PMBS values in the ictal, pre-, post-, and interictal phase subgroups.
Results
Interictal analyses between migraine patients and controls
Repeated measures ANOVA. Interictal migraine patients compared to headache-free controls.

PMBS response is the ratio between mean power in the interval from 1–3 sec after movement offset and mean power in the interval −3 to −1 sec before movement onset (baseline). Ratios were LN-transformed before statistical analysis. Within subject factors used were side (C3 vs. C4 electrode) and SM/M (sensorimotor vs. motor task). The between subjects factor is group (interictal migraine vs controls).
Post-movement beta synchronisation mean response/baseline ratios in interictal migraine subjects and headache-free controls.

PMBS response is the ratio between mean power in the interval from 1–3 sec after movement offset and mean power in the interval −3 to −1 sec before movement onset (baseline). Ratios were LN-transformed before statistical analysis and retransformed to mean ratios and mean ± SD for tabulation. Post-hoc two-sample Student’s t-tests (equal variance not assumed) are included. EEG from central electrodes C3 (left) and C4 (right). SM: sensorimotor test; M: motor test.
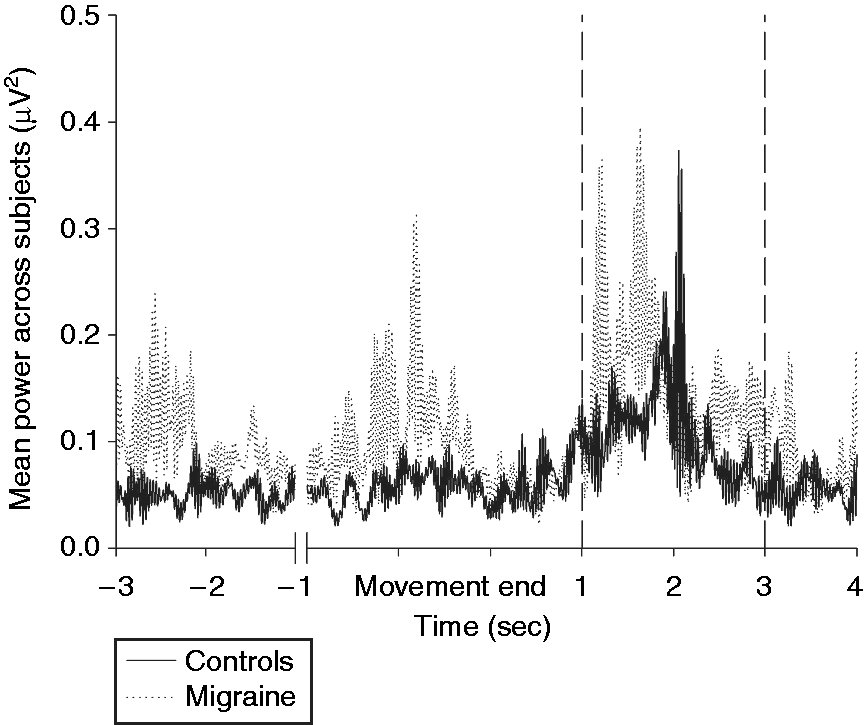
Grand mean power across subjects at the contralateral C3 electrode for the sensorimotor task in controls and interictal migraine patients. The first two seconds (−3 to −1) represent the pre-movement onset baseline. Broken vertical lines indicate the selected interval (1–3 seconds) for the post-movement period.
Paired analyses for preictal, ictal and postictal phases compared to the interictal period
Repeated measures ANOVA paired analysis in different phases of the migraine cycle (preictal, ictal and postictal, compared to a paired interictal recording).
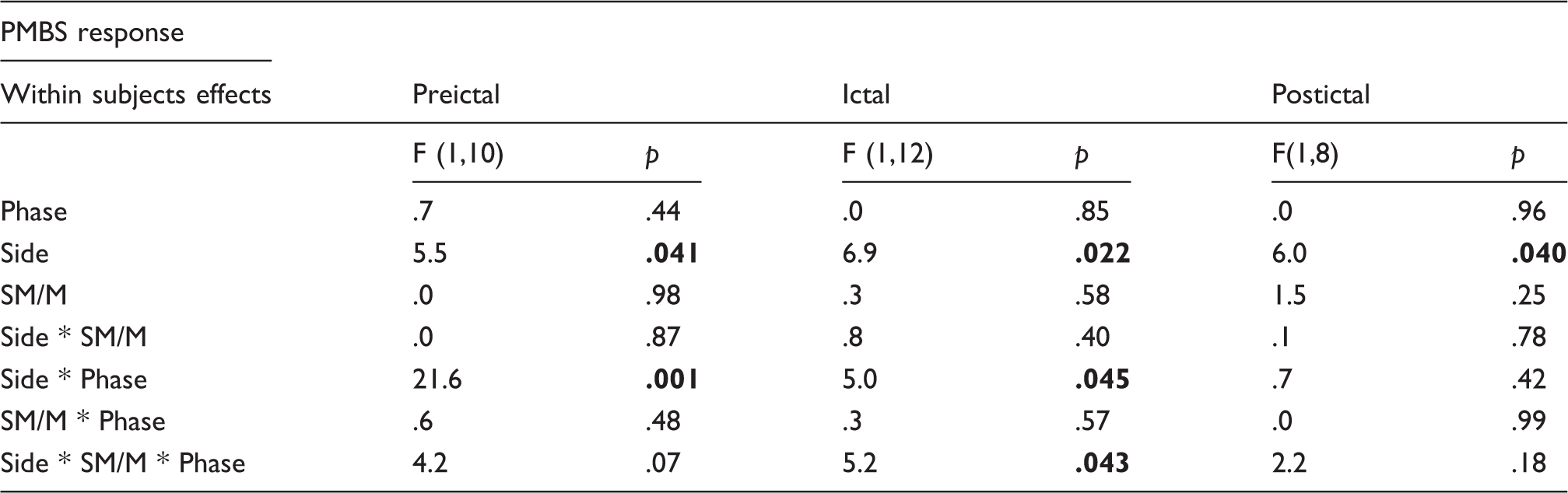
Paired analysis of subgroups preictal(< 36 h before migraine pain attack), ictal and postictal (< 36 h after migraine pain attack). PMBS response is the ratio between mean power in the interval from 1–3 sec after movement offset and mean power in the interval −3 to −1 sec before movement onset (baseline). Ratios were LN-transformed before statistical analysis. Factors used were side (C3 vs. C4 electrode), SM/M (sensorimotor vs. motor) and cyclic phase (preictal-interictal, ictal-interictal and postictal-interictal). Significant results are shown in bold.
Paired t-test for preictal-interictal, ictal-interictal and postictal-interictal comparisons.

PMBS response is the ratio between mean power in the interval from 1–3 sec after movement offset and mean power in the interval −3 to −1 sec before movement onset (baseline). Ratios were LN-transformed before statistical analysis and retransformed to mean ratios and mean ± SD for tabulation. Post-hoc paired Student’s t-tests are included. EEG from central electrodes C3 (left) and C4 (right) for the sensorimotor test and motor test. Significant results are shown in bold.

PMBS response to the sensorimotor task in paired migraine subgroups. The LN-transformed response variable has been retransformed to a mean ratio (post-movement/baseline) with SD for tabulation and graphic display. (a) PMBS response for ipsilateral C4. Significant difference with paired t-test in the ictal phase compared to the interictal phase (p = 0.045) (*). Tendency towards difference with paired t-test in the preictal phase compared to the interictal phase (p = 0.058). The C3–C4 difference for the sensorimotor test is highly significant (p = 0.042) between the preictal and interictal phase. (b) PMBS response for contralateral C3. No significant differences seen in the paired t-test.
Patients with right headache predominance had significantly lower ictal PMBS than patients with left predominance after motor tests, both contralateral over the left hemisphere (right predominance mean ratio = 1.01; left predominance mean ratio = 2.08; p = 0.001) and ipsilateral (right predominance mean ratio = 1.00; left predominance mean ratio = 1.95; p = 0.012).
Spearman rho correlation analysis of migraine cycle PMBS in ipsilateral (C4) sensorimotor cortex against clinical parameters.

Clinical data from questionnaire filled out by migraine patients compared to PMBS response with Spearman rho correlation analysis. Ipsilateral (C4) PMBS response to sensorimotor task (SM) and motor task (M). Clinical parameters are headache history duration (history), usual attack duration (duration), individual scores on headache frequency (0–4; frequency), usual headache intensity (0–4; intensity), individual scores on photophobia (0–2) and individual scores on phonophobia (0–2). Significant results are shown in bold.

Ipsilateral (C4) postictal PMBS response to motor test (M) and individual scores on headache intensity (0–4) (rho = −0.86, p = 0.001). The LN-transformed response variable has been retransformed to a ratio (post-movement/baseline) for tabulation and graphic display.
Discussion
The main finding in this blinded longitudinal study was a significantly lower sensorimotor PMBS response in the ictal phase compared to the interictal phase. A higher PMBS in the preictal phase compared to the interictal phase was also seen. Since PMBS may be related to a balance between inhibition from somatosensory afferents and intracortical inhibition (14,16,17), it is probably the level of cortical inhibition that is subject to cyclic modulation. This modulation, regulating the overall cortical excitability, may play a role in migraine attack initiation and continuation. In the present study, relative normalization of PMBS occurred in the postictal phase. Hence, altered intracortical inhibition and overall post-activation cortical excitability seem to normalize after migraine attacks.
However, our first working hypothesis, that migraine patients have increased PMBS and altered excitability in the interictal phase compared to healthy controls (1), could not be confirmed. Distinct differences could nevertheless be observed in the grand-mean time graphs, suggesting that a different analytical approach with briefer time-intervals (13,30) might reveal subtle interictal changes that could not be detected by the chosen PMBS-response variable.
Inhibitory neurons in primary hand motor cortex probably receive transcallosal inhibition from the contralateral motor cortex (31), most likely mediated by axons of layer 3 pyramidal neurons (32). The inhibition seems to be effected by interneurons receiving facilitatory interhemispheric projections (33,34). Based on rat experiments, cortical neurons dominated by thalamic inputs also seem to be affected by a change in transcallosal neural activity (35), possibly suggesting that interictal SSEP abnormalities in migraine can also be influenced by a change in interhemispheric inhibition. Reduced PMBS in the ipsilateral cortex has also been reported in amyotrophic lateral sclerosis, interpreted as impaired interhemispheric inhibition due to corpus callosum affection (18). Thus, a cyclic activity change in this interhemispheric transcallosal projection could explain why only ipsilateral PMBS was affected.
Structural corpus callosum abnormalities such as corpus callosum atrophy (36) and white matter axonal loss (37) have been shown in migraine patients, also correlating with disturbances of the anterior cingulate cortex, which is involved in pain processing (38). However, since PMBS was normal in interictal migraine, we found no evidence of any permanent deficit. Although structural changes may mark vulnerability in migraine, our results support a cyclic physiological change in interhemispheric inhibition from the preictal to the ictal state.
A recent SSEP study found reduced interictal lateral inhibition from ulnar and median nerve stimulation, thought to be a consequence of poor thalamocortical drive (39). Since lateral inhibition depends on short-range cortico-cortical connections confined within the contralateral hemisphere, one would expect to see a larger contralateral PMBS response to whole-hand movement in migraine if lateral inhibition had been compromised, but this was not found in the present study. The activation pattern by natural stimulation of sensory receptors during movement is also quite different from the synchronized volleys achieved by electrical nerve stimulation (39), and this large difference in methods may explain apparently different results. A future study of event-related PMBS-responses to individual finger movements might clarify this issue.
Our main result was only seen for the most complex task involving motor activation, tactile stimulation and cognitive evaluation. The movement-related afferent activity induced by the motor wrist flexion-extension was integral to both tasks. Hence, the present results do not suggest a general long-lasting change in cortical excitability. The proposed deficiency in interhemispheric inhibition seems to be particularly amplified during the cognitive evaluation of the complex SM stimulus. Cyclic alterations in the cognitive event-related potentials have also been described previously in migraine (40–42).
We observed lower ictal PMBS in patients with right-sided headache predominance over both the ipsilateral and contralateral sensorimotor cortex. Ictal somatosensory temporal discrimination (STD) has recently been shown to be decreased and related to headache lateralization in migraine patients (43). A similar finding was also seen in chronic migraine on days with and without headache (44), while no increase in STD was seen in patients with tension-type headache (45). Alterations of STD in ictal migraine and the findings in the present study may represent similar dysfunctions caused by deactivation of the sensorimotor cortex, possibly also increasing the risk of motor errors (46). Lateralized activation of the thalamic reticular nucleus has been shown to be operative in a cortical spreading depression rat model relevant to migraine (47). This abnormality may possibly be involved in the same somatosensory stimuli processing dysfunction.
Headache side has also been related to ipsilateral somatosensory aura symptoms (48), autonomic symptoms (49) corneal hypersensitivity (50), ictal nociceptive blink reflex latency (51) and frontal thermal hypersensitivity (52), in line with a proposed pathophysiological model based on meningeal sensory, autonomic and brainstem hyperactivity (53,54). Increased EEG delta activity has also been observed ipsilateral to headache in interictal migraine (23), possibly related to 3rd order thalamic neuron sensitization (55). Finally, patients with unilateral neuropathic pain had more restricted PMBS related to the painful body side ipsilateral to the movement (17). The latter observation resembles our preictal ipsilateral finding. These results suggest that PMBS asymmetry may be a relevant part of unilateral migraine and pain pathophysiology, possibly reflecting top-down pain-control mechanisms.
As somatosensory cortices are likely to be involved in sensory-discriminative aspects of pain (56), the preictal finding in the present study may be related to earlier findings of preattack pain hypersensitivity in migraine patients (52). Subcortical activation may follow a similar pattern. For instance, one recent fMRI study observed preictal increase of the response to gaseous and visual stimuli within the ipsilateral hypothalamus followed by ictal deactivation (57). These cyclic modulations may possibly also reflect a preictal increase in top-down inhibition of pain pathways (58).
The significant negative correlation between headache intensity and ipsilateral PMBS to the motor task in the postictal phase suggests that too-low levels of cortical post-movement inhibition relate to more pain during attacks. We only found this correlation for the ipsilateral response in parallel with our other ipsilateral findings, strengthening the notion that interhemispheric inhibition seems to be involved in pain control mechanisms terminating the attack in migraine. The observed postictal bilateral negative correlation between PMBS and photophobia and phonophobia also suggest that too-low levels of cortical post-movement inhibition relate to clinical sensory phobia. PMBS to the sensorimotor task did not correlate with headache intensity, possibly because cognitive and sensory evaluation also involve emotional aspects and brain structures that are not directly related to pain intensity. Phono/photophobia on the other hand, more complex symptoms of hypersensitivity, correlated with PBMS to the SM task. These observations strengthen the possibility that PMBS magnitude partly reflects, or correlates with, pain-reducing cortical mechanisms. The observed positive correlation between headache frequency and ipsilateral PMBS in the interictal phase suggests that interhemispheric inhibition may be potentiated by frequent headache, possibly reflecting interictal cortical defence mechanisms against future attacks. One study on repetitive transcranial magnetic stimulation and motor evoked potentials has found decreased interictal cortical activation, suggesting a lowered inhibitory threshold as a compensation for a low threshold for excitatory initiation of the migraine attack in subjects with higher headache frequency (6). Furthermore, reducing cortical excitability with inhibitory GABA-agonists seems to reduce the attack frequency of subjects with chronic migraine (59).
Some methodological details of the present study should be discussed. Multi directional movement of slightly different durations may cause ERD from one movement direction to occur simultaneously with ERS from another movement direction (29). However, to compensate for this, we decided to use a pre-movement onset baseline. Although lower beta band ERD may start about two seconds prior to a self-paced movement onset (11), our movements were triggered, not self-paced. Also, since the movement was initiated within one second after the light blink trigger in the vast majority of repetitions, the results should not be influenced by ERD interfering with the baseline.
We defined the post-movement period as 1–3 seconds after movement offset. The 1–3 second interval was shown in blinded pre-evaluation of the responses to be more appropriate for the present protocol. This period differs slightly from other studies that have reported PMBS to peak within the first second (11,14,30). However, these studies vary in design from ours by using self-paced movements, source-derivation EEG-reference, and briefer movements. Handedness could not be fully analysed because the vast majority of subjects were right-handed, but the exclusion of left-handed subjects did not change the main results.
An a priori selected beta band of 12–19 Hz (25,26) was used to avoid type 1 errors (60). Paired subgroups were rather small, suggesting that type II errors may have prevented our ability to detect small effects. Further larger longitudinal studies on migraine brain neurophysiology in different study populations are needed to replicate the findings from any single study.
Conclusion
Alterations of PMBS in the sensorimotor cortex throughout the migraine cycle may indicate that a dysfunction affecting these cortical areas is involved in the migraine attack cascade. Current understanding of the PMBS phenomenon suggests that our results represent asymmetrical cyclic modulation of cortical inhibition, involving lowered interhemispheric inhibition of the sensorimotor cortex during migraine attacks. Asymmetrical alterations in PMBS correlate with clinical headache predominance asymmetry, supporting an asymmetrical alteration in deactivation and interhemispheric inhibition of the sensorimotor cortex during attacks. Correlation with further clinical parameters substantiates the relevance of PMBS alterations as a part of migraine attack neurophysiology. Normalization of these changes seems to occur in the postictal phase. Longitudinal studies are advised to further examine these cyclic changes in cortical sensorimotor properties in migraine. Relevant experimental designs to further investigate these abnormalities include different stimulation protocols and modalities as well as prophylactic interventions.
Article highlights
Ictal decrease in post-movement beta event related synchronization may suggest a cyclic deactivation of the sensorimotor cortex in migraine. Findings suggest a dysfunction in intracortical inhibitory mechanisms and interhemispheric inhibition as a part of migraine attack initiation and continuation. Correlations between PMBS, headache side predominance and other clinical parameters strengthen the relevance of altered interhemispheric inhibition as a possible part of migraine neurophysiology.
Footnotes
Acknowledgements
The authors are most grateful for the extensive effort and contributions from both migraine patients and controls as well as for the assistance from Grethe Helde, Gøril Bruvik Gravdahl, Knut Hagen and Lars Jacob Stovner.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
