Abstract
Background
Clinical studies suggest a link between obesity and the primary headache disorder migraine. In our study we aimed to reveal the effect of obesity on meningeal nociceptor function in rats receiving a high-fat, high-sucrose diet.
Methods
Transient receptor potential ankyrin 1 (TRPA1) receptor activation-induced changes in meningeal blood flow, release of calcitonin gene-related peptide (CGRP) from trigeminal afferents and TRPA1 protein expression in the trigeminal ganglia were measured in control and obese rats. Metabolic parameters of the animals were assessed by measuring glucose and insulin homeostasis as well as plasma cytokine concentrations.
Results
The present experiments revealed an enhanced basal and TRPA1 receptor agonist-induced CGRP release from meningeal afferents of obese insulin-resistant rats and an attenuated CGRP release to potassium chloride. Obesity was also associated with an augmented vasodilatation in meningeal arteries after dural application of the TRPA1 agonist acrolein, a reduction in TRPA1 protein expression in the trigeminal ganglia and elevations in circulating proinflammatory cytokines IL-1β and IL-6 in addition to increased fasting blood glucose and insulin concentrations.
Conclusions
Our results suggest trigeminal sensitisation as a mechanism for enhanced headache susceptibility in obese individuals after chemical exposure of trigeminal nociceptors.
Keywords
Introduction
Obesity is one of the most significant global health issues. Clinical and population-based studies suggest a link between obesity and the common and disabling primary headache disorder migraine. Obesity may enhance the frequency and severity of headache attacks and appears to increase the risk for having migraine (1). Experimental results indicate multiple areas of overlap between migraine pathophysiology and the mechanisms related to excess body fat (2,3). Adipose tissue has significant endocrine functions by producing bioactive molecules that serve as regulators of metabolism and also possess immunoregulatory functions (4–6).
Primary headaches are associated with increased activity of the trigeminal nociceptive pathway (7). Earlier studies revealed that chemosensitive neurons expressing members of the transient receptor potential (TRP) receptor family play a crucial role in nociception and neurogenic vascular reactions in different tissues including the meninges (8–10). Activation of trigeminal chemosensitive afferents that innervate the dura mater facilitates pain signalling towards the central nervous system and elicits the release of neuropeptides, such as calcitonin gene-related peptide (CGRP) and substance P (SP), from their peripheral terminals leading to arterial vasodilatation, increase in vascular permeability and degranulation of mast cells (7). Peripheral events following the activation of dural chemosensitive nociceptors may generate positive feedback reactions that augment the initial vascular and nociceptive responses (11).
Enhanced activation of the trigeminovascular nociceptive pathway may result from sensitisation of meningeal nociceptors by cytokines and other inflammatory mediators. They have been shown to affect ion channels expressed in peripheral endings of trigeminal nociceptors, leading to increased excitability and sensitisation (12). Prolonged activation of meningeal primary afferents may also sensitise second-order neurons of the nociceptive pathway in the caudal trigeminal nucleus. Central sensitisation is considered a significant pathophysiological mechanism in the development of primary headaches (13).
The significance of CGRP in the pathophysiology of migraine is among the most widely discussed topics in the field of headache research (14). Clinical and experimental observations indicate a possible link between migraine and TRPA1 receptor activation-dependent release of CGRP from trigeminovascular nociceptive afferents. Further, activation of nasal mucosal trigeminal afferents which express the TRPA1 receptor has been suggested as a pathophysiological mechanism involved in the generation of headache induced by inhaled environmental irritants (15). The TRPA1 agonist acrolein is a component of tobacco smoke, vehicle exhaust and vapours of overheated cooking oils, which along with other TRPA1 agonists are known triggers of migraine attacks in susceptible individuals (16,17). Importantly, prolonged exposure to acrolein through inhalation has been demonstrated to sensitise the trigeminovascular system (18).
Obesity is often associated with chronic low-grade systemic inflammation characterised by enhanced levels of proinflammatory cytokines, reactive oxygen and nitrogen species (4,19,20). TRPA1 channels are potential targets of these agents. Modification of the TRPA1 receptor function leading to enhanced activity of the trigeminal nociceptors may be the mechanism underlying the activation of the nociceptive pathway and changes in meningeal blood flow in headache patients (16).
Although the aetiology of obesity is multifactorial, the Western-type diet is considered as a major contributor to the obesity pandemic (21). Therefore, animal models of diet-induced obesity are widely accepted approaches to mimic and, in turn, study human obesity and associated pathophysiological disturbances (22).
The present experiments were initiated in an attempt to reveal the effects of obesity on the TRPA1 receptor-mediated meningeal nociceptor function in rats maintained on a high-fat, high-sucrose (HFHS) diet. We measured, therefore, TRPA1 activation-induced changes in meningeal blood flow, CGRP release from trigeminal afferents and TRPA1 protein expression in trigeminal ganglia of control and obese animals. Metabolism was also assessed by examining glucose and insulin homeostasis as well as plasma cytokine concentrations.
Materials and methods
Animals and diet
Experiments were approved by the Ethical Committee for Animal Care of the University of Szeged and the University of Debrecen. Study procedures were carried out in accordance with the Directive 2010/63/EU of the European Parliament. All efforts were made to minimise the number of animals used and their suffering.
Male Sprague Dawley rats initially weighing 150–170 g (6 weeks old) were housed in an environmentally controlled room (12-hour light/dark cycle, 22 ± 2℃, 50%–70% relative humidity) and randomly divided into two groups. The control group was maintained on a regular diet consisting of standard laboratory chow (3.20 kcal/g, 59% carbohydrate, 32% protein, 9% fat; diet code: S8106-S011 SM R/M-Z + H, ssniff Spezialdiäten GmbH, Germany) and tap water. The experimental group received an HFHS diet composed of high-fat chow (4.56 kcal/g, 35% carbohydrate, 20% protein, 45% fat; diet code: 824018, Special Diets Services, UK) and tap water containing 5% sucrose. Diets were provided ad libitum for 20 weeks. All experiments were performed at the end of the dietary treatment period in 26-week-old animals when body weight (BW) was also measured.
Assessment of insulin sensitivity
The hyperinsulinaemic euglycaemic glucose clamp (HEGC) method was used to estimate insulin resistance by measuring the amount of glucose necessary to maintain normoglycaemia in the presence of increased insulin levels during a steady-state condition (23).
After an overnight fast, rats were anesthetised with thiopental sodium (100 mg/kg, intraperitoneally (i.p.)) and the trachea was cannulated to allow free breathing. The left and right jugular veins were cannulated for insulin (Humulin R, Eli Lilly, Indianapolis, IN, USA) and glucose (20%) infusion administered by means of an infusion pump. Blood sampling and monitoring of mean arterial pressure were performed through a cannula inserted into the right carotid artery. The body temperature of the rats was kept at 37.0–37.5℃. After a short stabilisation period, a continuous insulin infusion was commenced at a rate of 3 mU/kg/minutes (min) over 120 min during which the blood glucose was maintained at a euglycaemic level (5.5 ± 0.5 mmol/l) by adjusting the rate of glucose infusion. Blood glucose concentration was determined at 10-min intervals using a glucometer (Accu-Chek, Roche Diagnostics, Budaörs, Hungary). Steady-state condition was defined when the blood glucose level had stabilised for at least 20 min. In order to determine plasma insulin concentration, arterial blood samples were taken before the start of insulin infusion and during the steady-state period. Blood samples were centrifuged for 2 min at 4℃ and 10,000 g, and the plasma was aliquoted, frozen and stored at − 70℃ for later analysis. The average glucose infusion rate (GIR, mg/kg/min) needed to maintain euglycaemia during the steady state condition was used to characterise whole-body insulin sensitivity (24).
A homeostasis model assessment of insulin resistance (HOMA-IR) was calculated by using the data on fasting blood glucose (FBG, mmol/l) and fasting plasma insulin (FPI, µU/ml) levels obtained from the HEGC study as well as from a separate set of experiments aimed to determine circulating levels of plasma biomarkers. HOMA-IR was established as the product of FBG and FPI divided by a constant of 22.5 (25).
Analysis of plasma biomarkers (FBG, insulin, tumour necrosis factor α (TNFα), interleukin (IL)-1β and IL-6)
A separate set of experiments was performed to measure FBG, plasma insulin, TNFα, IL-1β and IL-6 concentrations. Overnight fasted rats were anesthetised with thiopental sodium (100 mg/kg, i.p.) and a cannula was placed into the carotid artery for blood sampling. After a short stabilisation period, blood glucose was determined by means of a glucometer, and blood samples were taken in Eppendorf cups. Samples were processed and stored as described above. Insulin concentrations of plasma samples, including those that were collected during the HEGC study, were determined by immunoradiometric assay (IRMA) using a commercially available insulin IRMA kit (Institute of Isotopes, Budapest, Hungary). TNFα, IL-1β (Thermo Scientific, Waltham, MA, USA, each) and IL-6 (Life Technologies, Carlsbad, CA, USA) were measured by enzyme-linked immunoassay (EIA) according to the manufacturer’s instructions.
Measurement of adiposity
The group of rats used for the assessment of metabolic parameters was also used later for the measurement of adiposity. After completing the experiments, intra-abdominal and epididymal white adipose tissue (WAT) fat pads were removed and weighed. Adiposity was characterised by expressing the sum of the WAT fat pads as a percentage of total BW.
In vivo recordings of meningeal blood flow
Changes in meningeal blood flow were measured in an open cranial window model (26). Control and obese rats were anesthetised with thiopental sodium (150 mg/kg, i.p.) and the trachea was cannulated to allow spontaneous breathing. Systemic blood pressure was recorded with a pressure transducer connected to a cannula inserted into the femoral artery. The body temperature was monitored by a rectal thermoprobe and was kept at 37–37.5℃ with a heating pad. The head of the rats was stabilised in a stereotaxic frame and a cranial window was made into the parietal bone by means of a saline-cooled drill to expose the dura mater (8,26). Blood flow was recorded with a needle-type probe of a laser Doppler flowmeter (Perimed, Sweden) positioned over a branch of the middle meningeal artery lying distant from visible cortical blood vessels. Blood flow was measured in perfusion units (PU). Data on meningeal blood flow and systemic blood pressure were processed with the Perisoft program (Perimed, Sweden). Stimulation of the dura mater was performed by topical application of acrolein (50, 100 and 300 µM), CGRP and histamine (both 100 µM) at a volume of 40 µl for 5 min followed by repeated washouts with synthetic interstitial fluid (SIF, containing in mM: 135 NaCl, 5 KCl, 1 MgCl2, 5 CaCl2, 10 glucose and 10 Hepes, pH 7.4) between them. In control rats the effects of pretreatments with the TRPA1 receptor antagonist HC-030031 (50 µM) and the CGRP receptor antagonist CGRP8–37 (100 µM) on acrolein-induced (300 µM) blood flow increase also were tested. Both antagonists were topically applied for 5 min prior to stimulation with acrolein (300 µM). HC-030031 was purchased from Tocris, Bristol, UK (stored as a 10 mM stock solution in dimethyl sulfoxide (DMSO)); all other drugs were from Sigma-Aldrich Chemie Gmbh (Germany) and were dissolved in SIF immediately before use. Basal blood flow was determined as the mean flow during 5 min prior to drug application. Percentage changes in meningeal blood flow in response to drug application were determined by comparing basal flow with the mean value of 1 min where change in blood flow was maximal within the 5-min application period.
In vitro measurement of CGRP release
An in vitro rat dura mater preparation was used to assess basal and stimulated release of CGRP (27). Control and obese rats were decapitated following deep anaesthesia with thiopental sodium (150 mg/kg i.p.). The skull was divided into halves along the midline after the removal of the skin and muscles. The cerebral hemispheres were removed, skull halves were washed at room temperature for 30 min in carbogen-gassed SIF, then settled in a humid chamber and the cranial fossae were filled with 300 µl SIF. Samples of the superfusate were collected with a micropipette at periods of 5 min for CGRP measurement. A control sample was taken after two consecutive washouts with SIF in order to determine basal CGRP release, then the dura was stimulated with acrolein at concentrations of 10, 50, 100 or 300 µM. In some control experiments the effect of the TRPA1 receptor antagonist HC-030031 (50 µM) pretreatment on acrolein-induced (300 µM) CGRP release was also measured. In the other skull half the effect of KCl (60 mM) application was compared with basal CGRP release.
A total of 100 µl of samples diluted with 25 µl EIA buffer were placed into Eppendorf cups and immediately frozen at –70℃ for subsequent analysis. The EIA method was used for CGRP determination (Bertin Pharma, France). The CGRP concentrations of the superfusates were expressed in pg/ml. Changes induced in CGRP release by acrolein and KCl were expressed as percentage changes relative to the basal release.
Measurement of TRPA1 protein expression in the trigeminal ganglion
Western blot analysis was used to determine TRPA1 protein expression in the trigeminal ganglia. Overnight fasted rats were anesthetised with thiopental sodium (150 mg/kg, i.p.) and decapitated. Trigeminal ganglia were removed and homogenised in ice-cold NP40 buffer (Thermo Fisher Scientific, Rockford, IL, USA) supplemented with 1 mM phenylmethylsulfonyl fluoride and protease inhibitor cocktail (both Sigma-Aldrich, St. Louis, MO, USA). Homogenates were sonicated twice for 10 seconds (sec) each and agitated constantly for two hours at 4℃. Lysates were centrifuged for 20 min at 13,680 g at 4℃. The protein concentration of the supernatant was determined by BCA Protein Assay (Thermo Fisher Scientific, Rockford, IL, USA). Whole-cell lysates (40-50 µg protein) were analysed on 10% sodium dodecyl sulphate (SDS) polyacrylamide gel under reducing conditions. Immunoblotting was carried out on a nitrocellulose membrane using a rabbit anti-TRPA1 antibody (1:2500, Alomone Labs, Jerusalem, Israel) together with β-actin (1:1000, Cell Signalling Technology, Danvers, MA, USA) overnight at 4℃ followed by incubation with donkey anti-rabbit immunoglobulin (Ig)G-horseradish peroxidase (HRP) (1:5000, Santa Cruz Biotechnology, Dallas, TX, USA) for an hour at room temperature. Immunoreactive bands were visualised by the SuperSignal West Pico Chemiluminescent Substrate (Thermo Fisher Scientific, Rockford, IL, USA) using a Gel Logic 1500 Imaging System (Kodak, Tokyo, Japan).
Statistics
All values were expressed as means ± SEM. Statistical analysis of the data was performed using Statistica 12 (StatSoft, Tulsa, OK, USA). For the statistical comparisons of CGRP and cytokine concentrations, meningeal blood flow changes and TRPA1 protein expression the Student’s t test was used for group sizes of n ≥ 10 and the Mann-Whitney U test for independent measurements of group sizes n < 10. One-way analysis of variance (ANOVA) followed by the Bonferroni test was used to compare metabolic parameters of the animals. A probability level of p < 0.05 was regarded as statistically significant.
Results
Effect of HFHS diet on BW, adiposity and metabolic parameters
Effect of high-fat, high-sucrose diet on the amount of intra-abdominal and epididymal fat pads and the ratio of white adipose tissue to body weight (WAT/BW).
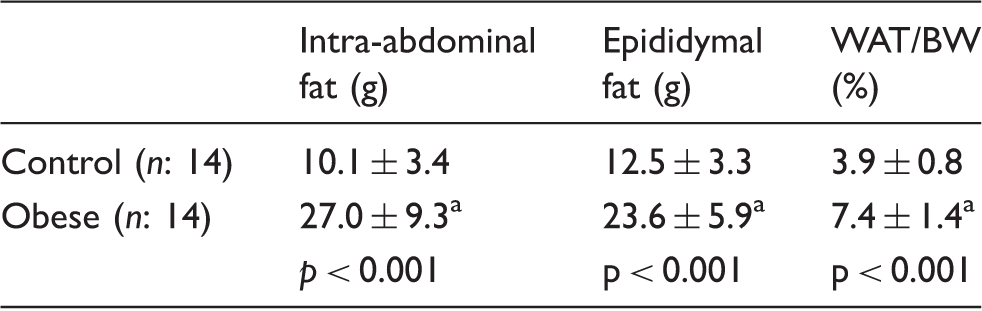
Statistically different from controls.
Effect of diet-induced obesity on fasting blood glucose (FBG) and fasting plasma insulin (FPI) concentrations, homeostasis model assessment of insulin resistance (HOMA-IR) index and glucose infusion rate (GIR) of the animals.

Statistically different from controls.
HFHS diet influenced differentially plasma cytokine levels. In obese animals significantly higher concentrations of IL-1β (p = 0.011) and IL-6 (p < 0.001) were measured, whereas TNFα levels were similar in control and obese rats (n = 9 for both groups, p = 0.26; Figure 1).
Effect of diet-induced obesity on plasma concentrations of tumour necrosis factor α (TNFα), interleukin-1β (IL-1β) and interleukin-6 (IL-6). The number of experiments is indicated in the bars. *Statistically different from the control.
Diet-induced obesity potentiates increases in meningeal blood flow elicited by acrolein
Basal blood flow measured in PU depends critically on the size of the arteries over which the probes of the laser Doppler flowmeter are positioned. In our experimental settings, therefore, we have carefully chosen identical segments of the medial meningeal artery for the blood flow measurements. The basal flow values were in the same range in control and obese rats amounting to 226.1 ± 20.2 and 214.4 ± 20.1 PU (p = 0.96), respectively. Topical administration of drugs activating TRPA1 receptors of trigeminal afferents (acrolein), or acting directly on endothelial- or smooth muscle cells of blood vessels (histamine and CGRP) increased meningeal blood flow both in control and obese rats. The maximal increase in blood flow could be observed generally in the third, and in a few cases in the fourth min of the 5-min drug application period independently of the type of stimulating agent.
Systemic blood pressure was slightly above the values measured earlier in non-obese animals under the same experimental conditions (8). It was 128 ± 17 and 134 ± 13 mmHg in control and obese rats, respectively. Drugs administered topically to the dura mater failed to influence systemic blood pressure.
In control rats acrolein induced a dose-dependent increase in meningeal blood flow that was significantly different from the controls at 100 (n = 10; p < 0.001) and 300 µM (n = 12; p < 0.001) concentrations. The blood flow-increasing effect of acrolein at 300 µM was abolished by the pretreatment of the dura mater with the TRPA1- (n = 8; p = 0.001) and CGRP receptor antagonists (n = 8; p = 0.001, Figure 2).
Changes in acrolein-, histamine- and calcitonin gene-related peptide (CGRP)-induced increases in meningeal blood flow. (a) Original recordings and (b) statistical evaluation indicating the maximal blood flow-increasing effects of topical applications of acrolein (50, 100 and 300 µM), histamine, CGRP (both 100 µM) and effects of TRPA1- (HC-030031, 50 µM) and CGRP receptor antagonist (CGRP8–37, 100 µM) on acrolein (300 µM)-induced meningeal vasodilatation. The number of experiments is indicated in the bars. *Statistically different from the basal flow, #Statistically different from the effect of acrolein at 300 µM, ‡Statistically different from controls.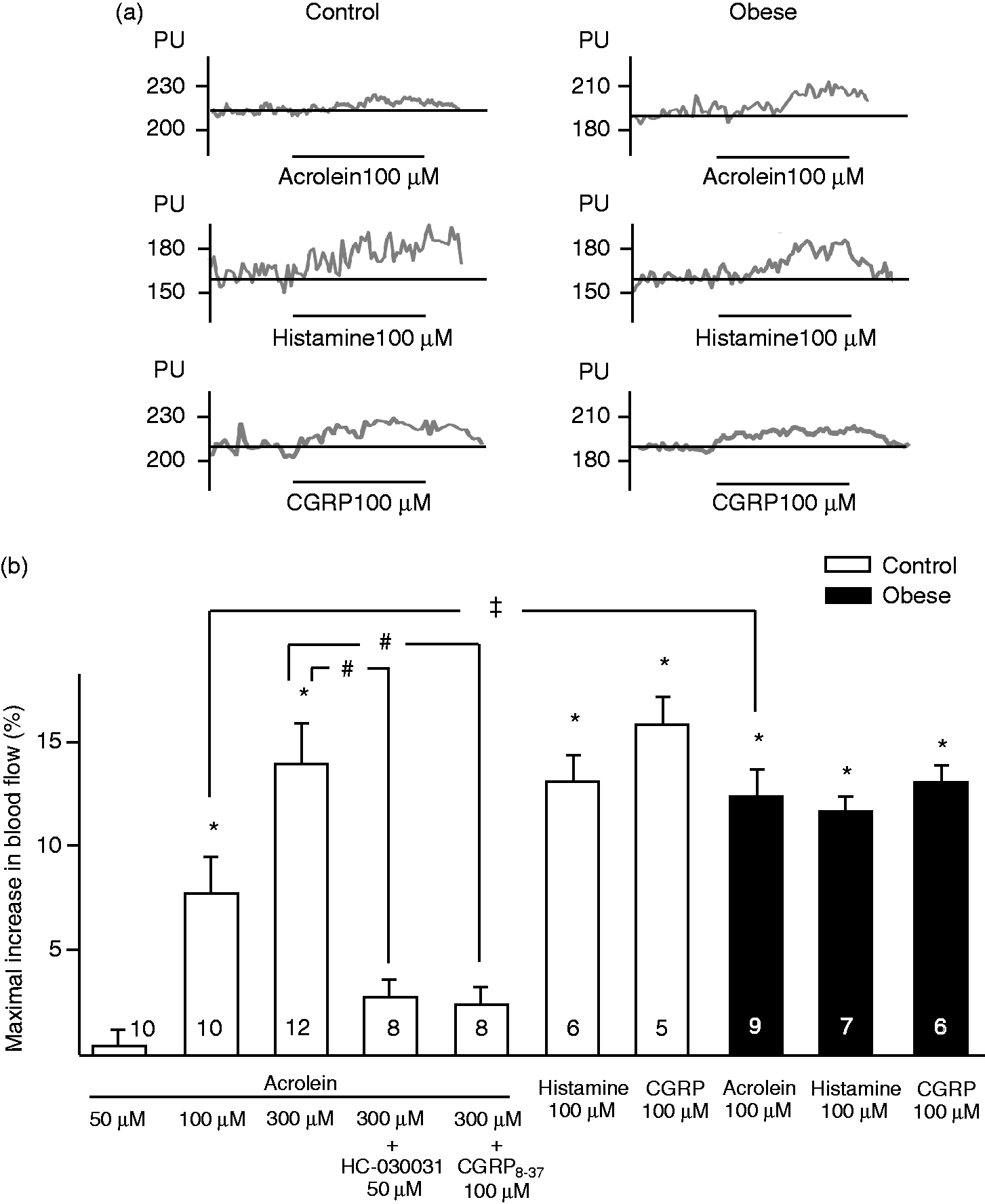
Dural administration of acrolein resulted in significantly higher increases in maximal meningeal blood flow in obese animals (control n = 10, obese n = 9; p = 0.049). Histamine- and CGRP-induced increases in meningeal blood flow were similar in control (n = 6 and 5, respectively) and obese rats (n = 7 and 6; p = 0.29 and 0.53, respectively, Figure 2).
Diet-induced obesity enhances basal and acrolein-stimulated CGRP release and attenuates the response to KCl
In the ex vivo dura mater preparation the basal release of CGRP was 16.03 ± 1.59 in control and 40.23 ±6.2 pg/ml in obese rats (p < 0.001). These data indicate a significantly increased spontaneous basal CGRP release in obese animals. Acrolein at a concentration of 10 and 50 µM failed to influence CGRP release in control animals (n = 7 and n = 6, p = 0.88 and p = 0.203, respectively). Acrolein at 10 µM concentration was also ineffective in obese animals (n = 7, p = 0.51). Higher concentrations of acrolein (100 and 300 µM) significantly increased the release of CGRP (control: n = 8, p < 0.001 for both acrolein concentrations; obese: n = 14, p = 0.001 for 100 µM). Acrolein-induced CGRP release was inhibited by pretreatment with the TRPA1 antagonist (n = 8, p = 0.001). Obese rats released significantly more CGRP as compared with the control group (p = 0.039, Figure 3).
Changes in acrolein- and KCl-induced release of calcitonin gene-related peptide (CGRP) from meningeal afferents. (a) The hemisected rat skull preparation used for measurement of CGRP release from meningeal afferents. (b) CGRP concentrations (normalised to the basal release) after topical applications of acrolein (10, 50, 100 and 300 µM), KCl (60 mM) and effect of transient receptor potential ankyrin 1 (TRPA1) receptor antagonist (HC-030031, 50 µM) on acrolein (300 µM)-induced CGRP release. The number of experiments is indicated in the bars. *Statistically different from the basal CGRP release, #Statistically different from the effect of acrolein at 300 µM, ‡Statistically different from controls.
KCl (60 mM) increased CGRP release to 307.4 ±38.3% (n = 8, p < 0.001) and 182.7 ± 18.2% (n = 13, p < 0.001) in control and obese animals, respectively. Although depolarisation-induced increase in CGRP release was significantly different from basal values in both groups, the high potassium-induced release was significantly lower in the obese rats as compared with the controls (p = 0.001, Figure 3).
Diet-induced obesity reduces TRPA1 protein expression in the trigeminal ganglia
The TRPA1 antibody detected a band at 110 kDa from whole-cell lysates of rat trigeminal ganglia. Following incubation of anti-TRPA1 antibody with its cognate peptide provided by the manufacturer, the band could no longer be detected indicating the specificity of the antibody for the TRPA1 protein. There was a significant reduction in the expression of this 110 kDa protein in the trigeminal ganglia of obese rats compared with the control group (control: n = 7, obese: n = 8, p = 0.048, Figure 4).
Expression of transient receptor potential ankyrin 1 (TRPA1) protein in the trigeminal ganglia. (a) Representative Western blot and (b) protein expression in control and obese animals normalised to β-actin. The number of experiments is indicated in the bars. *Statistically different from the control.
Discussion
The present study was initiated in an attempt to reveal possible mechanisms through which metabolic changes may affect the trigeminovascular system resulting in disturbances of trigeminal sensory functions, in particular the sensation of intracranial pain. Therefore, the effects of HFHS diet-induced obesity was studied on the trigeminovascular system by combining well-established methods of experimental headache research with measurements of metabolic parameters and plasma levels of inflammatory cytokines.
Rats maintained on an HFHS diet exhibited elevated FBG and insulin levels resulting in a significant increase in HOMA-IR index and a decreased GIR. Impairments in glucose and insulin homeostasis were associated with increased plasma levels of IL-1β and IL-6 (28).
Sensory neurogenic vascular reactions of dural blood vessels are reliable indicators of the efferent, local vascular functions of chemosensitive afferent nerves serving the dura mater (8,26,29). In the present study, administration of acrolein, a TRPA1 receptor agonist, resulted in an increase in meningeal blood flow that was inhibited by pretreatment of the dura mater with the TRPA1- or CGRP receptor antagonist, suggesting an activation of peptidergic meningeal chemosensitive afferent nerves. In vitro experiments furnished direct evidence for the release of CGRP by acrolein. These observations provide further support for acrolein, a common environmental pollutant, being an agonist on TRPA1 receptors of meningeal afferents initiating increases in dural blood flow implicated in the mechanism of intracranial pain (11,30). Probably the most intriguing finding of the present study was that, in obese animals, in comparison with the controls, dural application of acrolein resulted in significantly increased elevations of meningeal blood flow. In accord with this finding, acrolein-evoked in vitro release of CGRP was significantly increased in obese animals. Interestingly, in obese rats, the basal release of CGRP was also elevated. The meningeal vasodilatory effects of histamine and CGRP, which act directly on their endothelial and vascular smooth muscle receptors, were similar in control and obese animals.
In obese rats the markedly elevated levels of circulating IL-1β and IL-6 indicate a systemic inflammatory condition of the animals (4). These cytokines may activate and sensitise trigeminal afferents in multiple ways such as altering the expression and/or modifying the gating properties of ion channels (31). In vivo electrophysiological recordings demonstrated that both IL-1β and IL-6 can increase the excitability of meningeal nociceptors (32,33). IL-1β has been found to stimulate cyclooxygenase-2 and prostaglandin E2 synthesis as well as the release of CGRP in cultured trigeminal ganglion neurons (34). Inflammatory soup consisting of prostaglandin E2, bradykinin and histamine profoundly affects the electrophysiological properties of dural afferents leading to a decrease in the activation threshold and an increase in firing rate (35). Inflammatory soup has been shown to depolarise the resting membrane potential of meningeal afferents (31). IL-1β was able to exert similar effect in small trigeminal nociceptive neurons through the inactivation of voltage-gated potassium channels (36). Change in resting membrane potential is considered an important mechanism underlying the sensitisation of dural afferents evoked by inflammatory mediators leading to increased basal CGRP release (31). CGRP release induced by high KCl concentration requires the opening of voltage-gated calcium channels (37). The activity and/or expression of these channels may also be influenced by persistently elevated IL-1β and IL-6 levels possibly contributing to reduced CGRP release upon neuronal depolarisation with KCl (38). The IL-1β-induced increase in cyclooxygenase-2 activity, however, may result in the production of a variety of electrophilic molecules that can potentially modulate TRPA1 function (39,40). Our findings on unchanged levels of plasma TNFα in obese rats are in line with several reports showing that diet-induced obesity may increase TNFα concentration in the adipose and muscle tissue, but it does not necessarily lead to increased plasma levels of TNFα (41,42).
Although this aspect was not specifically investigated, oxidative and nitrosative stress may also modulate the observed neurovascular reactions in obese animals. Oxidative stress has been found to be increased in the nervous system of diet-induced obese mice that was correlated with the amount of adipose tissue (43). In our study, obese rats had significantly higher FBG concentrations accompanied by a marked hyperinsulinaemia whereas insulin resistance was also confirmed by increased HOMA-IR index and lower GIR value (44). High-fat diet-induced obesity with reduced glucose tolerance and elevated insulin levels has been shown to be associated with the accumulation of 4-hydroxynonenal (4-HNE), an important biomarker of lipid peroxidation, in primary sensory neurons (45). 4-HNE can selectively activate TRPA1 resulting in enhanced excitability of chemosensitive nociceptors and release of CGRP (46).
Since in HFHS obese rats impaired glucose and insulin homeostasis and increased levels of IL-1β and IL-6 were observed, it is reasonable to assume that changes in basal and stimulated CGRP release can be the final outcomes of intricate dose- and time-dependent interactions of the above factors resulting in sensitisation of dural afferents. This assumption is in line with previous findings showing an enhanced activity of the trigeminal pain pathway in obese mice (47,48).
Channel sensitisation, increased trafficking and/or membrane expression of the receptors are possible mechanisms of nociceptor sensitisation. Although data about the exact mechanisms of cytokine-induced sensitisation of TRPA1 receptors are sparse, knowledge concerning the cytokine effects on another member of the TRP receptor family, the transient receptor potential vanilloid 1 (TRPV1) receptor, which is highly colocalised with TRPA1 in dural afferents (49), may be relevant. Indeed, an increased trafficking of both TRPV1 and TRPA1 receptors induced by TNFα has been demonstrated in trigeminal neurons (50), while IL-1β and bradykinin sensitised TRPV1 receptor without increasing its membrane expression (51). Since in our obesity model an increase in plasma IL-1β concentration was associated with an unaltered TNFα level and a slightly reduced TRPA1 receptor expression, the augmented neurovascular responses following TRPA1 activation may be attributed to channel sensitisation rather than an altered expression and/or trafficking of the receptor.
TRPA1-immunoreactive perivascular axons form bundles in the dura mater (30), in which the exact number of immunoreactive afferents cannot be reliably determined. Therefore we applied the Western blot technique to provide (semi)quantitative data on the density of meningeal TRPA1 receptors. The reduced TRPA1 protein expression in the trigeminal ganglia of obese rats can be considered part of compensatory mechanisms due to persistently increased neuronal activity (52).
Recent findings have shown that collaterals of chemosensitive meningeal nociceptors project through the skull, innervate extracranial tissues, e.g. nasal mucosa, forming functional connections between extra- and intracranial structures (53). Sensitised TRPA1 channels of trigeminal afferents innervating the nasal mucosa are targets of inhaled irritants, such as acrolein, and their stimulation may lead to enhanced activity of the trigeminovascular nociceptive pathway in obese animals, and probably in humans.
In conclusion, this study demonstrates that HFHS-diet-induced obesity leads to the sensitisation of dural afferents and, consequently, increased neural and vascular responses in the trigeminovascular system upon TRPA1 activation. The findings may also bear relevance with respect to pathologies relating to obesity and environmental pollutants. First, an increased incidence of migraine headache is well documented in obese individuals (1). Second, several environmental pollutants have been shown to produce headache through activation of the TRPA1 receptor (54). The present study, by revealing changes in metabolic homeostasis, plasma cytokine levels and, in particular, sensitisation of TRPA1 receptors expressed by trigeminovascular afferents, provides novel insight into possible mechanism of headaches induced by environmental pollutants.
Article highlights
Diet-induced obesity leads to sensitisation of meningeal trigeminovascular afferents. Enhanced activity of the nociceptive pathway through obesity-induced sensitisation of trigeminal transient receptor potential ankyrin 1 (TRPA1) receptors may promote headache generation. Obesity-induced sensitisation of TRPA1 receptors is likely the result of altered gating properties of the receptor channel. Obesity increases basal and TRPA1 receptor-mediated release of calcitonin gene-related peptide (CGRP) from trigeminovascular afferents.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grant K-101873 of the Hungarian Scientific Research Fund (OTKA). AV was supported by a Hungarian Social Renewal Operation Program (grant number TÁMOP 4.1.2.E-13/1/KONV-2013-0010). BM was supported by the Gedeon Richter’s Talentum Foundation (grant number 1103 Budapest, Gyömrői út 19-21).
