Abstract
Background
The association between migraine and the incidence of ischemic stroke varies in different subgroups of patients. We aimed to clarify this association using a population-based database.
Method
A nationwide cohort study was conducted using data from the Taiwan National Health Insurance Research Database. Two cohorts were extracted: a neurologist-diagnosed migraine cohort, and a non-headache, propensity score-matched comparison cohort. All participants were enrolled in this study between 2005 and 2009, and were followed through the end of 2010, death, or the occurrence of ischemic stroke. Adjusted hazard ratios (aHRs) and 95% confidence intervals (CIs) were calculated with a Cox proportional hazards model to compare the between-group risks.
Results
Both cohorts (n = 119,017 each) were followed for a mean period of 3.6 ± 1.3 years. A total of 744 migraine patients (429,741 person-years) and 617 matched comparison individuals (436,141 person-years) developed ischemic stroke during the research period. Compared to the comparison cohort, patients with migraine were at an increased risk of ischemic stroke (aHR: 1.24, 95% CI: 1.12–1.38, p < 0.001). Subgroup analysis by age and sex revealed the highest risk in women aged ≤ 45 years (aHR: 3.44, 95% CI: 2.20–5.39, p < 0.001), especially among those with migraine with aura (aHR: 4.58, 95% CI: 2.45 – 8.56, p < 0.001). A trend for increased stroke risk was observed in men aged ≤ 45 years (aHR: 1.54, 95% CI: 0.96–2.48, p = 0.075).
Conclusion
Migraine is associated with an increased risk of ischemic stroke, especially in younger (age ≤ 45 years) women with migraine with aura. The trend toward ischemic stroke in younger men merits further exploration.
Introduction
Migraine is a common and disabling disorder, with an overall one-year prevalence of 8%–15% (4.5%–6% in men, 14%–18% in women). The incidence of migraine peaks at 25–55 years of age (1–3). Migraine is considered to be a neurovascular disease, in which the trigeminovascular system plays a major role (4). In addition, migraine is associated with other vascular disorders, including ischemic and hemorrhagic stroke (5–7), coronary artery disease (8) and peripheral arterial disorders (9).
Migraine is classified into migraine with aura (MA) and migraine without aura (MO). Early migraine studies reported an increased risk of ischemic stroke among migraine patients (5), and subsequent cohort studies confirmed this association (10,11). However, those studies showed that the increased risk of ischemic stroke existed exclusively in a specific subgroup of migraine patients, namely female patients with MA (7,10). Male patients with migraine, in contrast, were more prone to develop myocardial infarction, but not stroke (8).
Despite the reported link between migraine and ischemic stroke, some cohort studies have underrepresented younger migraine patients (e.g. age ≤ 50 years), in whom migraines are most prevalent (7,8). In addition, in a recent study the risk of ischemic stroke is increased in elderly migraine patients who smoke; however, the status of migraine (i.e. MA or MO) does not alter the association (12). In light of this background, we sought to clarify whether the risk of ischemic stroke in migraine patients is modulated by age, sex and status of migraine.
Methods
Data sources
Taiwan began the National Health Insurance (NHI) program in 1995 to finance health care for all of its citizens, and has a coverage rate of up to 98% (13). The NHI is a mandatory universal health insurance program that offers all Taiwanese residents comprehensive medical care coverage including outpatient, inpatient, emergency, dental, traditional Chinese medicine services, and prescription drugs. The diagnosis of diseases follows the codes in the International Classification of Diseases, ninth revision, clinical modification (ICD-9-CM), 2001 edition (14). This study used the NHI databases managed and publicly released by the National Health Research Institute, Taiwan. The multiple NHI databases (the NHI enrollment files, the claims data, and the registry for drug prescriptions) provide comprehensive utilization and enrollment information for all patients under the NHI program. All information that would potentially expose a specific individual patient to identification has been encrypted. The confidentiality of the data abides by the data regulations of the Bureau of National Health Insurance and the National Health Research Institute. The institutional review board approved this study.
Study population
The migraine cohort was composed of all patients, out of a population of approximate 23 million, who visited a neurologist and were given a diagnosis code of migraine (code 346.x) between January 2005 and December 2009. According to the ICD-9-CM, three subgroups of migraine were defined: MA, codes 346.00–346.01; MO, codes 346.10–346.11; and migraine unspecified, codes 346.90–346.91. Each migraine patient entered the cohort on the day a code of migraine was given by a neurologist. We enrolled people aged ≥ 20 years, and excluded individuals who had antecedent strokes, malignancy, or acquired immune deficiency syndrome (AIDS) between January 2004 and December 2009. We retrieved information of comorbid diseases known as risk factors for ischemic stroke, according to the INTERSTROKE study (15), including hypertension, diabetes mellitus, coronary artery disease and atrial fibrillation.
The matched comparison cohort was selected from a dataset of one million beneficiaries who were randomly sampled from the original NHIRD. In the comparison cohort, individuals with diagnosis of migraine (codes 346.x), tension-type headache (code 307.81), or headache (code 784.0) between January 2004 and December 2009 were excluded. Since patients in the comparison cohort had no headache diagnosis, the index date could not be assigned. Thus, we randomly give each of the participants a “pseudodiagnostic” date corresponding to the index date of one of the patients in the migraine cohort. Similar to the migraine cohorts, we enrolled those aged ≥ 20 years, and the same exclusion criteria also apply to the comparison cohort.
Propensity score matching
Propensity scores were calculated using multivariate logistic regression for the predicted probability of migraine status (Supplementary Table 1). For each migraine patient, we identified one non-headache, i.e. one without, comparison participant with a similar demographic characteristic, based on propensity score (±0.1) for the likelihood of a migraine diagnosis. Individuals who could not be matched were excluded from the study (Figure 1).
Enrollment of patients.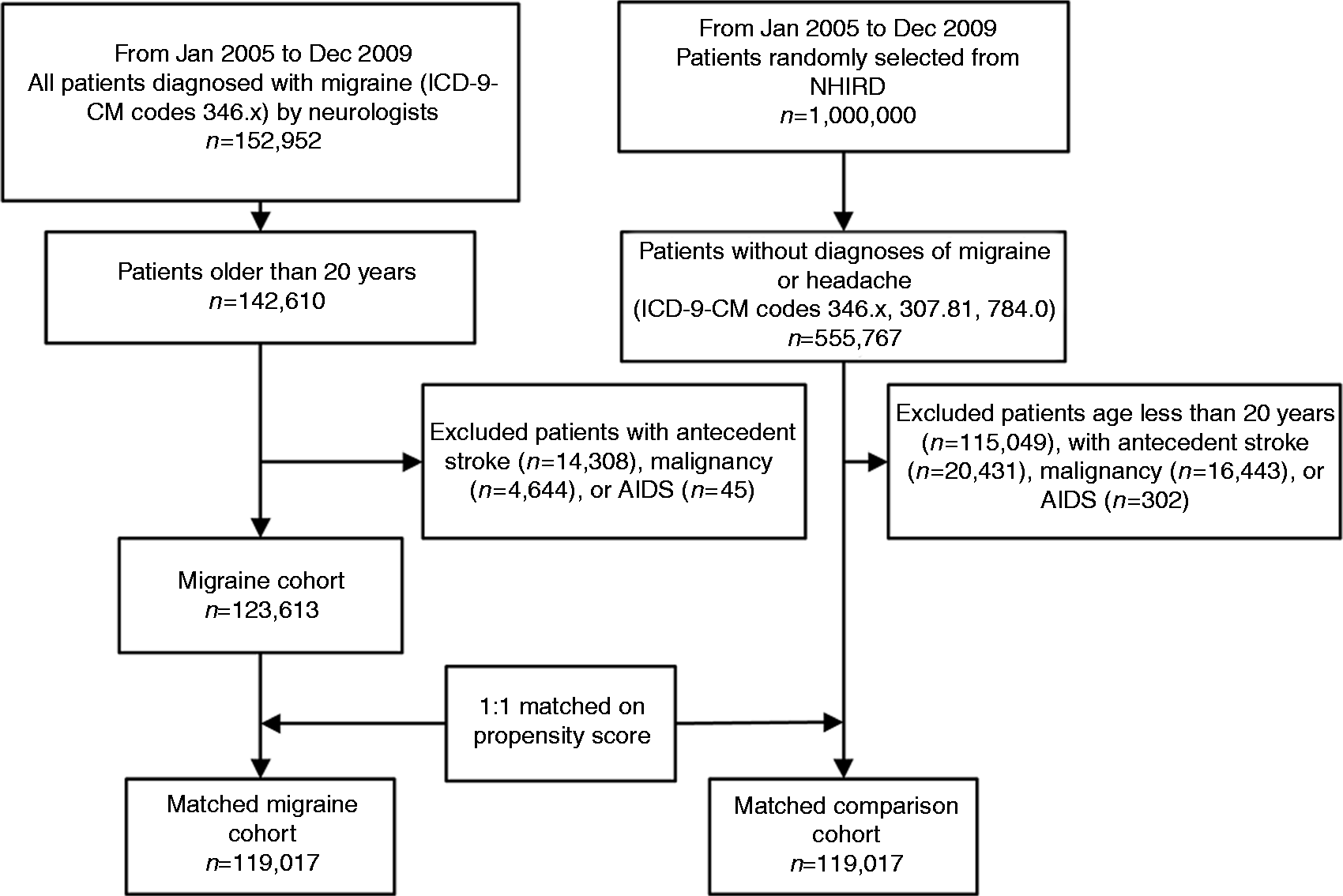
Main outcome and follow-up
The main outcome of ischemic stroke was defined as hospitalization claim with the diagnosis codes of 433–434. The accuracy of diagnosis of ischemic stroke with hospitalization in this database was 94% in a validation study (16). All participants in the migraine and comparison cohorts were followed up until the diagnosis of ischemic stroke, termination of NHI coverage, death, or December 2010, whichever came first.
Statistical analyses
The demographics both for the migraine patients and the comparison cohort were examined using descriptive statistics. Incidence rates (per 100,000 person-years) were calculated using Poisson distribution methods. The relative risk of ischemic stroke between the migraine and the comparison groups was calculated with the hazard ratio (HR) using the Cox regression models with a conditional approach using stratification. Adjusted HR (aHR) was calculated regarding propensity score as a continuous variable. The influences of age, Charlson Comorbidity Index (CCI), diabetes mellitus, coronary artery disease, hypertension and atrial fibrillation subgroups on the incidence of ischemic stroke were also calculated using the Cox regression model. Tests of interactions were performed in subgroup analysis using the likelihood ratio test. Sensitivity analysis was conducted to validate our results in those without oral contraceptive use.
The Microsoft SQL Server 2008 R2 (Microsoft Corp., Redmond, WA, USA) was used for extraction and computation of data, data linkage, processing, and sampling. All statistical analyses were performed using STATA statistical software version 12.0 for Windows (StataCorp., College Station, TX, USA). Statistical significance was defined as a p value of less than 0.05.
Results
Study cohorts
Demographic and clinical characteristics of patients.
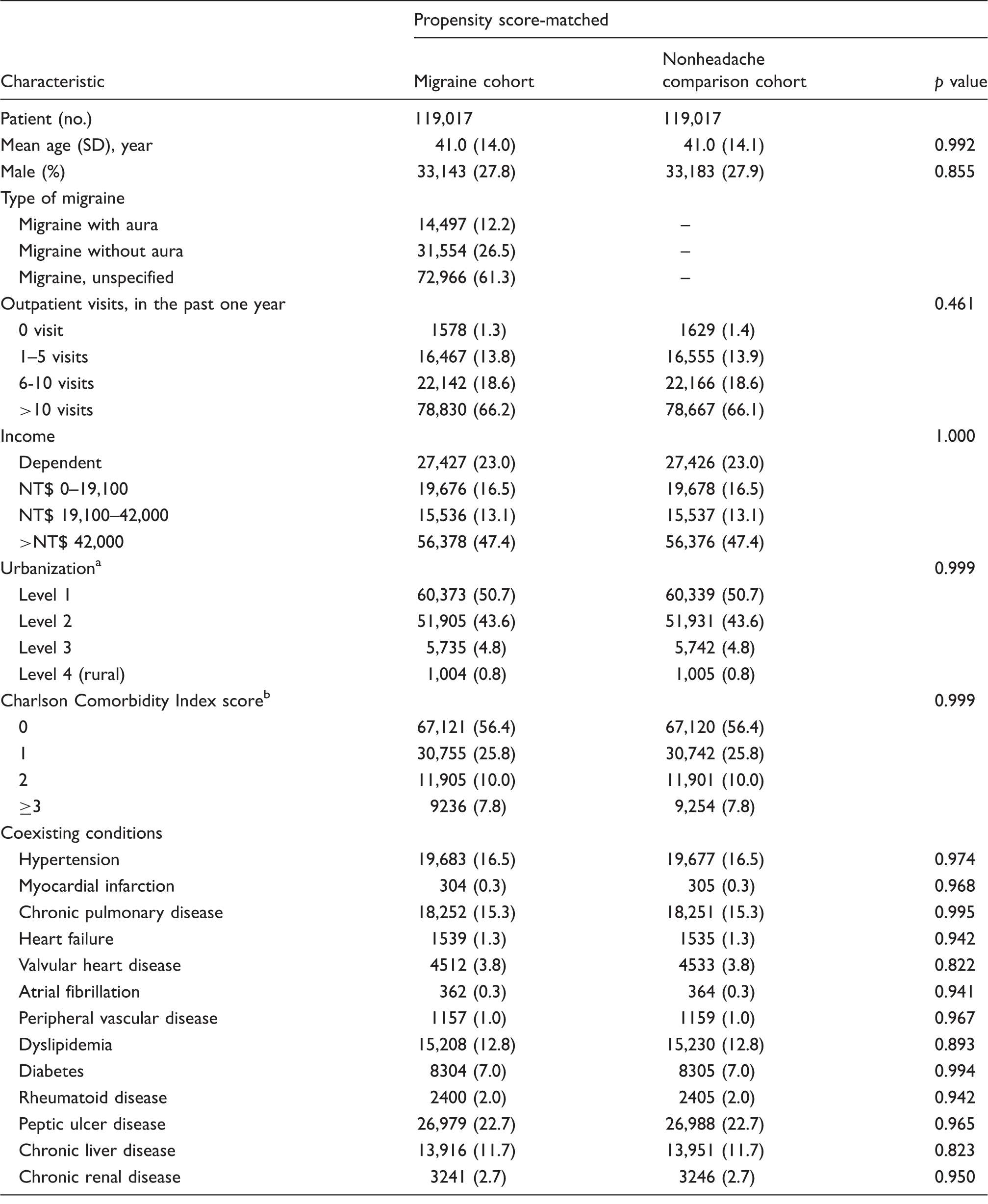
SD: standard deviation; NT$: new Taiwan dollars.
Urbanization levels in Taiwan are divided into four strata according to the Taiwan National Health Research Institute publications, ranging from level 1 (most urbanized areas) to level 4 (least urbanized areas).
Charlson Comorbidity Index (CCI) score is used to determine overall systemic health. With each increased level of CCI score, there is a stepwise increase in cumulative mortality.
Incidence of ischemic stroke
After a mean follow-up period of 3.6 ± 1.3 years (range: 1–6 years), 744 migraine patients (429,741 person-years) and 617 matched comparison individuals (436,141 person-years) had been newly diagnosed with ischemic stroke. Incidence rates of ischemic stroke were 173.1 and 141.5 per 100,000 person-years in the migraine and comparison cohort, respectively. Compared to the matched comparison cohort, the overall risk of ischemic stroke was increased among migraine patients (HR: 1.24, 95% CI: 1.12–1.38, p < 0.001). Among the 119,017 participants in each group, 4841 patients in the migraine group and 4899 in the comparison groups had early termination of follow-up due to either the termination of NHI coverage or death.
Age stratum and risk for ischemic stroke among migraine and nonheadache comparison cohorts.

Per 104 person-years.
Interaction p < 0.001. cAdjusted for propensity score.
HR: hazard ratio; CI: confidence interval.
Impacts of sex, age and migraine status on development of ischemic stroke
Incidence rates and hazard ratios for ischemic stroke events among migraine and nonheadache comparison cohorts.
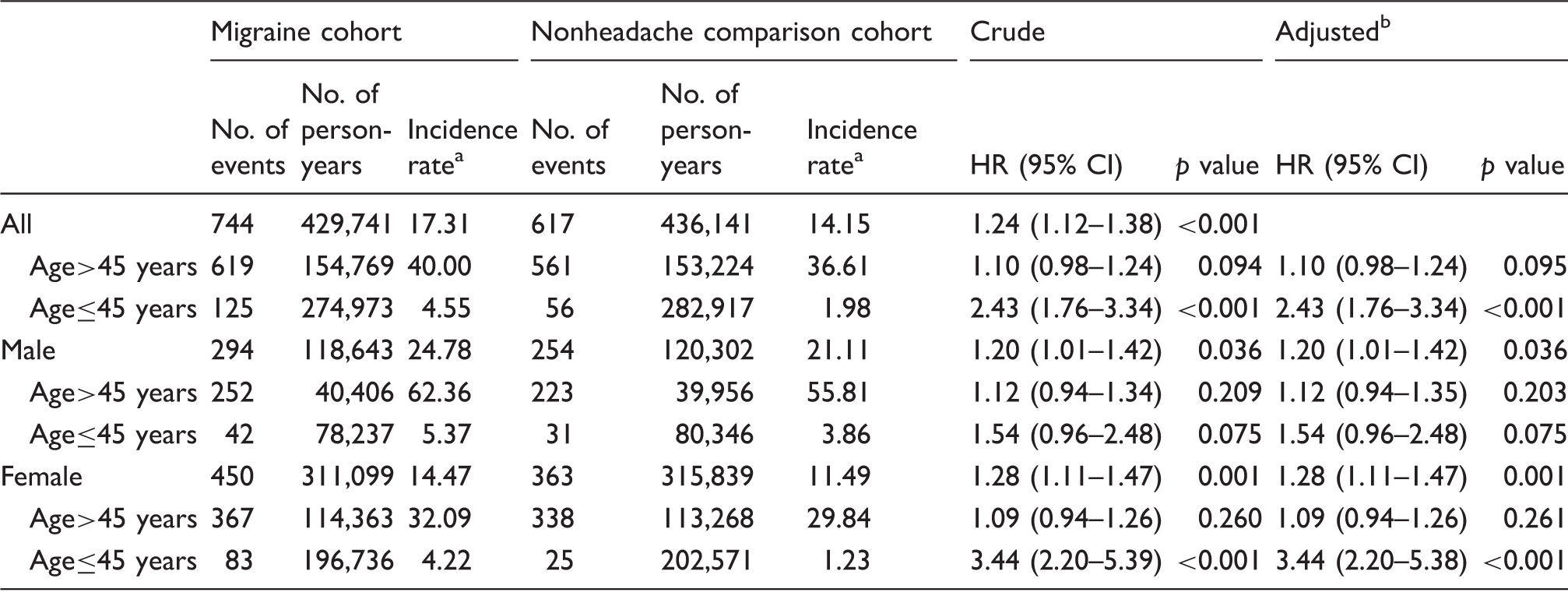
Per 104 person-years.
Adjusted for propensity score.
HR: hazard ratio; CI: confidence interval.
Risk of ischemic stroke among different categories of migraine patients in a propensity score-matched model.

Adjusted for propensity score.
HR: hazard ratio; CI: confidence interval.
Oral contraceptive use and ischemic stroke
Sensitivity analysis of the risks of ischemic stroke between migraine and nonheadache comparison cohorts.

Per 105 person-years.
CI: confidence interval.
Discussion
Results of this large-scale study confirm an association between migraine and the development of ischemic stroke. Moreover, this association was found to be modulated by age, sex and migraine subtype. Most previous studies of this association have concerned Western populations (7,10,17). Prevalence rates of MA and of stroke subtypes differ between Asians and Caucasians, with Asian populations demonstrating a lower prevalence of MA and higher incidence of hemorrhage stroke (3,18,19). Our study adds to the evidence that the differential risk of ischemic stroke in younger female patients with MA is universal across different ethnic groups.
In this study, the incidence of ischemic stroke in the comparison cohort was 145 per 100,000 person-years. This rate is slightly higher than that reported in an earlier population-based prospective cohort study (19). The difference may be due to the fact that Taiwanese individuals have universal health insurance coverage and easy access to medical care. The mean number of clinic visits per person-year reached 15.6 in 2012 (20).
In line with prior cohort studies, we found that the risk of ischemic stroke was increased in female patients with MA (aHR: 1.60, 95% CI: 1.08–2.38, p = 0.019), but not in female patients with MO (aHR: 1.08, 95% CI: 0.82–1.42, p = 0.566). This difference between individuals with MA versus MO is consistent with the findings of a recent meta-analysis (HR: 2.16 for MA vs. 1.23 for MO) (21). Among female participants ≤ 45 years old, the risk was even higher, and was exclusively associated with MA (aHR: 5.79, 95% CI: 1.69–19.89, p = 0.005), similar to the findings of Chang et al. (17). In the Women's Health Study, the risk of ischemic stroke did not differ between the MA and non-migraine cohorts until six years of follow-up (11). Our study, with a relatively short follow-up duration of 3.6 years compared to 10 years in the Women's Health Study, still identified an association between migraine and ischemic stroke. This difference might be due to differences in study design, study population, ethnic composition, or other unaccountable environmental factors.
In contrast with previous large cohort studies (8,10), we found an increased risk of ischemic stroke in men with migraine (aHR: 1.20, 95% CI: 1.01–1.42, p = 0.036). Subgroup analyses by age and migraine subtype revealed a trend toward the development of ischemic stroke in younger (age ≤ 45 years) male patients (aHR: 1.54, 95% CI: 0.96–2.48, p = 0.075). In the literature, younger male patients have been underrepresented or not analyzed separately (21). Thus, the fact that this association was not identified previously might have been due to low statistical power. Nevertheless, the trend toward ischemic stroke in younger men merits further exploration.
An age-modulated association has been reported in Dutch (cut-point of 45 years) (10) and American studies (highest risk among those aged 45–49 years) (7, 11). In addition, a recent study of elderly migraine patients failed to find any association between migraine and ischemic stroke in nonsmokers (12). This age-related differential risk might be explained by the fact that, despite a two-fold increased risk ratio among young MA patients, the absolute risk of ischemic stroke is still low in young people. As patients age, the contribution of MA to the incidence of ischemic stroke becomes relatively insignificant compared to other major risk factors of stroke that increase in prevalence with age, such as hypertension, diabetes mellitus, and atrial fibrillation. This speculation is supported by a previous observation that the increased relative risk of ischemic stroke in female patients with MA exists exclusively in those with the lowest Framingham risk score (7).
Although the underlying mechanism for the association between MA and ischemic stroke remains speculative, we can suggest some possibilities. First, MA patients have an unfavorable lipid profile compared to MO patients, which might contribute to the increased risk of ischemic stroke (10). Second, the increased risk is strongest in women with MA, and women with MA who use oral contraceptives are at an even higher risk for developing ischemic stroke (10). Thus, an interaction between MA and hormone changes might provide an additional risk for stroke. Third, patients with MA are prone to thrombosis. Serum levels of prothrombin factor 1.2, a marker of thrombin formation, are elevated specifically in MA but not MO patients (22). Finally, a genetic predisposition might contribute to the difference. Factor V Leiden G1691A and prothrombin G20210A are risk factors for stroke occurring in younger people, and these variants are overrepresented in MA patients compared to the normal population, or to MO patients (23). These hypothesized links require further validation.
This study has several strengths. Prior large cohort studies have enrolled relatively older individuals compared to the present study. The mean age of women in the MA subgroup of the Women's Health Study was 53.2 years (7), while the Northern Manhattan Study enrolled a population with an average age of 68 years (12). However, migraines are most prevalent among people aged 20–50 years (1,2,24). Our study population (mean age of 41 years) and the Genetic Epidemiology of Migraine (GEM) study (mean age of 41.9 years for migraine patients) (10) better represent the most prevalent age group of migraine patients. Still, all of these studies have confirmed that the association between MA and ischemic stroke is strongest in younger female rather than older female participants. Second, the diagnosis of migraine was defined by codes given by neurologists. In Taiwan, the accuracy of migraine diagnosis by neurologists is 91.6%, according to a validation study using the International Classification of Headache Disorders, second edition (ICHD-2) criteria (25). Third, an ischemic stroke was defined by hospitalization with ICD codes 433–434. The accuracy of hospitalization for ischemic stroke has been validated to be 94% (16).
This study also has several limitations. First, in claim-based studies such as this one, the date of entry does not coincide with the onset of migraine. Thus, whether the duration of migraine affects the outcome cannot be ascertained. Information regarding several risk factors for ischemic stroke, including smoking, waist-to-hip ratio, body mass index, dietary risk, physical activity and alcohol intake (15), were not accessible from the database. Migraine patients, especially those with MA, are more likely to smoke (26). These unaccounted-for risk factors, especially smoking, might modulate the association between migraine and stroke, and they require further investigation.
Second, we defined ischemic stroke in those with hospitalization claims due to the advantage of a validated diagnosis (16). Minor stroke might be underrepresented among these cases. In MA patients, stroke is typically milder and associated with a better outcome (27). Thus, the use of hospitalization claims might result in an underestimation, rather than an overestimation, of the association of MA patients with ischemic stroke. Third, all of the patients enrolled in this study had sought medical assistance for migraine. Patients with previous or nonactive migraine may be underrepresented. In addition, the disease coding does not reflect the heterogeneity of migraine. The frequency of migraine, or the status of chronic migraine, may modulate the association, but could not be assessed using this database. Fourth, there may be migraine patients who did not seek medical assistance in the comparison cohort, leading to an underestimation of the strength of the association between migraine and stroke. However, we excluded people diagnosed with any headache disorder from the comparison cohort, which would reduce this possibility. Finally, migraine was defined exclusively as neurologist-diagnosed migraine. Because neurologists also diagnosed most cases of ischemic stroke, our results are prone to Berkson's bias (28).
In summary, the results of this large cohort study confirm that migraine is associated with an increased risk of ischemic stroke. This association is strongest in younger (age ≤ 45 years) female patients with MA, but a trend also exists in younger (age ≤ 45 years) male patients. Because of differences in ethnic composition, caution should be exercised in generalizing our results to non-Asian populations. Thus, the age-, sex- and subgroup-specific associations we observed need to be further verified in future studies.
Clinical implications
Migraine is associated with a higher risk of developing ischemic stroke. The increased risk of ischemic stroke is greatest among younger (≤45 years) female patients having migraine with aura. Comorbid risk factors for ischemic stroke, such as hypertension and diabetes mellitus, cannot account for the increased risk of stroke. A trend toward an increased risk of ischemic stroke in younger men merits further investigation.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. All authors have completed the Unified Competing Interest form at ![]() (available on request from the corresponding author) and make the following declarations: K.-P. Peng, Y.-T. Chen, and C.-H. Tang report no disclosures; S.-J. Wang has served on the advisory boards of Allergan and Eli Lilly Taiwan. He has received speaking honoraria from local companies (Taiwan branches) of Pfizer, Eli Lilly and GSK. He has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital and Taiwan Headache Society. J.-L. Fuh is a member of a scientific advisory board of Eli Lilly, and has as well received research support from the Taiwan National Science Council, Taipei-Veterans General Hospital and Eli Lilly.
(available on request from the corresponding author) and make the following declarations: K.-P. Peng, Y.-T. Chen, and C.-H. Tang report no disclosures; S.-J. Wang has served on the advisory boards of Allergan and Eli Lilly Taiwan. He has received speaking honoraria from local companies (Taiwan branches) of Pfizer, Eli Lilly and GSK. He has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital and Taiwan Headache Society. J.-L. Fuh is a member of a scientific advisory board of Eli Lilly, and has as well received research support from the Taiwan National Science Council, Taipei-Veterans General Hospital and Eli Lilly.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from National Science Council of Taiwan (NSC 102‐2321‐B‐010‐030, 100‐2314‐B‐010‐018‐MY3, and 99‐2314‐B‐075‐036‐MY3), Taipei‐Veterans General Hospital (VGHUST102‐G7‐6‐1, V102C‐118, V102E9‐001), NSC support for the Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC 101‐2911‐I‐008‐001), Brain Research Center, National Yang‐Ming University, Ministry of Health and Welfare (MOHW104‐TDU‐B‐211‐113‐003), and a grant from Ministry of Education, Aim for the Top University Plan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
