Abstract
Background
Visual after-effects are illusions that occur after prolonged viewing of visual displays. The motion after-effect (MAE), for example, is an illusory impression of motion after viewing moving displays: subsequently, stationary displays appear to drift in the opposite direction. After-effects have been used extensively in basic vision research and in clinical settings, and are enhanced in migraine.
Objective
The objective of this article is to assess associations between (1) MAE duration and visual symptoms experienced during/between migraine/headache attacks, and (2) visual stimuli reported as migraine/headache triggers.
Methods
The MAE was elicited after viewing motion for 45 seconds. MAE duration was tested for three test contrast displays (high, medium, low). Participants also completed a headache questionnaire that included migraine/headache triggers.
Results
For each test contrast, the MAE was prolonged in migraine. MAE duration was associated with photophobia; visual triggers (flicker, striped patterns); and migraine or headache frequency.
Conclusions
Group differences on various visual tasks have been attributed to abnormal cortical processing in migraine, such as hyperexcitability, heightened responsiveness and/or a lack of intra-cortical inhibition. The results are not consistent with hyperexcitability simply from a general lack of inhibition. Alternative multi-stage models are discussed and suggestions for further research are recommended, including visual tests in clinical assessments/clinical trials.
Keywords
Introduction
Migraine is a common neurological condition that is characterised by recurrent and severe headaches. Most people will have experienced headaches where they can identify the cause, such as headaches resulting from stress, tiredness, dehydration or, in women, hormonal factors. For many, these headaches are easily resolved with rest, sleep, or over-the-counter medication. The same factors, however, can trigger migraine as well as other types of headache, as can certain environmental stimuli, such as visual patterns, flickering lights, noises and smells (1–5). In the case of migraine, the headache and associated symptoms are more serious and, in many cases, migraine can have a debilitating effect on a person’s everyday life, impacting on work, education, social and family activities, particularly when people susceptible to environmental or visual triggers encounter them unexpectedly. In this report, the term migraine refers to headaches and associated symptoms that meet the diagnostic criteria for migraine (see Method section), and the term headache refers to other, benign, headaches experienced by the non-migraine/control comparison group.
Stripes and flicker are ubiquitous in the environment. People can be exposed to flicker from sunlight through trees or gratings, from badly maintained lighting, by working in environments that are lit with older fluorescent lamps operated at 50 or 60 Hz, by using liquid crystal display (LCD) screens with a flickering back-light or cathode ray tube (CRT) screens that have refresh rates less than 100 Hz. Flicker appears in images on television (TV) and in the cinema and, more recently, in the images displayed on projection screens at subway/train stations and in pubs and bars. Stripes appear on clothing, escalator treads, gratings, window blinds and some art designed for public spaces. Glare can result from windows, from halogen or light-emitting diode (LED) spotlights, from LED car head and tail lights, and from other overhead lighting that is visible in the peripheral field of view. Inadvertent, and thereby unexpected, exposure to these migraine and headache triggers can severely disrupt a person’s quality of life as, if it triggers migraine or headache, it affects their ability to work, study, or have family or social interactions.
Symptoms associated with migraine, such as photophobia and phonophobia, nausea and vomiting, and intense, pulsating pain exacerbated by even routine physical activity, explain the frequent anecdotal reports that the best solution is to rest in a quiet, dark room and try to sleep. In migraine with aura, other transient neurological symptoms (commonly visual, sometimes somatosensory, sometimes involving speech) can further impact on people’s ability to interact with the world and the people around them. Aura symptoms usually precede the headache by up to 60 minutes and the sensory ones typically move or spread during that time.
Aura symptoms vary in severity. The visual symptoms usually involve a partial loss of vision, often in one quadrant or hemifield, which is preceded by positive symptoms superimposed on whatever the person is looking at. The positive symptoms include the classic fortification spectra, which is a jagged or zigzag collection of lines that starts centrally and grows, shimmering, over time. Others report simpler stars and phosphenes, or they have the impression of looking at the world through running water. People also report double vision, or tunnel vision, a loss of vision, or more elaborate distortions such as in the relative sizes of parts of images (or of the person’s own body image). Somatosensory aura symptoms typically involve pins and needles, tingling or numbness on one side of the face or body, but can extend to hemiplegia. The reported language difficulties are reminiscent of either of the aphasias described by Broca (difficulty finding appropriate words), or Wernicke (fluent output but little content). Evidently, people who experience these symptoms will have a loss of productivity at home, work or in education and a reduction in their quality of life beyond that which results from a severe headache.
The pathophysiology underlying the variety of symptoms involved in migraine is still not entirely understood despite numerous psychophysical, electrophysiological and imaging studies. Visual function has been frequently tested in migraine, in between attacks, due to (i) the ability of visual stimuli to induce a migraine attack, (ii) the intense sensitivity to light (photophobia) that patients typically experience during an attack and (iii) the fact that aura symptoms are commonly visual (4–8). Some people with migraine also report sensitivity to light in between attacks, and use sunglasses to try to alleviate it. Although these issues have motivated research into visual processing in migraine, there has been relatively little research into how they may all be related. Most of the literature has focussed on visual triggers (see Shepherd (5) for a review). The present study sought to assess associations between visual triggers, visual symptoms and performance on a visual task using the perception of motion.
Many visual tasks have been employed previously to compare performance in migraine and control groups, involving, for example, the perception of attributes such as colour, orientation with real and illusory lines, flicker, visual discomfort, as well as performance differences using visual search and visual masking tasks (reviewed in Shepherd (8)). There have also been a number of studies that have examined several aspects of the perception of motion. For example, performance on visual motion-processing paradigms such as pattern adaptation, threshold discrimination and threshold detection using local, global and relative motion tasks has been reported to differ between migraine and control groups. Generally, when there are group differences, the migraine group’s performance is impaired (9–12). Although different authors often propose different models of anomaly, there is a general consensus that abnormal cortical processing is an underlying factor in the pathophysiology of migraine and underlies these group differences in motion perception.
The experimental paradigm used here, pattern adaptation, has recently revealed large differences in performance between migraine and control groups (13,14). Pattern adaptation may involve adaptation at multiple stages within the visual system, from the retina to early and later cortical areas, but it certainly involves the cortex and reflects specific interactions between groups of neurons (13–16). Current understanding of these interactions makes pattern adaptation an ideal tool to assess proposed models of cortical function in migraine. In pattern adaptation, participants simply look at a display for some time, or ‘adapt’ to the display, and then selective effects of the adaptation on the perception of subsequent displays are examined. Any selective effects, or illusions, are described as after-effects of the adaptation, and have been used to infer the existence of neurons or pathways selective for particular attributes of the adapting display long before their existence was confirmed with single neuron recordings.
It is now known that, early in the visual system, the visual scene is effectively deconstructed, with specialised pathways and areas for the coding of different attributes such as motion, orientation, spatial frequency, depth, colour, and more. This separation begins as early as the retina and continues throughout the early and later visual cortical areas. One clear indication that some of the effects of adaptation are cortical is their ability to transfer inter-ocularly: that is, if the adapting display is viewed with one eye only, but the test display is presented to the other eye, the after-effect/illusion is still seen, although it may be at a weaker intensity (15,16). Binocular cells, which are activated by displays presented to either eye, are first found in abundance in the primary (striate/V1) visual cortex (17,18). After-effects have been used as a non-invasive way to assess the basic organisation of the visual system and, subsequently, to assess models of cortical function in clinical conditions such as epilepsy (19,20), schizophrenia (21,22), Parkinson’s disease (21) as well as migraine (13,14,23).
The motion after-effect (MAE) is the illusory impression of motion that is experienced after steadily gazing at a moving pattern. If an observer looks at a waterfall for a few minutes and then transfers their gaze to the foliage beside the fall, the foliage appears to drift upwards, the opposite direction to the original motion (15,24). The effect can be seen after prolonged exposure to coherent motion in any particular direction, or other moving displays such as those that expand, shrink, or rotate. Once the motion stops, any subsequently presented display seems to drift in the opposite direction.
The MAE reflects adaptation in direction selective neurons tuned to the direction of motion presented during the adapting display (8,25–28). Cortical neurons produce a steady low level of spontaneous activity when not engaged by any stimulus. If a visual display contains elements with a certain motion direction and speed that activates particular neurons then, initially, they will respond vigorously. Over time, however, their response declines and, when the pattern is removed, the neurons appear unresponsive for a period of time as they take time to ‘recover’. During that time, the spontaneous activity of all other neurons sensitive to different motion directions exceeds that of the suppressed neurons. This produces a biased distribution of spontaneous activity that, overall, is similar to activity produced by slow motion in the opposite direction and results in the perceived after-effect (29,30). How long the adapted neurons remain suppressed, and hence how long the illusion persists, depends on several factors, one of which is the contrast of the adapting and test displays. Keck et al. (31) reported that the magnitude of the MAE increased with increasing adaptation contrast or with decreasing test contrast, i.e. it was maximal for high adapting contrasts paired with low-test contrasts. It was concluded that the imbalance in activity between adapted and unadapted cells was greater for low-test contrasts than for high, which resulted in the prolonged MAE. Shepherd (13,14) found that the MAE was more pronounced in migraine, that is, it lasted longer than in the control group when using medium- or high-contrast adapting and test displays (Michelson contrasts of 30% (reference Shepherd (14)) or 78% (reference Shepherd (13))).
Various authors have reported increased pattern sensitivity in migraine, also referred to as pattern glare or visual discomfort (3,14,32–37). These terms refer to the perceptual distortions (colours, shadowy shapes, shimmer and/or motion) and discomfort that can be experienced when viewing high-contrast, mid-spatial frequency, striped patterns. Some of the perceptual distortions have been attributed to fixation instability and accommodative changes, whereas others have been attributed to an abnormal spread of cortical activation to neighbouring neurons due to the massive excitation generated by the striped pattern (see Shepherd (8) for further details). Shepherd (13,14) reported positive correlations between MAE duration and pattern sensitivity: the MAEs experienced by those who saw illusions in high-contrast striped patterns were longer than those who did not. These high-contrast patterns are reported to be capable of inducing migraine if viewed for prolonged periods of time. Shepherd (13,14) also reported positive correlations between the MAE and visual migraine triggers: the MAE experienced by those who reported visual stimuli could trigger their attacks were longer than those who did not.
Here, the relationships between the MAE and display contrast, various visual migraine triggers and visual symptoms were explored. Trials consisted of three test contrasts (high, medium and low). The adaptation contrast was kept constant (medium). Based on previous research (31), larger effects were predicted for low- compared to high-contrast test displays in both groups. Several group differences were expected: (i) the migraine groups (with and without aura) were predicted to have longer MAEs than the control group across all test contrast conditions; (ii) the MAEs were predicted to be longer for those in the migraine groups who reported visual triggers and visual symptoms; (iii) there would be no significant differences between the migraine sub-groups with and without aura (13,14). The earlier work used a composite measure of susceptibility to visual triggers, whereas here individual visual triggers and symptoms were examined separately.
Method
Participants
Twenty-two migraine and 11 control participants were recruited from advertisements and an existing migraine database at Birkbeck College, London. Participants received a small honorarium for their time and expenses. There were 11 migraine participants with visual aura (VA, nine female, two male, mean age ± one standard deviation (SD): 29 ± 5 years, range 22–36 years, mode 24 years, median 30 years); 11 migraine participants without aura (MO, 10 female, 1 male, age: 27 ± 6 years, range 21–41 years, mode 21 years, median 28 years) and 11 control participants (nine female, two male; age: 30 ± 6 years, range 23–42 years, mode 21 years, median 28 years). Sample size is consistent with previous research.
All participants completed either a migraine or a headache questionnaire, detailing the characteristics of their migraine/headache symptoms, their frequency and duration, and possible migraine/headache triggers. The trigger list included visual stimuli: flickering light; striped patterns; alternating light and shade (such as dappled sunshine, transitions from sunshine to shade or vice versa); and other visual stimuli that they volunteered. Examples of these other visual stimuli were lattices, glare, high contrasts (e.g. sun reflected off chrome or water), computer use, TV and cinema, particularly three-dimensional (3D) cinema. Participants were asked if each item commonly, occasionally or never triggered migraine or headache. The data were coded as yes (commonly or occasionally), or no, for each item.
All in the migraine group fulfilled the International Headache Society (IHS) criteria for migraine (38). None in the control group experienced regular or severe headaches that fulfilled IHS criteria. Of the control participants who reported having headaches, they were tension type, sinus related, or due to dehydration. All testing was performed when participants appeared symptom free and no participant had experienced a migraine/headache 48 hours on either side of the test session. None of the participants were on prophylactic medication for any condition, nor had they taken any acute medication within 48 hours of the test session. None reported having any other neurological condition, nor any condition that can affect eyesight. All participants had a monocular and binocular visual acuity of at least 20/25, with or without optometric correction.
The study received ethical approval from Birkbeck’s Department of Psychological Sciences Ethical Committee. Informed written consent was obtained from all participants in accordance with the Declaration of Helsinki (1991).
Apparatus/Materials
MAE
The displays were created using experimental scripts developed in C in conjunction with C routines from the Video Toolbox (39). The stimuli were presented on a 21-inch flat-screen CRT monitor (LaCie) connected to an Apple Macintosh G4 computer. The CRT monitor had a spatial and temporal resolution of 1280 × 960 pixels, and 100 Hz, respectively. Trials consisted of an adapting and test display that together elicited the MAE.
Adapting display
A 14-degree square window displayed random light and mid-grey pixels (average luminance = 30 cdm−2, Michelson contrast = 30%) moving coherently upwards at a speed of 3 degrees/second. The adapting display was presented for 45 seconds. Participants were seated 60 cm from the monitor in an otherwise dark room. The experiment consisted of 12 trials, divided into three blocks, one for each test display contrast (the adaptation contrast was always the same). Block order was randomised. Thus, the experiment had a mixed (3 × 3) quasi-experimental design, with contrast as the within-subjects factor and group as the between. The experiment was preceded by six practice trials (two for each test contrast level). During presentation of the adapting displays, participants were asked to look at a fixation point at the centre of the screen whilst paying attention to the whole display. Apart from a change in CRT size, the adapting displays (contrast, duration, size, speed) were identical to one of the conditions used previously by Shepherd (14). Test display contrast, however, differed to that earlier study: the medium test display contrast was the same in both studies, but, in this study, a higher and lower contrast test display was added (see below).
Test displays
Immediately after adaptation, participants were presented with a test display. Test displays contained random, stationary, light to dark-grey pixels, which resembled that of a snapshot taken of a detuned TV. Three different contrast test displays were used – high (Michelson contrast 78%), medium (30%) and low (0.1%). All test displays had the same mean luminance as the adapting display (30 cdm−2). The presentation of a test display immediately after the adapting display elicited the illusion of slow, downward motion. When the stationary test display appeared, participants were asked to try not to blink and to indicate when the illusory motion stopped by pressing a key on the computer keyboard. The experimental session lasted between 75 and 90 minutes. Participants initiated each trial with a key press and so could sit quietly between trials, in the darkened room, if they wished to pause or take a break. In between trials, the CRT displayed a uniform grey screen (luminance 30 cdm−2) together with a small, centrally located, information window asking participants to push the enter key to continue with the next trial. The information window also informed participants whether the next trial would be the same (which occurred four times in succession within each block of trials); or whether the contrast of the test displays was to change at the start of a new block of four trials. As with all the previous studies (11,13,14,23), an experimenter was present throughout the experimental session to ensure the participants understood the task and were looking at the adapting and test displays.
Results
The data were assessed using PASW statistics version 20 (SPSS Inc, Chicago, IL, USA). The data from each group were normally distributed (Kolmogorov-Smirnov tests, p > 0.05), so group differences were assessed with analysis of variance (ANOVA), t-tests, Pearson’s correlation coefficient (r) and the point-biserial correlation coefficient (rpb). When sphericity was violated in the ANOVA (which occurred for effects of contrast, see below), the degrees of freedom were adjusted using the Greenhouse-Geisser epsilon.
Average MAE durations for each group in each condition are shown in Figure 1. Several trends are clear. Overall, as expected, the MAE lasted longer in both migraine groups than in the control group for all three contrast test displays, and the MAE was greatest in those with VA. Secondly, high-contrast test displays produced the shortest MAEs and low contrast, the longest, for all three groups. This was also as expected. A three (group) × three (test display contrast) mixed ANOVA produced a significant main effect of group [F(2,30) = 6.5, p = 0.005] confirming the overall MAE duration (collapsed across test display contrast) was greater in the migraine groups. Three planned comparisons revealed that the overall MAE durations for the VA and MO groups differed significantly from the control group (VA vs C: p = 0.003; MO vs C: p = 0.028; one-tailed tests, Bonferroni corrected), whereas they did not differ significantly from each other (p = 0.93, two-tailed test, Bonferroni corrected). The main effect of contrast was significant, confirming that low contrast test displays elicited longer MAEs than high [F(1.7, 50.1) = 10.4, p < 0.001). Pairwise comparisons revealed that, collapsed across groups, the MAE for the low- and high-contrast test displays differed significantly (p = 0.002), as did the MAE for the medium- and high-contrast test displays (p = 0.02) whereas the MAE for the low- and medium-contrast test displays did not differ significantly (p = 0.21, all comparisons Bonferroni corrected). The group × contrast interaction was not significant [F(3.3, 50.1) < 1, p = 0.45].
MAE data: Means (±SE) of the MAE duration for the migraine and control groups for high-, medium- and low-contrast test displays. MAE: motion after-effect; VA: migraine with visual aura; MO: migraine without aura; C: control group; SE: standard error.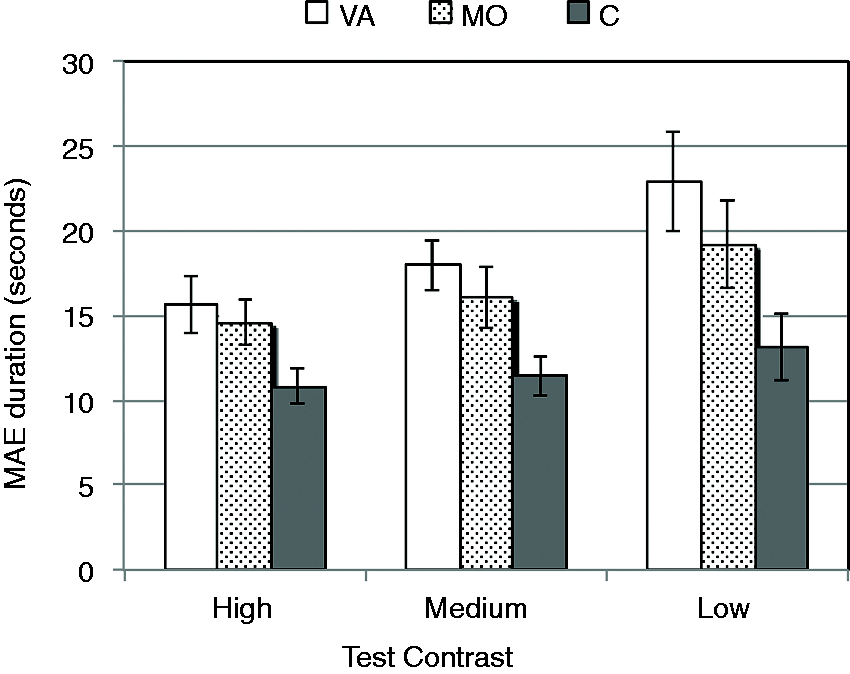
Columns labelled VA, MO, C: Migraine and headache characteristics related to light, visual triggers and migraine frequency.

Standard errors are in parentheses. Columns labelled r (Pearson’s correlation coefficient) and rpb (point-biserial correlation coefficient, for yes/no questionnaire answers) show the associations between each of the questionnaire items and the duration of the MAE with each contrast of the test displays (high, medium, low). Pearson’s and point biserial correlation coefficients. aSignificance at p < 0.05; **p < 0.005; bold upright: one-tailed tests, based on previous research; bold italic: two-tailed tests. N = number of participants; VA: visual aura; MO: migraine without aura; C: control; MAE: motion after-effect.
Shepherd (13,14) reported that the length of the MAE in migraine was greatest in those who reported having visual migraine triggers. This association was confirmed here for striped patterns and MAE duration with the medium- and low-contrast test displays (medium: rpb(22) = 0.52, p = 0.01; low: rpb(22) = 0.42, p = 0.03, one-tailed tests), and for flicker and MAE duration with the low-contrast test displays (rpb(22) = 0.36, p = 0.04, one-tailed, see Table 1). These associations persisted when the migraine and control groups were combined (Table 1), which will be returned to in the Discussion (striped patterns as a trigger correlated with MAE duration with the medium- and low-contrast test displays; flicker as a trigger correlated with MAE duration with low-contrast test displays: rpb(33) = 0.56, p = 0.001; rpb(33) = 0.52, p = 0.001; rpb(33) = 0.34, p = 0.03; respectively).
Photophobia during either a migraine or headache correlated significantly with the MAE duration for each test display contrast (high: rpb(33) = 0.32, p = 0.03; medium: rpb(33) = 0.43, p = 0.01; low: rpb(33) = 0.45, p = 0.01; one-tailed tests). The MAE duration for the medium- and low-contrast test displays also correlated significantly with the frequency of migraine and headache (three-month frequency, medium contrast: r(33) = 0.56, p = 0.001; low contrast: r(33) = 0.46, p = 0.01; 12-month frequency, medium contrast: r(33) = 0.46, p = 0.01; low contrast r(33) = 0.51, p = 0.004; two-tailed tests; cf. Shepherd (13,14)). See Table 1 for full details of the correlations between MAE duration for each test display contrast and each questionnaire item. For the migraine group alone, there was a significant association between the MAE duration for the medium-contrast test displays and the number of days since the last migraine attack (r(22) = –0.41, p = 0.03, one-tailed) and migraine frequency assessed over the last three months (r(22) = 0.46, p = 0.02, one-tailed). There was a trend for an association between the MAE duration for the medium-contrast test displays and migraine frequency assessed over the last 12 months, and there was a significant association between the MAE duration for the low-contrast test displays and migraine frequency (r(22) = 0.34, p = 0.06, r(22) = 0.44, p = 0.03, one-tailed tests).
Discussion
As expected, the MAE in the migraine groups lasted longer than in the control group for all test display contrasts, and the low-contrast test displays produced the longest MAEs for both groups. Furthermore, as predicted, the MAEs were longer for those with visual triggers (striped patterns and flicker) and visual symptoms (VA and photophobia). Nevertheless, as has been found before, the differences between the migraine sub-groups with and without aura were not statistically significant. Shepherd (13,14) suggested that (i) there may be a continuum of cortical anomaly in migraine regardless of aura symptoms, rather than qualitative differences between migraine subgroups, and that (ii) studies that do report differences between the migraine sub-groups, with and without aura, may have recruited participants for whom migraine classification co-varies with other factors, such as pattern sensitivity or susceptibility to visually triggered migraine. The associations between MAE duration and visual triggers reported here are consistent with these suggestions. As mentioned, the earlier work used a composite measure of susceptibility to visual triggers, whereas here individual visual triggers and visual symptoms were examined separately (Table 1). Photophobia and sensitivity to stripes and flicker, as migraine triggers, were all associated with the duration of the MAE: those who indicated these questionnaire items were migraine triggers had longer after-effects than those who did not.
Several members of the control group reported similar headache triggers (flicker, striped patterns, patterns of light and shade – transitions from light to dark, or vice versa or dappled sunshine – and, when asked to volunteer other headache triggers, cited visual items that were similar to those reported by the migraine group, such as, TV and cinema, computers, or bright sunshine when the sun is low in winter). This is consistent with a previous study of 180 participants (132 with migraine) which found that 60% of the migraine group, and 15% of the control group, cited similar visual stimuli as reliable migraine/headache triggers (5). Although visual triggers were more commonly endorsed by the migraine group, clearly similar features of the visual environment can induce headache as well, which would merit further study as it may lead to guidelines to influence the design of the environments in which people live and work to minimise the inadvertent triggering of both migraine and headache (see also Debney (1), Harle et al. (4) and Elias et al. (58)). This is returned to at the end of the Discussion.
The current finding of prolonged MAEs for low-, compared to high-, contrast test patterns is in line with those of Keck et al. (31), who also found longer MAEs for lower-contrast test patterns. As mentioned in the Introduction, the MAE is caused by a biased distribution of activity in direction-selective cells throughout the visual pathways from the retina to cortex, but certainly involving the cortex. The perception of something as stationary occurs only if cells responding to all directions exhibit the same level of activity. Once a set of direction-selective cells is suppressed, by adaptation, the overall distribution of activity in all cells tuned to all motion directions is biased and will give rise to an MAE. Thus, the after-effect results from a reduced response from adapted neurons, which is detectable against the activity generated in the whole population of direction-selective cells by the subsequent test displays (27–30,40). As soon as the adapting motion stops, however, the adapted cells start to recover and the length of that recovery, together with any residual response to the test patterns, determines the duration of the MAE. Since direction-selective cortical cells are also responsive to contrast, there would be a larger residual response in the adapted cells to the high-contrast test patterns than to the low, resulting in smaller MAEs for the high-contrast test patterns.
While speculative, this account can explain longer MAEs in migraine: if the residual response of the adapted neurons to each test display in the migraine group was lower than that in the control group, the result should be a more pronounced MAE in migraine, as reported here. Consistent with this suggestion, there are reports that contrast sensitivity is impaired in migraine (33,34) and is related to relative motion thresholds (11). If contrast sensitivity is impaired, people with migraine viewing patterns of any contrast may see those patterns as having a lower contrast. Neither contrast sensitivity nor relative motion thresholds, however, involve adaptation. Recent research has, consequently, examined contrast sensitivity and MAE duration in the same participants to determine any formal associations between these two aspects of perception (23).
Shepherd (8,14) discussed various models of cortical function in migraine and concluded that prolonged MAEs are not consistent with a general cortical hyperexcitability. An early model suggested that hyperexcitability, in migraine, could result from a lack of cortical inhibition (35,41,42). Various neurophysiological and pharmacological studies have shown that inhibition is not, however, involved in adaptation in the visual cortex (striate), at least in cats (43,44). A simple lack of cortical inhibition in migraine is unlikely to explain the group differences reported here. Shepherd (13) instead suggested prolonged MAEs in migraine were consistent with a lack, or extended suppression, of cortical excitatory connections, or increased cortical inhibition in migraine.
Another model of cortical hyperexcitability in migraine is that it leads to greater background noise in the visual system and a greater response to incoming signals (10). If both signal and noise were elevated in proportion in migraine, then there would be little reason to expect differences between the migraine and control groups. Any greater suppression from larger signals elicited by the adapting displays would be lost in the greater background noise elicited by the test displays.
A third model has proposed hyperexcitability in migraine that results in increased general noise in the visual system, without entailing a greater response to incoming signals (9). Greater noise, against which the MAE signal must be detected, should produce weaker MAEs, as the MAE signal would be more readily masked by the elevated background noise. Again, this model is not supported by the present data. Furthermore, tasks designed to measure internal noise in the visual system have found no differences between migraine and control groups (12).
Fourth, hyperexcitability could raise the activity of direction-selective cells to a uniformly higher rate without increasing variability. In this case, the neuronal response elicited by a person with migraine may be comparable with another without migraine viewing a higher-contrast pattern, analogous to the increase in firing rate with contrast that is observed in direction-selective cells in physiological studies (45). This proposal appears consistent with pronounced after-effects in migraine: stronger activity during adaptation could subsequently produce greater suppression in adapted cells. The test displays would elicit a uniformly higher level of activity in unadapted cells, so the dip in the activity of adapted cells should be readily detected. Because the MAE is maximal for low-contrast test displays combined with equal or higher-contrast adapting displays (30,31,46), however, this is also an unlikely explanation. Moreover, as mentioned above, it is not consistent with reports of impaired contrast sensitivity in migraine. Impaired contrast sensitivity should result in people with migraine perceiving the adapting and test display contrasts as lower, not higher.
An alternative explanation for prolonged after-effects is that they are related to the reported lack of habituation and even increased amplitudes of visual evoked potentials (potentiation) in migraine, which may result from low cortical preactivation or hypoexcitability (reviewed in Tibber and Shepherd (47)). Habituation and adaptation share certain similarities: habituation is a decline in response to repetitive stimuli, whereas adaptation is a decline in response to continuous stimuli. Both reduce redundancy, protect against response saturation and conserve energy. Despite these similarities, a lack of habituation is clearly not mirrored by a simple lack of adaptation. During the adaptation, however, a potentiation of response over time would mean that, by the end of the adaptation, neurons tuned to the adapting motion would have responded more strongly, resulting in a greater suppression. To assess this explanation, it would be useful to compare migraine and control groups’ performances for a range of shorter adaptation times (see, for example, van Wezel and Britten (27,28)). Ongoing research with adaptation times of 15, 30, 45 and 60 seconds has shown that MAEs occurred for each adaptation time; however, clear group differences appeared only with 45 and 60 seconds of adaptation. This is not consistent with a potentiation of response over time in migraine, but further research with a reasonable sample size is needed.
It is possible that one single model for differences in neuronal function in migraine is likely to be an over-simplification, and that different paradigms and visual tests, which tap different stages in the visual pathways, may reveal multiple types of altered neuronal processing (8,47). Tibber and Shepherd (47), using a colour adaptation task, reported differences that were consistent with either increased GABAergic inhibition, or increased GABAergic inhibition and glutaminergic excitation, at different sites of a particular retinal circuit. They suggested that conflicting models of migraine involving hyper- vs hypo-excitability, or increased vs decreased inhibition, might reflect differences in the circuitry that is sequestered by each particular experimental paradigm. That is, different tasks engage different processing streams, and different stages within each stream. Each may respond or adapt differently, and these responses or adaptive responses may differ between migraine and control groups in distinct ways at different stages within the visual pathways.
Recent studies on adaptation have described adaptation throughout the visual pathways, from the retina, through the lateral geniculate nucleus (LGN), to the visual cortex, that are qualitatively different at different stages, at least in the anaesthetised mouse and primate (59,62). Those authors point out that such effects are ubiquitous across species studied (from invertebrates to vertebrates), and across sensory modalities. It is likely, therefore, that they share certain similarities with human adaptation (see Larrson and Harrison (60) and Stocker and Simoncelli (61) for examples of inherited, and discrete, effects of adaptation demonstrable in humans). Adaptation at one level (e.g. the retina, the LGN) will propagate through subsequent processing stages yielding adaptation effects at higher stages that are inherited from earlier stages, yet the higher stages can also become adapted themselves potentially in a different way.
As an aside, many studies of the MAE use drifting sine-wave gratings as adapting and test stimuli and are, therefore, likely to show adaptation of different cell groups at different stages in the visual system as some may adapt to motion, some to the spatial frequency of the sine-wave grating, and some to their combination (e.g. Larrson and Harrison (60)), which complicates interpretation of effects of adaptation and site(s) of adaptation. Furthermore, adaptation to the spatial frequency component of gratings in V1 is likely to swamp adaptation effects to motion at either V1 or at later sites. This is one of the reasons why random dot displays were used in this study as adapting and test stimuli, rather than gratings. The second reason was to minimise the chance of triggering migraine by using striped patterns during adaptation. The study by Larrson and Harrison (60) is one of the few to compare motion adaptation with sine-wave gratings and with random dot displays and looked for the effects of adaptation at different stages of processing, but within the cortex only. They report separable effects that can be seen at V1 and V5, most clearly shown with the random dot rather than the grating stimuli.
Returning to models of adaptation in migraine, Thabet et al. (63) have recently presented a model of flicker adaptation at three stages within the visual system, in which differences between migraine and control groups could occur at each stage, which was developed from a model of adaptation to orientation (62). That model involves both precortical and cortical sites, and changes in the adaptability of both inhibitory and excitatory neurons at different levels. Adaptation of excitatory neurons at early/monocular sites results in a modest adaptation in those binocular cells to which they are connected. Adaptation of inhibitory cells at early/monocular sites produces increased excitability in those binocular cells or sites that they had been inhibiting. Inhibitory circuits are proposed to be broadly tuned, so adaptation leads to increased excitability in a broad range of binocular cells from disinhibition. Their data on flicker adaptation were well fit by this model, and they suggested their data indicated there is stronger adaptation in migraine, particularly at the two monocular sites. It is difficult to get direct psychophysical evidence for such a model, but further research could combine psychophysics with imaging, as suggested by Karanovic et al. (37). This model is consistent with the earlier suggestions that one single model for differences in neuronal function in migraine is likely to be an over-simplification, and that different paradigms and visual tests, which drive different neural circuits and stages in the visual pathways, may indicate multiple types of altered neuronal processing in migraine rather than one general one (8,47).
Nevertheless, the consistent pattern of group differences, particularly for tasks involving motion, suggests separate/additional clinical uses for visual tasks and questionnaires on visual symptoms regardless of the underlying model used to explain the group differences. Briefly, in clinical settings, it would be useful to include questions about visual triggers, as well as visual symptoms, when taking a patient’s headache history (see Mulleners et al. (56)). This would be beneficial when advising patients about identifying and avoiding environmental triggers. It would also be useful to include assessments of visual triggers and visual function in clinical trials. There are a growing number of reports that perceptual and electrophysiological measures can track both migraine periodicity and treatment outcome (33,48–55,57). Much of this research uses electrophysiological measures, which have few practical applications. Visual tests are simpler to administer and could be included in future clinical trials to replicate and extend these electrophysiological findings. There is recent interest in tracking changes in behavioural or electrophysiological measures throughout the migraine cycle (e.g. 52–54,57). In this study, there was a significant association between migraine frequency and MAE duration: the more frequent the migraine, the longer the MAE. Similarly, Shepherd et al. (11) found significant associations between a relative motion task and migraine frequency, the number of years experienced, and two measures of visual discomfort. If a simple visual test could be instrumentalised, it could result in an application patients could use at home to track their migraine cycle and, perhaps, take evasive action to thwart an impending migraine attack.
A motion task is likely to be the most promising type of visual task to pursue in an applied or clinical setting. While pattern sensitivity/visual discomfort, and flicker tasks also show fairly consistent group differences (8,11,32–34,36,37,41,47,63), they are unlikely to be useful for repeated testing to track the migraine cycle as they do not always correlate significantly with migraine frequency or the time elapsed since the last attack (11,36,37,63) and both can trigger migraine. Motion tasks using random dot displays, rather than drifting gratings, are likely to be more useful as they have also shown replicable group differences, and can show associations with (i) migraine frequency, (ii) the time elapsed since the last attack, (iii) reports of visual triggers and (iv) pattern sensitivity/visual discomfort (11,13,14) and they are not aversive to look at (9–14,23). Ongoing research is addressing this possibility, using repeated tests on a visual motion task together with questionnaires on visual triggers and visual symptoms. The present study has highlighted some similarities in headache triggers between migraine and control groups (see also Shepherd (5)), so a large cohort of people with and without migraine is being recruited, initially with an online experiment.
In conclusion, the present data have revealed prolonged neuronal suppression following 45 seconds of visual adaptation in migraine with and without VA, which results in a longer perception of the MAE. The association between photophobia during an attack and MAE duration simply reflects the group differences between people with and without migraine, since more in the migraine group reported photophobia with their headaches (Table 1, see also Sandor et al. (57)). The associations between the duration of the MAE and visual triggers, however, are consistent with earlier reports (13,14) and extend that work by identifying flicker and striped patterns as more relevant triggers than other visual stimuli. Further studies are recommended to examine the usefulness of visual tasks to predict response to migraine treatments, and as a non-invasive tool to track changes that occur throughout the migraine cycle. Motion tasks offer a promising place to start, given the consistency of reports showing differences between migraine and control groups (9–14,23). Further research is also recommended to better define environmental visual triggers and their relationships with visual symptoms and migraine and headache. For example, it is unclear why striped patterns, cited as a visual trigger, correlated to a greater extent with MAE duration than flicker, since flicker and motion perception appear more intimately linked than the perception of stripes and motion. Further refinements to the assessment of the nature of flickering and striped visual triggers, and their relationship with performance measures, is recommended. A more structured questionnaire on visual triggers would be useful. A better understanding of the types of visual or environmental triggers of migraine and headache may lead to guidelines for the design of the environment in which people live and work, to minimise exposure to headache triggers, similar to those guidelines that exist for photosensitive epilepsy (64). Such guidelines appear long overdue considering the much higher prevalence rates of migraine.
Article highlights
This study extends earlier work on motion perception in migraine by assessing one aspect of motion perception, the motion after-effect (MAE), together with an assessment of visual symptoms (visual aura, photophobia and light sensitivity) and visual triggers (flicker, stripes, patterns of light and shade, or other visual stimuli such as computer screens, cinema and high-contrast reflections). Patients with migraine who also have visual symptoms and visual migraine triggers show, inter-ictally, the largest differences in MAE duration, compared to the control group. This study replicates earlier reports of enhanced visual after-effects in migraine, showing that this simple visual test is capable of revealing large group differences, and thus may be a test that is useful to include in clinical trials or to track changes during the migraine cycle. This study confirms the usefulness of recording additional measures when performing visual tests in migraine, if the aim of the research is to provide evidence for or against models of anomalous visual processing in migraine.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
