Abstract
Background
It has been hypothesized that a constitutionally narrow cavernous sinus might predispose individuals to cluster headache. Cavernous sinus dimensions, however, have never been assessed.
Methods
In this case–control study, we measured the dimensions of the cavernous sinus, skull base, internal carotid and pituitary gland with high-resolution T2-weighted magnetic resonance imaging in 25 episodic, 24 chronic and 13 probable cluster headache patients, 8 chronic paroxysmal hemicrania patients and 22 headache-free controls. Dimensions were compared between groups, correcting for age, sex and transcranial diameter.
Results
On qualitative inspection, no relevant pathology or anatomic variants that were previously associated with cluster headache or chronic paroxysmal hemicranias were observed in the cavernous sinus or paracavernous structures. The left-to-right transcranial diameter at the temporal fossa level (mean ± SD) was larger in the headache groups (episodic cluster headache: 147.5 ± 7.3 mm, p = 0.044; chronic cluster headache: 150.2 ± 7.3 mm, p < 0.001; probable cluster headache: 146.0 ± 5.3 mm, p = 0.012; and chronic paroxysmal hemicrania: 145.2 ± 9.4 mm, p = 0.044) compared with controls (140.2 ± 8.0 mm). After adjusting for transcranial diameter and correcting for multiple comparisons, there were no differences in the dimensions of the cavernous sinus and surrounding structures between headache patients and controls.
Conclusion
Patients with cluster headache or chronic paroxysmal hemicrania had wider skulls than headache-free controls, but the proportional dimensions of the cavernous sinus were similar.
Introduction
Trigeminal autonomic cephalalgias (TACs) such as cluster headache and paroxysmal hemicrania are characterized by recurrent, severe, short-lasting attacks of unilateral headache accompanied by ipsilateral facial autonomic symptoms (1,2). Both forms of TACs may present either in an episodic form, characterized by periods of several weeks to months during which many attacks occur alternating with attack-free periods of several months to years, or a chronic form, in which attacks continue recurring without (long) attack-free periods. In probable cluster headache, patients fulfil all but one of the diagnostic criteria for cluster headache (2).
The cavernous sinus has been put forward as a possible key area in the pathophysiology of cluster headache (3). Trigeminal nociceptor excitation might initiate cavernous sterile inflammation and trigeminal-parasympathetic cavernous internal carotid artery vasodilatation (4), which could obliterate cavernous venous outflow. Such processes might compress the internal carotid sympathetic plexus, thereby explaining the unilateral headache, parasympathetic discharge and sympathetic dysfunction during cluster headache attacks (4,5).
It has been suggested that a narrow cavernous sinus, either constitutionally or acquired due to structural lesions, might predispose an individual to cluster headache (6–8). Here we present the first study assessing the structure and dimensions of the cavernous sinus with high-resolution magnetic resonance imaging (MRI) in cluster headache and chronic paroxysmal hemicrania.
Materials and methods
Subjects
In total, 98 subjects with episodic (n = 25), chronic (n = 25) or probable (n = 15) cluster headache, chronic paroxysmal hemicrania (n = 8) or without a history of headache (n = 25) took part in the study. Patients were diagnosed at the Department of Neurology by two experienced headache experts (GGS and JAvV) according to the International Classification of Headache Disorders: 2nd edition (ICHD-2 (9)). All patients also fulfilled the criteria of the International Classification of Headache Disorders, 3rd edition (beta version) (ICHD-3-beta (2)). Subjects with probable cluster headache fulfilled all but one of the ICHD-2 and ICHD-3-beta diagnostic criteria for cluster headache (attack duration >180 minutes, n = 12; absence of autonomic symptoms, n = 3) (2,9,10). The local medical ethics committee of Leiden University Medical Center approved the study and all subjects gave written informed consent.
MRI acquisition and analysis
Whole-brain 3D T1-weighted turbo field echo (repetition time (TR)/echo time (TE): 7.4/3.4 ms; 160 axial 1.0-mm continuous slices; 260-mm field of view (FOV); acquisition matrix: 256; flip angle: 8°), combined proton density and T2-weighted fast spin echo (TR/TE: 3000/(27/120) ms; 48 axial 3-mm continuous slices; 220-mm FOV; acquisition matrix: 256 × 220) and fluid-attenuated inversion recovery images (TR/TE: 8000/100 ms; inversion time: 2000 ms; 48 axial 3-mm continuous slices; 220-mm FOV; acquisition matrix: 256 × 191) were acquired at a 1.5-T Philips Medical System Scanner (NT-ACS, Philips, Best, The Netherlands) in order to exclude relevant gross pathology outside the cavernous sinus region. Furthermore, high-resolution T2-weighted spin echo images of the sellar region (TR/TE: 2000/120 ms; 80 axial 1.0-mm slices; 0.5-mm slice overlap; 200-mm FOV; acquisition matrix: 256 × 256; 0.78 × 0.78-mm pixel resolution; flip angle: 90°) were obtained. For obtaining measurements, these images were reoriented with image contrast at comparable levels using multiplanar reformation in Vitrea (Vital Images, Inc., Plymouth, MN, USA). Correct orientation was defined as having the optic chiasm as a horizontal line and the pituitary gland with the caudal part of the infundibulum in the same coronal slide, with all structures positioned as symmetrically as possible.
An experienced neuroradiologist (MCK), who was blinded for headache diagnosis and clinical data, systematically read all of the available images of the brain and cavernous sinus on a radiological viewing station in order to identify structural abnormalities of the anatomic structures in the brain, sella, cavernous sinus or surrounding skull base.
For quantitative analyses, reoriented images were interpreted by two observers (HSB and MAMS, both blinded to all clinical data). The quantitative analysis consisted of 13 measurements in coronal and sagittal view (see Figure 1). As a reference, the transcranial (left-to-right) diameter was measured in the coronal plane at the temporal fossa level (at the level of the carotid arteries) (1a). Another skull base measure entailed the diameter of the sellar compartment (1b). In the cavernous sinus, cranial–caudal (2a), medial–lateral (2b), supracarotid (2c), subcarotid (2d) and midline–inferolateral (2e) diameters were measured. Further measurements included left-to-right (3a) and cranial–caudal (3b) internal carotid artery (ICA) diameters cranial–caudal (4a), left-to-right (4b), and anterior–posterior (4c) diameters of the pituitary and the length of infundibulum (4d). The cavernous sinus and ICA diameters were measured bilaterally. Pituitary volume was estimated after left-to-right, anterior–posterior and cranial–caudal pituitary diameters were measured, applying Cavalieri’s principle in the following equation (11):

We mirrored all left and right measurements of patients with predominant left-sided headache as if they had right-sided headache. For main analyses, cavernous sinus dimensions ipsilateral to the headache in TAC patients were compared with the mean of the left and right cavernous sinus dimensions in controls. As approximately 14% of subjects with cluster headache experience side shifts or contralateral attacks (12,13), we performed sub-analyses for those subjects who had only experienced strictly unilateral headache during their disease history. As sex is a main predictor for transcranial diameter (with males having larger skull sizes compared with females) and sex was unequally distributed among the subject groups, we also performed sex-stratified sub-analyses.
Statistical analyses
SPSS (version 16.0; SPSS, Inc., Chicago, IL, USA) was used for the statistical analyses. In order to assess inter- and intra-rater reproducibility, each measurement was obtained by both raters or twice by the same rater in 20 subjects and intra-class correlation coefficients (ICCs) were calculated. All ICCs lay in the range of 0.57–0.97 (see Supplementary Table 1 for exact ICCs), indicating fair to excellent reproducibility for all measurements (14).
Multiple linear regression models controlling for sex, age and skull diameter (as a correction for head size) were applied for all measurements in order to compare headache patients with controls. The association between headache diagnosis and transcranial diameter at the temporal fossa level was determined by a linear regression model controlling for sex and age. p-values <0.05 were considered significant. Despite the explorative nature of this study, we also corrected p-values for multiple comparisons using the Benjamini and Hochberg false-discovery rate (FDR) correction (15).
Results
Demographic characteristics of subjects included in analyses.
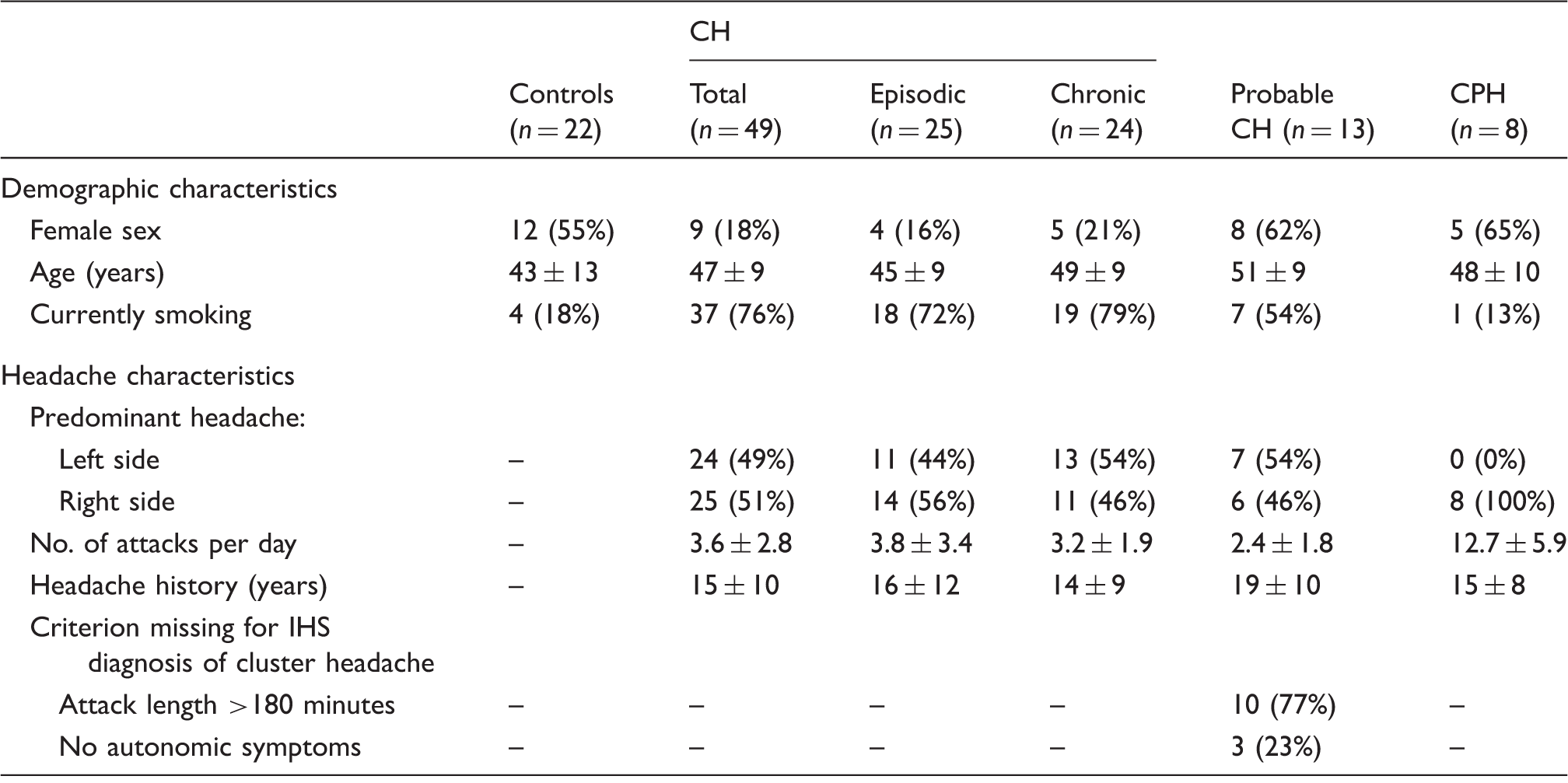
Mean ± SD for continuous variables, number (%) for categorical variables.
CH: cluster headache; CPH: chronic paroxysmal hemicrania.
Incidental and pathological findings on magnetic resonance imaging of subjects included in analyses.
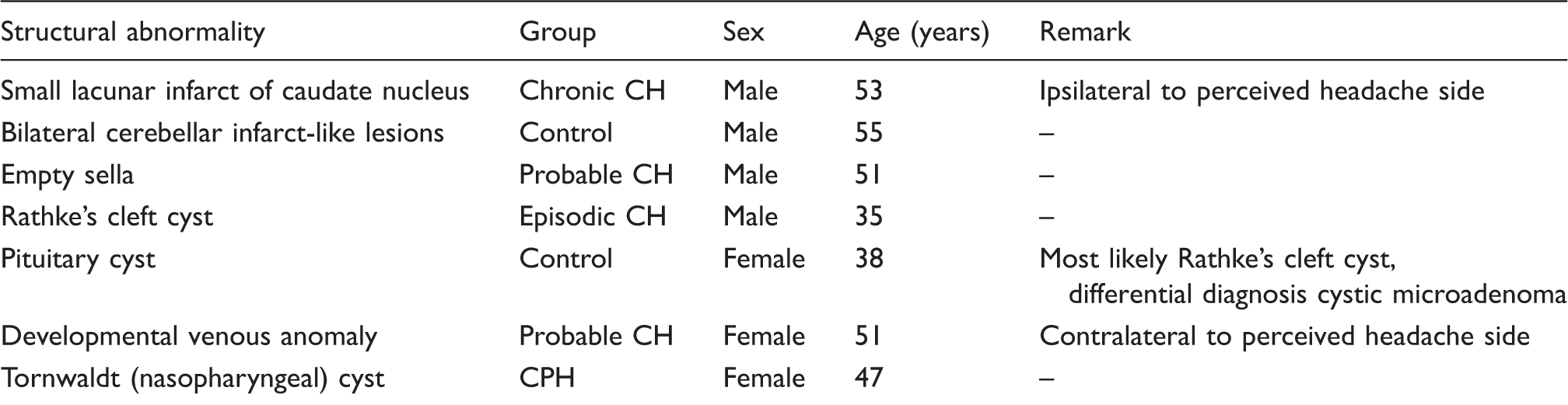
CH: cluster headache; CPH: chronic paroxysmal hemicrania.
Dimensions of the cavernous sinus and neighbouring structures of cluster headache and chronic paroxysmal hemicrania patients and headache-free controls.

All measurement in millimetres, unless stated otherwise. Denotation is means ± SDs. Values for controls are means of left and right measurements. Values for trigeminal autonomic cephalalgia measurements are ipsilateral to the headache side. Number–letter combinations correspond to those used in Figure 1.
p < 0.05, multiple regression analysis controlling for sex and age, uncorrected for multiple comparisons.
Discussion
This is the first study using high-resolution MRI to specifically assess the aspect and dimensions of the cavernous sinus in cluster headache and chronic paroxysmal headache.
Despite the acquisition of high-resolution T2-weighted 1.5-T MRI, we did not find pathological lesions in the cavernous sinus or its neighbouring structures as have been previously associated with TACs (8), which is in line with a previous study using conventional sequences in 0.5-T and 1.5-T magnetic resonance systems (7). However, we cannot exclude that structural abnormalities would have shown up using a more extensive MRI protocol including gadolinium-enhanced T1-weighted images of the sellar region and magnetic resonance angiography of the intracranial and cervical vasculature.
We failed to find any evidence of a constitutionally narrow cavernous sinus region in these TACs, even when combining the results for all 49 (episodic and chronic) cluster headache patients together, which would provide 90% power for demonstrating a 15% difference in cavernous sinus diameter at alpha = 0.05 versus controls. Our results did not change when leaving out subjects who experienced side shifts or contralateral attacks at some point in their disease history.
Subjects with cluster headache and chronic paroxysmal hemicrania did have a wider skull size than in headache-free controls. Although an explanation for this finding remains purely speculative, it seems to be in accordance with observations of so-called leonine facial features in cluster headache, as described several decades ago (16).
Article highlights
Patients with cluster headache or chronic paroxysmal hemicrania have wider skulls than headache-free controls. The proportional dimensions of the cavernous sinus were similar between patients with cluster headache and chronic paroxysmal hemicrania. This opposes the theory of a narrowed cavernous sinus in cluster headache pathophysiology.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: MD Ferrari receives trial support from Medtronic outside of the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an unrestricted grant of the Asclepiade Foundation.

