Abstract
Objective
The objective of this article is to determine whether cutaneous allodynia (CA) influences the response to treatment with occipital transcutaneous electrical stimulation (OTES) in chronic migraine (CM) and chronic tension-type headache (CTTH).
Methods
One hundred and sixty consecutive patients with CM or CTTH were randomized to be treated with real or sham OTES stimulation three times a day for two consecutive weeks. All patients completed the validated 12-item allodynia symptom checklist for assessing the presence and the severity of CA during headache attack. Primary end-point was change (≥50%) in number of monthly headache-free days.
Results
There was a significant difference in the percentage of responders in the real OTES compared with sham OTES group (p <0.001). Importantly, there was not a significant change of monthly headache-free days in the allodynic patients with CM and CTTH treated both with real and sham OTES, while the number of headache-free days per month was significantly reduced in the real (86%) but not in the sham group (7%) of non-allodynic patients with CTTH and CM.
Conclusions
Severe CA is associated with decreased response to treatment with OTES in patients with CM and CTTH.
Keywords
Introduction
Individuals with chronic tension-type headache (CTTH) and chronic migraine (CM) have headache more than 15 days per month for more than three months (1). These patients presenting with chronic headaches often overuse acute-headache medications, and a minority of them do not respond to any of the known preventive therapies (2–4). Hence, either invasive or non-invasive peripheral neuromodulation procedures (5–7) have been introduced to treat these headache sufferers with chronic headaches that become drug resistant (8).
Transcutaneous electrical stimulation (TES), a non-invasive peripheral neuromodulation procedure, has been reported to be effective in the management of headaches (9–11). By contrast, TES elicited only a placebo response in chronic pain (12), and a recent systematic review showed weaker evidence of efficacy of treatment with TES in chronic headaches (13). These conflicting results may be due to confounding factors, such as factors that can influence the response to treatment with TES in headache sufferers.
It is recognized that cutaneous allodynia (CA), a perception of pain when a non-noxious stimulus is applied to normal skin, is associated with primary chronic headaches (14,15). Since it has been suggested that the presence of CA during a headache attack may influence the response to triptans in migraine sufferers (16), we hypothesized that severe CA could decrease the analgesic effect of TES in chronic headaches. To address this hypothesis, we have investigated both the severity of CA and the response to preventive treatment with TES in headache sufferers presenting with CM and CTTH.
Methods
Participants
Two hundred and fifty-one headache sufferers underwent screening for eligibility for inclusion in the study; 91 of these headache sufferers were ineligible. A total of 160 headache sufferers were included in this study: 77 had chronic migraine (CM) and 83 had chronic tension-type headache (CTTH). Institutional review board approval was obtained for the study, and written informed consent was obtained from all the subjects.
Patients
A total of 160 headache sufferers (33 men and 127 women aged 18–73 years, mean 41 years) were enrolled between December 2009 and February 2013 among the outpatients attending the Institute of Neurology in Catanzaro. Patients fulfilling the following criteria were included: chronic headache for at least two years, CTTH or CM (International Classification of Headache Disorders, second edition (ICHD-II)) (17), pain involving the occipital region, at least four attacks per week, failure on at least three well-conducted preventive drug treatments. Exclusion criteria were use of a preventive treatment in the last two months, previous TES treatment (because this could affect the sham TES credibility), severe anxiety and mood depression, severe sleep disturbances, and associated disabling neurologic or other psychiatric disorders.
The validated 12-item allodynia symptom check lists (ASC-12) (18) were administered during an headache attack both at baseline and during the follow-up. Allodynia symptoms and score on CA severity defined the following categories: none (0−2), mild (3−5), moderate (6−8) and severe (9 or higher). Pain and disability were assessed with a 0−10 visual analog scale (VAS) and a Migraine Disability Assessment (MIDAS) questionnaire at baseline and during the follow-up. Mood and anxiety were assessed with Beck Depression Inventory-II (BDI-II) and Hamilton Anxiety Rating Scale (HARS) at baseline and during the follow-up. BDI-II total scores served as indicator of the occurrence and severity of the symptoms of depression. Higher scores (range 29–63) on BDI-II served to indicate a severe level of depressive symptoms.
Neurostimulation
For real and sham occipital TES (OTES) therapy, identical devices were used. The electrostimulator (Genesy 3000, Globus Italia) generated biphasic rectangular impulses with the following parameters: pulse width 250 µs, frequency 40 Hz, intensity 20 mA. The stimulator buzzed identically during real and sham OTES, but no current was actually delivered to the electrodes in sham OTES. Real and sham OTES were delivered with 50 mm× 90 mm self-adhesive electrodes placed on the occipital region covering the greater occipital nerves bilaterally. Instructions to patients were the same, hence it was not possible to distinguish real from sham OTES without testing both treatments.
General procedure
In our headache center, the blinded therapist administered OTES during the treatment period in the outpatient clinic. The neurostimulation session lasted 30 minutes, and was administered three times a day for two consecutive weeks. Given that patients had chronic headaches, OTES was administered more often during the symptomatic phase. The stimulation parameters, however, weren’t modified during the symptomatic phase of the patients. All patients were permitted to use acute medications at the same levels prior to baseline, but other methods of pain control or new preventive treatments were prohibited.
Study design and outcome measures
We performed a prospective, blinded, randomized and placebo-controlled study (Figure 1). The principal investigator (F.B.), who was not involved in treating and assessing outcome of patients, randomized the eligible patients into real or sham groups in a 2:1 ratio. The 2:1 randomization was chosen to minimize the number of patients who received no stimulation, and to maximize the number of patients who received real stimulation for determination of CA-related effects on the response to treatment with OTES. To guarantee the concealment of allocation, we observed the following procedures: the research assistant, who had access to the randomization list and who was not involved in recruiting, treating, or assessing outcome of the patients, delivered real OTES or sham OTES devices to the therapist; the therapist administered OTES during the treatment period in the outpatient clinic; the investigators who assessed outcome made no inquiries concerning stimulation intensity levels or experienced sensations, and avoided physical contact between patients during the visits; patients were instructed to address questions concerning the device to the research assistant throughout the study; the investigators who assessed outcome, the patients, and the therapist were all blinded. The baseline evaluation was followed by a 14-day treatment period with intermediate visits after 30 days and a final visit at 60 days. Patients filled in diaries recording headache occurrence and acute medications use one month before treatment and during the two months post-treatment. Blinded investigators created the database from the headache diaries filled in by the patients. Duration of OTES use served as a measure for compliance and was recorded by the therapist, who was trained to do so. Patients’ satisfaction with group allocation and treatment result were assessed during the treatment period and during a visit at 30 and 60 days.
Flow diagram from screening to study completion. Of the 251 patients prescreened, 91 were ineligible.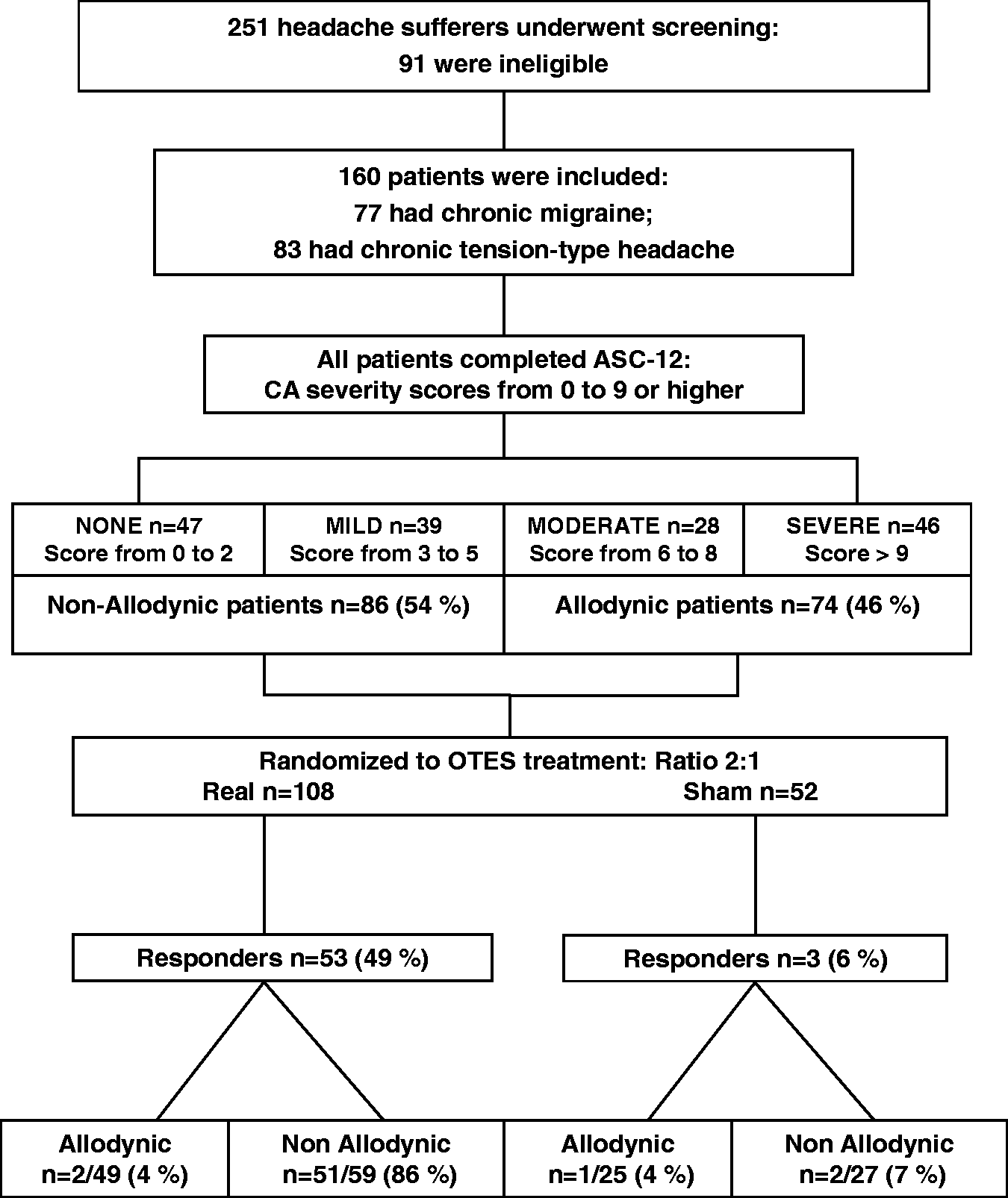
The primary end-point was considered the change in the number of headache-free days per month. A respondent was considered a subject who achieved a 50% or greater reduction in number of headache days per month. Other secondary outcomes were: 1) change in total headache days per month compared with baseline; 2) change in mean reduction of headache days per month compared with baseline; 3) change in monthly acute medications use compared with baseline.
This clinical study has not been registered with Clinicaltrials.gov nor with EudraCT.
Statistical analysis
Statistical analysis was performed with R Statistical Software (R for Unix/Linux, version 2.15.1, The R Foundation for Statistical Computing, 2012). The influence of the headache diagnosis, OTES treatment, CA and medication overuse on the responders-to-non-responders ratio was assessed by means of a multivariate analysis involving logistic regression on a generalized linear model. Differences in monthly headache days between evaluations at baseline, at 30 days and after 60 days in the real and sham groups both of allodynic and non-allodynic patients with CM and CTTH were compared using a single-tailed Wilcoxon rank sum test. Differences between treatments at baseline and after 30 and 60 days were assessed using single-tailed Mann-Whitney U test. Differences in BDI and HARS between both real and sham groups and allodynic and non-allodynic groups at baseline were assessed by using Mann-Whitney test and unpaired t-test.
The Wilcoxon rank sum test and the paired t-test were used to assess differences in BDI, HARS, VAS and MIDAS severity scores at 30 days and after 60 days compared to baseline. Differences in allodynia distribution within responders were evaluated by means of Fisher’s exact test, while asymmetry in allodynia distribution between the real and sham OTES groups was assessed using a stratified analysis with Mantel-Haenszel test. The χ2 test was used to compare both the patients’ satisfaction and the change in number of responders at 30 days and after 60 days. The monthly change in acute medications use at 30 days and after 60 days was evaluated using the McNemar’s test. When not explicitly specified, all p values were two tailed, and α level was set at p < 0.05.
Results
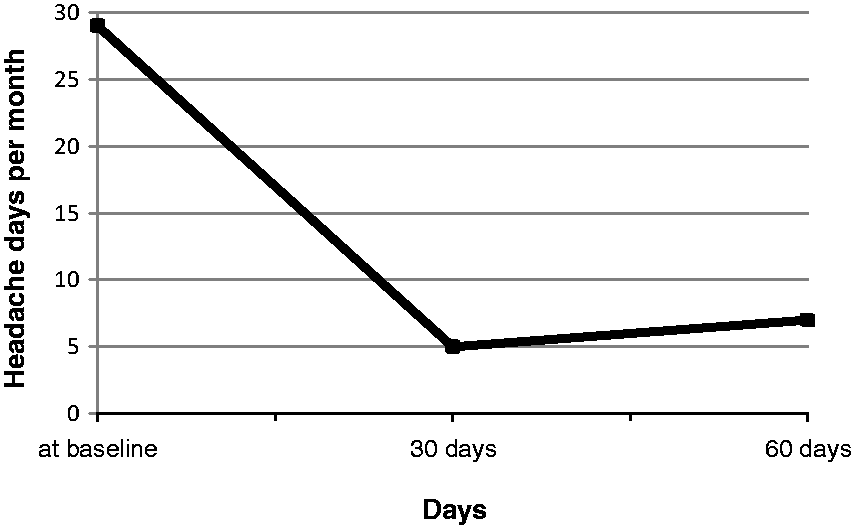
The characteristics of the 160 headache sufferers are summarized in Table 1. One hundred and eight patients (including 54 patients with CM and 54 patients with CTTH) underwent treatment with real OTES, and sham OTES was administered to 23 patients with CM and 29 with CTTH (Table 2). There was a significant difference in the percentage of responders in the real OTES compared with sham OTES group (p < 0.001). There were 53 (49%) respondents among 108 patients treated with real OTES, including 46 (43%) with CTTH and seven (6%) with CM, while only one (2%) patient with CM and two (4%) patients with CTTH had a significant treatment response to sham OTES. Moreover, based on the ASC-12 CA severity scores the patients were grouped in either a non-allodynic group (no or mild CA with a score of 0−5) or an allodynic group (moderate or severe cutaneous allodynia with a score above 6). The non-allodynic group consisted of 86 patients, including 78 with CTTH and eight with CM. The allodynic group consisted of 74 patients, including 69 with CM and five with CTTH. Forty-nine allodynic and 59 non-allodynic patients were treated with real stimulation, while sham stimulation was administered to 25 allodynic and 27 non-allodynic patients (Table 3). There was no significant change in the number of headache-free days in 47 of 49 (96%) allodynic patients in the real group and in 24 of 25 (96%) allodynic patients in the sham group (Figure 2). Of note, treatment with real OTES caused an average increase in headache pain intensity in allodynic patients (Table 2). Monthly headache-free days were significantly reduced in 51 out of the 59 (86%) non-allodynic patients in the real group, and in only two of 27 (7%) non-allodynic patients in the sham group. Comparison between total headache days at 30 and 60 days showed that the efficacy of real OTES lasted 60 days in two-thirds of respondents (Figure 3).
The response to treatment with occipital transcutaneous electrical stimulation in 160 headache sufferers grouped according to 12-item allodynia symptom checklist severity scores. The duration of efficacy of treatment with occipital transcutaneous electrical stimulation lasted about 60 days in two-thirds of the responders. Demographic and characteristics of the 160 headache sufferers. F: female; M: male; ASC-12: validated 12-item allodynia symptom checklists; CA: cutaneous allodynia. Response to treatment with real or sham occipital transcutaneous electrical stimulation (OTES) in 160 headache sufferers grouped according to diagnosis of headache. p < 0.001 compared to Real group in CTTH; p < 0.001 compared to Sham group in CTTH; p = <0.01 compared to baseline; p < 0.002 compared to baseline; p = 0.008 compared to baseline; p = <0.001 compared to baseline; p = <0.001 compared to baseline. F: female; M: male; CTTH: chronic tension-type headache; VAS: visual analog scale; MIDAS: Migraine Disability Assessment; BDI II: Beck Depression Inventory II; HARS: Hamilton Anxiety Rating Scale. Response to treatment with real or sham occipital transcutaneous electrical stimulation (OTES) in 160 headache sufferers grouped according to 12-item allodynia symptom checklist (ASC-12) severity scores. p = <0.008 compared to baseline; p = <0.007 compared to baseline; p = <0.001 compared to baseline; p = <0.001 compared to baseline; p = <0.001 compared to baseline; p = <0.001 compared to baseline; p = <0.001 compared to baseline; p < 0.001 compared to baseline; p < 0.001 compared to baseline. VAS: visual analog scale; MIDAS: migraine disability assessment; BDI II: Beck Depression Inventory II; HARS: Hamilton Anxiety Rating Scale.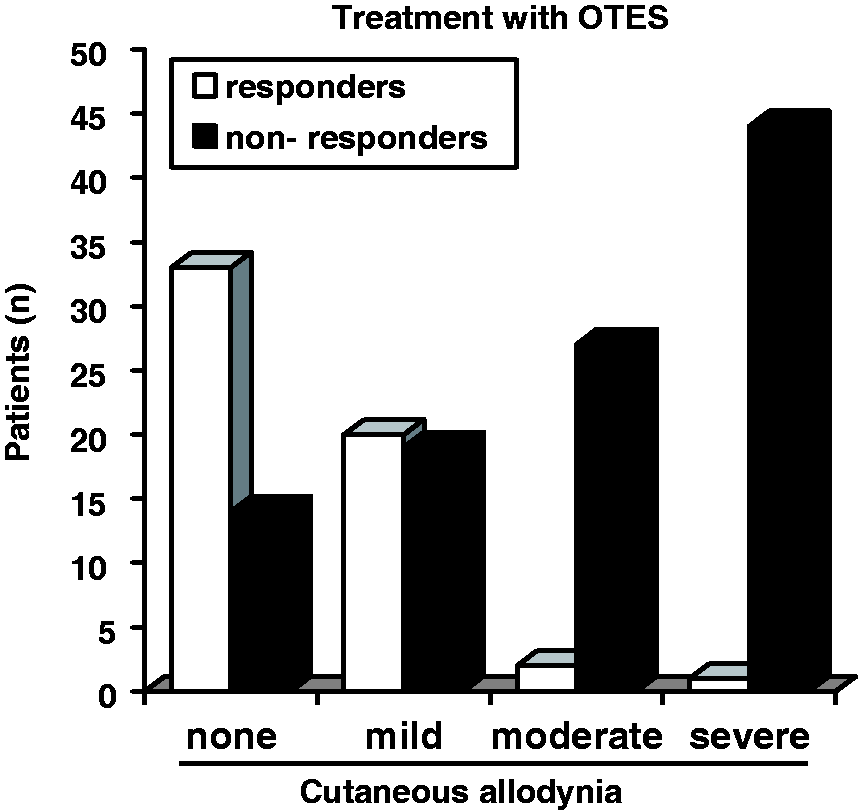



The results of the multivariate analysis showed that headache diagnosis and medication overuse did not influence the response to OTES treatment, while the outcome of patients was significantly influenced by CA (p = 0.00016), OTES treatment (p = 0.003) and by CA and OTES treatment interaction (p = 0.004). The model coefficients (with 95% confidence intervals) were CA 4.12 (2.04–9.78); OTES treatment 3 (1.49–7.13); interaction of CA and OTES treatment: 2.97 (1.28–6.26). These data indicated that CA was an independent predictive factor of efficacy of real OTES treatment (Table 3). After real OTES treatment, a slight change in ASC-12 CA severity scores was recorded only in few allodynic patients, but this change in CA score was not significantly different compared with baseline. The distribution of acute medications use did not differ significantly for allodynic compared with non-allodynic patients, and there was no significant effect of medication overuse on the outcome (Table 3), while after OTES treatment, either acute medications use or MIDAS scores in responders differed significantly compared with baseline (Tables 2 and 3). For anxiety and mood, there were not significant differences between real and sham groups (p = 0.6; p = 0.21), and between the allodynic and non-allodynic groups (p = 0.055; p = 0.49) at baseline. During the follow-up, there were significant differences in HARS and BDI II scores for responders compared to baseline (Tables 2 and 3). For compliance there was no significant difference between the real and sham groups during OTES treatment. Importantly, the observance of OTES treatment in allodynic patients and non-allodynic patients was not significantly different (Table 3). The proportion of satisfied patients did not differ between the real and sham groups during OTES treatment, but there were significant differences in patients’ satisfaction with treatment results for real compared to sham OTES at 30 days (chi-square = 11, p = 0.0009) and after 60 days (chi-square = 15, p = 0.0001). Neither adverse events nor side effects occurred in the real or sham group.
Discussion
This randomized, placebo-controlled study provides evidence that severe CA is associated with decreased response to treatment with OTES in patients with CM and CTTH, and it demonstrates the efficacy of OTES in non-allodynic headache sufferers presenting with CTTH and CM. These findings suggest that the response to OTES is related to severity of CA in CM and CTTH. To our knowledge, this study is the first evidence that severity of CA is an influencing factor on the response to preventive treatment in chronic headaches.
In the present series CA resulted an independent predictive factor of the efficacy of real OTES treatment in CM and CTTH. Indeed, the majority of non-respondents to treatment with real OTES were allodynic patients with CM, but there was also a lack of treatment response to real OTES in allodynic patients with CTTH. Given that non-respondent patients had severe CA, the presence of severe CA may be the explanation of decreased response to treatment with OTES in our series of patients presenting with CM or CTTH. It is recognized that severe CA is highest in transformed migraine, but it can also occur in other primary chronic headaches and severe episodic CTTH (15). CA represents a peripheral manifestation of increased responsiveness of central pain neurons during a headache attack, and the degree of this central sensitization reflects the severity of CA (19). Hence, we hypothesize that administration of electrical stimuli on cutaneous areas of increased skin sensitivity lead to an increase of the central sensitization, which in turn causes the increase of pain in allodynic headache sufferers. In fact, real OTES caused an increase in headache pain intensity in more severe allodynic headache sufferers of the present series. This latter observation may explain the lack of effectiveness of TES in headache sufferers found in some studies (13). Indeed, the severity of CA in these headache sufferers was not reported by the authors. Thus, the presence of severe CA in those headache sufferers could be an explanation for the lack of effectiveness of TES in these studies. Moreover, to avoid possible confounders such as factors influencing both the central sensitization and the response to treatment in headache sufferers, we excluded in our study patients with severe anxiety, mood depression, or sleep disturbances. Furthermore, the distribution of acute medications use was not significantly different between allodynic and non-allodynic patients, and there was no significant effect of medication overuse on the outcome. Hence, anxiety, mood depression, sleep disturbances, or medication overuse may not be considered a possible cause of scarce response to real OTES treatment in allodynic patients. In addition, the observance of the treatment in allodynic patients and non-allodynic patients was not different. These latter data suggest that the lack of efficacy in allodynic patients was not due to the lack of tolerance in these patients. To avoid another possible confounder due to different difficulty in managing pain among patients, we recruited headache sufferers who had failure on at least three well-conducted preventive drug treatments, and both allodynic and non-allodynic patients presented the same difficulty in managing chronic pain. Hence, the scarce response to OTES treatment in allodynic patients may not be due to a generic difficulty in managing pain in these patients. The given data and the above-mentioned studies support our finding that severe CA is associated with diminished treatment response to real OTES treatment in headache sufferers presenting with either CM or CTTH, suggesting that severity of CA may be an influencing factor on the analgesic effect of OTES in these patients.
Confirming these latter observations, primary outcome measures also indicated that real OTES was superior to sham stimulation for the prevention of headache in our series of non-allodynic patients. Although the majority of these respondents to treatment with real OTES were non-allodynic patients with CTTH, we documented also the analgesic effect of treatment with real OTES in non-allodynic patients with CM. Given the low prevalence and lower severity of CA among these respondents, a possible explanation of the beneficial effects of OTES in our series of headache sufferers could be the lack of severe CA in the respondents. However, the OTES mode of action remains unclear in these non-allodynic respondents and a theory may be only hypothesized. Considering that central sensitization is generated by prolonged nociceptive input from pericranial myofascial structures in patients with CTTH (20), we hypothesize that administration of electrical stimuli on cutaneous areas covering occipital pericranial myofascial tissues stopped the prolonged nociceptive input generating the central sensitization in our non-allodynic patients with CTTH. This hypothesis is supported by the evidence demonstrating that central sensitization is due to loss of gray matter brain structures involved in pain processing in CTTH (21) and the reverse of central sensitization leading to a decrease in pain by using low-frequency TES in patients with CTTH (22), while the beneficial effects of OTES in our non-allodynic patients with CM may be due to transcutaneous electrical stimulation of the greater occipital nerve. The occipital nerve stimulation has been shown to produce a pain modulation via the peripheral and central nervous systems, resulting in pain relief in patients with CM (23–25). Interestingly, a recent study reported that supraorbital TES was an effective preventive treatment in episodic migraine patients (26). Considering that severe CA is less common in episodic migraine (15), and the presence and severity of CA were not reported by the authors, the absence of CA in those respondent migraine sufferers cannot be excluded. Moreover, after OTES treatment there were significant differences in pain, disability, acute medications use, anxiety and mood, for non-allodynic responders compared with baseline. These results proved the efficacy of OTES treatment in these patients. Taken together these observations support our finding that preventive treatment with OTES is effective in non-allodynic headache sufferers presenting with CTTH and CM.
In the present study, the analgesic effect produced by real OTES treatment was reduced when we applied electrical stimuli on cutaneous areas of pain and allodynia in patients with CM and CTTH. Although we hypothesized the different mechanisms of action of OTES treatment in allodynic and non-allodynic patients, the mechanisms underlying pain relief by OTES in patients with CM and CTTH remain unclear. It is now recognized (27) that TES produces both segmental and descending inhibition, the release of endogenous opioids, and modulation of autonomic activity. These data suggest that several central mechanisms may play a role in relief of pain by TES in chronic headaches (28). In addition, there was an increase of headache pain intensity in some severely allodynic patients. Since specific opioids receptors are activated to produce analgesia with TES (29), a possible explanation of these CA-related effects on OTES treatment could be the lack of release of endogenous opioids in severely allodynic patients. Considering that a descending inhibition is activated by TES (27), it seems less probable that in these allodynic patients the headache worsening after OTES treatment may be caused by a sort of nocebo effect due to a mechanism of descending facilitation. We believe that further research concerning OTES treatment for patients with chronic headache should particularly target the mechanisms underlying pain relief.
In this study, the results did not show a significant analgesic effect of placebo treatment. In addition, the comparison of the treatment response to real OTES between patients with CM and CTTH including both allodynic and non-allodynic patients indicated a significant analgesic effect of treatment with real OTES only in non-allodynic headache sufferers. Moreover, the treatment with real OTES and placebo were the same, and it was not possible to distinguish real from sham OTES without testing both treatments. Thus, we provided an adequate control for electrical stimulation, and thereby we eliminated an eventual active placebo effect of the treatment with OTES in these patients.
Despite the fact that we observed methodologic procedures to avoid the risk of unblinding and to guarantee the concealment of allocation, partial unblinding due to the difference in experienced sensations between real and sham stimulation may have occurred in our study. We do not believe, however, that unblinding markedly influenced our results for the following reasons. The lack of difference in both the compliance rates and the proportion of satisfied patients between real and sham groups during OTES treatment does not indicate the occurrence of a massive unblinding in our study. If this were the case, one would expect a lower compliance and satisfaction in the sham group. The percentage of responders in the sham group also does not favor a massive unblinding in our study. The responder rate for sham OTES treatment was within the range of those reported for other trials with peripheral neurostimulation devices (25,26). Considering that unblinding is inversely proportional to the percentage of responders in a sham group, it was not considered massive in the Occipital Nerve Stimulation for the Treatment of Intractable Migraine (ONSTIM) (25) and Prevention of Migraine by Supraorbital Transcutaneous Neurostimulation Using the Cefaly® Device (PREMICE) (26) trials. Thus, unblinding may not be pronounced in our study. We are fully aware, however, that unblinding may be a possible weakness of all neurostimulation trials because of the difference in experienced sensations between real and sham stimulation. To avoid this risk of unblinding, the use of a special sham stimulation device that is active only for the first 30–60 seconds could be useful in future neurostimulation trials.
We believe that the results of our study may also be relevant to studies of new peripheral neuromodulation procedures in headache sufferers. However, this was a single-center study, and neither the long-term response to OTES nor the CA-related effects on the response to repeated OTES treatments were evaluated in these patients.
Finally, the present data highlight that the severity of CA may be an influencing factor on the response to preventive treatment with OTES in headache sufferers presenting with CM and CTTH, suggesting that CA should be considered in the management of these patients in clinical practice.
Clinical implications
Severe cutaneous allodynia (CA) decreases the response to preventive treatment with occipital transcutaneous electrical stimulation (OTES) in chronic migraine (CM) and chronic tension-type headache (CTTH). OTES increases the head pain intensity in severely allodynic patients with CM and CTTH. OTES is effective in non-allodynic patients with CTTH and CM. The response to OTES administered 30 minutes three times a day bilaterally for two weeks lasts about 60 days. Severity of CA is an influencing factor on the response to preventive treatment with peripheral neuromodulation procedures in CM and CTTH, and CA should be considered in the management of chronic headaches in clinical practice.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgment
We are grateful to the patients who participated in the study.
