Abstract
Background
There is a considerable amount of practice variation in managing migraines in emergency settings, and evidence-based therapies are often not used first line.
Methods
A peer-reviewed search of databases (MEDLINE, Embase, CENTRAL) was carried out to identify randomized and quasi-randomized controlled trials of interventions for acute pain relief in adults presenting with migraine to emergency settings. Where possible, data were pooled into meta-analyses.
Results
Two independent reviewers screened 831 titles and abstracts for eligibility. Three independent reviewers subsequently evaluated 120 full text articles for inclusion, of which 44 were included. Individual studies were then assigned a US Preventive Services Task Force quality rating. The GRADE scheme was used to assign a level of evidence and recommendation strength for each intervention.
Interpretation
We strongly recommend the use of prochlorperazine based on a high level of evidence, lysine acetylsalicylic acid, metoclopramide and sumatriptan, based on a moderate level of evidence, and ketorolac, based on a low level of evidence. We weakly recommend the use of chlorpromazine based on a moderate level of evidence, and ergotamine, dihydroergotamine, lidocaine intranasal and meperidine, based on a low level of evidence. We found evidence to recommend strongly against the use of dexamethasone, based on a moderate level of evidence, and granisetron, haloperidol and trimethobenzamide based on a low level of evidence. Based on moderate-quality evidence, we recommend weakly against the use of acetaminophen and magnesium sulfate. Based on low-quality evidence, we recommend weakly against the use of diclofenac, droperidol, lidocaine intravenous, lysine clonixinate, morphine, propofol, sodium valproate and tramadol.
Introduction
Migraine is a common disorder, with prevalence rates among Canadian adults in the range of 23%–26% in women and 7.8%–10% in men (1–3). Migraines negatively affect quality of life (4,5) and engender significant disability (4,6–8). A study by the World Health Organization showed that migraine is the third most prevalent medical disorder and the eighth most disabling worldwide (9). Migraine also results in a large cost to society, through associated health care costs and loss of productive time (10–14).
Headaches are one of the most common reasons for visiting the emergency department (ED). In a United States (US)-based study, headache was the first listed diagnosis in 2.4% of ED visits, and 34.6% of headache visits were accounted for by migraine (15). One study found that patients presenting with migraine or other headaches had the third highest self-reported pain scores among all patients presenting to the ED with a painful condition (16).
There is a significant amount of practice variation in terms of the management of migraine in the ED (17–19). The use of therapies for migraine is variable and many practitioners are not using evidence-based therapies first line (19–21).
Objectives
The primary objective of this systematic review is to describe and evaluate the evidence from randomized controlled trials (RCTs) for the effectiveness and tolerability of pharmacologic or nutraceutical interventions in the acute treatment of migraine pain in adults presenting to the ED or other similar settings.
Methods
A detailed description of the methodology and results is available in the online Supplementary Material.
Criteria for considering studies for this review
Studies were limited to double-blind prospective RCTs of patients receiving treatment for migraine in the ED or in other similar settings. Studies in outpatient clinics were included if parenteral therapy was used to acutely treat migraine. The target population was adults aged 18 and over. Only studies using either the International Headache Society diagnostic criteria for migraine (22) or the Ad Hoc criteria (23) were included. Included studies were limited to pharmacologic or nutraceutical agents that are commercially available, although not all agents reviewed are available in Canada. Only studies with at least one outcome measure pertaining to acute pain relief were included.
Search methods for identification of studies
MEDLINE and In-Process & Other Non-Indexed Citations (1946 to 2013 March Week 1), Embase (1980 to 2013 Week 11) and EBM Reviews—Cochrane Central Register of Controlled Trials (February 2013) were searched on February 20, 2013, using the OvidSP interface. A librarian (Margaret Sampson, MLIS, PhD, AHIP) developed the MEDLINE search strategy, which was peer reviewed by a second librarian (Janey Joyce, MLS) using the Peer Review of Electronic Search Strategies (PRESS) standard (24). The MEDLINE strategy was then adapted for the other databases. Searches were limited to RCTs or systematic reviews. The Cochrane HSSS sensitivity- and precision-maximizing version (2008 revision) (25) and the systematic review subset (26) were used in MEDLINE. The United Kingdom (UK) Cochrane Centre search for trials (26) and the Scottish Intercollegiate Guidelines Network (SIGN) search filter for systematic reviews (27) were used in Embase. Searches were not restricted by language of publication. Search strategies are presented in the online Supplementary Material. Systematic reviews and included RCTs were reviewed for relevant references.
Data collection and analysis
Two independent reviewers (SLO and SNC) used the title, keywords and abstract to determine the study’s eligibility for inclusion in the systematic review. Subsequently, three independent reviewers (SLO, SNC and GD) used standardized forms to evaluate potentially eligible studies using the abstract and/or the entire publication in order to make a final determination of eligibility for inclusion.
Each study was graded according to its risk of bias, as outlined in the Cochrane Handbook for Systematic Reviews of Interventions (28). The final rating of individual studies according to their methodological quality was carried out as per the US Preventive Services Task Force criteria. This system analyzes the quality of RCTs according to seven different domains. Studies are rated as “good” if they meet all criteria. If there are criteria that are not met, but the study contains no fatal flaw that would invalidate its results, then the study is rated as “fair.” Studies with fatal flaws are rated as “poor” (29).
Groups of two or more trials without significant clinical heterogeneity were combined in meta-analyses. Odds ratios were calculated for the outcome of interest, with confidence intervals set at 95% for both the individual studies and the pooled odds ratio. Chi square tests were used to obtain estimates of statistical heterogeneity. Where chi square results were indicative of significant heterogeneity (p ≤ 0.1), the study results were pooled using a random-effects model. Where the studies were homogenous (p > 0.1), the results were combined using the Mantel-Haenszel methods.
GRADE quality of evidence levels.

GRADE: Grading of Recommendations Assessment, Development and Evaluation Working Group.
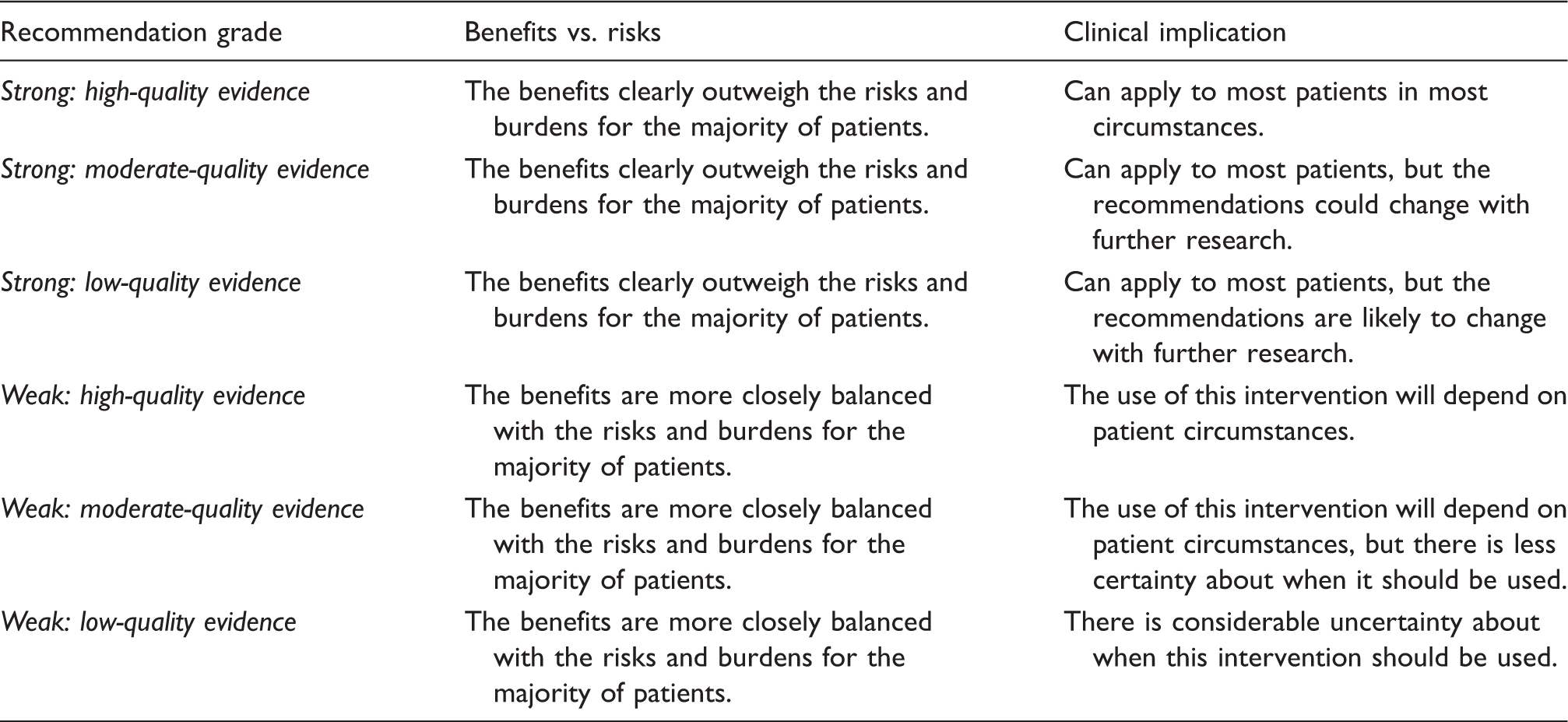
Recommendation grades used in this systematic review.
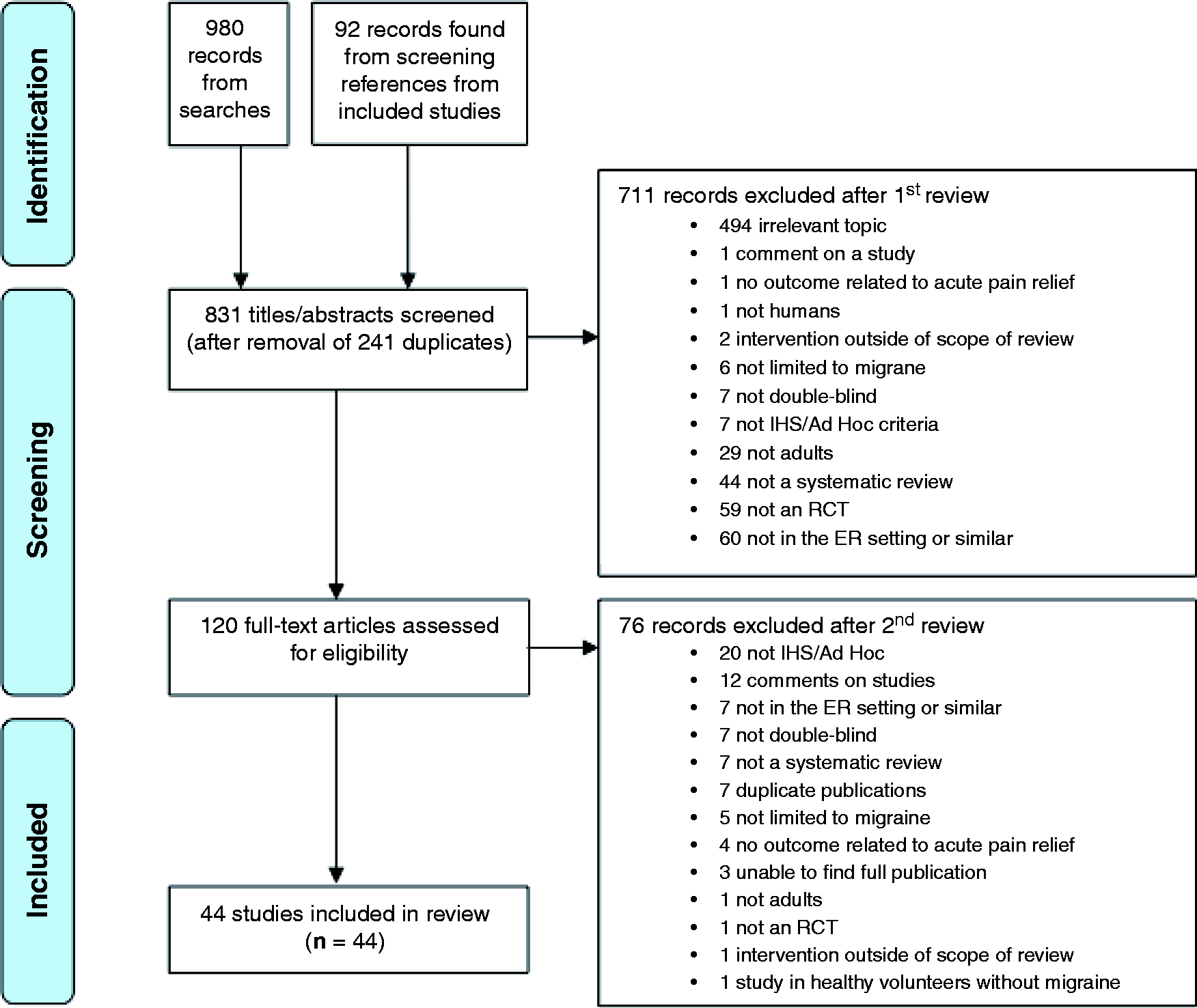
PRISMA diagram.
Results
1. Sumatriptan
Strong recommendation, moderate-quality evidence
We recommend that clinicians offer subcutaneous (SC) sumatriptan to eligible patients presenting with acute migraine. Based on the literature, we recommend sumatriptan 6 mg SC.
There was consistent evidence, in the form of seven RCTs, all of “fair” quality, to show that SC sumatriptan is more effective than placebo (32–39). The results of these trials were pooled together into a meta-analysis (Figures 2 and 3).
Meta-analysis of subcutaneous sumatriptan vs. placebo: percentage of patients achieving pain freedom. Meta-analysis of subcutaneous sumatriptan vs. placebo: percentage of patients achieving pain relief.

Three trials found that sumatriptan was inferior to strong active comparators. A “fair” quality RCT failed to find a difference between SC sumatriptan and metoclopramide in the primary outcome, but metoclopramide was superior to sumatriptan in some of the secondary outcomes (39). Prochlorperazine was superior to SC sumatriptan in a “good” quality trial (40). Intranasal ketorolac was superior to intranasal sumatriptan in a “fair” quality RCT (41).
Two trials found sumatriptan to be superior to active comparators. A “poor” quality RCT showed superiority of SC sumatriptan over SC dihydroergotamine in terms of rapid pain relief, although the dihydroergotamine patients were more likely to have sustained pain relief at 24 hours (42). A “fair” quality RCT, powered only to detect superiority of trimethobenzamide over SC sumatriptan, failed to show any statistically significant difference between trimethobenzamide and sumatriptan (43).
The recommendation is strong because there is consistent evidence showing that sumatriptan is more effective than placebo in this setting. Although there are trials showing superiority of prochlorperazine and metoclopramide over sumatriptan, these two agents are very strong comparators for this indication, thereby making it difficult to discount sumatriptan’s efficacy based on these results. Because the recommendation is restricted to SC sumatriptan, the intranasal trial did not impact the assessment. The body of evidence was downgraded to moderate because half of the sumatriptan studies were carried out in a clinic-based setting, and the evidence derived from those studies is indirect when one is extrapolating it to an ED setting.
2. Phenothiazines: prochlorperazine
Strong recommendation, high-quality evidence
We recommend that clinicians offer prochlorperazine to eligible patients with acute migraine. Based on the literature, we recommend prochlorperazine at a dose of 10 mg intravenously (IV).
A “fair” quality RCT found that prochlorperazine yielded more clinical success than both metoclopramide and placebo. However, two of the prochlorperazine patients (8%) had dystonic reactions for which they were excluded from analyses (44). In a very small, “fair” quality pilot RCT, rectal prochlorperazine was superior to rectal placebo in terms of efficacy (45). A “good” quality RCT found IV prochlorperazine to be more effective than SC sumatriptan and placebo (40). Another “good” quality trial failed to show a difference in efficacy between IV prochlorperazine and metoclopramide, and akathisia rates were equal (13% of each group) (46). IV prochloperazine was also superior to octreotide in a “fair” quality trial, but did yield higher rates of sedation and akathisia (47). Finally, a small “poor” quality RCT found IV prochlorperazine to be superior to sodium valproate (48).
As above, we found high-quality evidence for the efficacy of prochlorperazine. Although the reported incidence of extrapyramidal side effects is significant, we feel that the efficacy of prochlorperazine outweighs the risk of side effects in the majority of patients.
3. Phenothiazines: chlorpromazine
Weak recommendation, moderate-quality evidence
We recommend that clinicians offer chlorpromazine for the acute treatment of migraine. The most effective dose of chlorpromazine for this indication is not well established. Based on the evidence available, we recommend chlorpromazine 0.1 mg/kg–25 mg IV.
IV chlorpromazine was significantly more effective than placebo at yielding pain relief in a “fair” quality trial, but side effects were common with chlorpromazine, especially drowsiness and postural hypotension (49). Another “fair” quality RCT found no difference in efficacy nor incidence of side effects between IV chlorpromazine and metoclopramide (50). A small, “poor” quality RCT failed to show any difference between IV chlorpromazine and intramuscular (IM) ketorolac (51).
We found moderate-quality evidence supporting the efficacy of chlorpromazine in acute migraine. The strength of this recommendation is weak based on the fact that side effects are common with chlorpromazine.
4. Other antiemetics: metoclopramide
Strong recommendation, moderate-quality evidence
We recommend that clinicians offer metoclopramide to patients presenting with acute migraine. We recommend metoclopramide at a dose of 10–20 mg IV for the acute treatment of migraine.
In a small “poor” quality RCT, intravenous metoclopramide resulted in greater pain relief than placebo (52). A “fair” quality RCT found IV metoclopramide and placebo to be less efficacious than prochlorperazine, and two patients (8%) in the metoclopramide group were excluded from analysis because of dystonic reactions (44). A “good” quality RCT comparing IV metoclopramide and prochlorperazine failed to find any difference in efficacy, and 13% of patients in each group experienced akathisia (46). There was no difference in efficacy nor side effect rates when comparing IV metoclopramide and chlorpromazine in a “fair” quality trial (49). Another “fair” quality RCT failed to show any difference between IV metoclopramide and SC sumatriptan in terms of the primary outcome, but rates of two-hour pain freedom and use of rescue medications favored metoclopramide, with no difference in the incidence of side effects between the groups (39). There was no difference in the primary outcome when comparing IV metoclopramide to magnesium sulfate and placebo in a “poor” quality RCT, and one patient in the metoclopramide group (3%) developed a dystonic reaction (53). Finally, a “good” quality dose-finding trial found no difference in the primary outcome nor in akathisia rates when comparing IV metoclopramide at doses of 10 mg, 20 mg and 40 mg (54). Our searches of clinical trial registries identified an unpublished RCT comparing metoclopramide to droperidol for the acute treatment of migraine in the ED (clinicaltrials.gov identifier NCT01406860).
Based on the above, we found moderate-quality evidence supporting the efficacy of metoclopramide for this indication. This body of literature included multiple studies that had significant methodological flaws, as well as one unpublished trial. However, overall, the evidence for the efficacy of metoclopramide outweighs the risk of extrapyramidal side effects, in the majority of patients.
5. Other antiemetics: granisetron
Strong recommendation, low-quality evidence
We recommend against the use of granisetron for the acute treatment of migraine.
In a very small and “poor” quality trial, no significant differences were found when comparing IV granisetron to placebo in terms of the primary outcome, and the incidence of constipation was higher among patients receiving granisetron (55).
6. Other antiemetics: trimethobenzamide
Strong recommendation, moderate-quality evidence
We recommend against the use of trimethobenzamide for the treatment of acute migraine.
We found moderate-quality evidence that trimethobenzamide is inferior to sumatriptan. A “fair” quality trial comparing IV trimethobenzamide to SC sumatriptan was discontinued after interim analyses found that trimethobenzamide was very unlikely to show greater efficacy than sumatriptan (43).
7. First-generation antipsychotics: droperidol
Weak recommendation, low-quality evidence
We recommend against the use of droperidol for the acute treatment of migraine.
In a large but “poor” quality RCT, IM droperidol was superior to placebo in terms of efficacy, but akathisia rates were high (56). Another “poor” quality trial compared IM droperidol to meperidine and found no difference in efficacy between the two groups, with 13.3% of the droperidol patients reporting akathisia (57). As noted above, we identified an unpublished RCT comparing metoclopramide to droperidol for the acute treatment of migraine in the ED (clinicaltrials.gov identifier NCT01406860).
The risk of akathisia is significant with this medication and is felt to outweigh any potential benefit it may have in the acute treatment of migraine.
8. First-generation antipsychotics: haloperidol
Strong recommendation, low-quality evidence
We recommend against the use of haloperidol for the acute treatment of migraine.
IV haloperidol was superior to placebo in reducing migraine pain in a “poor” quality RCT, but the incidence of side effects with haloperidol was very high (58). The side effects of haloperidol are therefore felt to outweigh any benefit it may have based on the current literature.
9. Nonsteroidal anti-inflammatory drugs (NSAIDs): ketorolac
Strong recommendation, low-quality evidence
We recommend that clinicians offer ketorolac to eligible patients presenting with migraine to the ED or similar settings. Ketorolac can be given IM at a dose of 60 mg or IV at a dose of 30 mg IV, based on the current evidence.
We found low-quality evidence to support the efficacy of ketorolac in the acute treatment of migraine. The evidence is derived from a group of small studies with methodological flaws and imprecise results. In a “fair” quality RCT, IM ketorolac and meperidine did not differ in efficacy, nor in incidence of side effects (59). IM ketorolac was less effective than meperidine in a small “poor” quality trial, and only minor side effects were reported (60). The results of these two trials comparing ketorolac and meperidine were pooled into a meta-analysis, and are displayed in Figure 4.
Meta-analysis of ketorolac vs. meperidine: percentage of patients achieving pain freedom.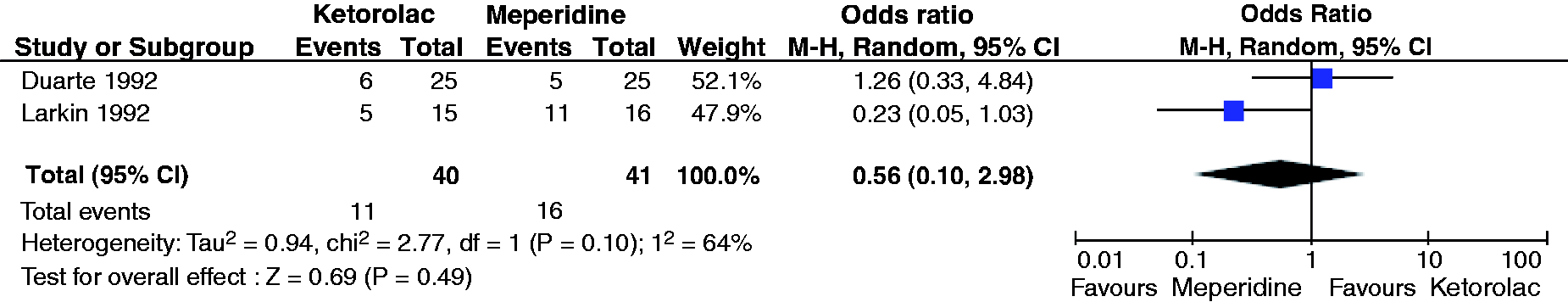
IV ketorolac was more effective than intranasal sumatriptan in a small, but “fair” quality trial (41). A small, “poor” quality trial comparing IM ketorolac and IV chlorpromazine failed to show any difference in efficacy between the two agents (51).
Overall, the evidence seems in favor of efficacy for ketorolac and the side effect profile appears to be relatively benign.
10. NSAIDs: diclofenac
Weak recommendation, low-quality evidence
We recommend against the use of IM diclofenac for the acute treatment of migraine.
In a “poor” quality RCT, IM diclofenac was compared to IV placebo and there were no significant differences in the primary outcome between the groups, other than in small subgroup analyses. However, diclofenac appeared to be more efficacious than placebo in some of the secondary outcomes (61). IM diclofenac and tramadol did not differ in efficacy outcomes in a small “poor” quality trial (62). In another “poor” quality RCT, IM diclofenac appeared to be less effective than intramuscular dihydroergotamine, but it is unclear if the results were statistically significant (63).
The balance between benefits and side effects of IM diclofenac is not entirely clear, but appears to disfavor diclofenac for this indication.
11. NSAIDs: lysine clonixinate
Weak recommendation, low-quality evidence
We recommend against the use of IV lysine clonixinate for the acute treatment of migraine.
In a small, “poor” quality trial, IV lysine clonixinate was superior to placebo in terms of efficacy, but was associated with a higher incidence of side effects (64).
Because the evidence for lysine clonixinate’s efficacy is weak and there are significant side effects associated with its use, we are recommending against its use for migraine.
12. Opioids: morphine
Weak recommendation, low-quality evidence
We recommend against the use of morphine for the acute treatment of migraine.
A “poor” quality trial compared IV morphine to dexamethasone and it appears as though morphine was less effective in reducing migraine pain. However, the results reported in the text contrast those in the graphical display (65).
Because of the degree of methodological shortcomings in the one included study, it is difficult to draw conclusions about morphine for this indication. With the current evidence, we feel that the risks of morphine most likely outweigh any potential benefit it may have for the acute treatment of migraine.
13. Opioids: tramadol
Weak recommendation, low-quality evidence
We recommend against the use of tramadol for the acute treatment of migraine.
A small, “poor” quality RCT failed to find a difference in efficacy between IM tramadol and diclofenac (62).
Based on the limited and low-quality evidence, we feel that the risks of tramadol most likely outweigh any potential benefits it may have for the treatment of acute migraine.
14. Opioids: meperidine
Weak recommendation, low-quality evidence
We recommend that clinicians offer meperidine to eligible patients presenting with acute migraine to the ED or similar settings. Meperidine can be used in doses of 75–100 mg IM.
A “fair” quality RCT did not find any difference between IM meperidine and ketorolac in terms of efficacy, although the incidence of side effects was higher in the meperidine group (59). IM meperidine appeared to be more efficacious than ketorolac, with a comparable incidence of side effects, in a “poor” quality trial (60). The results of these two trials comparing meperidine and ketorolac were pooled in a meta-analysis and the results are displayed in Figure 4. Finally, IM meperidine and droperidol did not differ in terms of efficacy in a small, “poor” quality RCT (57).
We found low-quality evidence that meperidine has similar efficacy to ketorolac and droperidol for the treatment of acute migraine. The balance of risks and benefits appears to be slightly in favor of meperidine’s efficacy over its side effects based on the current evidence.
15. Other analgesics: acetaminophen
Weak recommendation, moderate-quality evidence
We recommend against the use of acetaminophen for the treatment of acute migraine.
We found moderate-quality evidence to refute the use of IV acetaminophen. A “fair” quality trial found no difference between IV acetaminophen and placebo, with several minor side effects reported among the acetaminophen group (66).
We therefore feel that the side effects of acetaminophen outweigh any potential benefit it may have for the acute treatment of migraine.
16. Other analgesics: lysine acetylsalicylic acid (L-ASA)
Strong recommendation, moderate-quality evidence
We recommend that clinicians offer L-ASA, at a dose of 1–1.8 g IV, to eligible patients presenting with migraine to the ED or similar settings.
We found moderate-quality evidence that L-ASA is effective for acute migraine treatment. In a “poor” quality trial, IV L-ASA was superior to IM ergotamine in achieving rapid pain relief, but the groups did not differ in terms of efficacy by two hours. Side effects were less common in the L-ASA group (67). A “fair” quality RCT showed that IV L-ASA was superior to placebo, but inferior to SC sumatriptan in terms of efficacy. L-ASA was well tolerated in that trial, with a lower incidence of side effects than sumatriptan (34).
Lysine-ASA therefore appears to have some efficacy and a favorable side effect profile.
17. Ergot alkaloids: ergotamine
Weak recommendation, low-quality evidence
We recommend the use of ergotamine for the acute treatment of migraine in patients presenting to the ED or similar settings. Ergotamine can be given to appropriate patients at a dose of 0.5 mg SC.
We found low-quality evidence that ergotamine may be effective for this indication. A “poor” quality trial comparing SC ergotamine to IV L-ASA showed that L-ASA resulted in higher response rates at one hour, but that the groups did not differ in terms of efficacy by two hours. The ergotamine group had a higher incidence of side effects, namely related to increased nausea and vomiting (67).
The balance between the benefits and side effects of ergotamine is not as clear as would be necessary for a strong recommendation and the quality of the evidence is low.
18. Ergot alkaloids: dihydroergotamine
Weak recommendation, low-quality evidence
We recommend that clinicians offer dihydroergotamine, at a dose of 1 mg SC or IM, to patients with acute migraine presenting to the ED or similar settings.
We found low-quality evidence, from two “poor” quality RCTs, that dihydroergotamine is likely effective in acutely treating migraine. IM dihydroergotamine was more effective than diclofenac in a “poor” quality RCT, although only raw data were reported and it is unclear if the results were statistically significant (63). Another “poor” quality trial showed that SC sumatriptan yielded higher rates of pain relief than dihydroergotamine at one and two hours, but that the efficacy did not differ between the groups by three hours, and by 24 hours, recurrence rates were lower with dihydroergotamine. Incidence of side effects was higher with dihydroergotamine (42). Searches of clinical trial registries identified one unpublished RCT comparing dihydroergotamine to dexamethasone for the acute treatment of migraine in the ED (clinicaltrials.gov identifier NCT00733382).
Although the evidence seems to indicate some efficacy, because of its significant side effects, the balance of the risks and benefits with this drug is less clear.
19. Other: lidocaine
Weak recommendation, low-quality evidence
We recommend that clinicians offer intranasal lidocaine, at a dose of 40–80 mg, for the treatment of acute migraine. However, we recommend against the use of IV lidocaine for this indication.
A small, “poor” quality trial found no difference between intranasal lidocaine and placebo when each was combined with IV prochlorperazine (68). Another “poor” quality RCT showed that intranasal lidocaine was superior to placebo but was associated with more side effects, all related to the irritative properties of lidocaine (69). IV lidocaine was no different from placebo in terms of efficacy in a small but “fair” quality trial (70).
The side effect profile of intranasal lidocaine appears to be relatively benign, thus providing some evidence that the possible efficacy of intranasal lidocaine may outweigh its risks. We are recommending against the use of IV lidocaine because we found low-quality evidence, from one “fair” quality trial, that it is no different from placebo for this indication.
20. Other: propofol
Weak recommendation, low-quality evidence
We recommend against the use of propofol for the treatment of acute migraine in patients presenting to the ED or similar settings.
We found low-quality evidence that propofol is superior to dexamethasone for this indication. A “poor” quality trial found IV propofol to be superior to dexamethasone in terms of pain relief, but 44.4% of the propofol group experienced side effects, some of which were severe (e.g. slurred speech) (71).
Based on this evidence, we feel that any potential benefit of propofol is outweighed by its significant side effects.
21. Other: magnesium sulfate (MgSO4)
Weak recommendation, moderate-quality evidence
We recommend against the use of MgSO4 for the acute treatment of migraine.
We found moderate-quality evidence that MgSO4 lacks efficacy for this indication. IV MgSO4 was superior to placebo in a very small, “poor” quality trial published only as an abstract (72). A “fair” quality trial comparing IV MgSO4 to placebo found only subgroup differences in terms of the primary outcome, but it appeared as though MgSO4 was superior to placebo in several secondary efficacy outcomes (73). A “good” quality trial found that IV MgSO4 was inferior to placebo in terms of several efficacy outcomes and yielded a higher incidence of side effects, flushing being the most common (74). IV MgSO4, metoclopramide and placebo did not differ in efficacy in a “poor” quality trial, and flushing was the only side effect reported among the MgSO4 group (53).
We feel that the evidence does not support efficacy of MgSO4 for this indication. We are therefore recommending against its use for this indication, despite a relatively benign side effect profile.
22. Other: dexamethasone
Strong recommendation, moderate-quality evidence
We recommend against the use of dexamethasone for the acute treatment of migraine pain in the ED or similar settings. Note that this recommendation does not address dexamethasone’s use in preventing migraine recurrence, which has been well studied in the setting of acute migraine but is beyond the scope of this review.
We found moderate-quality evidence that dexamethasone is not effective in acutely treating migraine pain. IV dexamethasone was compared to placebo in a large, “good” quality RCT, and there was no difference between the groups in any of the efficacy outcomes nor in terms of side effects (75). A “poor” quality RCT reported superiority of IV dexamethasone over morphine, but the graphical reporting of results was discrepant and it is therefore impossible to draw conclusions from this trial (65). Another “poor” quality trial found IV propofol to be superior to dexamethasone for migraine relief (72). As noted above, one unpublished RCT comparing dexamethasone to dihydroergotamine was identified in our searches of clinical trial registries (clinicaltrials.gov identifier NCT00733382).
Although its side effect profile appears relatively benign, its lack of efficacy precludes a recommendation for its use for the acute treatment of migraine pain.
23. Other: octreotide
Weak recommendation, moderate-quality evidence
We recommend against the use of octreotide for the acute treatment of migraine.
A “fair” quality RCT showed that IV octreotide was inferior to prochlorperazine in terms of efficacy, but was better tolerated (47).
We are therefore providing a weak recommendation against the use of octreotide, given that it was relatively well tolerated and prochlorperazine is a very strong comparator.
24. Other: sodium valproate
Weak recommendation, low-quality evidence
We recommend against the use of IV sodium valproate for the acute treatment of migraine.
We found low-quality evidence, from one “poor” quality RCT, that IV sodium valproate is inferior to IV prochlorperazine for this indication (48).
The strength of the recommendation is weak because prochlorperazine is a strong active comparator and the quality of the trial was “poor.”
Conclusions
Summary
Summary of recommendations.
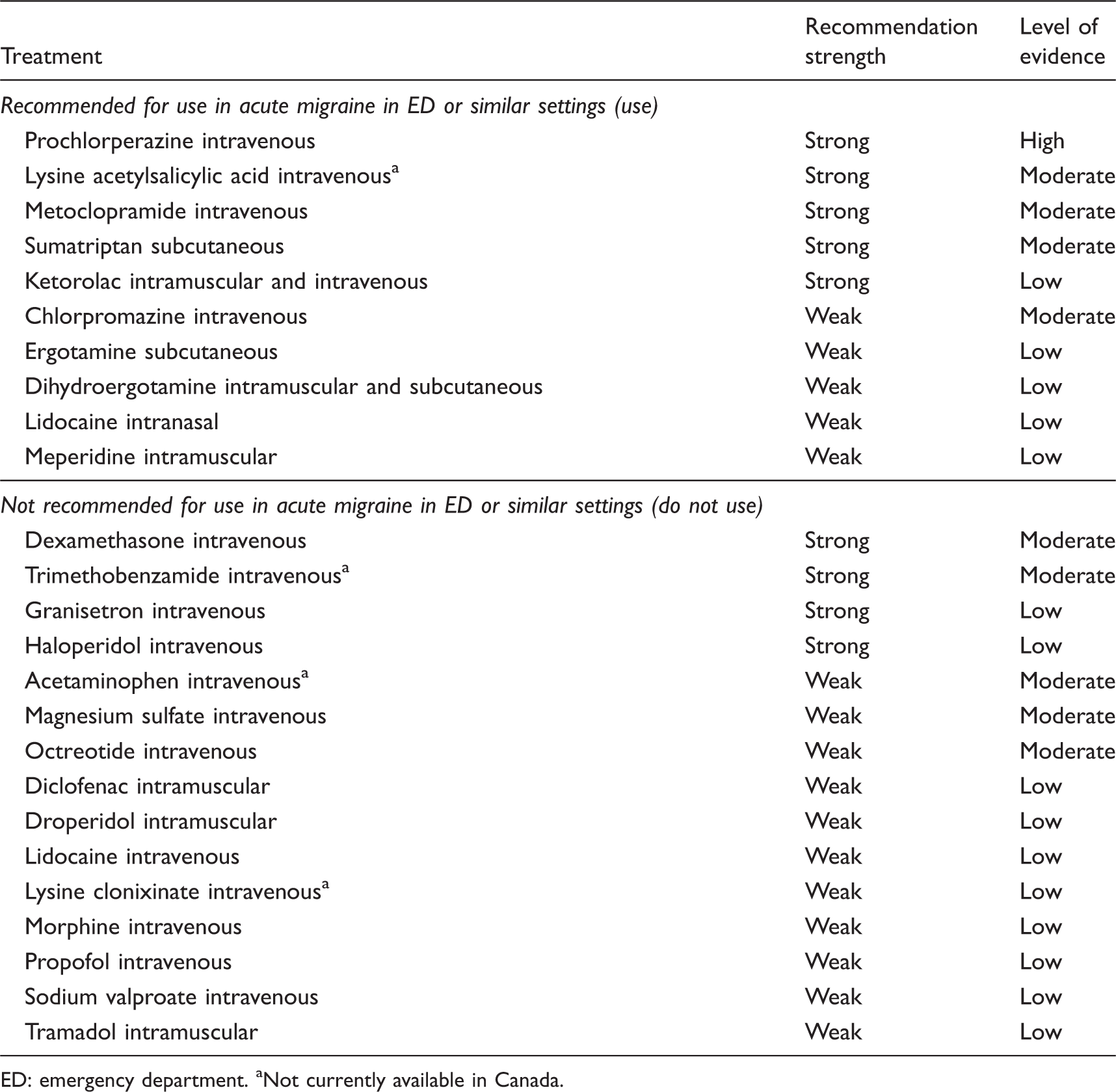
ED: emergency department. aNot currently available in Canada.
Clinical comments
The evidence reviewed is generally of low quality and lacks a sufficient amount of active comparator trials to guide clinicians in choosing between evidence-based therapies. Clinicians should consider individual patient factors and preferences in choosing therapies for the acute treatment of migraine in emergency settings.
Four therapies currently available in North America are strongly recommended for use in this systematic review: prochlorperazine, sumatriptan, metoclopramide and ketorolac. When deciding to prescribe prochlorperazine for acute migraine treatment, clinicians can consider concurrently administering diphenhydramine in order to prevent extrapyramidal side effects. In addition, prochlorperazine should not be used in patients with other risk factors for extrapyramidal symptoms. Sumatriptan is likely to be more effective in patients presenting within the first few hours of their migraine (76), and should not be used first line if patients have already taken a triptan in the past 24 hours. In clinical practice, metoclopramide and ketorolac are often administered together. As with prochlorperazine, metoclopramide should be avoided in patients with other risk factors for extrapyramidal symptoms. Although dihydroergotamine received only a weak recommendation for use given low-quality evidence and a significant side effect profile, it is commonly used in practice by headache experts, and is a reasonable option to use first line in the appropriate patient. If used, dihydroergotamine should be coadministered with an antiemetic. It should be avoided in patients with vascular disease, given its significant vasoconstrictor effect (77).
In the acute treatment of migraine in emergency settings, clinicians should not only administer medications but also consider hydration with IV fluids. Migraine patients often present with photophobia and phonophobia, so efforts should be made to provide them with a dark and quiet place to rest. Patients with nausea should be treated with an antiemetic agent. These ancillary measures are essential components of the acute treatment of migraine, and should be considered for every patient.
Ultimately, reducing the number of emergency visits for migraine is the goal. There is ample evidence to suggest that migraine prophylaxis and prescription of migraine-specific rescue therapies reduces resource use in general, and specifically ED visits (78–80). Therefore, clinicians caring for patients with migraine on an outpatient basis should endeavor to optimize prophylactic and rescue therapy, so as to prevent emergency visits in the first place.
Limitations
We chose to include only studies that used International Headache Society criteria for migraine, or, for older studies, the Ad Hoc criteria. This resulted in a significant number of otherwise eligible studies being excluded based on how they selected participants. Although this may be considered a limitation of our systematic review, we felt it necessary to exclude studies that did not use validated criteria for migraine, because of the risk of including studies with a heterogeneous population composed of patients with other headache types.
Another potential limitation of our systematic review relates to our inability to locate three publications that were identified in the search. This introduces a potential for selection bias.
Future research
We believe that our systematic review has uncovered many areas of potential future research because, in general, this body of evidence is of low quality. Future studies should address the use of interventions that, based on the current literature, have low-quality evidence and relatively favorable side effect profiles. In addition, the literature is lacking evidence regarding the use of combination therapies. Furthermore, although some studies addressed the use of repeated dosing of the intervention in question, overall, there is a lack of evidence for how to treat patients who are refractory to first-line medications. Finally, the literature does not address the question of whether subgroups of migraine patients may respond differentially to the interventions. For example, it is unclear as to whether patients presenting with a longer duration of migraine, or different intensity (i.e. moderate or severe), will respond to the interventions in the same manner as those presenting earlier. This is especially relevant with the case of sumatriptan, given that there is evidence that the triptans are most effective if administered early during the migraine attack (76). Also, it is unclear as to whether migraine without aura and migraine with aura patients respond differently to the interventions prescribed in the ED or similar settings.
Clinical implications
A wide variety of therapies have been studied for the treatment of acute migraine pain in emergency settings. Overall, the quality of evidence is low. Five therapies received strong recommendations based on the results of this systematic review, with varying levels of evidence quality: prochlorperazine, lysine-acetylsalicylic acid, metoclopramide, sumatriptan and ketorolac. There is an important need for further research in this area in order to guide evidence-based clinical decision making for patients presenting to emergency settings with migraine.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
Serena Orr has nothing to declare.
Werner Becker has served on medical advisory boards for Merck, Allergan, Pfizer, Tribute, ElectroCore, Amgen, AGA Medical, and St Jude. He has received speaker honoraria from Teva, Serono, Merck, Allergan, Tribute, and Pfizer, and research support in multicenter clinical trials from Allergan, Medtronic, AGA Medical, and Merck.
Michel Aubé and Jeptha Davenport have nothing to declare.
Esma Dilli has received a speaker honorarium from Allergan, UCB, Merck, Tribute and Johnson and Johnson. She has served on medical advisory boards for Allergan and Tribute.
David Dodick is the editor-in-chief of Cephalalgia. After submission of the manuscript, an acting editor-in-chief is selected by the managing editor of the journal and at no time during the peer-review or editorial process is Dr Dodick involved in any editorial decision regarding this manuscript. Within the past 24 months, Dr Dodick has provided consulting services for Allergan, Amgen, Alder, Arteaus, Pfizer, Merck, ENeura, NuPathe, Eli Lilly & Company, Autonomic Technologies, WL Gore, Ethicon J&J, Zogenix, Supernus, Labrys, and Bristol Myers Squibb. Dr Dodick has received funding for travel, speaking, or editorial activities from the following: CogniMed, Scientiae, IntraMed, SAGE Publications, Sun Pharma, Allergan, Lippincott Williams & Wilkins, Oxford University Press, Cambridge University Press, Miller Medical, and Annenberg Center for Health Sciences. He received publishing royalties for Wolff’s Headache and Other Head Pain, 8th edition (Oxford University Press, 2009) and Handbook of Headache (Cambridge University Press, 2010).
Rose Giammarco sits on the advisory board for Allergan and has previously been on the advisory board for Pfizer and Johnson & Johnson. She has received support for clinical trials and honoraria from Allergan.
Jonathan Gladstone has served on advisory boards for Allergan, ElectroCore, Johnson & Johnson, Merck, Pfizer, and Tribute. He has received honoraria via unrestricted educational grants from Allergan, Johnson & Johnson, Merck, and Pfizer for developing and presenting Headache Continuing Medical Education (CME).
Elizabeth Leroux has served on medical advisory boards for Merck, Allergan, Pfizer, Tribute, and ElectroCore. She has received speaker honoraria from Teva, Merck, Allergan, Tribute, Purdue, Johnson & Johnson, and Pfizer, and research support for an investigator-initiated clinical trial from Allergan.
Heather Pim and Garth Dickinson have nothing to declare.
Suzanne N. Christie has received grants and research support from Biogen-Idec and Novartis. She has also received speaker honoraria from Allergan, Johnson & Johnson, Tribute, and Teva Neuroscience. She has received consulting fees from Allergan, Tribute, and Biogen-Idec.
Acknowledgments
We would like to thank Margaret Sampson, MLIS, PhD, AHIP, for developing the electronic search strategies, and Janey Joyce, MLS, for peer reviewing the MEDLINE search. We would also like to thank Peter Lugomirski, MD, and Mindy Rush, MD, for their assistance with translation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
