Abstract
Introduction
Occipital nerve stimulation (ONS) may provide pain relief in migraine patients. In this double-blinded trial we investigated the significance of paresthesia and possible placebo effects.
Methods
Patients already treated with ONS reporting stable treatment effect were included. “Effective stimulation,” “subthreshold stimulation” and “no stimulation” were compared. Patients cycled through all three treatment groups. Outcome was measured using the visual analog scale (VAS) for pain, McGill Pain Questionnaire and SF-36.
Results
Eight patients were included, mean preoperative VAS was 8.20 ± 1.22. A significant improvement in pain was observed in favor of suprathreshold stimulation compared to subthreshold stimulation (1.98 ± 1.56 vs 5.65 ± 2.11). Pain also significantly improved under subthreshold stimulation compared to no stimulation (5.65 ± 2.11 vs 8.45 ± 0.99). No changes in SF-36 were observed.
Conclusions
Paresthesia is not required to achieve pain reduction but suprathreshold stimulation yields better results, underlining the significance of stimulation parameter customization.
Introduction
Neurostimulation modulating functional pathways involved in primary headache pathogenesis represents a rather new therapeutic approach for primary headache disorders.
As in other headache disorders, the main rationale for occipital nerve stimulation (ONS) in migraine is the anatomo-functional convergence of cervical (especially C2), somatic trigeminal, and dural trigeminovascular afferent neurons on second-order nociceptors in the trigeminocervical complex (1). But the lack of standard surgical technique, standard stimulation parameters and accepted guidelines for patient selection impedes comparison between studies, resulting in many unsolved questions (2–8).
The potential bias in patient self-assessment due to paresthesia generally experienced during supra-threshold stimulation complicates evaluating treatment efficacy. The presence of paresthesia implies that placebo effect cannot be eliminated, making blinded studies complex to design, and whether paresthesia is required for efficient treatment is still under discussion. No double-blind studies addressing these issues have been published to date.
Led by our clinical experience, we hypothesized that subthreshold stimulation would be less effective than suprathreshold stimulation. Secondly, we hypothesized that in the no stimulation group, no treatment effect would be seen proving treatment effect at three months.
Methods
Study design
Migraine patients treated with ONS were asked to participate in the study. To reduce potential effects of impedance changes, only patients reporting stable and significant (> 30%) pain relief for at least three months postoperatively were included. Further inclusion criteria: age 18 years or older, stable medication, and written informed consent. Exclusion criteria: alternative therapy during course of the study and other medical disorders that would confound the study.
The study includes three treatment groups: “effective stimulation” (Group 1), “subthreshold stimulation” (Group 2) with the amplitude set just below perception level, and “no stimulation” (Group 3) with the implantable pulse generator (IPG) set to the lowest possible amplitude.
Written informed consent was obtained from each patient. Ethics approval for the study was obtained from the local board of the Heinrich-Heine University of Düsseldorf, Germany.
Randomization and course of the study
Randomization was performed using Matlab’s “rand” algorithm. A fixed randomization table was generated before the start of the study.
Each patient cycled through all three groups in random order. Each stimulation design was tested for seven days. Patient and physician examiner were blinded to the results of randomization and therefore treatment allocation. Randomization, as well as programming of the required stimulation settings, was performed by a study nurse. At the end of the study, the patient’s original primary stimulation parameters were reprogrammed.
Stimulation parameters
To preserve blinding, the amplitude was successively decreased and then set either to subthreshold or no stimulation amperage. This allows blinding of the patients in Group 2 and 3 only; no blinding can be achieved in Group 1. For technical reasons inherent to the neurostimulation system, the IPG had to be set to the minimal output possible of 0.1 mA in the no stimulation group (Group 3).
Outcome assessment
Outcome was assessed on the last day of each cycle by the treating physician including the visual analog scale (VAS), the McGill Pain Questionnaire (German version (9), MPQ) and the Short-Form-36 (SF-36) questionnaire (RAND Cooperation, German version, v1). Primary outcome was reduction in pain; secondary outcome measure was increased quality of life. Statistics were performed using IBM SPSS Statistics 19 (IBM Corp. Int.) and SigmaPlot 12 (Systat Software, CA, USA). The two-tailored level of statistical significance stipulated was 0.05 and 0.01, respectively. Since the data did not show a Gaussian distribution, the Friedman test was performed for screening and the Wilcoxon rank sum test was performed for paired treatment group comparison.
Results
Patient population
Fifteen patients being treated with ONS for migraine in 2012 were assessed for eligibility, and seven patients were excluded (five for insufficient/inconsistent treatment effect, one declined to participate, one with recent changes in medication).
All eight patients finally included received bilateral ONS lead implantation (Octrode™, St Jude Medical (SJM), St. Paul, MN, USA), and all patients fulfilled the International Headache Society (IHS) criteria for chronic migraine. Mean preoperative VAS score collected in an outpatient setting prior to surgery (±SD) was 8.20 ± 1.22. This declined to 1.35 ± 0.84 under ONS with optimal stimulation parameters prior to the start of the study (mean 119 ± 25 days following implantation). Mean pre-study MPQ values were 6.21 ± 3.52. These values were taken as the baseline pain level and the baseline treatment effect, respectively.
Study results
Cephalgia improved substantially with effective stimulation of the occipital nerves in all patients. Statistical screening resulted in highly significant differences between groups for VAS and MPQ (p < 0.0001 both) and significant differences in SF-36 (p = 0.012). In intergroup comparison, the mean preoperative VAS score (±SD) was 8.20 ± 1.22 and improved highly significantly with pre-study stimulation to 1.35 ± 0.84 (p < 0.0001). Pain also highly significantly improved with subthreshold stimulation (Group 2) compared with no stimulation (Group 3), with a VAS score (±SD) of 5.65 ± 2.11 and 8.45 ± 0.99, respectively (p = 0.0031). In addition, a highly significant improvement in pain was found in favor of effective stimulation (Group 1) when comparing this group with subthreshold stimulation (Group 2), suggesting that suprathreshold stimulation is more effective at controlling pain (p = 0.0003, Figure 1). No significant difference was observed between pre-study and Group 1 pain levels (p = 0.091).
Pain severity measured on the visual analog scale (VAS) between the three treatment groups and the pre-study optimal stimulation parameters (*** = highly significant, ns = not significant, whiskers represent minimum/maximum).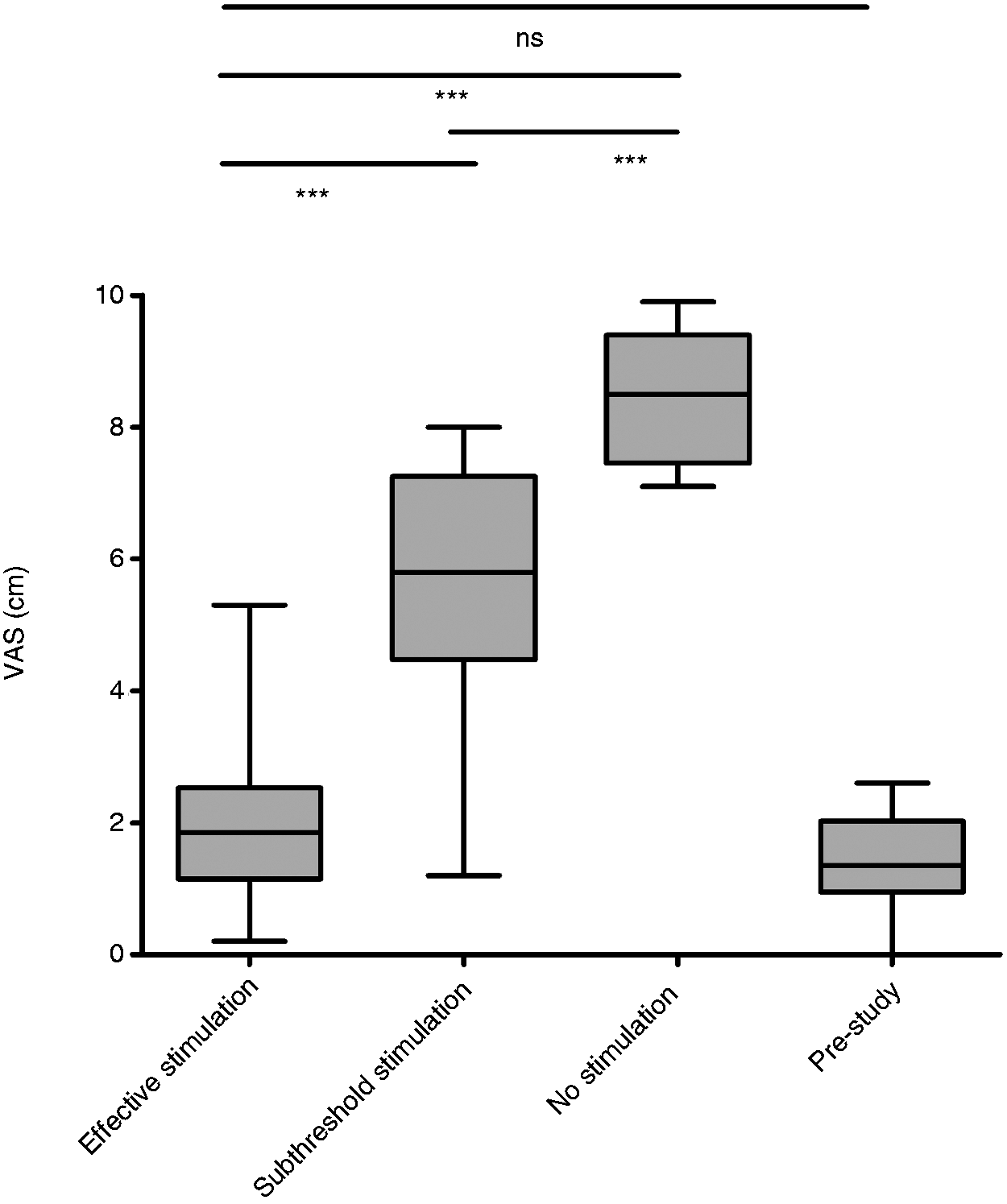
Similar results were seen in pain assessment using the MPQ. Median MPQ score (±SD) was 8.37 ± 3.70 in Group 1 versus 17.38 ± 3.42 in Group 2, and 26.04 ± 4.66 in Group 3 (no stimulation), resulting in highly significant differences between all groups (p = 0.0004 and p = 0.0011, respectively, Figure 2).
Pain severity measured by the McGill Pain Questionnaire between the treatment of the three groups and the pre-study best stimulation setting (*** = highly significant, ns = not significant, whiskers represent minimum/maximum).
Summary of data and statistical results.

VAS: visual analog scale; MPQ: McGill Pain Questionnaire; SF-36: Short-Form-36; SD: standard deviation; CI: confidence interval.
Mean pulsewidth was 335.8 µs (100-450, SD 120.7), and mean frequency was 37.9 Hz (30–80, SD 16.2). Pulsewidth, frequency and contact configuration remained unchanged during the study; mean voltage applied was 2.6 V (0.4–6.7, SD 1.8) in Group 1 and 1.3 V (0.3–3.1, SD 0.8) in Group 2 (p = 0.093, n.s.).
There were no technical malfunctions or complications.
Discussion
Two key findings are presented in this study: 1) ONS produces reproducible and reversible pain relief in patients suffering from chronic migraine and 2) paresthesia is not mandatory for effective treatment.
ONS is effective both in supra- and subthreshold stimulation, whereas higher-amplitude stimulation (which elicits paresthesia) results in better pain control. This emphasizes the significance of meticulous IPG programming and examination of side effects such as paresthesia in ONS patients. In previous studies on ONS efficacy (Precision Implantable Stimulator for Migraine (PRISM), OnStim and the recent publication by Silberstein (8)) the primary endpoint (reduction in VAS) was not reached. But reduction in pain might not be the best outcome parameter, at least not in all chronic headache disorders. In cluster headache, reduction in headache frequency is more appropriate. The short follow-up following implantation commonly reported might add to the problem, as in our experience treatment effect of ONS can be validated only following a “running-in time” unique to this stimulation technique.
It is still under vigorous debate as to whether the mechanism of action in ONS is based on peripheral nerve field stimulation or on retrograde activation of the pain matrix. Functional imaging studies indicate that central processing is involved with the target neurons located in the trigeminocervical complex, the thalamus, as well as the pain matrix (10–12).
The persistence of treatment effect in the absence of paresthesia found in our study supports the assumption of central pain perception modulation by ONS. Similar dose-related effects of neurostimulation have been shown previously in vagal nerve stimulation studies (13). This is additionally supported by positron-emission tomography (PET) studies on stimulation amplitude and changes in regional cerebral activity (12). Similar studies on ONS parameter settings and correlation with functional and metabolic imaging are planned (personal communication, 2012).
These studies might elucidate the quick loss and recovery of ONS efficacy observed in our study if changes are applied to a running system.
We observed no changes in quality of life measured by SF-36, most likely because the questionnaire encompasses a number of items that are unlikely to change during a one-week period.
One technical limitation was encountered during the study: Inherent to the design of the IPG used, stimulation could not be completely turned off to achieve the no stimulation condition (Group 3). The stimulation amplitude could be set only to the minimal output of 0.1 mA. However, this amplitude is not likely to induce any effect. Since the mean pain level in this group was close to the level prior to ONS surgery (8.45 vs. 8.22), we therefore assume that Group 3 resembles the real no stimulation condition.
The second limitation to this study is the missing blinding of the patients in Group 1 due to paresthesia. We believe the results, especially the good pain relief in Group 2 compared to Group 3, indicate the successful blinding in Groups 2 and 3 and therefore deliver authentic results.
A third limitation is the small number of patients included and missing washout periods between the different stimulation settings. The possibility of prolonged effects exceeding active stimulation must be taken into account, although the statistical effects are moderated by randomization.
Alongside clinical diagnostics and patient history, the indication for ONS surgery should be carefully assessed by an interdisciplinary team, including headache specialists and functional neurosurgeons as proposed by National Institute for Health Care Excellence (NICE, NICE interventional procedure guidance 452).
Today, ONS is considered an innovative and promising treatment for different primary headache disorders, although many aspects remain unclear. Additional positive preliminary results have been reported for the management of occipital neuralgia, hemicrania continua and facial pain.
This study was not designed to provide evidence of overall efficacy of ONS, but to exclude a potential placebo effect of treatment as far as possible and elucidate the role of paresthesia. Only patients reporting a stable treatment effect were included, but the results are encouraging since the reproducibility of treatment effect could be proven. The second-tier role of paresthesia in treatment efficacy supports the assumption of central pain modulation by ONS.
Conclusions
ONS delivers consistent and reproducible results in the treatment of distinct medically intractable migraine. Paresthesia is not required to achieve pain reduction, but suprathreshold stimulation yields better results, highlighting the significance of stimulation parameter customization. The positive effects of ONS in this cohort were shown to be mainly stimulation dependent, thereby excluding a significant placebo effect. Further investigations are required regarding long-term outcome, optimization of the surgical technique, and especially patient selection. A specialized interdisciplinary team is mandatory to successfully establish this promising technique. Refinement of stimulation equipment and the development of outcome predictors will most likely further improve clinical results.
Clinical implications
Occipital nerve stimulation (ONS) delivers consistent and reproducible results in the treatment of distinct medically intractable migraine. Paresthesia is not required to achieve pain reduction, but suprathreshold stimulation yields better pain control. The second-tier role of paresthesia in treatment efficacy supports the assumption of central pain modulation by ONS.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
JV and PJS are consultants for SJM, receiving payment for preparing and giving educational presentations, as well as reimbursement for travel expenses. GB has received a fellowship grant from SJM. CW and SS are consultants for SJM and Spinal Modulation Inc, receiving payment for preparing and giving educational presentations, as well as reimbursement for travel expenses. AG is a consultant for Allergan, MSD, Bayer, Teva, Pfizer, Weber und Weber, Mundipharma and Gruenenthal. LK has nothing to declare.
