Abstract
Background
Prior studies demonstrate reduced cortical thickness and volume in migraineurs. However, the effect of age on cortical thickness has not been assessed in migraineurs. In this study we investigated whether the process of aging on cortical thickness affects migraineurs differently compared to age-matched healthy controls, i.e. whether aging exacerbates cortical thinning in migraineurs.
Methods
Cortical thickness was estimated using a general linear model vertex-by-vertex approach for 32 healthy controls (mean age = 35.3 years; SD = 11.6) and 27 episodic migraine patients (mean age = 33.6 years; SD = 12.3). Results were modeled using a main effect analysis to estimate the effect of age on cortical thickness for each group separately, and an age-by-group analysis to estimate differences in age-related cortical thinning between migraine patients and normal controls.
Results
Although migraineurs and normal controls both have expected age-related thinning in many regions along the cortical mantle, migraineurs have age-related thinning of regions that do not thin in healthy controls, including: bilateral postcentral, right fusiform, and right temporal pole areas. Cortical thinning of these regions is more prominent with advancing age.
Conclusion
Results suggest that migraine is associated with atypical cortical aging, suggesting that the migraine disease process interacts with aging to affect cortical integrity.
Introduction
Migraine is a debilitating disorder that consists of recurrent headache, hypersensitivities to lights, sounds and odors, nausea and vomiting. Approximately 12% of people experience migraine each year in North America, with women being three times more likely to develop migraine compared to men (1–5). Migraine usually begins during the late childhood years to early twenties and typically persists for at least several decades (5). Although the clinical manifestations of migraine are well described, its pathophysiology remains poorly understood. However, a growing number of migraine imaging studies demonstrate atypical structure and function of a complex network of pain processing areas, commonly termed the “pain matrix,” including sensory-discriminative, cognitive, and affective regions of pain processing (6–10).
Anatomical studies using voxel-based morphometry or vertex-wise analysis methods have reported a reduction of cortical volume and thickness for migraine subjects in areas involved in pain processing (8,11,12), and several studies have hypothesized that recurrent activation of areas associated with pain would eventually lead to this cortical decline. As such, the argument has been made that migraine could be a progressive disorder, i.e. that repeated migraine attacks over an individual’s lifetime could induce a negative and compounding effect on gray matter (GM) health (10). Although several studies have documented a correlation between cortical atrophy and migraine frequency and duration (10,13,14), more recently published studies have found the opposite, i.e. the absence of an interaction between migraine frequency and brain structure decline (8,15,16). Another prominent factor that negatively affects cortical integrity is age. With advancing age, cortical thickness decreases (17–19). Several studies have assessed the effect of specific disease processes on the aging brain (20–25) and reported accelerated or atypical age-related decline (atrophy) in pain disorders (22,23,25), autism (24), schizophrenia (21), and Alzheimer’s disease (20) relative to healthy, normally aging controls. As such, it is possible that the aging brain is more vulnerable to disease states than the younger brain (26). However, the effect of age on regional thickness patterns has not been directly assessed in migraine.
Here, we examined the effects of aging in migraine patients and control subjects and explore the age-by-group interactions using a vertex-wise general linear model (GLM) to estimate cortical thickness along the entire cortical mantle. We hypothesized that the migraine disease process interacts with brain aging, resulting in abnormal cortical aging patterns of the adult migraine brain.
Methods
Subjects
This study evaluated cortical thickness in 32 healthy control subjects (25 females, seven males; mean age 35.3 years; age range 20–60 years; SD 11.6) and 27 episodic migraine subjects (22 females, five males; mean age 33.6 years; age range 18–64 years; SD 12.3). Although 32 episodic migraine subjects were originally evaluated, four subjects had to be excluded because they were unable to tolerate the magnetic resonance (MR) scanning session and a single subject had to be excluded because of a brain abnormality identified on T1 imaging, thus leaving 27 episodic migraineurs in the final analysis.
All subjects had given informed written consent prior to participation in accordance with the institutional review board guidelines of Washington University Medical Center. All migraine subjects met International Classification of Headache Disorders II (ICHD-II) diagnostic criteria for episodic migraine (27). Migraine subjects were not taking migraine prophylactic medications and were not overusing migraine abortive medications according to ICHD-II criteria. All migraine subjects reported having migraine symptoms for at least three years. Nine of the 27 migraine patients reported visual auras, while the rest reported no migraine-related auras. Magnetic resonance imaging (MRI) was conducted when migraine patients were headache free for at least 48 hours. Control subjects were healthy community-dwelling volunteers without history of migraine or headache disorders other than infrequent tension-type headache (<3 headache days/month). Twenty-four control subjects reported at least one tension-type headache a month, and eight subjects denied experiencing tension-type headaches. Control subjects were similar in age (each migraine subject was within four years of age relative to a healthy control) with proportionate male/female ratios. All participants completed the Beck Depression Inventory (BDI-II) and the state/trait anxiety inventory (STAI, Form Y-1 and Form Y-2) to assess symptoms of depression and anxiety (28,29). Subjects were screened for history of head trauma, psychiatric disorders, chronic or acute pain disorders (other than migraine for patients) and on occurrence excluded from this study. All subjects had normal T1 and T2 scans, including the absence of white matter (WM) T2 hyperintensities.
MRI acquisition parameters
All images were obtained on a Siemens MAGNETOM Trio 3 T scanner (Erlangen, Germany) with total imaging matrix (TIM) technology using a 12-channel head matrix coil and Food and Drug Administration (FDA)-approved sequences. Structural scans included a high-resolution three-dimensional (3D) T1-weighted sagittal magnetization-prepared rapid gradient echo (MP-RAGE) series (echo time (TE) = 3.16 ms, repetition time (TR) = 2.4 s, inversion time (TI) = 1000 ms, flip angle = 8 degrees, 176 continuous slices with 1 × 1 × 1 mm voxels, 256-mm field of view (FOV), acquisition matrix 256 × 256) and T2-weighted images in an axial plane TE = 88 ms, TR = 6280 ms, flip angle = 120 degrees, 1 × 1 × 4 mm voxels, 256-mm FOV, 36 slices, acquisition matrix 256 × 256.
Cortical reconstruction and segmentation
T1 MP-RAGE sequence image processing was performed using the FreeSurfer image analysis suite (version 5.3), which is documented and freely available for download online (http://surfer.nmr.mgh.harvard.edu/). To avoid postprocessing irregularities between workstations, all image postprocessing was conducted using a single Mac workstation running OS X Lion 10.7.5 software. The methodology for this procedure is described in detail in prior papers (30–41). In short, processing includes skull stripping (41), automated Talairach transformation, segmentation of subcortical WM and GM structures (34,38), intensity normalization (42), tessellation of brain boundaries, automated topology correction (41), and surface deformation (30–32).
For statistical interpretation, we used surface-based group analysis tools within FreeSurfer (QDEC) to investigate cortical thickness differences between migraine and control subjects. Cortical thickness was interpreted as a distance measured from the boundary of the GM and WM to the boundary of the GM/cerebral spinal fluid at each vertex along the brain surface (32). Cortical thickness maps were smoothed using a smoothing kernel of 15 mm full width at half maximum (FWHM).
FreeSurfer output of cortical thickness was manually inspected to ensure that GM and WM boundaries were correctly delineated before subject data were included for further analysis. This is a time-consuming but important procedure that requires up to one hour per subject, but it validates that none of the subjects included in this study had significant errors in estimating cortical thickness throughout the brain.
Statistical analysis
Demographic group characteristics and estimates for total intracranial volume (ICV), and total GM and WM volume were assessed using independent sample t tests (two tailed). Gender differences were assessed using a chi-square test. First, for migraine subjects and healthy controls, regional cortical thickness-to-age interactions were analyzed within FreeSurfer (QDEC) using a GLM whole-brain vertex-wise analysis. This was conducted for each group and each hemisphere separately. Statistical significance levels were set at p < 0.01, uncorrected. Next, an age-by-group interaction analysis was conducted in order to estimate whether cortical thinning was more strongly related to age for one group versus another. In order to ascertain that depression (BDI-II), anxiety (STAI), and migraine burden (calculated as years with migraine × headache frequency) were not contributors to group differences they were entered as covariates and statistically controlled for within the GLM model. To control for multiple comparisons, statistical significance levels were cluster-corrected for both hemispheres using a Monte Carlo simulation (43) of p < 0.025. Lastly, to visually graph regional differences in the age-related thinning slopes of migraine subjects and healthy controls, mean cortical thickness of selected regions surviving the p < 0.025 cluster correction was estimated. These regions were exported into SPSS 21.0 (SPSS Inc, Chicago, IL) for further analysis. To test whether the mean thickness-to-age correlation slopes between both groups differed significantly from each other, we used a Web utility (http://quantpsy.org) that converts the correlation coefficients into a z score using Fisher’s r-to-z transformation (44) and a formula described by Cohen and Cohen (45) (formula 2.8.5, page 54) that estimates significance levels of correlation coefficients taking into account the sample sizes of both subject groups.
Results
Subject demographics and clinical characteristics of migraineurs and healthy controls.

Migraine onset refers to the age (in years) when migraine symptoms first started. Disease duration was calculated as the number of years lived with migraine. Headache frequency is indicated in days per month. f: female; m: male; BDI: Beck Depression Inventory; SD: standard deviation; n/a: not applicable.
Additionally, in order to estimate overall cortical thickness differences between groups, a GLM analysis was conducted using age as a covariate of no interest. Following Monte Carlo cluster correction for both hemispheres (p < 0.025), results indicated no surviving clusters in the left or right hemisphere, thus indicating no significant overall cortical thickness group differences.
Main effect analysis of age-related thinning
First, we computed the effect of age on cortical thickness for each subject group separately (Figure 1). Healthy controls and episodic migraineurs had widespread bilateral cortical thinning with advancing age over frontal and temporal regions. There were no significant areas of age-related thickening in the migraine group. In the control group, a small cluster of left lateral occipital thickening was found.
Age-related thinning and thickening patterns shown for healthy controls and migraine subjects for the right (a) and left (b) hemisphere separately. The color blue indicates age-related thinning and red indicates age-related thickening along the cortical mantle. In order to visually demonstrate the widespread age-related changes, significance thresholds were set at p < 0.01, uncorrected.
Age-by-group interaction
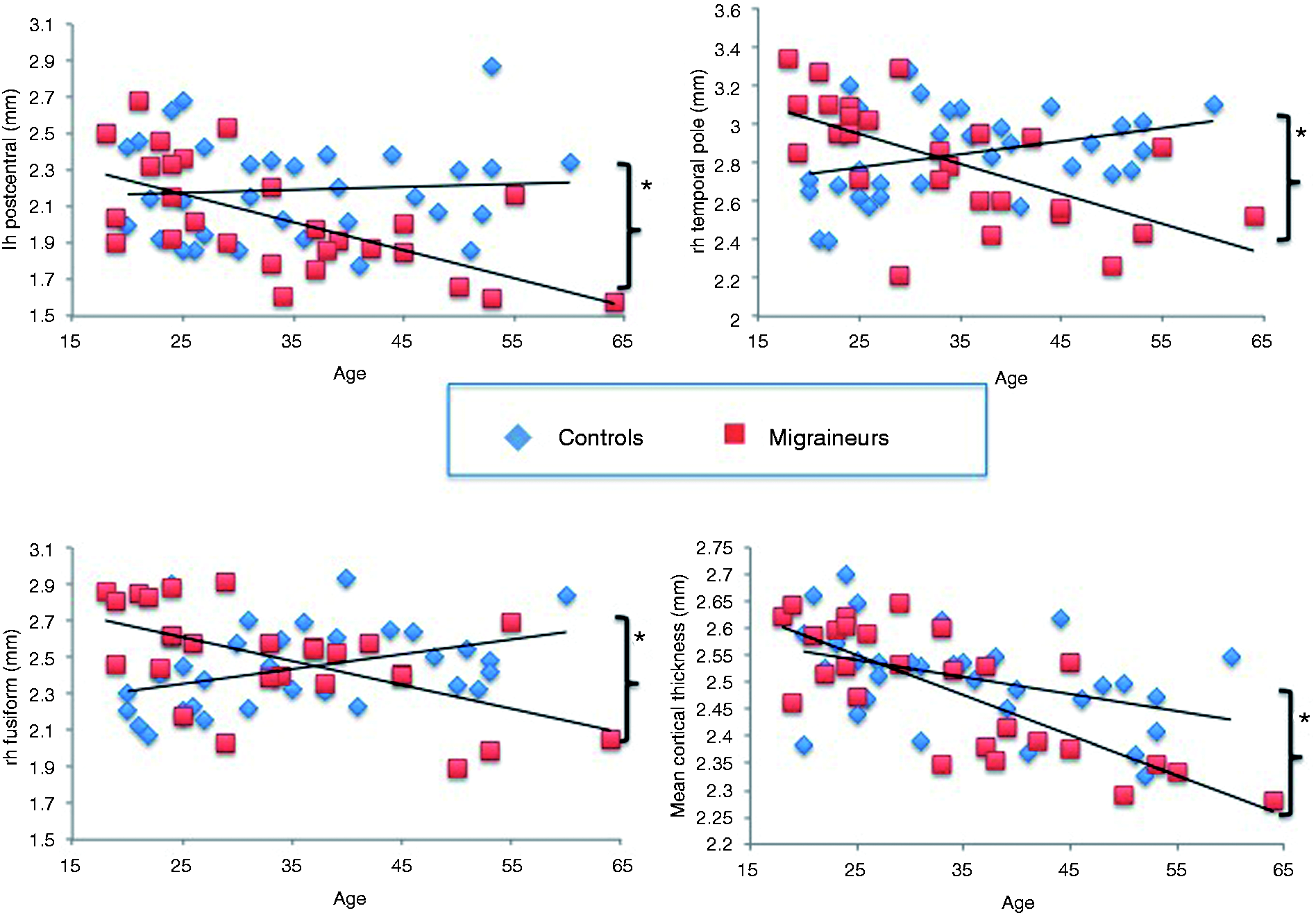
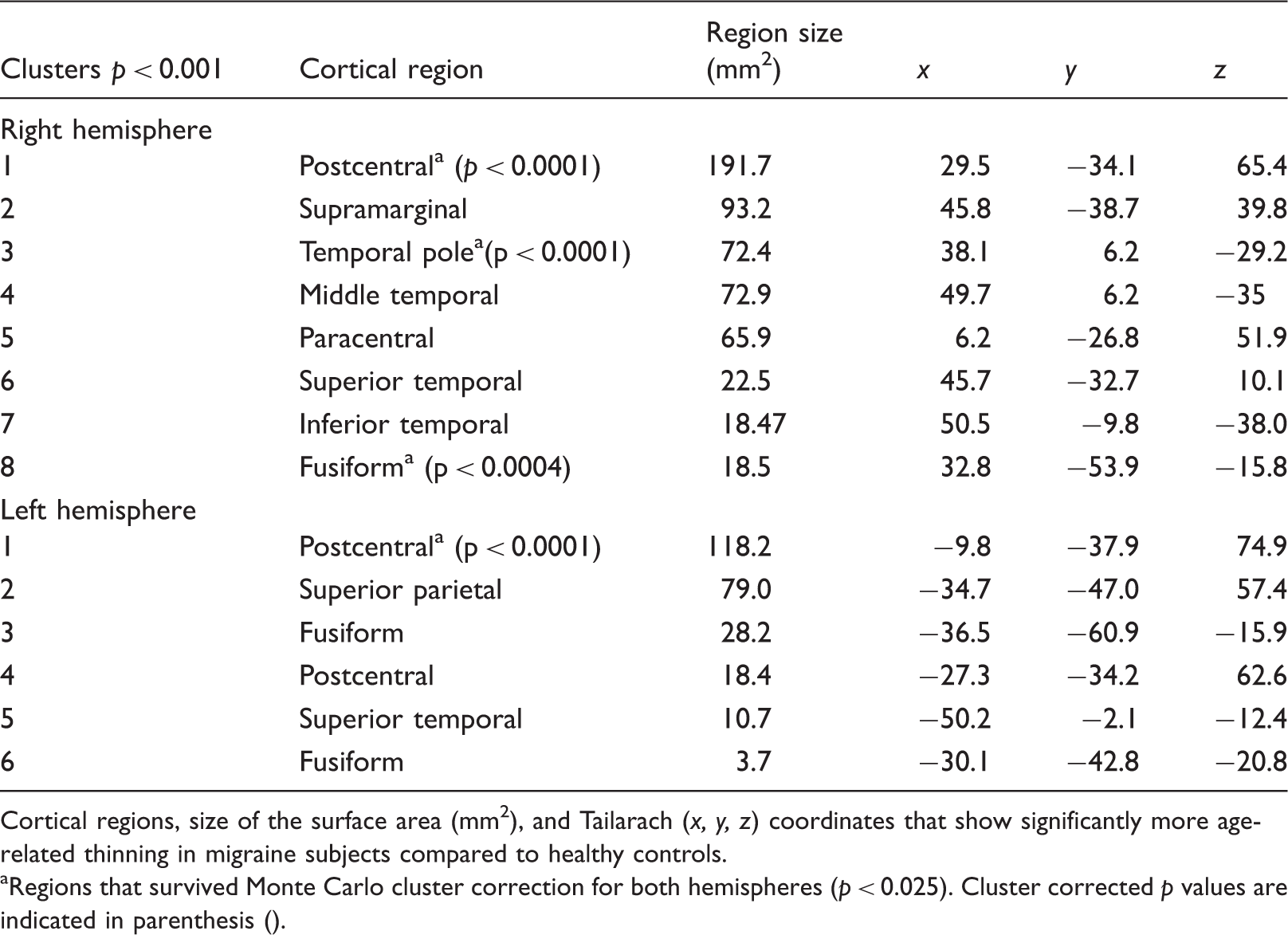
There were significant group differences in the age-by-cortical thickness correlations (Figure 2). The episodic migraine group had progressive age-related cortical thinning compared to control subjects in a variety of bilateral regions, although slightly more pronounced in the right hemisphere. Cluster-corrected regions of age-related thinning included those within the bilateral postcentral, right fusiform and right temporal pole. See Table 2 for a complete list of these cortical regions. Slopes of cortical thinning according to age were calculated for mean cortical thickness for selected regions surviving cluster correction. Regions surviving cluster correction showed significant differences of cortical thinning in migraine patients and healthy controls. Scatterplots in Figure 3 demonstrate the age-group interaction of mean cortical thickness (calculated across both hemispheres), right fusiform, left postcentral, and right temporal pole cortical thickness with age. Migraineurs show significant age-related thinning in the left postcentral (r = −0.624; p = 0.001), right temporal pole (r = −0.631; p < 0.0001), and right fusiform (r = −0.569, p = 0.002) region. In contrast, healthy control subjects indicate no significant thinning in the left postcentral area (r = 0.070; p = 0.702), mild, but nonsignificant thickening in the right temporal pole (r = 0.348; p = 0.051), and significant thickening by age in the right fusiform (r = 0.410, p = 0.020). Aging slopes for the left postcentral (p = 0.0037), right temporal pole (p < 0.0001), and the right fusiform (p < 0.0001) are significantly different between migraineurs and healthy controls. Both groups indicate significant age-related mean cortical thinning across both hemispheres (migraineurs: r = −0.785, p < 0.0001; healthy controls: r = −0.411, p = 0.019). Aging slopes are significantly different between groups (p = 0.024), with migraineurs showing steeper overall cortical thinning compared to healthy controls. Overall, our results indicate that migraine patients have steeper slopes of cortical thinning with advancing age whereas no cortical thinning was apparent in the healthy control group for left postcentral, right temporal pole, and right fusiform regions. Interestingly, although during young adulthood cortical thickness was similar between migraine and healthy control groups, with migraineurs showing perhaps slightly increased cortical thickness, this pattern reverses past the third decade of life, when migraineurs demonstrate accelerated cortical thinning relative to healthy controls.
Age-by-group interaction in migraine subjects and healthy controls for the right and left hemisphere separately. For illustration, significance thresholds were set at p < 0.01, uncorrected. Light blue areas encircled in red survive cluster correction (p < 0.025). The color blue indicates regions where migraine subjects show more age-related cortical decline compared to healthy controls. The color red indicates the opposite. One (a) and (b) show the inflated maps of the lateral brain surface, 2) dorsal view, and 3) ventral view of the left and right hemisphere. For healthy controls and migraine patients, mean cortical thickness was calculated for regions surviving cluster correction (p < 0.025). rPC: right postcentral; lPC: left postcentral; rFS: right fusiform; rTP: right temporal pole. Scatterplots indicating differences in age-related slopes for migraine subjects (red) and healthy controls (blue). *Indicates significant differences in the correlation slopes between healthy controls and migraine patients. Right and left hemisphere clusters are reported separately. Cortical regions, size of the surface area (mm2), and Tailarach (x, y, z) coordinates that show significantly more age-related thinning in migraine subjects compared to healthy controls. aRegions that survived Monte Carlo cluster correction for both hemispheres (p < 0.025). Cluster corrected p values are indicated in parenthesis ().
Discussion
The main finding of this study is that migraine interacts with the aging process, resulting in localized regions of cortical thinning in temporal (right fusiform and right temporal pole) and parietal (bilateral postcentral) regions.
Continuous decline of cortical thickness with advancing age is a well-established process (18,19,46). Although our results of predominant frontal thinning in healthy controls and migraineurs are in agreement with several normal aging studies (17–19,47), those studies have also found widespread areas of cortical thinning throughout the cortex. Unlike our study, which included age ranges 20 through 60, those studies had larger sample sizes including age ranges of subjects from late childhood through the eighth decade of life, and were henceforth able to show more pronounced cortical thinning patterns than demonstrated by our study.
Prior studies have investigated age-related cortical thinning and volume loss in a variety of disease states and hypothesized that disease-specific factors may modulate healthy aging patterns. For example, compared to healthy controls, steeper slopes of cortical thinning have been found in healthy carriers of the apolipoprotein E gene (ApoE) (20), in granulin (GRN) gene carriers (48), and in patients with temporomandibular joint disorder (22). Similarly, accelerated volume decline with advancing age was noted in patients with schizophrenia (49).
Since migraine typically begins in late childhood to early adulthood and lasts for at least many decades, it is hypothesized that migraine-specific clinical aspects such as disease duration and migraine frequency may have a cumulative or progressive effect on cortical decline (9). Several studies to date, using voxel-based morphometry, investigated the effect of disease duration on cortical integrity in migraine subjects. Schmitz et al. showed a positive correlation between disease duration and WM density in the cerebellum as well as decreased GM density in the brain stem and lentiform nucleus while controlling for age (10). Similarly, results by Rocca et al. demonstrated a negative correlation between putamen volume and disease duration in pediatric migraine, but an absence of a relationship between fusiform atrophy and disease duration (15). The study reported herein is novel since it examined the effects of age on cortical thickness in episodic migraine subjects. Although cortical thickness and volume both provide measurements of changes in GM integrity, these measures do not necessarily track each other (50). It is suggested that cortical thickness might provide a more reliable measure of cortical integrity (51) and better detection of slight GM reductions.
Our results indicate that migraine patients, but not healthy control subjects, show age-associated thinning of temporal (right fusiform and right temporal pole) and parietal (bilateral postcentral) cortex. We conclude that these regions may undergo age-related modification in response to the migraine disease process. Our findings are in agreement with several other studies that found abnormal GM aging in patients with chronic pain due to temporomandibular disorder (TMD) (22) and fibromyalgia (23,25), albeit age-related atrophy patterns were demonstrated for different regions (thalamus, cingulate, insular cortex).
Our study identified advanced age-related cortical thinning of the postcentral region in migraineurs, which is part of the primary somatosensory cortex, predominantly participating in sensory-discriminative pain processing. Functional magnetic resonance imaging (fMRI) studies demonstrate activation of postcentral regions in response to painful stimulation (52,53). Our finding of postcentral thinning with advancing age in migraineurs is in accord with other studies showing cortical volume loss and cortical thinning in patients with other types of chronic pain (54,55). However, it is noteworthy that the location of bilateral postcentral cortex thinning demonstrated in this study does not seem to be confined to areas subserving processing of head pain.
Our results also demonstrate age-related cortical thinning in temporal regions in episodic migraineurs. The fusiform is a region important for assigning emotional valance (56) and imagery (57). Increased activation in the fusiform gyrus has been reported in chronic pain subjects during mental visualization of pain (57), and fusiform gyrus GM volume reductions have been identified in cluster headache patients who were between bouts (58). Fusiform atrophy was also identified in children with migraine, the extent of atrophy not correlating with migraine duration (15). The presence of fusiform atrophy in very young migraineurs combined with the absence of a correlation to migraine duration lead the authors to hypothesize that the fusiform abnormalities might serve as a migraine disease biomarker.
Structural and functional temporal pole abnormalities are shown in several migraine studies to date. Using positron emission tomography (PET) imaging, increased activation in the temporal pole was first demonstrated for migraineurs during the acute migraine phase relative to migraineurs in the interictal state (59). These data were corroborated in a more recent fMRI study that demonstrated temporal pole hyperexcitability as well as increased functional connectivity to other brain regions for migraineurs in response to painful heat stimulation (60). Temporal pole activation increased in migraineurs during the ictal state, leading the authors to suggest that the temporal pole might become sensitized by repeated migraine attacks. Similarly, increased regional homogeneity in the temporal pole was found using resting-state fMRI for migraineurs with longer disease durations relative to migraineurs with shorter disease durations (61). It is of note that migraine subjects with longer disease durations were significantly older than those with shorter disease durations, which complicates the interpretation of the findings. Lastly, in a study investigating the interregional correlations of cortical networks, results indicated altered temporal pole structural connectivity in female migraine patients relative to healthy control subjects (62).
In summary, this study demonstrates that migraine patients show structural alterations of temporal and parietal regions and that cortical decline of these regions becomes more pronounced with advancing age. Although the physiological mechanisms underlying cortical thinning are not completely understood, thinning may reflect cytoarchitectural changes of neuronal density or synaptic pruning, possibilities that need to be addressed in future studies. It is noteworthy that migraine prevalence starts declining in the fifth decade (5) and that migraine symptoms often become “milder” with decreasing frequency during this stage of life (63). Future longitudinal studies are needed to evaluate whether age-related cortical thinning contributes to the decline and/or remission of migraine symptoms in older patients.
There are several limitations in this study. First, although the results of this study are intriguing, they should be interpreted with caution because of the relatively small sample size and cross-sectional design. Inferences toward a larger population base can only be warranted using a longitudinal design with a larger study population.
Migraine is a complex disorder that usually has an onset in early adulthood and continues for many years thereafter. Many studies have attempted to correctly assess the “migraine burden,” which we calculated as years with migraine × headache frequency. It should be noted that this is a rough estimate and potentially flawed measure of lifetime headache burden. This measure assumes that the headache frequency reported by the patient remains the same (or is unaltered) over many years. In order to have a more precise understanding of the headache pattern over a patient’s lifetime, a longitudinal assessment would facilitate the interpretation of how declining, progressing, or stable patterns of attack frequency over someone’s lifetime are related to structural decline.
Possibly confounding variables that we could not assess in our analysis are the influence of medication on cortical thickness. Although we enrolled only subjects who did not use preventive migraine medication, and were headache free at the time of the scan (and thus not in need of taking pain-related medication), we cannot account for the effect of medication that subjects used on occasion to treat migraine attacks. Since opioid dependency is indicated to decrease regional brain volume (64), we included only subjects who were not taking scheduled opioid medication. Only one migraine participant included in this study treated migraines with opiates on occasion.
Conclusion
Our results suggest that migraine interacts with age to induce microstructural brain changes. Migraine patients show age-related cortical thinning patterns regardless of disease duration and frequency.
Clinical implications
Migraine interacts with age to induce microstructural brain changes. Migraineurs show age-related thinning in regions that do not thin in healthy controls. Localized regions of cortical thinning in migraineurs include temporal (right fusiform and right temporal pole) and parietal (bilateral postcentral) regions.
Footnotes
Funding
This work was supported by a grant from the National Institutes of Health (K23NS070891) to TJS.
Conflict of interest
None declared.
Authorship statement
We declare that all authors made significant contributions toward the study design, acquisition, and interpretation of the data. All authors were involved in drafting and revising the manuscript, and approved the final version to be published.
Acknowledgments
We would like to thank the study participants and imaging technicians for their time commitment and dedication to this study.
