Abstract
Background
Migraine patients have an increased photic-driving response. This ‘H-response’ (HR) has potential diagnostic value but it is time consuming.
Aim
The aim of the study was to establish a fast and standardized test for the study of migraine biology and treatment.
Methods
We studied 11 migraine patients and 11 matched control participants. We used stroboscope flashes with a ‘chirp’-like linear frequency-increase from 10 to 40 Hz. EEG was recorded from occipital electrodes. Power spectral density was calculated for the stimulus response and corrected for baseline. An HR-estimator was calculated as the average power between 18 and 26 Hz in the stimulation-frequency window.
Results
There was a significant difference for single (p < 0.05) and for 10 averaged recordings (p < 0.01) between migraineurs and controls, and a high inter-test reliability (Cronbach’s alpha = 0.94).
Conclusion
Chirp-like stimulation to study the H-response is reliable and efficient and might therefore have a potential for acute interventional studies in migraine research.
Introduction
Migraine is disabling headache, usually associated with nausea and sensory hypersensitivity, affecting up to 15% of the population. In 1959, Golla and Winter described a test to discriminate between patients with episodic disabling headaches and headache-free controls (1) using a custom-built amplifier to quantify photic driving responses. When they were recording EEG during steady-state stimulation from 10 to 24 Hz, they found that the EEG of the headache patients was driven to frequencies above 18 Hz (‘H-response’), whereas in healthy controls was limited to about 14 Hz. More than 40 years later Chorlton and Kane (2) confirmed the high specificity and relatively high sensitivity of the ‘H-response’ test, applying the classification criteria for migraine of the International Headache Society. A review of the literature regarding electroencephalographic research in migraine showed the prominent photic driving response at high flash rates in migraine patients to be consistently reported (3). Only a recent study by Bjork et al. showed conflicting results and suggested that the driving response of migraine patients might be overestimated because of altered cortical information processing depending on the temporal proximity to migraine attack, as well as the influence of habituation among controls (4).
We studied the H-response applying a short, fast and dynamic stimulus, which might be used in the clinical setting, or as a tool for acute interventional or longitudinal studies in migraine.
Methods
Subjects
This study has been approved by the joint research ethics committee of the Institute of Neurology and the National Hospital for Neurology and Neurosurgery, Queen Square, London, UK. Written informed consent was obtained from all participants. All subjects were devoid of any other medical condition and had no personal or family history of epilepsy. In regard to the high frequency of tension-type headache in the general population, it was not possible to find enough control participants who never had any headaches in their lives.
Characteristics of migraine patients and controls without migraine headaches.

Significant group differences. VRS: verbal rating scale.
All recordings were made in the headache-free interval, at least 3 days before and after an attack. This was checked by personal, phone or email communication.
Stimulus design
A special series of a single flash-stimuli was administered, which is similar in temporal characteristics to the ‘chirp’ of a cricket. The frequency increased every four flashes by 1 Hz, resulting in the chirp-like pattern (Figure 1). The stimuli included 124 flashes, covering a range from 10 to 40 Hz, and a duration of nearly 6 seconds. The interstimulus-interval was 10 to 15 seconds.
The ‘chirp-stimulus’, increasing photic stimulation from 10 to 40 Hz with four flashes for every frequency step and total duration of 6 seconds.
Recording equipment
Subjects were seated in an armchair, in a quiet room with dimmed light, with eyes closed 20 cm in front of a Xenon flash lamp. Stimuli were presented with a SLE CPS-20 photic stimulator (SLE Diagnostics, Croydon, UK). Stimuli were triggered by a custom-made MATLAB (version 6.5, release 13, MathWorks, Natick, USA) program using the Cogent2000v1.24 toolbox (Functional imaging laboratory, London, UK). We used standard EEG equipment (Micromed, Treviso, Italy) with 9-mm stick-on silver cup electrodes. The active electrode was placed on O1 and O2 according to the standard 10–20 system and referred to the right ear lobe. The ground electrode was placed on the nasion. Sampling frequency was 256 Hz, with analogue filters ranging from 3 to 70 Hz.
Data analysis
EEG recordings were processed using custom written MATLAB programs. For the analysis we used a total of 12 seconds per sweep, with 3 seconds baseline before the chirp-stimulation and 3 seconds afterwards.
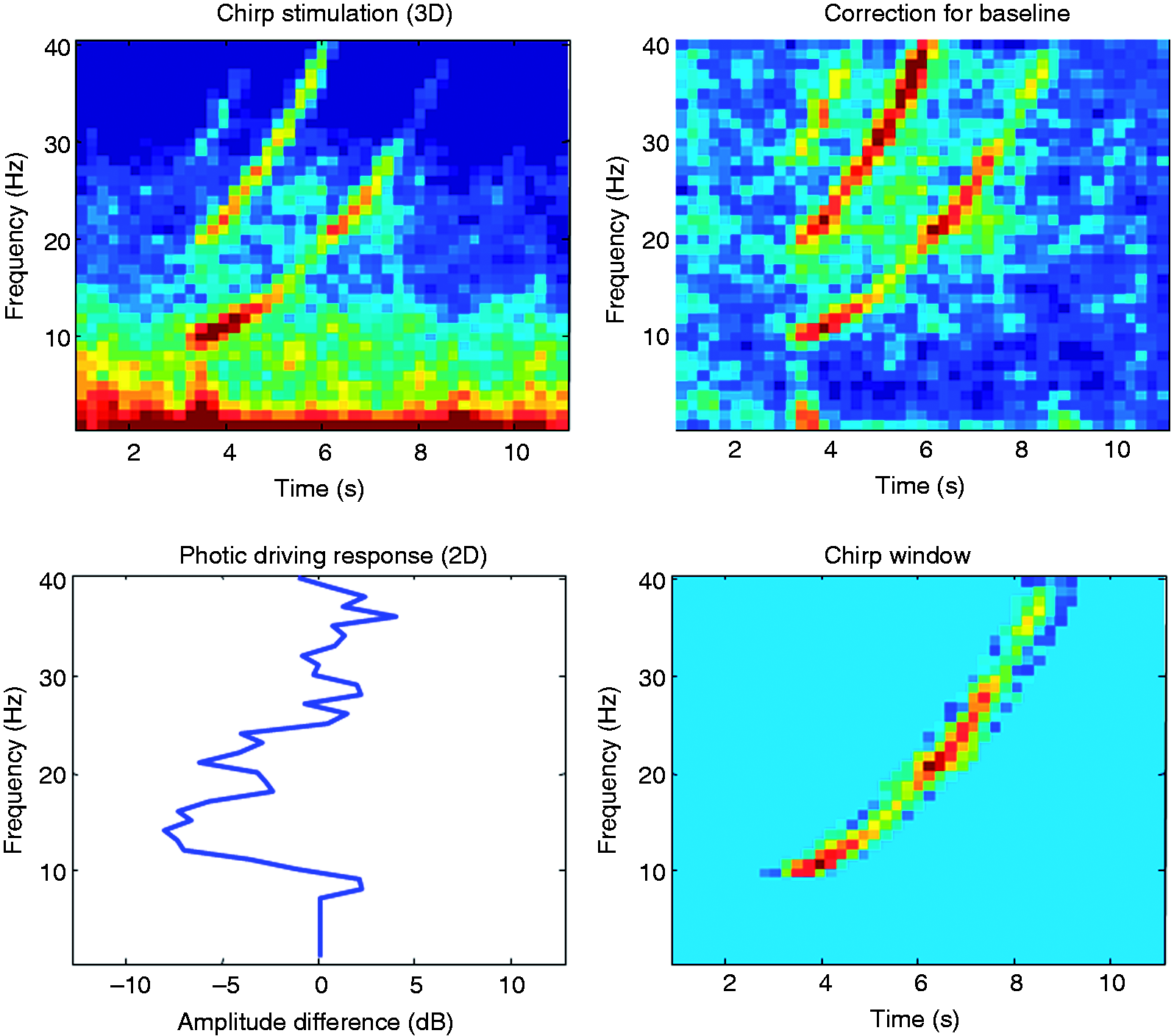
The HR-estimator was calculated as following (see also Figure 2):
For every single sweep of 12 seconds, power spectral density (PSD) was calculated in the time/frequency window, using fast fourier transformation (FFT), with a window of 128, an overlap of 64 and a resolution of 128. The PSD data of every single sweep was corrected for baseline (averaged 3 seconds before the stimulation). Thereafter the power in a predefined time window of the sweep (time between the four flashes plus 200 ms) was averaged for every stimulation frequency from 10 to 40 Hz to receive a single power estimate. The HR-estimator was defined as the average EEG power between 18 and 26 Hz for every single sweep. The figure shows the calculation of the HR-estimator in a clockwise mode. The time-frequency analysis of a single sweep of chirp stimulation was corrected for baseline. An occipital EEG recording for a single sweep of 12 seconds (baseline 3 seconds, stimulation 6 seconds, post-stimulation 3 seconds) of a typical migraine patient. In a next step the power in a predefined time window of the sweep was averaged for every stimulation frequency from 10 to 40 Hz to receive a single power estimate (2D).
Statistical analysis
Independent samples Student’s t-tests were used to compare HR-estimators between patients and controls. Pearson’s r was used for correlation and Cronbach’s alpha for reliability analysis. All results were considered significant at the 5% level (p < 0.05). Statistical calculations were carried out with MATLAB or SPSS (Release 11.5 for Windows, SPSS Inc., Chicago, USA).
Results
In all participants, photic driving could be elicited. Looking at the EEG power spectrum, in the range from 10 to 18 Hz, photic driving was similar in both groups, with an amplitude difference when compared to baseline of 3 dB. In the range from 19 to 26 Hz, the power spectrum was markedly different, with a more pronounced photic driving in this frequency range in the migraine group, and a trend towards more photic driving response above 26 Hz. There was a significant group difference for the HR-estimator between migraineurs and control participants (p < 0.05). The HR-estimator was reliable over 10 repetitions with a Cronbach’s alpha of 0.94 (n of cases = 22, n of items = 10).
Discussion
In this study we used a novel type of stimulus design to elicit an H-response, applying flash stimuli with increasing frequencies from 10 to 40 Hz. We were able to discriminate migraine patients from healthy volunteers, finding a higher photic-driving power in migraineurs in the lower beta-frequency range. The 6-second ‘chirp response’ proved to be reliable and, in contrast to the previous types of steady-state stimulation, may be administered efficiently in clinical or research setting. It adds another information processing aspect than the well described habituation deficit to the study of brain function in migraine (5). In line with previous studies, our data showed the highest discriminating power in the frequency range of 20 to 25 Hz.
Currently, the gold standard to diagnose migraine is the clinical information in the framework of the International Classification of Headache Disorders. However, the sensitivity (86.4%) and specificity (97.5%) in the study of Chorlton and Kane was good for the identification of migraine with and without aura, but not for other migraine subtypes (2). The discriminating value of the H-response between migraine and tension-type headaches has been questioned by de Tommaso et al. who studied 120 migraineurs, 64 tension-type headache patients and 51 controls (5). In their study there was no difference of the mean amplitude of the first harmonic elicited by flash stimulation in the 15 to 27 Hz range between different types of headache. They suggested a common neuronal dysfunction in the two studied headache subtypes, or poor discrimination using ICHD-II criteria. Allowing tension-type headaches in our control group, we still had a significant group difference in the photic-driving response in the low beta frequency range, suggesting different underlying information processing in the groups we studied. We do not have any reliability data for repeated testing on different days. However, we found good intra-individual reliability of the H-response within the same day of recording. Therefore ‘chirp stimulation’ may be a valuable tool for interventional research with longitudinal design.
In regard to confounding factors, all subjects were at least 3 days before or after an attack, what minimizes the influence of the interictal state on information processing characteristics (6). As photic-driving responses decrease with age (7), this should not influence the interpretation of our results, where migraine patients were slightly older than the control participants. The sex differences are expected to be small, although higher photic-driving is reported in women, depending on their hormonal state (8).
Using our stimulus design, it was possible to avoid interference with habituation processes that have been shown to be abnormal in visual processing in migraineurs (9). In analogy to the interictal lack of habituation the photic driving response of migraineurs may be explained by abnormal cortical information processing. As a result of the dynamic design of the chirp stimulus the brain is forced to react with fast adaptive changes. This makes it less likely to be affected by habituation processes, although based in revised concepts of habituation it cannot be excluded (10). Angelini et al. investigated phase synchronization in EEG recordings from migraine patients (11). They found enhanced alpha band phase synchronization in the presence of visual stimuli and concluded that migraine patients have an overactive regulatory mechanism to external stimuli. In a recent study we showed that amplitudes of single trial visual evoked potentials were explained almost entirely by increases in local amplitude, implying a higher energy demand (12). Other authors showed a variation of evoked potentials towards a migraine attack (13).
These findings may suggest that the migraine brain is ‘running at higher levels’ when stimulated interictally and therefore might consume more energy. The normalization of both photic driving and evoked potentials during the attack may point towards a kind of ‘valve’ mechanism of the migraine attack itself.
Clinical implications
Migraine is a clinically relevant disorder, affecting up to 15% of the general population. The pathophysiology is still not understood and there are no clinical tests for the diagnosis. This study provides new insights into cortical information processing in migraine patients and offers a new research tool for the investigation of abnormal perceptive mechanisms.
Footnotes
Acknowledgements
The work was conducted at the UCL Institute of Neurology, London UK. We thank David Holder for the opportunity to use his electrophysiology lab.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
