Abstract
Background
The aetiology behind chronic post-traumatic headache (CPTH) after mild head injury is unclear and management is complicated. In order to optimize treatment strategies we aimed to characterize a CPTH population.
Methods
Ninety patients with CPTH and 45 patients with chronic primary headaches were enrolled from the Danish Headache Center. All patients were interviewed about demographic and headache data. They completed the Harvard Trauma Questionnaire (HTQ), Rivermead Post Concussion Symptoms Questionnaire, SF-36 and a headache diary.
Results
The CPTH group experienced more cognitive (p < 0.001) and somatic symptoms (p = 0.048) and rated their self-perceived health as more affected in terms of physical function (p = 0.036), physical role function (p = 0.012) and social function (p = 0.012) than the control group. Surprisingly, 31% of the CPTH group had a score equal to or above the cut-off score for having post-traumatic stress disorder (PTSD) according to the HTQ. In terms of demographics and headache, the groups were comparable except the CPTH group were more often without affiliation to the labour market (p < 0.001).
Conclusions
The loss of work capacity and high levels of disability for the CPTH patients suggests directions for further research into what important factors are embedded in the patients’ PTSD symptoms and might explain their prolonged illness.
Keywords
Introduction
Diagnostic criteria of chronic post-traumatic headache attributed to mild head injury (5.2.2) according to ICHD-2 (2).

Reports of the incidence of post-traumatic headache (PTH) after trauma differ widely, from 30% to 90%, probably because of different definitions and study methodologies (6). In the Akerhus population-based study of chronic headache among 30-44 year olds, the prevalence of CPTH after mild head injury was reported to be 0.15% in men and 0.20% in women (7). In the Danish Headache Center (8), a tertiary headache unit, the prevalence of CPTH patients in 2011 was 10% of all newly referred patients; however, most studies exclude CPTH patients so the exact prevalence of CPTH following a concussion is difficult to establish (5,7,9).
CPTH is the most prevalent symptom after a concussion and is a cardinal symptom in the definition of the post-concussional syndrome. In this paper concussion is chosen as a representation of the terms: mild head injury and mild traumatic brain injury (mTBI) (6). Besides CPTH, the concussive patient may also present a wide range of emotional, cognitive and other somatic symptoms in relation to their concussion. These symptoms are, however, often unspecific, difficult to measure and frequently reported among the general population, too (9). The controversies around the diagnostic criteria are well known in terms of type of trauma causing the headache as well as type of headache in relation to the trauma. The lack of clear-cut clinical features, the definition of the term ‘chronic’ (10) and the time frame may also confuse the issue.
Clear correlation between severity of injury and the persistence of symptoms over time is lacking and has led to hypotheses of a psychological aetiology to CPTH. The hypotheses can be grouped into pre-morbidity (11) and the patients’ interpretation of the impact of their trauma, that is post-traumatic stress disorder (PTSD) (12). Both hypotheses lack research, but in this study we focus on the patients’ interpretation of their trauma. The studies by Hickling et al. (13) and Chibnall et al. (12) found that it is important to evaluate PTH patients for the presence of PTSD, because they found that 29-75% of their CPTH patients have PTSD.
Chronic headache also has a major impact on quality of life (QoL) (14). However, in a study by Marcus of CPTH patients compared with non-traumatic headache patients, no significant difference was found in the SF-36 when days with headache were taken into account (15).
In contrast to primary headache disorders (16), to our knowledge, no economic cost studies have been made on CPTH, but a study by Leibson et al. (17) reported that concussive patients had an average medical annual cost of $11,725 for the period 1-5 years after trauma. The total cost of CPTH also includes the issue of whether or not CPTH is simply a product of the legal litigation, and that no recovery can be expected before a compensation settlement has been paid. That is still a point of contention in the field of CPTH (18).
The present report is the first part of a larger clinical CPTH study. The aim here is to describe a CPTH population and, in detail, to compare this population with other chronic headache patients in terms of headache characteristics, medication, demographic data, QoL and post-concussion symptoms. In addition, we aim to focus on PTSD and possible characteristics for patients who developed prolonged CPTH and to discuss implications for psychological treatment strategies for this patient group.
Methods
Patients with CPTH referred to the Danish Headache Center (DHC), a multidisciplinary, tertiary headache centre (8) between June 2008 and August 2011 were identified by the headache specialists and referred to the research psychologist and first author (DK). Eligible participants were patients who were diagnosed with CPTH attributed to mild injury according to ICHD-II (2) (Table 1), and who were interested in possible psychological treatment of their headache. The selected patients were required to be adults aged 18-65 years and without other neurological or psychiatric disorders. In cases where neuroimaging (MR/CT-scan) had been conducted after their head trauma, it had to be normal without signs of contusions or other traumatic brain lesions.
Exclusion criteria were pregnancy, present medication and/or substance overuse as defined by ICHD-II, a history of pre-existing primary headache defined as more than 12 days of tension-type headache annually and/or more than one migraine attacks per month in the last year. Patients developing CPTH in relation to a whiplash injury were also excluded. The study was approved by the regional ethics committee (H-1-2011-FSP).
In total, 155 patients with CPTH were screened and interviewed by the first author (DK) (Figure 1). Ninety patients were included and underwent a detailed and structured interview about their head trauma, current headache, general demographic features and litigation/insurance status.
Flow diagram of CPTH patients.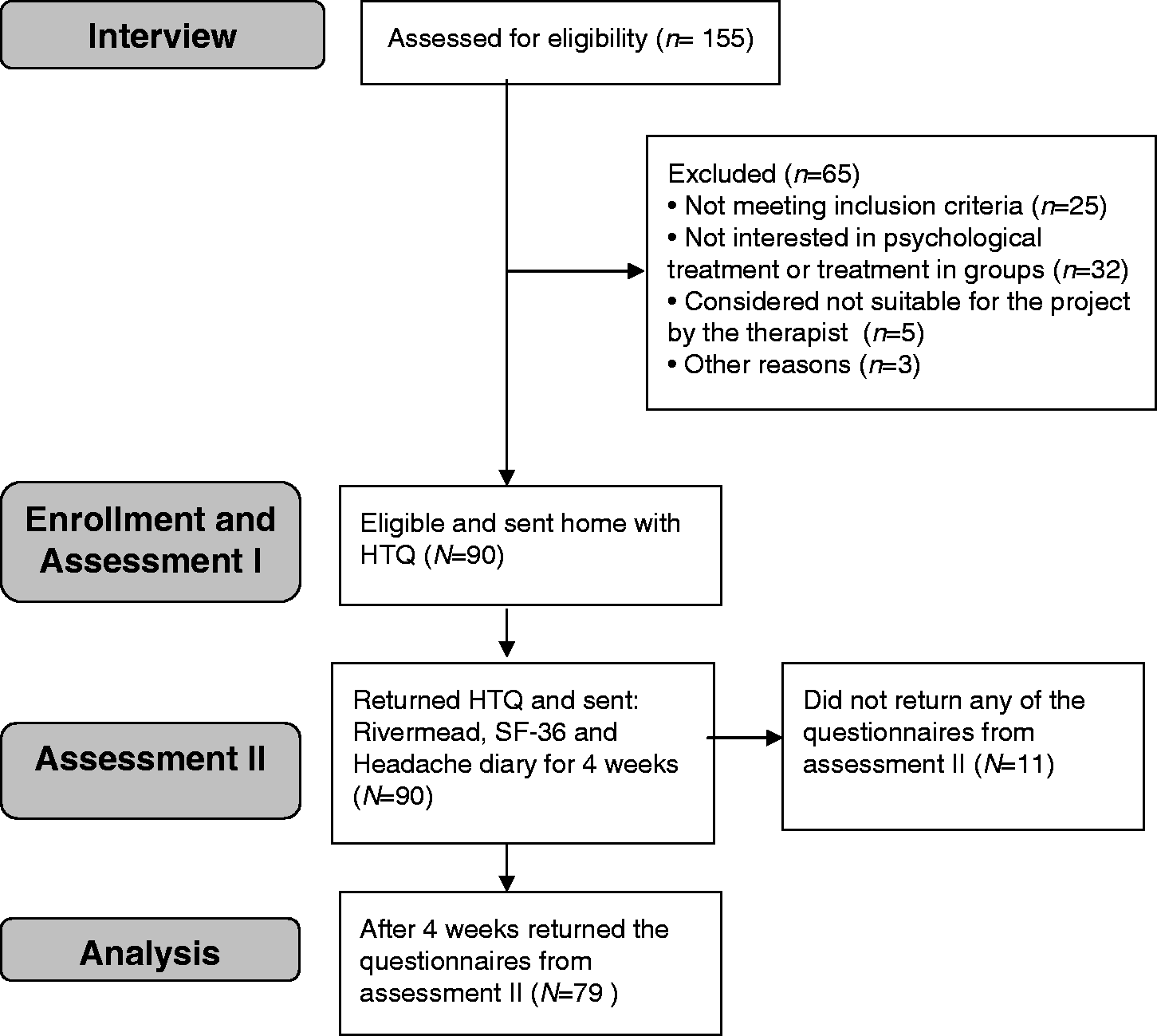
After the interview the patients were asked to complete the four validated questionnaires (the Harvard Trauma Questionnaire, Rivermead Post Concussion Symptoms Questionnaire, SF-36 and a headache diary) at home and to return them in a prepaid envelope. Patients who did not return the questionnaires after two reminders were excluded.
We aimed to have a consecutive group of patients with primary headaches as control subjects so 45 patients already enrolled in the Danish Headache Center with chronic primary headaches such as migraine, CTTH or cluster headache, but without PTH and without MOH were given the Rivermead Post Concussion Symptoms Questionnaire and SF-36 serving as a control group. The same exclusion criteria were used, and their headache should not have developed in connection with a head trauma. Overall, the control group was matched with the patient group according to sex, age and number of headache days.
Assessment
Headache
Headache was recorded in a 4-week baseline period at the beginning of Assessment II (see Figure 1) via a basic headache diary (19), which has been validated (20) and which records the duration and intensity of any headache on a daily basis for CPTH patients. In case of missing values CPTH patients were interviewed in detail about frequency, duration and intensity of their headaches through the preceding 4 weeks. Similarly, the control group was ascertained and interviewed about their headaches. The primary outcome variable was the area-under-the-headache curve (AUC) and calculated as the sum of the daily recordings of headache duration multiplied by headache intensity.
Post-traumatic stress disorder
The Harvard Trauma Questionnaire-Part IV (HTQ) (21) was used to assess and measure the presence and severity of possible PTSD symptoms in relation to the trauma causing the headache (and was therefore not given to the control group). The HTQ includes 17 of the PTSD symptoms in the DSM-IV (22). Also the intensity of the three core symptom groups: re-experiencing (5 items), avoidance (7 items) and hyper-arousal (5 items) are measured. The items are scored on a 4-point Likert scale (from 1 = ‘not present’ to 4 = ‘very often present’). An item score must be ≥3 to count as a symptom for PTSD.
Concussion
The Rivermead Post Concussion Symptoms Questionnaire (RPQ) (23) is a validated (24) questionnaire that measures the severity and number of cognitive, emotional and somatic symptoms commonly experienced after concussion. The 16 symptoms are rated on a 5-point Likert scale (from 0 = ‘not experienced at all’ to 4 = ‘a severe problem’) and summed to a total score (range 0-64) excluding ratings of 1 as recommended by King et al. (23). The symptoms were divided into three sub-scales: cognitive, emotional and somatic according to Potter et al. (25), but following Sigurdardottir et al. (26) we added the original symptom ‘double vision’ to the somatic scale, because a large proportion of our participants experienced that symptom. To make the subscales comparable, the total score from each subscale was divided by its number of items.
Health-related quality of life
Health-related quality of life was measured with the Danish version of the SF-36 (27). This consists of 36-questions on perceived health-related quality of life, with answers being divided into eight scales measuring health concepts: physical functioning (PF), role physical (RP), role emotional (RE), social functioning (SF), bodily pain (BP), mental health (MH), vitality (VT) and general health (GH). On each scale the score ranged from 0 to 100, with a high score indicating better perceived health (28,29).
Statistical methods
Data were analyzed with SPSS version 19.0. Average values of continuous variables are described by means and standard deviations (SD). Categorical variables are described by frequencies and percentages. Chi-square analysis was used in the case of categorical variables and for frequency analyses. The Welch t-test was used when large differences in SD were seen (e.g. in months with headache and headache index). Mc-Nemar's test was used for the marginal frequencies of binary outcomes of occupation (before/after trauma) within the CPTH group. We used the independent t-test for group comparisons on the Rivermead and SF-36 scales, which did not deviate markedly from normality, and Bonferroni adjustments of alpha levels to correct for the large number of comparisons being made. For hypothesis generation, analysis of variance (ANOVA) was used for within the CPTH group. In all cases p < 0.05 (two-tailed) was considered to be statistically significant.
Results
Demographic characteristics
Demographic data.

Data are presented as number of patients (%) unless otherwise stated.
p < 0.05; **p < 0.01; ***p < 0.001.
CPTH: chronic post-traumatic headache.
The ‘typical’ patient with CPTH was a 34-year-old woman with a headache history for 27 months. She was married, and had education to the level of skilled labour. Before her trauma she had been employed full-time, but was either on sick leave or with an unresolved social situation after the trauma. The only significant demographic differences between the CPTH and control groups were that more CPTH patients lived singly (p = 0.028) and the control subjects were more often currently employed (p < 0.001). Within the control group the patients with cluster headache were more often living as singles (p < 0.001) than the other control patients. For the CPTH group, their occupation status had also changed significantly (p < 0.001) from 89% being employed full-time before the trauma to only 23% after the trauma (Figure 2). When adjusted for time with headache being over versus under 2 years, no difference in occupation was seen (95% CI −0.275 to 0.151; p = 0.6).
Occupation before trauma and currently.
Trauma characteristics and compensation
Most traumas were caused by falls (45%) followed by traffic accidents (24%) (Figure 3). Half of the patients who had sustained CPTH through traffic accidents were cyclists/people on mopeds having solo accidents, 18% were pedestrians. Thirty-two per cent (n = 7) of the patients had been involved in car accidents, but they reported that they were bumped into from the side (no rear-impact). Seventy-six per cent of the patients had at least one neuroimaging at some point after their trauma. At the time of inclusion, 51% of the CPTH patients had an ongoing insurance case in relation to their trauma. Seventeen patients were assessed to have suffered permanent injury to the extent that they were awarded, on average, a sum corresponding to 8% (SD 3.28) of their cumulated insurance rate.
Cause of the trauma.
Headache
Headache characteristics.
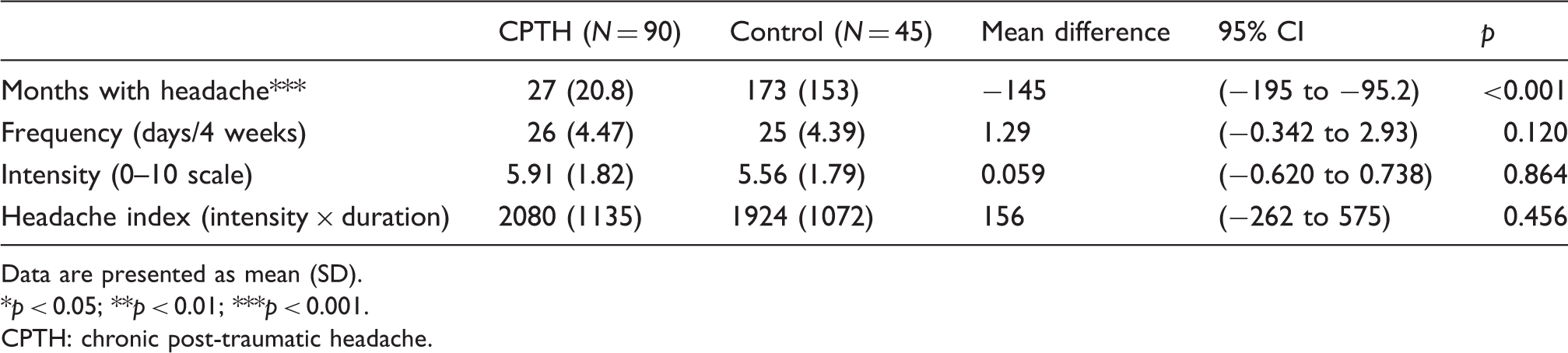
Data are presented as mean (SD).
p < 0.05; **p < 0.01; ***p < 0.001.
CPTH: chronic post-traumatic headache.
Clinical headache presentation.

Data are presented as number of patients (%).
p < 0.05; **p < 0.01; ***p < 0.001.
CPTH: chronic post-traumatic headache.
Pharmacological treatment
Pharmacological treatment.

Data are presented as number of patients (%).
p values based on Fisher’s Exact Test between CPTH and Control groups.
p < 0.05; **p < 0.01; ***p < 0.001.
Includes only multiple types of different treatment categories (e.g. ‘Triptans’ and ‘Analgesics and/or NSAIDs’), patients may have used more than one treatment from the same class. Because 32% of the patients were using treatments from two or three categories, the total numbers of patients using the specific category are listed; including patients using more than one category.
CPTH: chronic post-traumatic headache; NSAID: non-steroidal anti-inflammatory drugs.
Regarding the use of pharmacological treatments, no sex differences were seen between the groups, other than that more women than men were taking triptans (p = 0.036). In relation to other chronic headache patients, the control group used significantly (p = 0.015) more triptans but less analgesics (p = 0.002) than the CPTH group.
Psychological stress and QoL
Even though the head trauma itself was characterized as minor, 31% of the CPTH group had a score equal to or above the cut-off score for having a PTSD according to the HTQ. The mean total score of the symptoms for re-experiencing was 8.6 (SD 3.2), avoidance 15.0 (SD 5.0) and hyper-arousal 13.4 (SD 3.4), and the total score of all symptoms was 37.0 (SD 10.0).
The RPQ demonstrates that CPTH patients had a higher total score (p = 0.012), higher impact on the cognitive (p < 0.001) and somatic factors (p = 0.048) compared with the control group, whereas no difference between the two groups was seen on the emotional factor.
Description of QoL and post concussive symptoms.
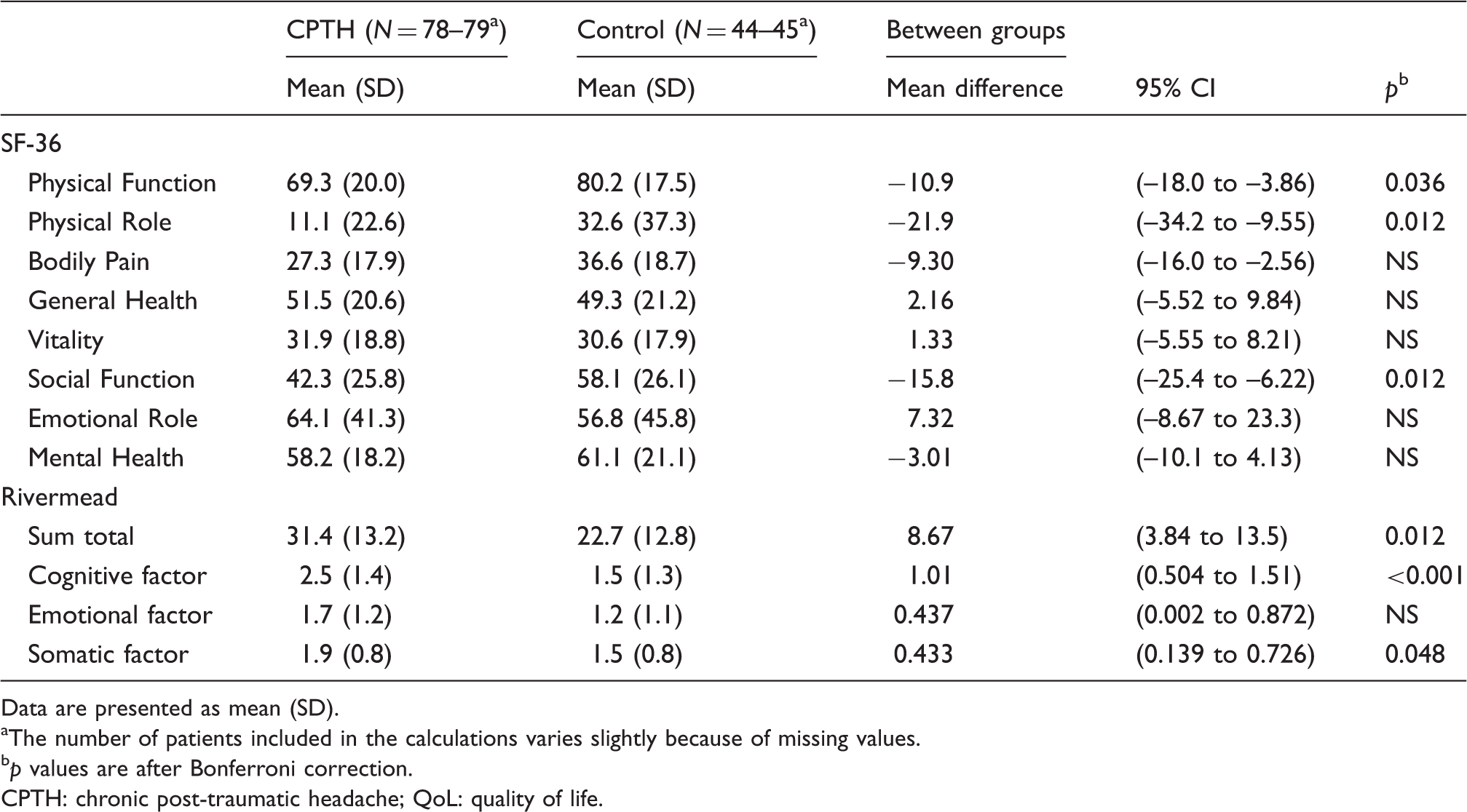
Data are presented as mean (SD).
The number of patients included in the calculations varies slightly because of missing values.
p values are after Bonferroni correction.
CPTH: chronic post-traumatic headache; QoL: quality of life.
Multiple factors
For hypothesis generation, a series of 12 multiple regression analyses were run, one on each of the 12 scores from the SF-36 and Rivermead questionnaire as dependent variables in turn. Sex, age, time with headache, PTSD, applying for financial compensation and being in a work situation were entered as independent variables (co-variants/fixed factors). A linear regression resulted in only PTSD having a significant (when adjusted for Bonferroni) relationship with the following dependent variables: Rivermead somatic score of 0.644 units (95% CI 0.278 to 1.01, p = 0.012), Rivermead total sum score of 13.4 units (95% CI 7.56 to 19.2, p < 0.001), Rivermead emotional score of 1.17 units (95% CI 0.616 to 1.73, p < 0.001), SF-36 on the score of physical function −12.1 units (95% CI −20.8 to −3.36, p < 0.001) and SF-36 on the score of general health −16.5 units (95% CI −25.9 to −7.02, p < 0.001).
Discussion
The main findings of our study were that the CPTH group had significantly less affiliation to the labour market compared with the matched headache control group, whereas there were no differences on most of the demographic characteristic and the two groups were equally burdened by headache. Despite these similarities, the CPTH group experienced significantly more cognitive and somatic symptoms than the control group, but at the same time reported the same level of emotional symptoms. The CPTH group also rated their self-perceived health as more affected in terms of physical health, whereas psychological health was very similar in the two groups with the exception of social function.
Clinical presentation
In our study the majority of the CPTH patients (97%) presented with tension-type like headache and only 28% a migraine-like headache either with or without aura. This is not consistent with the study by Lucas et al. (3), who found that migraine was the most frequent headache type after concussion. A causal explanation for that could be that our study did not include patients suffering from primary headache prior to their concussion, whereas Lucas et al. included patients with pre-injury headache. They found that 17.5% of their sample had a history of pre-injury headache prior to mTBI, more than half endorsing symptoms consistent with migraine or probable migraine headache. In a study by Hass (4) that has a similar setting and method to our study, it was reported that 80% had a tension-type like headache and only 21% met the criteria for migraine without aura. Additionally, a prior study by Baandrup et al. (5) from our research group showed comparable results (62% had tension-type like headache, 29% had migraine or probable migraine). Another interesting result from that study is that 42% of the patients at referral time for the DHC fulfilled the criteria for MOH in combination with their PTH. In our study only 13% have had a MOH history during the PTH. A possible reason for that could also be that since the study by Baandrup et al. in 2003, general awareness in the Danish population of the risk of developing a MOH has advanced.
Broadly, it is difficult to compare the above mentioned studies because different sample selections have been used. Also, different definitions of PTH and concussion have been applied as well as the inclusion or exclusion of patients with pre-injury headache and/or MOH. Major strengths of our study are the standardized use of ICHD-II and diagnostic diaries for classifying patients, the controlled design and the fact that we excluded CPTH patients with a prior headache history.
The patient’s use of headache medication was also in focus in our study. Comparing the two groups, no differences were found between patients who did not use medication or used medication from one or more treatment categories. We found that CPTH patients significantly more often used simple analgesics, but less triptan than the control group. A reason for the increased use of analgesics could be that CPTH patients, because of the trauma, experienced other physical pain as well and that they to a greater extent suffered from tension-type like headache, making analgesics a first treatment choice.
In a review, Zasler (30) pointed out that the pharmacological management of PTH in general relies on treatment guidelines for the primary headache, one reason for this being the lack of specific research into the pharmacological management of CPTH. Similarly, in clinical practice, these patients are often found to be very difficult to manage and to respond rather negatively to the offered pharmacological treatment – an attitude with which we concur.
Concussion symptoms and QoL
When CPTH patients are seen in our clinic, additional to headache, other somatic, cognitive and emotional symptoms are also reported. That they score higher on the somatic and cognitive factors is therefore not surprising. These results are consistent with a study by Smith-Seemiller et al. (31), who compared patients with chronic pain with patients with mTBI not further specified with regards to headache. Parallel with our CPTH group, their mTBI group also had more cognitive problems than the chronic pain group. This indicates that the cognitive symptoms reported by our CPTH patients might be caused by trauma mechanisms and not the perceived pain itself. Smith-Seemiller et al. also found that patients with pain were more affected on the emotional factor than patients with mTBI who might not experience pain in combination with their mTBI. Compared with our groups they were equally burdened on the emotional factor, which indicates that it might be the headache pain itself and not the concussion that raises the emotional factor.
That pain, including headache, influences QoL has been reported in numerous studies (14). We find that physical function, role physical and social function burden the CPTH patients significantly more than the control group. In a comparable study by Marcus (15), the results indicated as in our study, that the trauma-caused headaches tended to lower physical function more than the non-traumatic headaches. The questions that form the physical function scale are formulated in a way that does not embrace the limitations caused by headache. Instead, the function scale focuses on activities of daily life such as walking one block and dressing oneself. When CPTH patients still score low on the physical scale, it can also be because of other trauma-related pain problems.
The questions within the physical role scale describe the limits, time and energy used at work or in domestic work. The elevated cognitive and somatic deficits experienced by the CPTH group could partly explain their lowered score on the physical role.
The questions embedded in the social function scale are focused on social activities. In interviews CPTH patients often mention that not only is the headache a limiting factor, but they also feel stigmatized by not having a job and by having an invisible disability. Not being able to focus on a conversation for very long time and tiring very quickly add further limitations to their social life.
Psychological stress
The possible association between PTSD and CPTH is complex and probably multifactorial. In our study a relatively large number (31%) of CPTH patients had a score equal to or above the cut-off score for having a PTSD according to the HTQ. This result is consistent with a study of whiplash patients by Elmose et al. (32), who found that 23% had a PTSD (diagnosed according to DSM-IV) and the CPTH study by Chibnall et al. (12) where 29% had the diagnosis (DSM-III). Even though chronic headache patients may not have experienced a physical or psychological trauma in relation to the onset of their headache, Peterlin et al. (33) found that the frequency of PTSD is higher in migraineurs (30%) than in the general population, whereas Ifergane et al. (34) were unable to confirm an overrepresentation of PTSD in migraineurs. The HTQ was chosen because we found it was the best questionnaire to capture the possible psychological traumas the CPTH patients had experienced. As a result of the phrasing of the questions, we were unable to use it for our control group. According to the study by Peterlin et al. (33), no differences in terms of PTSD symptoms should have been expected between our two patient groups.
Whether the PTSD diagnosis is relevant for CPTH or the individual factors embedded in the PTSD is not clarified. Our study was not designed to answer that question but rather to point to the direction of psychological stress playing a significant role in CPTH. The PTSD (or the factors embedded in the questionnaire) was the only variable in the linear regression that was associated with increased symptoms measured by the Rivermead and a reduced QoL as measured by the SF-36.
In terms of screening patients and treatment planning, it could therefore be relevant to have a closer look at the underlying factors behind the PTSD, as these factors may be predictive of prolonged CPTH.
Health care costs
Those of our CPTH patients who were seeking injury compensation did not appear more burdened by post-concussive symptoms, and did not have a lower QoL, than patients not actively seeking compensation. A reason for this could be that in Denmark there is a relatively low insurance compensation rate (0-15%) for concussions compared with other countries, and furthermore there is a fairly easy access to health-related public income compensation making the question of whether compensation slows down recovery a more complex issue.
When both groups are so comparable in terms of demographics, affiliation to work prior to trauma and headache characteristics, then why are CPTH patients significantly less employed after the trauma than our control group? The major difference between the two groups is the length of time that they have been suffering headaches; the control subjects having a duration of 172 months and the CPTH patients only 27 months.
A hypothesis could be that if the patients had adequate time to recover after their trauma they would once again be a part of the work force. However, in a post hoc analysis with a separation of the CPTH group according to headache duration (under vs. over 2 years), no differences were seen between the groups’ work affiliation, nor had the headache duration an effect on number of symptoms in relation to concussion (Rivermead) or QoL (SF-36). Hence, other pathways must be explored.
Limitations
In the definition of PTH there is an ongoing discussion as to whether the diagnosis of concussion should require the patient to have been unconscious for some brief time. The majority of the patients in this study did not seek medical treatment immediately after their trauma, which itself happened under circumstances where they were alone and experiencing a stressful event. As with most other CPTH studies, not knowing patients’ state of consciousness could be a confounder, but despite this uncertainty we included patients fulfilling the ICHD-2 criteria as our focus was on the disabling effect of CPTH. Because the samples of both CPTH and control patients were drawn from a specialized headache centre where patients with chronic and rare headache types are seen, the results may not be representative for patients less burdened by CPTH. Our control subjects were drawn consecutively from patients with primary headaches and inclusion of cluster patients did not affect the overall analysis or conclusion of the study. No separate analyses have been made on patients dropping out after the screening interview.
Conclusion
CPTH patients have fairly similar clinical presentation to patients with chronic primary headaches but their somatic and cognitive symptoms are elevated and their ability to work and QoL are markedly reduced. The question of compensation and litigation played apparently a minor role in our study population, although this issue is open to further investigation.
Thirty-one per cent of our CPTH patients also had a score equal to or above the cut-off score for having a PTSD according to the HTQ, indicating a high level of psychological stress and possibly a poorer prognosis. Because of the overall high degree of disability for the CPTH patients, loss of working capacity and high indirect health care costs, we find it of major importance to further characterize the symptoms embedded in the PTSD diagnosis. Whether pre-morbid personality is important for development of CPTH should also be investigated. Such information would assist in optimizing treatment strategies for these complicated and refractory patients in the future.
Clinical implications
The CPTH group had significantly less affiliation to the labour market compared with a matched control group with the same impact of headache. The CPTH group experienced significantly more cognitive and somatic symptoms than the control group. 31% of the CPTH group had a score equal to or above the cut-off score for having a PTSD according to the Harvard Trauma Questionnaire indicating a high level of psychological stress and possibly a poorer prognosis.
Footnotes
Funding
The research received grants from Lundbeck (R67-A6507) and Helsefonden (2099B033).
Conflict of interest
DK has received honoraria and travel funding from Allergan and Pfizer. RJ; TT and HF have not received any funding in terms of this study.
