Abstract
Objectives
Our objective was to determine odorants that trigger migraine attacks and the time of onset of headache after exposure.
Methods
Migraine or tension-type headache patients, diagnosed according to the criteria of the International Classification of Headache Disorders-II, were interviewed about lifetime prevalence of headaches triggered by odors and time of onset of pain, after exposure of the patient to the odor.
Results
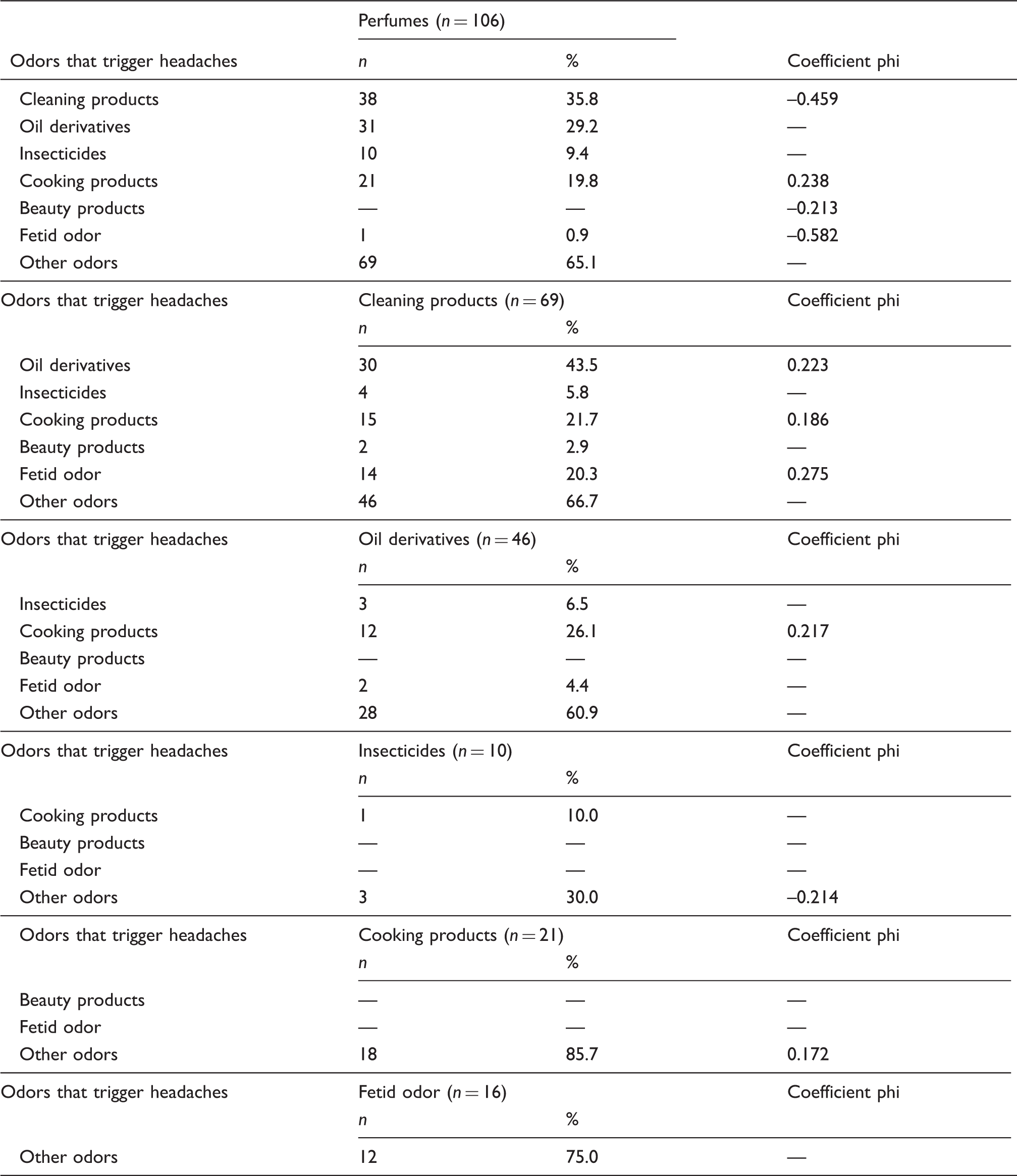
We studied 200 migraine patients and 200 tension-type headache patients. There were odor-triggered headaches after 25.5 ± 1.9 minutes of exposure in 70.0% (140/200) of migraine patients and none with tension-type headache, which ran at low sensitivity (70.0%, 95% CI 63.1–76.2) and high specificity (100.0%, 95% CI 97.6–100.0). Odor-triggered headaches are distributed in the following order of frequency: perfumes (106/140, 75.7%), paints (59/140, 42.1%), gasoline (40/140, 28.6%) and bleach (38/140, 27.1%). There was significance in the association of odor-triggered migraine, especially among perfume with cleaning (phi = –0.459), cooking (phi = 0.238), beauty products (phi = –0.213) and foul odors (phi = –0.582).
Conclusions
Odorants, isolated or in association, especially perfume, may trigger migraine attacks after a few minutes of exposure.
Introduction
In general, odors are a result of the action of volatile molecules, either organic or mineral, with distinct physicochemical properties that, acting as a stimulus to the olfactory system, trigger impulses that are transmitted to the brain (1,2).
There are three known characteristics of odors: the character or quality, corresponding to a rating scale of measurement in which a person recognizes the odor as similar to one of his or her own repertoire; the intensity and concentration, which are equivalent to the strength of the perception of odor; and the hedonic tone assessment, defined as the measurement of the pleasantness of odor (3).
Odorant substances well tolerated by the general population may trigger headache attacks in migraine patients (4–8), even in low concentrations.
This fact is relevant because in migraine, a chronic neurological disease with 15.2% prevalence in Brazil (9), headaches are associated with nausea or vomiting, photophobia, phonophobia, dizziness and osmophobia (4,10,11), with an intensity that may incapacitate the patient from performing daily activities.
The migraine attacks may be triggered by various factors, including odors. However, odors as migraine triggers have been insufficiently studied. This is the first study to specifically determine the odorant substances that trigger migraine attacks and the time of onset of headache after exposure.
Patients and methods
Study design and patients
This was a retrospective study with comparison of groups. The study population comprised a nonrandom and convenience sampling, consisting of the first 200 migraine patients and 200 tension-type headache patients (comparison group) treated at a headache clinic in the city of Teresina in northeastern Brazil. Data for this study were collected from August to December 2011.
Inclusion and exclusion criteria
Patients aged between 18 and 60 years, with migraine or tension-type headache, according to the diagnostic criteria of International Classification of Headache Disorders, second edition (ICHD)-II (12), and presence of headache in the last six months at a frequency less than 15 days per month were included in the study.
In order to obtain consistent and valid data, the study excluded patients without a headache in the last six months, association between migraine and tension-type headaches, concomitantly or at different times, the presence of other primary or secondary headaches, the presence of associated diseases or the use of medications that might impair olfaction, and pregnant women.
Data collection
The patients underwent a thorough standard medical history performed by a headache specialist to record the typical characteristics of their pain over the past six months. Having met the criteria for inclusion and exclusion, patients were invited to participate in the study by signing the informed consent form. There was, then, a structured interview based on a questionnaire to investigate the research objectives; excluded at this stage were uncooperative patients or those who answered the interview incompletely.
The interview consisted of questioning the patients about odorant substances that trigger headaches at least once in a lifetime, as well as time of onset of pain after exposure. Such substances were divided into eight categories: fetid odor, perfumes, oil derivatives, insecticides, cleaning, culinary and beauty products, among others.
Statistical analysis
Once the information was organized in the database, the Statistical Package for Social Sciences (SPSS®) version 17.0 for statistical analysis was used. The chi square test with Yates correction, Fisher’s exact test and Mann-Whitney test for differences between averages of unpaired samples were used, besides the phi coefficient of contingency for nominal variables, assuming a significance level of 0.05. Sensitivity and specificity parameters and positive and negative predictive value were determined.
Ethical aspects
This study was approved by the Ethics in Research Involving Human Subjects Committee at the State University of Piauí, Brazil, protocol number 049/11 and the National Ethics in Research System, registry number 439715. All patients signed the informed consent form.
Results
There were 400 patients averaging 38.6 ± 9.9 in age (95% confidence interval (CI) 37.7–39.6) and ranging from 22 to 58 years old, 219 (54.7%) of whom were women, which corresponded to the sex ratio of 0.83:1 male/female.
Distribution of sex and age according to diagnosis of 200 patients with migraine and 200 with tension-type headache.

TTH: tension-type headache; CI: confidence interval. ap value based on the chi square test. bp value by the Mann-Whitney test for average difference test between averages of unpaired samples.
The triggering of headaches by odors occurred in 140/200 (70.0%) migraine patients and in none of the tension-type headache patients (0/200), and it had low sensitivity (70.0%, 95% CI 63.1–76.2) and high specificity (100.0%, 95% CI 97.6–100.0). The pain occurred 25.5 ± 1.9 minutes after exposure of the patient to odor. The median latency was equal to 10 minutes, meaning that in 50% patients, the pain appeared before 10 minutes and in 50%, after 10 minutes of exposure.
Distribution of odors and latency times to trigger headache, isolated or in association, in 140 migraine patients.

They were not able to describe perfumes in 82 cases (77.4%); cooking products, four cases (19.0%); fetid odors, four cases (25.0%); and insecticides, one case (10.0%).
Association between odors that trigger headaches in 140 patients with migraine.
Discussion
In migraine, the attacks may be triggered by many identifiable factors and, in most cases, in multiples for the same patients. Among these factors, the ones that most frequently trigger pain are stress, hormonal changes (ovulatory or premenstrual period, menopause and hormone replacement therapy), deprivation or excessive sleep, eating habits (long fasting periods or ingestion of certain foods, food additives or alcohol), physical exertion (sexual activity, coughing, sneezing, bowel movement and weight lifting) (10,13–16). Besides these factors, many patients may present with headache attacks triggered by sensorial stimulation, such as luminosity, noises or exposure to some types of odorant substances (5,7,10,17–22).
The two groups were not randomly selected. Thus, migraine occurred more often in women and tension-type headache in men. Despite the predominance of women in studies on odors (7,18,20) and evidence that women outnumber men in olfactory identification (23,24), we do not believe that this has an impact on our results because a study (25) that evaluated the role of odors in migraine exclusively in men showed that 48% have headaches triggered by odors, and mainly by perfume, similar to women.
The olfactory processing is altered during headache attacks in migraine patients, suggesting that there are specific neuronal connections between the olfactory and trigeminal nociceptive system (26) because most odors are bimodal, i.e. activating trigeminal and olfactory nerves (27). The trigger mechanism of the headache by odors in tension-type headache patients is unknown. A study by functional magnetic resonance imaging (fMRI) during headache attacks in migraine patients and normal subjects showed increased activity of the limbic system and brainstem in response to olfactory stimulation only in migraine patients (26).
There is another hypothesis that the olfactory stimulus excites the locus coeruleus in migraine patients and causes norepinephrine release. Consequently, substance P and calcitonin gene-related peptide (CGRP) are released. These two potent and inflammatory vasodilatory substances trigger the painful phenomenon (28).
Recent studies have shown that inhalation of certain odors can cause severe attacks of headache through stimulation of transient receptor potential ankyrin 1 (TRPA1), a nonselective cation channel expressed in sensory neurons, and activation of the trigeminovascular system. Consequently, there are nociceptive responses and calcium-dependent release of CGRP from trigeminal nerve terminals in the dura mater. Furthermore, TRPA1 activation can activate dural nociceptors and lead to central sensitization and cutaneous allodynia (29,30).
It was confirmed that odorant substances triggered migraine attacks in most of the patients at least once in a lifetime. Headache was typical of migraine patients, i.e. it was associated with nausea and/or vomiting, photophobia and phonophobia. A high percentage of headaches triggered by odors was also found by other authors (31,32). However, there was disagreement in other studies in which the authors reported different percentages ranging from 25.3% to 46.0% (4,13,18).
In the literature review, we found no studies in which tension-type headache patients describe attacks triggered by odors. Tension-type headache patients may report intolerance to odors during crises, but no headache was triggered by odors. These findings confirmed that odors are used to differentiate migraine from tension-type headache (16,18).
Other authors (18) report that osmophobia during migraine attacks includes different odors, while the olfactory stimuli as trigger is more individualized, since in most cases only one odor is identified.
There is a correlation between headache triggered by odors and osmophobia, both during the attack and in the period between episodes. In our experience, in the study we are carrying out, osmophobia is present prospectively in 86% (172/200) of migraine patients during attacks, and in 24% (48/200) in the period between attacks. Headache triggered by odors was observed in 74.4% (128/172) of those who had osmophobia during the attack, and in 87.5% (42/48) when the osmophobia occurred in the period between attacks. The patients reported intolerance to any odor.
In the present study, we observed that the olfactory stimulus was individualized. However, in many patients the headache was triggered by more than one odor, but this occurred at different times.
The characteristics of intensity, character and hedonic tone of odors were not applied as criteria to determine the onset of pain. However, the hedonic tone seems to be the most important since there was a predominance of perfume, a pleasant odor. Several authors have reported similar results when they affirm that perfumes, more frequently trigger pain, followed by oil derivatives and cleaning and cooking products (4,7,10,13,18,19).
Although most of our patients have reported perfume as the main odorant substance triggering headaches, only a minority was able to distinguish the fragrance as being from a floral or citric source. In a similar study in which there was a detailing of fragrances, the type of perfume was also distinguished by a minority of patients (18).
It was also possible to discriminate statistically significant associations from several categories of odorant substances in the triggering of headaches, but this occurred at different times. In other studies, there were also associations with two or more odors in percentages ranging from 63.8% to 64.7% of the patients (10,18).
Many patients had headache attacks triggered by more than one odor but not concurrently. We have calculated the latency between exposure to an odor and onset of headache only for patients who cited a single odor. From these latencies, it was possible to calculate the mean latency of the group of odors. Therefore, one single latency was recorded in the study protocol (Figure 1).
Study protocol.
In our study, pain occurred within minutes after patient exposure to the odor. These data are confirmed by a single study conducted to determine the time of the onset of pain, in which its authors reported that 68.4% of the patients presented headache in less than 20 minutes after exposure to the odors (10).
Conclusions
The odorant substances, isolated or in association, that triggered migraine attacks, ranked by frequency were: perfumes, paints, gasoline and bleach. Headache occurred within a few minutes after exposure to the odor.
Clinical implications
Odors trigger headaches in migraine but not in tension-type headache patients. Odors trigger migraine attacks after a few minutes of exposure.
Footnotes
Acknowledgment
The authors thank Laís Guimarães Vieira, statistical consultant, for her invaluable statistical help.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
