Abstract
Aim
Craniomandibular disorders (CMD) are widespread, but we know little about the cerebral representations associated with this pain syndrome and nothing about changes in cerebral representations of occlusion induced by common therapy approaches.
Methods
In a longitudinal therapy study, we applied functional magnetic resonance imaging (fMRI) in 14 patients with mild CMD during occlusal movements. fMRI, pain scoring, kinematic investigations of occlusal movements, and jaw muscle electromyography (EMG) were measured before and after two weeks of therapy with an individually optimized Michigan splint.
Results
The patients’ subjective pain ratings decreased, and the symmetry of condylar movements increased over the period of therapy. After therapy, EMG of the jaw muscles demonstrated more relaxed resting conditions and increased activity during maximal occlusion. fMRI during occlusion showed an activation decrease in the right anterior insula and right cerebellum over the course of therapy. Correlation analysis between pain score and fMRI activation decreases identified right anterior insula, left posterior insula and left cerebellar hemisphere. Left cerebellar and right primary motor activation magnitude was negatively associated with symmetry of the condylar movements.
Conclusions
Our findings highlight the impact of the anterior insula for the internal monitoring and the anticipation of temporomandibular joint (TMJ) pain. In addition, an increase of symmetry of condylar movements after therapy has been associated with a decrease of activation magnitude in primary motor and cerebellar regions.
Keywords
Introduction
Craniomandibular disorders (CMD), also called temporomandibular disorders (TMD), are a widespread medical problem in dental practice (1). The prevalence of CMD symptoms varies between 3.2% and 17.6%, depending on the study design, symptoms evaluated and other parameters, such as the age of the group and country of investigation (2). Specifically, “myofacial pain” has a prevalence of 10% (3). In general CMD shows a female predominance (2). The treatment is most frequently performed using occlusal splints. Currently, its efficacy is controversially discussed, but in a double-blinded, controlled, randomized clinical trial over six months the efficacy of two types of occlusal splints showed to be superior for reducing pain to that of a non-occlusal (without an effect on occlusion) splint (4). Commonly, it is recommended that occlusal stabilization splints be worn at night (1).
CMD has a high comorbidity with other typical chronic pain symptoms, and orofacial pain is often accompanied by widespread pain (5), supporting the idea of a multifactorial etiology. Therefore, central cerebral processes in these patients compared to other patients suffering from chronic pain symptoms or the impact of cerebral representation on temporomandibular movements by therapeutic approaches has recently been a matter of debate. We have already investigated the impact of occlusal splints on the representation of occlusion in healthy subjects (6). In one case study, we proposed a procedure for testing representation changes in CMD patients using a longitudinal approach (7). In the present study, we first focused on the impact of occlusal splint usage over a two-week period on the cerebral representation of occlusion and reported clinical symptoms in a group of patients with mild CMD. A second aim of this study was to investigate whether changes in clinical characteristics over the course of therapy, such as pain, jaw muscle activity during occlusion, and movement kinematics during jaw-open and -closing movements, are associated with cerebral activation changes in occlusion over time. We expected to observe these changes in regions relevant for pain processing (insula, primary and secondary somatosensory cortex, and cingulate cortex (8), but also in areas processing sensorimotor control of occlusal movements (primary motor cortex (M1), superior parietal lobe, cerebellar hemispheres (6)). Thus, we applied a region of interest (ROI) analysis after measuring functional magnetic resonance imaging (fMRI) during occlusal movements with and without an individually optimized Michigan splint. In addition, we measured subjective pain intensity, movement kinematics during occlusion, and superficial electromyography (EMG) of the jaw muscles before and after a two-week therapy period with the splint.
Materials and methods
Participants and clinical tests
Fifteen participants (14 females, one male) aged between 21 and 53 years (mean age 26.73 ± 8.80 years) with CMD were recruited by the dental clinic of the University of Greifswald, section for CMD. They had no history of neurological and psychological illnesses. Eleven patients were classified as Ia, one as Ib (predominantly muscular problems) and three as IIa (disc displacement with reduction) in the revised Research Diagnostic Criteria of Temporomandibular Disorders (RDC/TMD (9)). Classification was performed by an experienced CMD specialist (B.K.). Before and after therapy subjects were asked to judge their pain intensity on a 10-digit ordinal scale numbered from 0 (no pain) to 10 (unbearable pain). We had to exclude one female patient (RDC/TMD: IIa) from our study because of unrealistic pain ratings (PRE: 10, POST: 0) and anxious behavior during fMRI measurements. The remaining participants were: 13 females, one male, average age: 25.71 ± 8.17. The median of pain score over all subjects included before therapy was five. Additionally, we used the “Graded Chronic Pain Status” (GCS) questionnaire (10) and the “Mandibular Function Impairment Questionnaire” (MFIQ (11)) in their German versions to classify the subjects before therapy. Both are reliable and valid measuring tools (GCS (12); MFIQ (13)). Concerning the GCS, seven participants were classified as level I (low disability and intensity), six as level II (moderate disability and intensity) and one as level III (severe disability and intensity). The MFIQ scored 11 patients at a qualitative level of function impairment of I and three patients with II.
Intervention
For each patient an individually optimized maxillary stabilization appliance of the Michigan type was produced. Patients wore the splint at night (average over the first 13 days: 8.80 ± 0.42 hours/day) during about two weeks (average: 16.29 ± 5.50 days). Over this period none of the subjects took any pain medication, which was controlled by the initial instruction and a later questionnaire. Informed consent was obtained from the patients and approval was given by the ethical committee of the medical faculty of the University of Greifswald.
Measurements of temporomandibular muscles
Bipolar surface electrodes measured, bilaterally, the EMG in the region above the masseter muscles. Electrodes (diameter 14 mm; center to center 20 mm; Noraxon Dual Electrodes, Noraxon, Scottsdale, AZ, USA) were placed parallel to the longitudinal axis of the masseter muscles. The reference electrode was positioned postauricular on the right mastoid region. The EMG signals were differentially amplified (EMG-8 Bluetooth Measuring System for Recording Muscle Activity of Zebris Medical, Isny, Germany) and sampled at 1.000 Hz per channel. Data were analyzed with WinJaw software (Zebris Medical, Isny, Germany). Muscle activity was measured at rest in a relaxed mandibular position with the incorporated splint. A second measurement took place during maximal voluntary teeth clenching (MVC) in the intercuspal position (maximal meshing of cusps of opposing teeth). The signals were averaged, with muscle activity assessed as the root mean square (RMS) of the amplitude (unit: µV). The resulting values of right and left masseter muscles were averaged at rest and during MVC.
Kinematographic measurements
We used the Jaw Motion Analyzer (JMA, Zebris Medical, Isny, Germany), an ultrasonic-based measuring system that functions as a navigation system by applying a sampling frequency of 80 Hz. We recorded the position of the mandible in space and time in relation to arbitrary reference planes. The mandible was opened three times from the intercuspal position up to the maximal mouth-open position and closed again. In a healthy and relaxed subject physiologically the right and the left condyle move forward and downward approximately symmetrically and homogenously and at the same time. Asymmetric changes in one or both condyle paths result in rotational slewing of the intercondyle axis to the right and/or left side (14).
The maximal angles of these rotations were measured during three jaw-open and -closing movements and averaged for the pre- and post-measurements. An angle of zero degrees means an ideal physiological situation—the greater the degree of the rotation angle, the more asymmetric condylar movements. For the correlation analysis with fMRI-activation magnitude, we calculated the differences in asymmetry (absolute values of condylar asymmetry) per time (Pre- minus Post-). This resulted in negative values if the symmetry was worse after therapy and positive values for an improvement of symmetry (see Figure 1).
(a) Time course of pain ratings (VAS) in the pain diary. Both movement pain (p < 0.005) and pain at rest (p < 0.05) were significantly reduced over therapy. (b) Occlusion resulted in increased activation in the right cerebellar hemisphere (MNI coordinates: e.g. 27, –57, –18) and right anterior insula (MNI coordinates: 39, 21, 3) before compared to after therapy. VAS: visual analog scale; MNI: Montreal Neurological Institute.
fMRI paradigms
Participants were asked to perform three different tasks before and after therapy: “occlusion”, “splint” and “ball.” During the “occlusion” task participants opened and closed their jaw in a relaxed manner but predefined frequency of one Hz (visual pacing by a pulsating circle, shown during both rest and activity). The same tapping movements were performed on the stabilization splint by the participants in the “splint” task. Finally, during the “ball” task these movements were again performed on a rubber ball of 4 cm diameter attached to an air pressure transducer that fed the signal to an acquisition device (Varioport, Becker Meditec, Karlsruhe, Germany) and later off-line data evaluation. The ball task was used for testing comparable movements of occlusion during the pre- and post-measurement. Because this condition was described as aversive we did not use this task for fMRI-data evaluation. All three tasks were performed in a block design consisting of nine blocks for each run, including five “rest” blocks and four “action” blocks, each lasting 20 seconds (10 scans) summing up to a scanning time per task of three minutes. The order of the paradigm investigated (“ball,” “occlusion,” “splint”) was pseudo-randomized to avoid habituation effects between tasks. Subjects were instructed immediately before each task. They were asked to perform repetitive 1 Hz occlusal movements during “action” blocks indicated with a green screen and paced by a white circle flashing with 1 Hz. A blue screen presented with the flashing circle indicated rest. Color contrasts to the white circle were balanced, and pulsating circles were presented during both rest and activation in order to avoid differences in visual presentation between tasks. Visual stimuli were back-projected onto a translucent screen, which participants viewed through a mirror mounted on the head coil. The position of the head was fixed using special pillars as head holders in order to fill out spatial distances between head and coil.
fMRI scanning
fMRI measurements were performed with a 3 T scanner (Siemens Verio, Erlangen, Germany) equipped with a 32-channel head coil. The functional images consisted of echo-planar images (EPI) with a repetition time (TR) of 2000 ms, an echo time (TE) of 30 ms, a flip angle α of 90° and field of view (FOV) of 192 × 192 mm2. Each volume consisted of 34 slices with a voxel size of 3 × 3 × 3 mm3 with a 1-mm gap between them. The first two volumes of each session were discarded to allow for T1 equilibration effects. Thirty-four phase and magnitude images were acquired in the same FOV by a gradient echo sequence with TR = 488 ms, TE (1) = 4.92 ms, TE (2) = 7.38 ms and α = 60° to calculate a field map aiming at correcting geometric distortions in the EPI images. Additionally, we measured a T1-weighted high-resolution structural whole-head image using a three-dimensional Magnetization Prepared Rapid Gradient Echo sequence (MPrage, TR = 1900 ms, TE = 2,52 ms, 176 sagittal slices, voxel size 1 × 1 × 1 mm).
Data reduction and statistical analysis
fMRI data
Spatial preprocessing and data analysis were performed with SPM8 (Wellcome Department of Imaging Neuroscience, London, UK) implemented in MATLAB (MathWorks Inc, Natick, MA, USA). Unwarping of geometrically distorted EPIs was performed in the phase encoding direction using the FieldMap Toolbox. Each time series was realigned and resliced after unwarping in phase encoding direction (anterior/posterior). The T1-weighted fMRI scans were coregistered to the functional images, segmented and normalized into the Montreal Neurological Institute (MNI) space, which allowed the pre-post comparison and the usage of anatomical masks. To correct for intensity inhomogeneity, EPIs were smoothed with a Gaussian filter of 6 mm full width half maximum (FWHM). To correct for low-frequency components, a high-pass filter with a cut-off of 128 s was applied.
For each participant a design matrix was created. The event intervals within each trial were modeled as boxcar functions convolved with a canonical hemodynamic response function. Individual statistical maps were calculated for the main effects. Contrast images of each participant were then used for group statistics calculated as random-effects analysis at the second level, which takes variance between participants into account.
We used an ROI approach (whole brain: p < 0.001, uncorrected) with a correction for multiple comparisons within the ROIs with p < 0.05 (family-wise error; FWE). For the ROI analysis we selected areas known to be involved in the processing of pain (insula, primary and secondary somatosensory cortex and cingulate cortex), but also areas processing sensorimotor control of occlusal movements (M1, superior parietal lobe, cerebellar hemispheres). We therefore used cytoarchitectural probability masks (ANATOMY,http://www.fz-juelich.de/inm/inm-1/DE/Forschung/_docs/Gehirnkarten/gehirnkarten_node.html). If there was no appropriate mask available (anterior insula, cerebellar hemisphere) we referred our analysis to the “Automated Anatomical Labeling” software (AAL). We further subdivided the insula mask (AAL) into its anterior and posterior part. In order to analyze changes over time induced by the therapy in general, we compared the measurements per time (“PRE minus POST” and “POST minus PRE”) for each condition (“occlusion” and “splint”) separately using paired t tests. Decrease of pain and decrease of degree of the rotation angle (movement kinematography) were both correlated with changes in fMRI maps for the occlusion task using SPM8 multiple regression in preselected ROIs (see above).
Evaluation of performance, kinematography and EMG
Significance of changes in pain ratings over time (ratings before each fMRI scanning in an ordinal scale) was tested with one-sample non-parametric testing (Wilcoxon test). Changes in pain scores during rest and movement (10 occlusion movements) as assessed with a visual analog scale (VAS) in a pain diary were averaged for the first two (days 1 and 2) and the last two days (days 13 and 14) for each subject and compared with paired t tests. Changes in symmetry of condylar movements (rotation angle) were compared using paired t tests between pre- and post-measurements. Correlation analysis was performed between pain decrease and decrease of degree of the rotation angle using Kendall tau-b test.
Performance data (pressure ball) were evaluated with MATLAB version 7.4, and we used paired t tests to compare possible differences in performance and amplitude of EMG data between pre- and post-measurements (SPSS, version 20.0)
Results
Ratings, EMG, kinematography and performance
After therapy, the subjective pain experience, as assessed by an ordinal scale before fMRI measurement, was reduced from a median of five points to a median of two points (Z = 3.38; p = 0.001). Relief in pain, as assessed using a VAS (diary), was greater for movement pain (t(13) = 3.71; p < 0.005) than for resting pain (t(13) = 2.81; p < 0.05; Figure 2(a)). The angle of rotation significantly decreased from 3.58 ± 1.79° to 3.15 ± 1.47° (t(13) = 1.80, p < 0.05, one sided). Additionally, pain decrease was associated with an increase in the symmetry of condylar movements (decrease of the rotation angle; tau-b = 0.49; p < 0.05). The average EMG muscle activity of the temporomandibular muscles (in the region of the right and left masseter muscles) with the splint in a relaxed position was reduced from 6.25 ± 3.97 µV to 3.51 ± 1.52 µV (t(12) = 2.36, p < 0.05) over the course of therapy. Furthermore, EMG activity during biting with maximal energy with intercuspidation increased from 219.71 ± 181.99 µV to 272.81 ± 232.21 µV (t(13)) = 2.81, p < 0.05; Table 1). The task performance during the occlusion condition, controlled by a pneumatic ball, showed no relevant differences between the pre- and post-measurement for both performance frequency (t(13) = 0.95; n.s.) and pressure amplitude (t(13) = 0.06; n.s.).
Pain decrease over the course of therapy was associated with a decrease in activation (parameter estimates) in right anterior insula (r = 0.85; MNI coordinates: 33, 18, 21). MNI: Montreal Neurological Institute. Bilaterally evoked EMG data above the jaw muscle skin assumed to be elicited from the masseter muscles of CMD patients. EMG: electromyography; CMD: craniomandibular disorders; SD: standard deviation; MVC: maximal voluntary teeth clenching.

fMRI data
Comparison over time
fMRI effects over therapy period: (PRE-POST(occlusion)); cut-off: p < 0.001.

aFWE: family-wise error correction for the respective mask. fMRI: functional magnetic resonance imaging; MNI: Montreal Neurological Institute; ROI: region of interest.
Correlation analysis
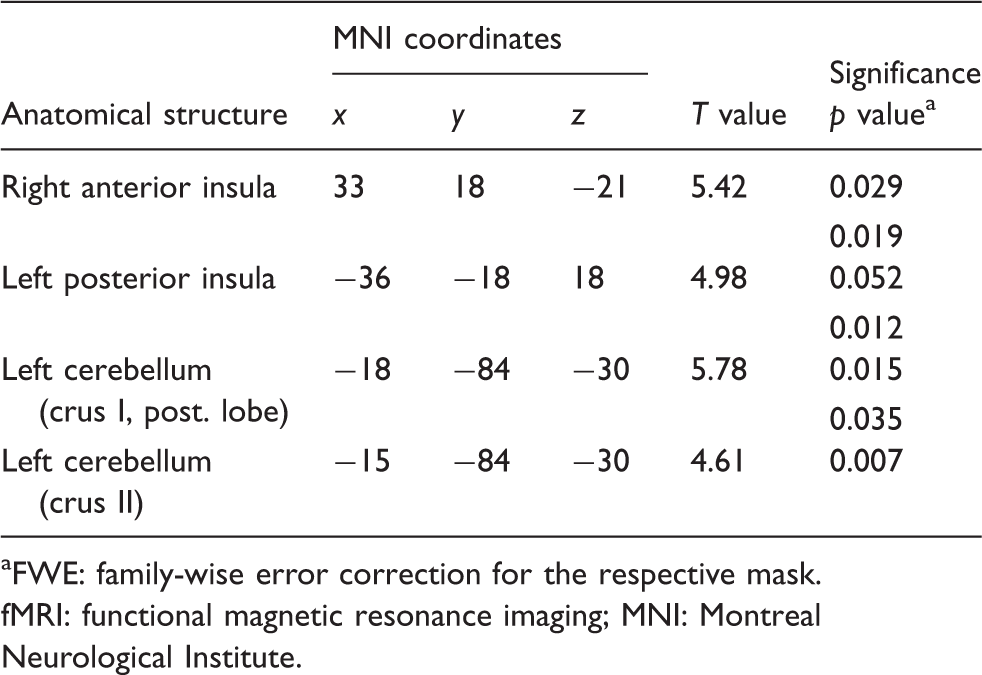
Pain reduction (assessment shortly before fMRI measurements) correlated positively with changes in blood oxygenation level-dependent (BOLD) magnitude during the therapy in the right anterior insula (t = 5.42; MNI coordinates (x, y, z): 33, 18, −21; Figure 3), the left posterior insula (t = 4.98; −36, −18, 18) and the left hemisphere of the cerebellum (Crus I (t = 5.78; −18, −84, −30), Crus II (t = 4.61; −15, −84, −30); Table 3). Increased symmetry of condylar movements (decreased angle of rotation) correlated positively with decreased activation in the left cerebellar hemisphere (Larsell’s HVI: t = 6.53; −12, −78, −21) and the right precentral gyrus (t = 5.87; 51, 3, 30) over the course of therapy (Figure 1).
(a) Top: Schematic drawing of the temporomandibular joint (TMJ) rotation angle deviation measurement per time. Bars represent simultaneous phases of movement of the intercondylar axis from maximal opening (lower bar) to the intercuspidal position (upper bar). When the rotation angle per time between the left and right TMJ is symmetric, parallel lines in the coordination diagram are plotted. Below: One example of losing movement patterns during pre- and post-measurements. The planes are more parallel during the post-measurement and therefore path of movement is almost ideal. (b) Activation in left cerebellum (Larsell's lobule HVI; MNI coordinates: –12, –78, –21) and right precentral gyrus (MNI coordinates: 51, 3, 30) showed a positive correlation with changes in the rotation angle over the therapy period. (c) Plotted associations between BOLD-effect magnitude (parameter estimates) over time and changes in the rotation angle over time. The more the angle of rotation decreased (corresponding to increased symmetry), the more the activation decreased in the left cerebellar hemisphere (r = 0.88; MNI coordinates: –12, –78, –21). Lower values than 0 means that symmetry did decrease over time. This was the fact in three subjects, who all showed low changes in fMRI activation for M1 and cerebellar hemisphere. MNI: Montreal Neurological Institute; BOLD: blood oxygenation level-dependent; fMRI: functional magnetic resonance imaging; M1: primary motor cortex. Positive correlation analyses between the fMRI effects over the therapy period (PRE-POST (occlusion)) and pain reduction; cut-off: p < 0.001. aFWE: family-wise error correction for the respective mask. fMRI: functional magnetic resonance imaging; MNI: Montreal Neurological Institute.
Discussion
Our study demonstrated that occlusal Michigan splint therapy for approximately two weeks in CMD patients was accompanied by subjective pain relief and decreased activation in the right anterior insula and right cerebellar hemisphere. Correlation analysis confirmed an association of the right anterior insula with the pain ratings. These associations with pain were also significant for the left posterior insula and the left cerebellar hemisphere. During therapy, the jaw musculature relaxed during rest and showed a marked increase in EMG activity during maximal occlusion. Finally, increased condylar movement symmetry correlated with decreases in the left cerebellum (VI) and right precentral gyrus.
Insula activation and anticipation of pain
Associations between right anterior insular activation and pain ratings in CMD patients were in keeping with findings in other patients with chronic pain. It has been described that activity in the anterior insula is associated with the affective somatosensory components of painful stimuli (15,16) their anticipation (17). Furthermore, chronic pain has shown an increased affective contribution compared to acute pain (18). Neuropathic chronic pain also shows increased activation in the right caudal anterior insula compared to experimentally induced pain (19). According to Craig (20), primary interoceptive representation takes place in the dorsal posterior insula and engenders distinct highly resolved feelings from the body. Processes within the right anterior insula appear to provide the basis of emotional awareness. This theory might explain why anterior insular activation is focused in the right hemisphere in our study, whereas additional activation has been identified in the left posterior insula, which is known to show a positive correlation with objective stimulus intensity and some sensory-discriminative characteristics (16).
More general effects over time (PRE minus POST measurements) were seen only in the right anterior insula. It has been reported that activation in this part of the insula is associated with the anticipation of emotionally aversive stimuli (painful stimuli (17)) and with unpleasantness of, for example, dyspnea (21). In another study (22), attended painful stimulation of the right or left hand was associated with larger volumes of right anterior insula activity compared to distracted painful stimulation, where it was reduced. This confirms the observation that attention/distraction primarily affects the right anterior insula. Perhaps right anterior insular activity before therapy represents a general heightened aversive emotional arousal state in our patients, such as their uncertainty regarding whether the jaw movement tasks would increase their perceived pain, although they were told the execution should not cause any additional pain.
Cerebellar and precentral activation and movement kinematics
It has been demonstrated before that the insertion of an occlusal splint results in a redistribution of the condyle-fossa distances in the temporomandibular joint (TMJ) (23). In our study we found a decrease of rotation angle during occlusion after the therapy intervention and a reduction of pain associated with a decrease of a rotation angle. The more the asymmetry of TMJ movement was decreased after therapy, the more pain was reduced during intervention. In addition we found a positive correlation of increased symmetry of condylar movements (decreased angle of rotation) with decreased activation in the left cerebellar hemisphere.
The cerebellum is well known for its involvement in motor function and, consequently, is active during chewing and biting paradigms (6,24). Sensorimotor mapping of the human cerebellum by Grodd and colleagues (25) revealed patterns of activation during repetitive movements of the lips and tongue in bilateral Larsell’s lobule HVI, which is similar to what we observed during occlusal movements. Similarly, Grabski and colleagues (26) observed activation in the superior cerebellar hemispheres (−16, −62, −18 and 20, −60, −20) during jaw movements in the proximity of our activity maximum in the right cerebellar hemisphere (27, −57, −18) in the PRE- minus POST-occlusion contrast.
An increase of cerebellar activity in the right posterior lobe before therapy indicates higher sensorimotor control processes before therapy. Wearing an occlusal splint alters somatosensory input thereby inducing changes in motor patterns during occlusion. The cerebellum also plays an important role in the early stages of motor learning (e.g. Steele and Penhune (27)). More specifically, our study revealed increased cerebellar activity in the right cerebellar hemisphere (Larsell’s lobule HVI) at the beginning of the therapy period. Furthermore, Larsell’s lobule HVI in the left cerebellar hemisphere was associated with changes in the symmetry of condylar movements. For this region an involvement in early motor learning has also been described (27), which may respond well to increased attentional demand. We also found that increases in the symmetry of condylar movements correlated with decreases in activation in the right precentral gyrus in terms of the height of the motor representation of jaw movements. In keeping with this, it has been reported that M1 correlates negatively with improvements in motor performance (27). Recent analysis focused on cerebellar involvement in pain procession (28,29), and our correlations between cerebellar activation and pain support these observations.
Relaxation and focused activation in the EMG of the jaw muscles
After splint therapy, we found increased relaxation of the jaw muscles during rest and a marked increase in EMG activity of the jaw muscles during MVC. These findings fit well with those of others showing decreased EMG amplitudes in masticatory muscles during clenching tasks in CMD patients compared to healthy controls (30,31). In addition, increased EMG activity in the resting position has been demonstrated in the anterior temporalis muscles (30). Experimentally induced pain in the masticatory muscles is capable of mimicking CMD symptoms and reducing EMG activity during maximal voluntary bite force (32) and chewing paradigms (33). Thus, the reduction in ipsilateral masseter muscle activity decreases with increasing distance from the location of the hypertonic (painful) infusion (33).
In our study, right anterior insular activity correlated positively with pain. Specific activation in this area is reportedly associated with the anticipation of pain (17). Therefore, it is also conceivable that decreased muscle activity during MVC is, to a certain extent, a learned strategy to avoid pain strengthening. After the successful therapy period, patients may not fear the pain and were, thus, able to mobilize increased bite force.
Nevertheless, it must be taken into account that the majority of our patients predominantly expressed myogenous problems. There is some controversial discussion regarding whether these patients might be clearly differentiated from healthy subjects by EMG indices (34).
Overall, this study provides some insights that might have clinical implications, too. First, anterior insula is an area associated with anticipation of aversive experience. In our next study, we will therefore focus on the affective components of TMJ pain. The splint might provide additional security in guiding TMJ movements that reduce fear of painful experience and after the training period might increase trust in performing TMJ movements. This is also expressed in the EMG amplitude increase of masticatory muscles during maximal occlusion after therapy. Second, we found an association of an increase of symmetry of TMJ movements and a decrease in motor effort, as indicated by decreased activation magnitude in the right M1 and left cerebellar hemisphere. Guidance of movements by the training with the occlusal splint results in more symmetric TMJ movements and effortless neural control of occlusion. According to these findings, successful therapy should decrease fear from moving the TMJ, decrease anticipation of painful experience and increase symmetry and movement guidance during occlusion.
Limitations of the study
First, our sample size was relatively small, resulting in less statistical power. Therefore, only the peaks of activation differences may have been detected. Second, the therapy duration was very short. Other studies investigating stabilization splint therapy in TMD normally use periods of treatment between four and 12 weeks (35). However, the pain diaries indicated the largest effect during the first week of therapy. Finally, most participants included in our study suffered only from mild CMD. On the one hand, this allowed us ethically to prescribe several important instructions, such as not allowing any pain modulation medication during the therapy period. On the other hand, mild CMD patients may not be the best representative group for investigating typical chronic CMD pain.
Conclusion
Overall, our study shows that successful CMD therapy using occlusal splints is associated with two primary modifications in the neural processing of occlusion. The first involves changes in the internal monitoring and anticipation of pain in the anterior insula. The second is a homogenizing of condylar symmetry associated with changes in motor processing in the right primary motor and left cerebellar hemispheres. Both processes should be investigated in detail in future therapy studies for this frequent pain syndrome.
Clinical implications
A decline of temporomandibular joint (TMJ) pain is associated with a decrease in insula representation. An increase in symmetry of articulation is associated with decreased cerebellar and primary motor representation during occlusion. Successful therapy of TMJ pain is associated not only with decreased pain during rest and occlusion but also with increased symmetry of occlusion and altered electromyography (EMG) parameters. Knowledge of functional parameters might help to further optimize therapeutic procedures of TMJ pain.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
