Abstract
Background:
Triptans represent the best treatment option for most migraine attacks, although this is not as well studied as it might be in controlled trials. Their efficacy and tolerability vary, both between agents, and from patient to patient, with about 30%–40% of patients not responding adequately to therapy. As yet unexplained, the failure of one triptan does not predict failure with another, and therefore triptan nonresponders cannot be defined as individuals who have failed a single triptan. Five clinical studies provide evidence that switching from a triptan that is ineffective to a second one can result in effective treatment in a proportion of patients. Systematic studies investigating whether there are patients who do not respond to all triptans in all formulations are lacking.
Methods:
Here we discuss the importance of identifying triptan nonresponders, the literature supporting their existence, and the issues to be resolved to design trials to investigate this.
Conclusion:
So far, no scientific data about the presence of a triptan nonresponder population are available. We propose a pragmatic study design to assess the existence of this subpopulation, recognizing the complexity of the question and the likelihood that more than one issue is at play in nonresponders.
Introduction
Migraine is a common, chronic disorder with debilitating features that reduces the quality of life of sufferers. The societal impact has been ameliorated with the advent of triptans, serotonin 5-HT1B/1D receptor agonists. At present there are seven marketed triptans. These compounds share a common pharmacology yet have distinctive pharmacokinetic and pharmacodynamic profiles, in addition to different formulations, such as oral, nasal and injectable. Triptans currently represent the best treatment option for migraine attacks, although their efficacy and tolerability vary, both between agents and from patient to patient (1). These distinctions give clinicians choices with which to tailor therapy in the individual, although at present it is not possible to predict how an individual will respond to a particular triptan (2).
Given modern trends in evidence-based medicine, pharmacogenetics and an interest in disease subsets with unmet therapeutic needs, triptans now pose a challenge: Are there migraine patients who do not respond to this class of medicine at all? This question has not been hitherto thoroughly investigated. This contrasts with the general view of neurologists that such patients exist. It would therefore be important, in our opinion, to identify nonresponse to encourage industry to devise new innovative medications, and to understand better the pathophysiology of such attacks. Indeed, as triptans work relatively well for a substantial proportion of patients, superiority trials seem a challenge.
Designing a trial in triptan nonresponders may be of benefit both for industry and for patients. In parallel, it would be useful to investigate whether triptan nonresponders could benefit from known medications already on the market with distinct pharmacology, such as nonsteroidal anti-inflammatory drugs (NSAIDs). It would also be useful to investigate clinical markers of triptan nonresponsiveness to be able to identify these patients early in the clinical setting. It has been suggested that patients who respond poorly to rizatriptan display fewer premonitory symptoms (3) or that migraineurs with unilateral cranial autonomic symptoms seem to respond to sumatriptan or rizatriptan better than other migraineurs (4,5). Yet these studies focus on single triptans, and it is not known whether these populations would respond better to other triptans or other formulations. Last, identifying triptan nonresponders could allow investigation of pharmacogenetic polymorphisms correlated with poor response or allow characterization of a particular migraine subtype with potential pathophysiological implications. Overall, there are many potential benefits from identifying these patients.
What do we define as a triptan responder?
To understand which patients, if any, are triptan nonresponders, we should first understand which patients respond to triptans. In the literature a number of outcomes have been used to determine the effectiveness of triptans. Single-attack endpoints include pain relief or pain freedom at two hours, recurrence rate, sustained response (partial or total) for 24 hours, tolerability, satisfaction, reduction of associated symptoms and others. The majority of trials on symptomatic treatment in migraine studied only one attack, while consistency of response is an important element to patients. Multiple-attack endpoints, such as consistency of response defined as positive outcome in at least two out of three treated attacks, have also been proposed to define the response to triptan (1). It is not surprising that the response rate to any triptan is highly dependent on the clinical endpoint chosen. Thus, depending on the endpoint, a patient can be or cannot be defined as a triptan responder.
Furthermore, some factors can modify the responsiveness to the single triptan (in a positive or negative fashion) independently of the outcome chosen. Some suggested factors include: concomitant use of other medications, migraine preventives (6), opioids (7) or overuse of analgesics (8), inadequate dosing (8), previous use of other triptans (9), use of the medication late in an attack/baseline severity of pain (3), incomplete absorption (due to concomitant gastric stasis or vomiting (10)), and pharmacogenetic differences (11,12). The issue is further complicated by the attack variability: menstrual-related migraine, early morning migraine, migraine with early nausea or gastric atony, migraine with aura, or with or without premonitory symptoms. These may co-exist in a single patient and their attacks may respond differently to a triptan or indeed across triptans. The above-mentioned variables imply that some migraine attacks may be responsive to triptans while others may not in a single patient.
Lastly, it should be highlighted that the placebo effect in migraine therapy has an important role in triptan response and can be a strong confounding factor when defining true triptan responders. Importantly, some efficacy outcomes display a far lower placebo effect rate compared to others
What do we define as a triptan nonresponder?
It is generally accepted that failure with one triptan does not necessarily predict the failure with another, and therefore triptan class nonresponders cannot be defined as individuals who have failed a single line of treatment. Five clinical studies provide evidence that switching from a triptan that is ineffective to a second one can result in positive treatment (13–17), although one can make criticisms of the study designs used. In these studies, the investigators assessed the efficacy of a second, and in one case a third, triptan in patients considered nonresponders to a first triptan. Of these five studies just two prospectively reassessed the response to the first drug (sumatriptan) (13,16), while in the others the nonresponsiveness was retrospectively obtained through patient report. None used a placebo-controlled reassessment. Interestingly, when responsiveness to the first triptan was evaluated with a prospective trial, approximately one-third of self-described sumatriptan nonresponders ended up to be responders, despite the fact that the treatment with sumatriptan was open label. In the two prospective studies a self-reported population of sumatriptan nonresponders was re-challenged with sumatriptan, and if no response was observed, a second triptan (almotriptan or naratriptan) was administered. In the first of the two studies (16), when evaluating pain freedom at two hours, the second triptan (almotriptan) had a similar response rate as that reported for an unselected migraine population (33% vs approx. 35% (1)). In contrast, in the second study (13) the response rate to the second triptan (naratriptan), considering the same endpoint, was much lower (6% vs approx. 22% (1)). The data from these studies are inconclusive and, at best, suggest that triptan nonresponse may not be consistent across the class and highlights two problems in research in this area: the lack of a placebo control and the possibility that, under particular circumstances, a second exposure to the same triptan may lead to a different response in the same patient. Unfortunately, the data do not answer the crucial question: Is the failure of one triptan a negative predictive factor for the responsiveness to a second triptan? As discussed below, the answer to this question could shed light on the presence or absence of a triptan nonresponder population.
Other investigations of patients who have failed more than one triptan are present in the literature, although data cannot be easily extracted. For instance, in a recent manuscript evaluating the fixed combination sumatriptan-naproxen (approved in the United States), patients enrolled had on average failed 3.3 triptans because of inefficacy or intolerability (18). Yet, patients were retrospectively evaluated for previous triptan efficacy and tolerability and were highly heterogeneous for clinical and pharmacological characteristics.
In conclusion, therefore, the presence of individuals who do not respond to multiple triptans is at present anecdotal and should be formally investigated in a clinical trial.
How should we look for triptan nonresponders?
A number of different study protocols could be envisaged to investigate the existence of triptan nonresponders. Moreover, a number of features might be considered desirable for the results to have a useful clinical impact.
First, it might be important to investigate a reliable, robust endpoint that is least affected by the placebo effect. Among these, pain free at two hours would appear best, and has the advantage of being desirable by patients (19). Although 24-hour pain-free responses may be more desirable, their population limitations make studies largely unfeasible. Furthermore, intra-patient consistency of response for pain free would be ideal, as it has been shown to increase data reliability and patient satisfaction (1). While consistency of pain free is among one of the most difficult endpoints to achieve, it is certainly highly desirable as a clinically meaningful outcome. Therefore, using this measure would also help identify those patients for whom novel therapies are warranted. Secondly, the trial should be prospective, and ideally in a treatment-naïve population, using measures and tools indicated in the International Headache Society Guidelines (20), such as the attack report form (diary) that the patient has to fill out during a migraine attack. Triptan-naïve patients may be difficult to access and this certainly introduces design issues, such as unblinding by side effects. Thirdly, patients should be screened to minimize the presence of the factors that can modify the response to treatment. The patient should also be advised to take the triptan as soon as they recognize the headache as a migraine, as the more severe the baseline pain is, the less effective is the medication. Unfortunately treatment of mild pain increases placebo response rates (21). Furthermore, the trial should discount attacks present at waking for homogeneity.
Which study design would be best to investigate the presence or absence of a triptan nonresponder population?
The presence of seven triptans in different formulations does not allow an exhaustive cross-over study. Therefore, the ideal option is a single therapeutic path that includes all triptans (and formulations). A patient should be challenged with the first triptan; if nonresponsive he or she should be administered a second triptan, and so on. A patient failing all triptans should be defined as a triptan nonresponder. Financial constraints or patient compliance make an exhaustive approach impractical.
Therefore, if the headache community considers revealing a triptan nonresponder population important, a novel and pragmatic approach will likely be necessary. For example, if we postulate that a triptan nonresponder population exists, then we could hypothesize that a patient not responding to the first triptan offered should have a reduced chance of responding to a second triptan (Figure 1(a)); a patient failing the first two triptans proposed should have an even lower chance to respond to the third triptan, and so on. On the other hand, response rates should be unchanged through lines of treatment in the absence of a triptan nonresponder population (i.e. a previous failure of triptans is not a negative predictor for future treatments with other triptans) (Figure 1(b)). According to the number of lines of treatment, the extrapolation of the incidence of triptan nonresponsiveness could be more precise. One approach would envisage at least three lines of treatment. The lines of treatment would be pragmatic, as most physicians would argue that after three triptans the chance for a patient to respond is very small. As the placebo effect could be a strong confounding effect, it would be important, as stated above, to use the outcome measure that is least susceptible. Furthermore, which triptans are used and in which order might, potentially, affect the results (13,16). We feel that the triptans to be used would be dependent on the secondary outcomes that the community is interested in. On the other hand, the order of administration should be controlled for, and therefore the study should be designed as a cross-over randomized trial. It could be argued that generic, cheaper triptans should be included, although that is a societal and not an intellectual issue. Therefore, although the study design is straightforward and does not differ from routine clinical practice, concerted and organized action from different groups would be required to achieve a sufficient power size.
Hypothetical results of a trial sequentially administering triptans to patients who have not responded to previous treatment(s) in the presence (a) or absence (b) of a triptan nonresponder population. The response was intended as pain free at two hours, which is fulfilled by about one-third of patients. Figure 1(a). As a triptan nonresponder population exists, the proportion of patients belonging to this category will increase in the subsequent lines of treatment. The scheme assumes (arbitrarily) that half of the patients not responding to a first challenge with a triptan belong to the triptan nonresponder population, and that as the rate of response to a single triptan in the triptan-sensitive population is 50% (33 patients out of the 66 patients who are not resistant to triptans), this rate will be maintained also in subsequent challenges with different triptans. The response rate in the overall population will therefore decrease after each challenge (33.3% of the naïve population will respond, 25% of the patients failing the first triptan will respond in the second challenge and 16.7% of the patients failing the first two triptans will respond in the third challenge). Figure 1(b). As a triptan nonresponder population does not exist, the proportion of patients responding to a triptan at subsequent challenges will be unchanged. As above, the overall response rate for each triptan is set at 33.3% (1).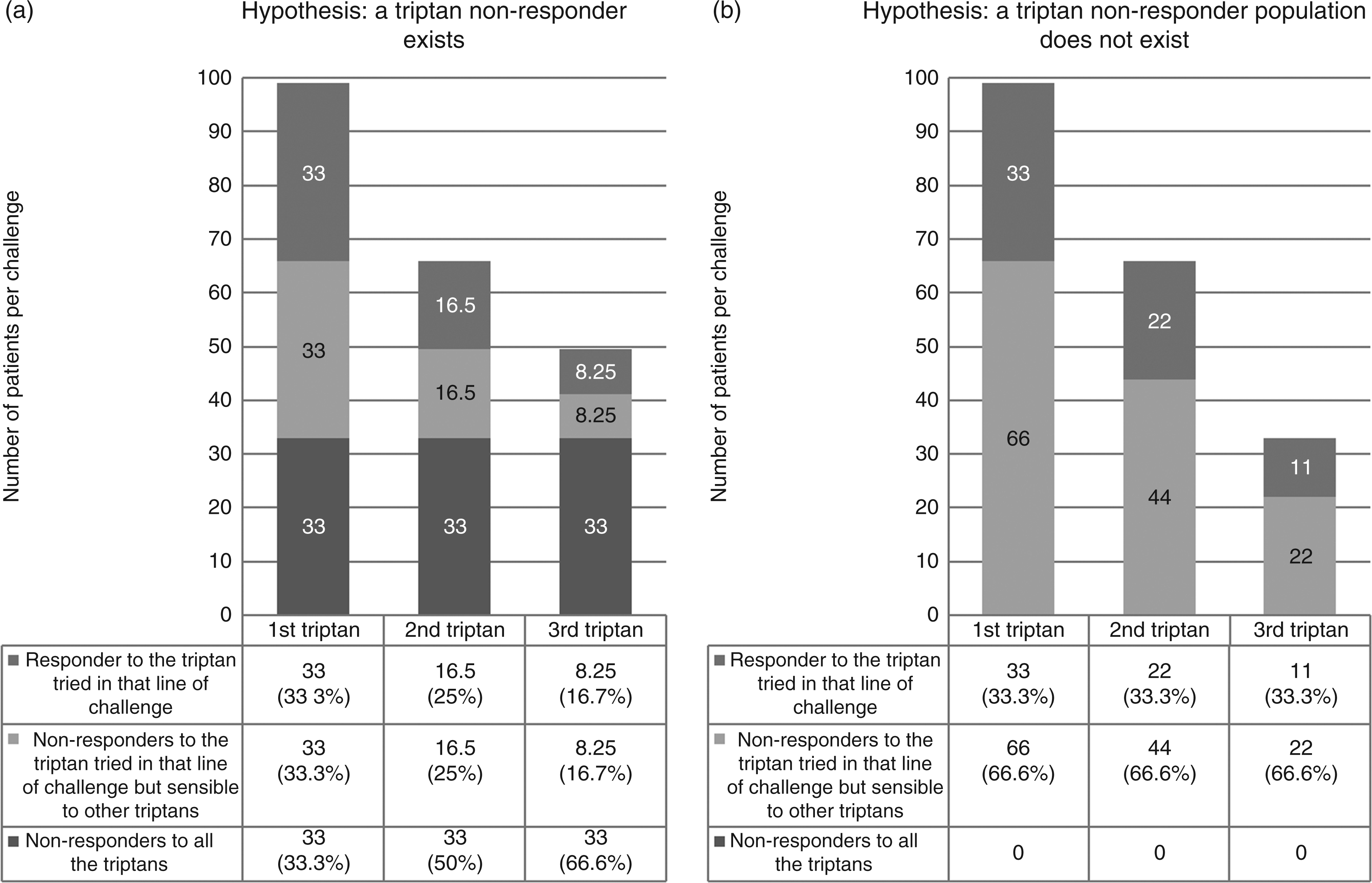
The effort required to complete the design described above might pose a limit in its feasibility. If this were the case, given the lack of any solid data in the literature, a multicenter prospective clinical practice study could represent an option to gather preliminary evidence both on the response rates and on the order of treatment.
Conclusions
Although triptans are the best symptomatic treatment for migraine, one of the most common and disabling neurological condition, many patients do not respond to the triptan that is prescribed to them. Perhaps surprisingly we know very little about the reasons behind these failures and about the patient population that does not benefit from triptans.
The present article suggests that the community should investigate which migraine populations are at present not treated adequately by triptans. In this setting, on scientific merit, we propose pain free at two hours in two attacks out of three as the cut-off between responders and nonresponders. Yet, it is important to consider whether this cut-off is adequate in the clinical setting. For instance, a number of patients do not have a pain-free response at two hours but are nonetheless satisfied with the treatment. In the opposite scenario, a number of patients are inconsistent responders, i.e. a minority of their attacks are not adequately treated. These latter patients would be classified as responders, but indeed their disease presents unmet therapeutic needs. It is important to note that the choice on where to draw the cut-off could have strong repercussions on the new drugs that might be developed in the future and for which patients.
We are confident that efforts to identify and investigate nonresponse to triptans would be helpful for many subjects, including physicians (expert in headache or not), clinical and basic researchers, pharmacological companies, health care providers, and of course, patients.
Clinical implications
Triptans represent the best treatment option for migraine attacks, yet a relevant number of migraine patients do not respond to the triptan they are exposed to. Few studies have investigated whether a patient not responding to a first triptan may have benefits from a second one; none have assessed whether some patients respond poorly to all triptans. Therefore, at present we do not know if a triptan nonresponder population exists, its frequency, whether these patients have a particular migraine endophenotype and/or endogenotype. Here we discuss the importance of identifying triptan nonresponders, the literature supporting their existence, and the issues to be resolved to design trials to investigate this. Such studies would benefit many, including clinical and basic researchers, as well as health systems.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
MV, AAG, ST and GN have no conflicts of interest. PJG is on the boards of Allergan, Colucid, MAP Pharmaceuticals, Merck, Sharpe and Dohme, eNeura, Neuroaxon, Autonomic Technologies Inc, Boston Scientific, Eli Lilly, Medtronic, Linde Gases, Arteaus, AlderBio and BristolMyerSquibb. He has consulted for Gammacore, Pfizer, Nevrocorp, Lundbeck, Zogenix, Impax and Dr Reddy, and has been compensated for expert legal testimony. He has received grant support from GlaxoSmithKline, MAP, MSD, eNeura and Amgen. He has received honoraria for speaking from MSD, Pfizer, Allergan and Mennarini, and payment for editorial work from
