Abstract
Background:
Several studies, but not all, of primarily middle-aged or younger adults have suggested that the common MTHFR C677T variant is a genetic risk factor for migraine with aura (MA). Here, we consider whether this variant is associated with MA risk in an older non-clinical population (AGES-Reykjavik cohort).
Methods:
Participants are a sub-sample (n = 1976) of subjects from the Reykjavik Study (RS; mean age 50) and its continuation, AGES-RS (mean age 76). We estimated the relative odds of MA in TT versus CC carriers using multinomial logistic regression. As both MA and the TT genotype may be linked with modestly reduced longevity, we performed a simple simulation to illustrate the effect that selective survival may have had on our observed gene–disease association.
Results:
TT versus CC carriers were at marginally reduced odds of MA (ORTT 0.55 (0.3–1.0), p = 0.07), significantly for women (ORTT 0.45 (0.2–0.9), p = 0.03). Assuming the ‘true’ (e.g. mid-life) effect of the TT genotype is ORTT 1.26, from a recent meta-analysis, our simulation suggested that if 25-year mortality had been (hypothetically) 13% higher in MA subjects with the TT versus CC genotype, the measured effect of the TT genotype on MA would have been attenuated to non-significance (e.g. ORTT 1.00). Our observed protective effect was consistent with the most extreme selective mortality scenario, in which essentially all of the previously reported increased mortality in MA subjects was (hypothetically) found in CT or TT carriers.
Conclusion:
The MTHFR 677TT genotype was associated with marginally reduced risk of MA in our older population. Our simulation illustrated how even modest selective survival might obscure the apparent effect of a genetic or other risk factor in older populations. We speculate that some of the heterogeneity previously observed for this particular genetic variant may be due to age range differences in the studied populations.
Introduction
The MTHFR gene on chromosome 1p36.3 encodes 5,10-methylenetetrahydrofolate reductase (MTHFR), a key enzyme in the folate metabolic pathway. A common functional variant in this enzyme, C677T, has been described in many populations (1). Carriers of the TT genotype have approximately 50% reduced enzymatic activity and consequently moderately increased total homocysteine, one of the major players in the folate metabolic pathway (2). This variant (or elevated homocysteine) has been associated with risk of various neurological disorders including Alzheimer-type dementia, Parkinson’s disease, epilepsy, and migraine with aura (MA) (3–5). In particular, the C677T variant is possibly the best studied and most replicated genetic risk factor for a common migraine type (MA) (6–20), although there has been considerable heterogeneity in the results to date (21,22).
Both MA and the MTHFR C677T variant may be related to longevity. As recently reported in the Reykjavik Study (RS) cohort, subjects with mid-life MA were at approximately 18% increased risk of all-cause mortality over 25 years of follow-up compared with those without headache (23). The TT genotype has also been reported to be less prevalent in older compared with younger subjects in several populations (Figure 1) – indirectly suggesting an adverse effect on longevity – although it has been suggested this variant may be protective in populations with mandated folic acid fortification (24).
Proportion of younger versus older subjects who are MTHFR 677TT carriers as reported in selected population-based studies.
We considered whether the MTHFR 677TT genotype was associated with increased risk of MA in a sub-sample of 1976 men and women who participated in the population-based RS (1967–1991; mean age 50), which was followed by the Age Gene/Environment Susceptibility-Reykjavik Study (AGES-RS; mean age 76). Anderson et al. recently proposed a general model to estimate SNP effect size erosion due to selective survival using three highly lethal diseases (intracerebral hemorrhage, ischemic stroke, and myocardial infarction) (25). Given the age of our study population, we wondered whether our late-life gene-association study might be influenced by selectively reduced participation of MA subjects with the TT genotype. These data led us to consider the broader question of the degree of selective survival that would be sufficient to erode or even reverse a ‘true’ genetic or other risk factor when measured in an older study population, even in a disease that may be associated with only modestly reduced longevity.
Methods
Study population
Detailed descriptions of the RS and AGES-RS have been published previously (26–30). In brief, the RS is a population-based cohort study established in 1967 by the Icelandic Heart Association to prospectively study cardiovascular disease in Iceland (26). In 2002, the RS continued as the AGES-RS to examine risk factors, genetic susceptibility, and gene/environment interactions in relation to disease and disability in old age (27). In the present genetic sub-study, 1976 men and women (mean age 76, range 66–93) were genotyped for the MTHFR C677T polymorphism. The AGES-RS was approved by the Icelandic National Bioethics Committees (VSN-00-063, 00-063-V8+1), which acts as the Institutional Review Board for the Icelandic Heart Association, the Data Protection Authority, and by the Institutional Review Board for the US National Institute on Aging, National Institutes of Health. Written informed consent was obtained from all participants.
Headache classification
Subjects were asked about headache symptoms in the mid-life RS interview. The mid-life headache classification and associated survival by headache type for this cohort has been described in detail (23). Briefly, those reporting headache once or more per month were asked follow-on questions about five features typical of migraine; those with two or more non-aura features were designated as migraineurs without aura (MO); those reporting either visual symptoms or numbness before headaches were designated as migraine with aura (MA). Subjects were classified into four mutually exclusive groups: no headache (n = 1357), non-migraine headache (NMH) (n = 367), migraine with aura (n = 167) and migraine without aura (n = 85). Subjects meeting both criteria for migraine with and without aura were included in the migraine with aura group.
Genotyping
DNA was isolated from peripheral blood lymphocytes according to the method developed by Scotlab Bioscience (Kirshaws Road, Coatbridge, UK). The proprietary GoldenGate assay by Illumina Genotyping Services (Illumina Inc., San Diego, CA, USA) was used for genotyping.
Analysis
We determined whether late-life genotype frequencies were in Hardy-Weinberg equilibrium overall and by headache categories. We calculated adjusted odds ratios (AORs) as an estimate of the relative risk of headache (NMH, MO, MA) in those with the CT or TT genotype compared with those with the CC genotype, adjusting for age (at blood draw for genotyping), sex, and duration of follow-up, using multinomial logistic regression.
Selective survival simulation
The prevalence of mid-life migraine has been previously reported for the Reykjavik cohort (23). We assumed that mid-life genotype frequencies by headache groups, if we had measured them, would be consistent with two assumptions: 10% of controls would be TT carriers (1), and the ‘true’ (e.g. mid-life) association between MTHFR C677T genotype and MA was as reported in a recent meta-analysis (22) using the additive model.
Using the calculated hypothetical mid-life genotype frequencies as baseline, we assumed that 25-year mortality risk in the MA group was different for those who were TT versus CT versus CC carriers. We first assumed, in the absence of selective survival, that MA subjects (all genotypes) were at 18% increased risk of 25-year mortality compared with controls (RRMA-All 1.18 (1.1–1.3), p < 0.05) as previously reported (23). This is a slightly lower age and sex-adjusted RR than the fully adjusted relative mortality estimate (RR 1.21 (1.1–1.3), p < 0.05) from this study, as here we want to estimate the hypothetical effect of selective survival per se without accounting for the potential explanatory or mediating factors such as the cardiovascular risk factors that were included in the fully adjusted models. We also note that the other published mortality risk estimates for MA, to our knowledge, were not appropriate estimates for this simulation as they were based on cause-specific mortality or did not provide a pooled estimate for women and men (31–33). We assumed no increased risk of mortality in subjects with headache other than MA, as previously reported (23,34) and, for simplicity, we assumed no increased risk of mortality in subjects with the TT genotype without MA.
Using the observed mortality in controls as baseline, the observed age- and sex-adjusted risk of mortality in subjects with MA compared with controls (23), and the hypothetical mid-life genotype frequencies, we calculated late-life genotype frequencies that would result from various levels of selective survival in MA subjects based on genotype. We held the overall RR of mortality in MA subjects constant at 1.18 and assumed that the increased risk of mortality in CT carriers would be half that of TT carriers. We then calculated ORs based on the resulting late-life genotype frequencies.
Results
Observed late-life MTHFR C677T genotype frequencies by mid-life migraine status in the AGES-Reykjavik Study.
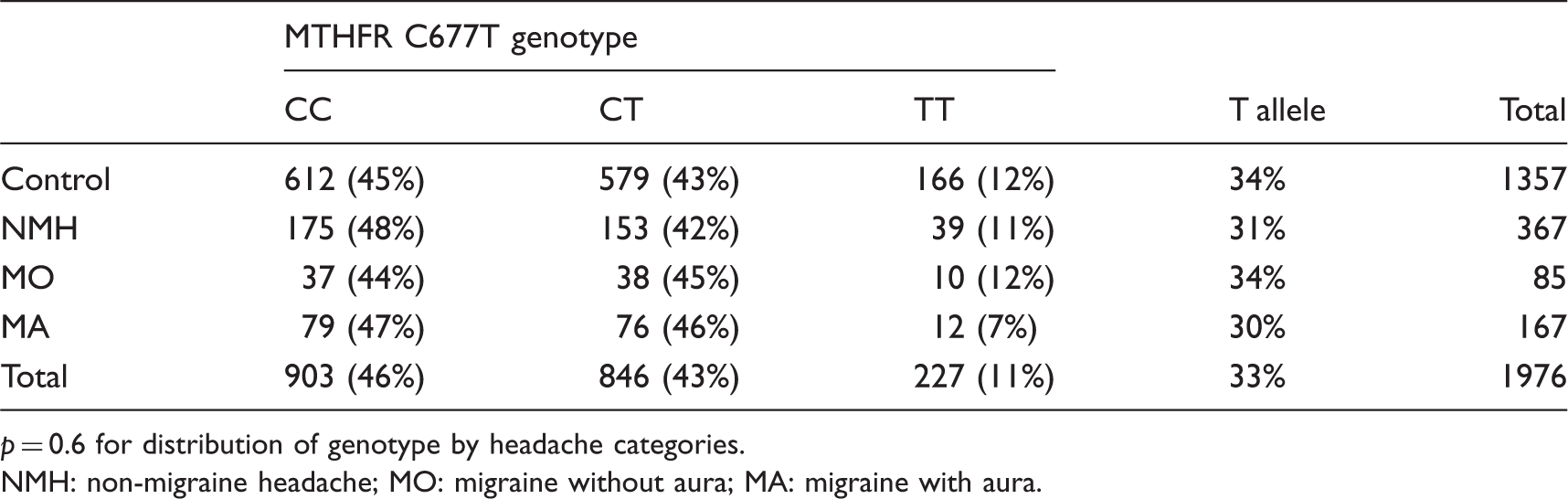
p = 0.6 for distribution of genotype by headache categories.
NMH: non-migraine headache; MO: migraine without aura; MA: migraine with aura.
Adjusted odds of migraine in subjects with the CT or TT genotypes relative to the CC genotype in the AGES-Reykjavik Study.
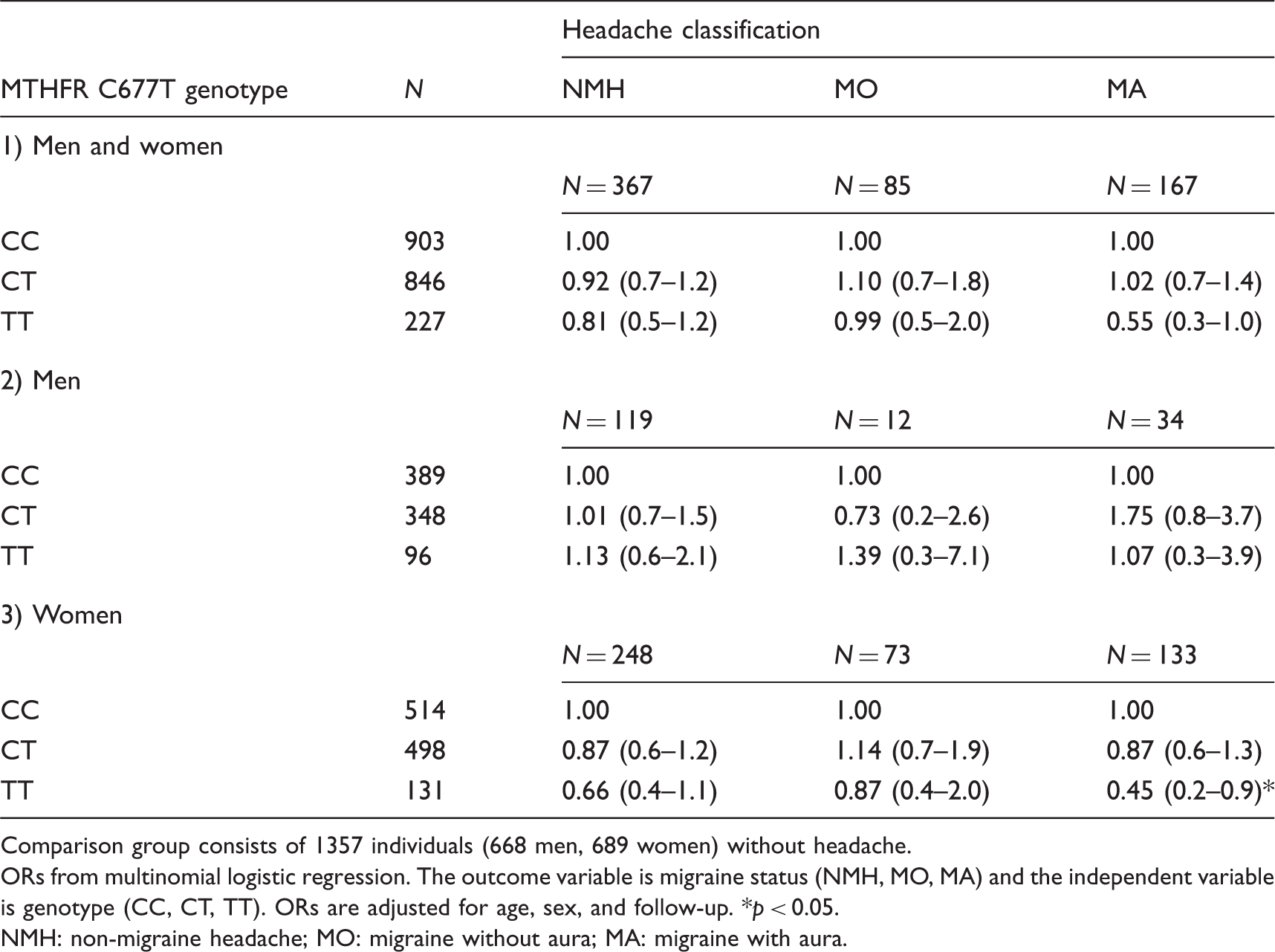
Comparison group consists of 1357 individuals (668 men, 689 women) without headache.
ORs from multinomial logistic regression. The outcome variable is migraine status (NMH, MO, MA) and the independent variable is genotype (CC, CT, TT). ORs are adjusted for age, sex, and follow-up. *p < 0.05.
NMH: non-migraine headache; MO: migraine without aura; MA: migraine with aura.
Hypothetical mid-life MTHFR C677T genotype distribution in the original Reykjavik Study cohort.

Percentages are rounded.
A: Actual prevalence of migraine in 18,725 subjects from the Reykjavik cohort at average age 50 (range 33–65) (from Gudmundsson et al., 2010 (23)).
B: Calculated hypothetical mid-life genotype distribution by headache type based on two assumptions: The prevalence of the TT genotype in controls is 10% (Botto and Yang 2000 (1)) and the ‘true’ relative odds of MA in TT versus CC carriers is 1.26, the pooled OR from one dozen studies as previously reported (Schürks et al., 2010 (22)).
NMH: non-migraine headache; MO: migraine without aura; MA: migraine with aura.
Illustration of how selective survival in subjects with a chronic disease (migraine with aura) based on genotype would change the apparent effect of the genotype in late-life: the AGES-Reykjavik Study.

Explanation of Table 4.
For the purpose of this simulation, ORTT 1.26 is assumed to be the ‘true’ measure of effect for the TT genotype on migraine with aura (MA) based on a recent meta-analysis (Schürks et al., 2010 (22)) (i.e. odds of MA are assumed to be 26% higher in TT versus CC carriers in younger populations, based on pooled OR 1.13 (0.97–1.32) per T allele). RRMA 1.18 is the observed age and sex-adjusted 25-year mortality rate ratio in MA subjects relative to controls as previously reported for the Reykjavik Study (Gudmundsson et al., 2010 (23)).
In the baseline scenario (column A), survival in the MA group relative to controls is assumed to be unrelated to genotype (e.g. RRMA-CC = RRMA-CT = RRMA-TT 1.18). In Scenario B, the indicated survival assumptions in the MA group by genotype produce a complete erosion of the evident effect of the TT genotype (Scenario B: ORTT 1.0). Scenario C survival assumptions produce the marginally protective effect for the TT genotype reported in the Women’s Health Study, in which subjects were on average age 55 at genotyping (Schürks et al., 2008 (12)) Scenario D survival assumptions produce the effect for the TT genotype reported herein, in which subjects were on average age 75 at genotyping.
Discussion
Selection bias is an ‘error due to systematic differences in characteristics between those who are selected for study and those who are not’ (35,36). Selection bias due to selective survival is a particular concern when the study population is elderly. If the disease of interest is associated with reduced longevity, there will naturally be fewer subjects with the disease (cases) in older study populations. Similarly, if the risk factor of interest (say, genotype ‘X’) is associated with reduced longevity, then there will naturally be fewer subjects in the study population with genotype X. If the reduced survival in cases is independent of genotype (or equivalently if the reduced survival by genotype is independent of case status) then the selection bias is considered ‘non-differential.’ The greater threat to study validity occurs with differential selection bias, meaning in this example that survival is additionally or exclusively affected in subjects with both the disease and genotype X. In this latter case genotype X might be over-represented in younger cases (e.g. associated with increased risk) but under-represented in older cases (e.g. associated with decreased risk) (25).
Although the MTHFR C677T variant is one of the few replicated candidate genetic risk factors for a common migraine type (21), we found in this older population that the TT genotype was marginally protective for MA. A recent meta-analysis (22) suggested that the pooled measure of effect for the TT genotype on MA was in the OR 1.3 range (additive model). There was evidence of heterogeneity by study location, which was ascribed to differences in ethnicity, but may additionally reflect regional environmental or dietary factors such as folic acid supplementation that influence the expression of MTHFR C677T genotype (37–40). Our study population includes European Caucasians and there is no mandatory folic acid fortification of food stuffs in Iceland.
We speculate that the different age ranges of the studied populations may also have contributed to this heterogeneity. Extant studies have generally not reported their results in a way that would allow a robust consideration of an age effect. For example, when considering the effect of selective survival it is not sufficient to consider only the mean age of the study population. The age distribution should also be considered as we would expect to find the greatest effect of differential survival in the older populations.
The recent reports from the Women’s Health Study (WHS) are of particular interest. That study of women (mean age 54 years, with about 10% 65 years or older) who had no history of cardiovascular disease or cancer showed at baseline that the TT genotype was less prevalent (e.g. was protective) in the women with MA compared with controls (12,41). Over 11 years of follow-up, women with MA had an increased risk of incident fatal and non-fatal cardiovascular disease (RR 2.06 (1.5–2.8)) (12). The risk was stronger in the women with MA and the TT genotype (RR 3.66 (1.7–7.9)) compared with women with MA and the CC (RR 2.39 (1.56–3.68)) or CT (RR 1.48 (0.89–2.47)) genotypes, although confidence intervals overlapped. These findings are consistent with our hypothesis of selective mortality or (additionally in this case) morbidity given their exclusion of women with prevalent cardiovascular disease. Arguing against this is the finding reported in a follow-up study suggesting that the protective effect of the TT genotype was most evident for women with less frequent attacks (42).
Anderson et al. recently proposed a general model to estimate SNP effect size erosion due to selective survival for three highly lethal diseases (intracerebral hemorrhage, ischemic stroke, and myocardial infarction) (25). Here we show how even modest selective mortality over a long period of follow-up, such as might occur in MA, could hypothetically erode or reverse the measured effect of a genetic or other risk factor when studied in an older population. In such cases, the degree to which the observed measurement of effect will generalize to younger populations, and vice versa, will depend not only on the age distribution of the target study population, but also on the magnitude of the association in younger populations, the natural history of the disease or risk factor, and whether participation is selectively affected in subjects with both the disease and risk factor of interest.
Clinical implications
The common MTHFR C677T variant has been linked with risk of migraine with aura in several studies. We found a protective effect for the TT genotype in our older population. In a simple simulation based on reported mortality history in migraineurs from our cohort, we illustrated how even modest selective mortality (in migraineurs with aura who are TT carriers) could hide the ‘true’ effect of a risk factor when measured in an older population.
Footnotes
Acknowledgment
We would like to thank the participants of the study and the Icelandic Heart Association clinic staff for their invaluable contribution.
Funding
This study was supported by a grant from the National Institutes of Health (N01-AG-1-2100), National Institute on Aging Intramural Research Program, the Hjartavernd (the Icelandic Heart Association) and the Althingi (the Icelandic Parliament).
Conflict of interest
None declared.
