Abstract
Migraineurs have an interictal sympathetic nervous system (SNS) hypofunctionality and hypersensitivity to adrenergic amines. The GNAS1 T393C polymorphism has been associated with a distinct SNS sensitivity in healthy subjects. We tested GNAS1 T393C variant in two independent sets of subjects. In the case-control subset, 365 migraine patients [194 with aura (MA)] vs. 347 healthy controls were studied. A significant excess of the CC genotype was found in migraneurs (31.2%) as opposed to controls (20.2%; P = 0.003). Using a logistic regression model corrected for sex, the CC genotype conferred a general risk for migraine twice [odds ratio (OR) 1.79, 95% confidence interval (CI) 1.27-2.53; P = 0.001] higher than CT/TT genotypes. Using parents from 117 migraine families, a marginally significant trend for association could be observed (P = 0.025), but the transmission disequilibrium test for alleles maternally transmitted failed to demonstrate familial association. In this subgroup, CC genotype conferred a risk for migraine over twice (OR 2.20; 95% CI 1.14-4.40; P = 0.019) higher than TT/TC genotypes. In conclusion, the GNAS1 T393C variant is associated with migraine, which suggests a genetic basis for its higher SNS sensitivity.
Keywords
Introduction
Migraine genetics are complex and largely unknown. There are only three genes (CACNA1A (1), ATP1A2 (2) and SCN1A (3)) identified for migraine, corresponding to the hemiplegic variants. Genome-wide scans performed to date have found significant linkage to some chromosomes (1, (4), 4 (5, 6), 5 (7), 6 (8), 11 (9), 14 (10), 15 (11), 18 (12), X (13)) for the regular forms of migraine, with (MA) and without aura (MoA). On the other hand, some association studies have suggested that common polymorphisms may influence the risk of migraine. These polymorphisms correspond to genetic variants of genes coding for receptors, amine transporters or enzymes implicated in the pathophysiology of migraine (for review see Estevez and Gardner (14)). Migraine has been considered to be a disorder of sympathethic disbalance. A relative baseline or interictal autonomic deficit, with marked hypersensitivity to endogenous amines, has been described (15). Most pain and catecholaminergic receptors have a heptahelical structure and use a G-protein transduction system. G-proteins consist of a family of α, β and γ subunits, the βγ forming a functional dimer. On activation of heptahelical membrane receptors, the α subunit releases guanosine diphosphate in exchange for guanosine triphosphate and both α and βγ subunits dissociate and can modulate a huge variety of targets such as ion channels, phospholipase C isoforms and adenyl cyclase isoforms, to name but a few (16).
Nevertheless, the G-protein system has scarcely been studied in migraine (17). Galeotti et al. (17) have shown that the inhibition of cAMP production by inhibitory G-proteins (Gi) is lower in headache (MA, MoA and cluster) patients, suggesting a lower inhibitory tone that predisposes to neuronal hyperexcitability. As they pointed out, elevated cAMP plasma levels in headache sufferers indicate an imbalance between the inhibitory and stimulatory system of adenyl cyclase, mediated by Gi and stimulant G-proteins (Gs), respectively. Inhibitory G-proteins seem to be expressed in peripheral lymphocytes in lower quantity in migraineurs than in healthy controls or patients with other painful disorders, but Gsα mRNA levels in lymphocytes are normal (18). β-Adrenergic receptors coupled to Gs protein (βAR-Gs) stimulate adenyl cyclase in cardiac and vascular smooth muscles. Migraineurs show basal, interictal adenosine (cAMP) plasma levels four times higher than controls or than in other chronic painful disorders (17). Hence, it is expected that mutations which ultimately affect the function of G-proteins must have a significant effect on intracellular signal transduction. The GNAS1 T393C polymorphism is located on exon 5 of the GNAS1 locus on chromosome 20 (19), which is under complex genetic control and harbours four imprinted transcripts (NESP55, XLαs, 1A and GNAS1). NESP55 and GNAS1 transcripts are expressed from the maternal allele. Exon 5 is common to all the transcripts.
We have analysed the potential association between migraine and the GNAS1 T393C polymorphism, which encodes for Gαs protein.
Methods
Subjects
We recruited two independent sets of subjects. For the case–control subset, 365 consecutive, unrelated, migraine patients attending our general Neurological clinic (aged 38.6 ± 15.0 years) and 347 healthy controls with no headache history (aged 38.9 ± 13.9 years) were available (Table 1). All were White and lived in Cantabria, northern Spain. Controls were recruited among patients' friends. Patients with a history of stroke (n = 5) were excluded to avoid misinterpretation of aural symptoms, as were controls with any type of migraine (eight cases with possible migraine). All cases and healthy controls were interviewed by an experienced neurologist. The migraine patients were diagnosed as having International Headache Society MoA (n = 171; aged 41.9 ± 16.5 years) and MA (n = 194; aged 35.7 ± 13.0 years). Forty-three MA patients had also experienced MoA attacks. For the family-based association study, 558 subjects belonging to 117 pedigrees were recruited and genotyped. To gain power, MA and MoA were treated jointly as ‘affected’. Informed consent was obtained from all patients and controls. The protocol had been approved by our local ethics committee.
Genotypic and allelic distribution among clinical subgroups of GNAS1 T393C polymorphism

For genotypic distribution:
For allelic distribution:
χ2 for MG/controls = 11.477 (2 d.f.); P = 0.003 P = 0.003
χ2 for MA/MoA/controls = 12.704 (4 d.f.); P = 0.013 P = 0.012
χ2 for MG males/control males = 6.515 (2 d.f.); P = 0.038, NS after correction P = 0.023 (OR 1.57; 1.06, 2.31), NS after correction
χ2 = 3.838 (2 d.f.); P = 0.147, NS P = 0.194, NS
Genetic analysis
We performed allelic discrimination to detect the GNAS1 T393C polymorphism using fluorogenic 3′-minor groove binding probes (MGB) in a real-time polymerase chain reaction (PCR) assay (20). The PCR was carried out on an ABI Prism 7000 thermocycler (Applied Biosystems, Foster City, CA, USA) using fluorescently labelled probes (200 n
Statistics
Case–control study
Analyses were made using a logistic regression model to assess whether the GNAS1 T393C polymorphism significantly contributed to prediction of migraine in general, MA and MoA. We also analysed the influence of this polymorphism on association with menstruation and age at onset. Menstrual migraine was assessed in women after asking them if menstruation was an unequivocal precipitating factor for migraine. Allele-wise and genotype-wise comparisons were made according to grouping TT vs. CC/TC and TT/TC vs. CC. As the sample was not perfectly matched for sex, this variable was introduced in every logistic regression equation. Here we considered significant association when P-values were ≤0.01, after Bonferroni's correction for multiple testing, since other hypotheses—main migraine subtypes, sex, age at onset and influence of menstruation—were tested. For general significance, the P-value was set at 0.05. For categorical analyses, we report overall χ2 test of association (α = 0.05).
Family study
Here we performed two distinct types of analysis. First, the transmission disequilibrium test (TDT) was used in those families composed of trios in which both parents were available, with at least one of them heterozygous for the marker. Since GNAS1 is maternally imprinted, we analysed only the transmission of maternal alleles, using the ETDT program (21). The ETDT program allows for analysis of only those alleles paternally or maternally transmitted. Second, unrelated subjects (parents) from this group were also used as an independent case–control subset. Here and in ETDT analyses, α-type error was set at 0.025, since parents were tested twice in the second case–control subset and in the TDT analysis.
Results
Case–control study
Both controls (P = 0.537) and cases (P = 0.051) fit the Hardy–Weinberg equilibrium. We observed significant differences in the genotypic distribution of the GNAS1 T393C polymorphism for migraineurs (χ2 = 11.81; 2 d.f., P = 0.003) compared with controls, even after stratifying the sample into MA and MoA (χ2 = 12.704, 4 d.f.; P = 0.013; Table 1). Numerically, there were more CC genotypes in MA than in MoA, but this difference was not significant. Using logistic regression analysis, after correction for sex, GNAS1 T393C significantly contributed to migraine prediction [odds ratio (OR) 1.79, 95% confidence interval (CI) 1.27-2.53; P = 0.001 for CC genotype vs. TC/TT genotypes; OR 1.78, 95% CI 1.2-2.7; P = 0.007 for CC genotype vs. TT]. This effect was even higher for MA vs. controls (OR 1.99, 95% CI 1.34-2.97; P = 0.001). No OR estimates for CC/CT genotypes vs. the TT genotype were significant. Nevertheless, the C allele was more frequent in migraineurs (54.3%) than in controls (45.8%; χ2 = 8.57, P = 0.003), conferring a non-adjusted OR for migraine of 1.37 (CI 1.1-1.7; P = 0.003). These data are slightly higher for MA than for MoA (see Table 1). GNAS1 T393C was not associated with migraine endophenotypes, such as association with menstrual migraine (Table 1) or age at onset (data not shown). Statistical power for association of migraine with the GNAS1 T393C polymorphism reached 0.81 for a significance level of 0.01 (22, 23).
Family study
We genotyped a total of 558 familial subjects (384 affected; 174 unaffected) belonging to 117 pedigrees (Table 2). There were 320 founders (194 genotyped). Using this group of founders as an independent case–control sample, we found an excess of CC genotypes in those having migraine (χ2 = 5.795, 2 d.f.; P = 0.055, NS, and P = 0.024 for trend). After grouping CT/TT genotypes to gain power, the CC genotype conferred an over twofold risk for migraine among founders using their respective spouse as a control (OR 2.20, 95% CI 1.14-4.40; P = 0.019). Statistical power for association of migraine with the GNAS1 T393C polymorphism using this subset of patients reached 0.69 for a significance level of 0.05 (22, 23). TDT analysis of those alleles maternally transmitted gave no significant results (Table 3).
GNAS1 T393C genotypic distribution in familial subjects∗
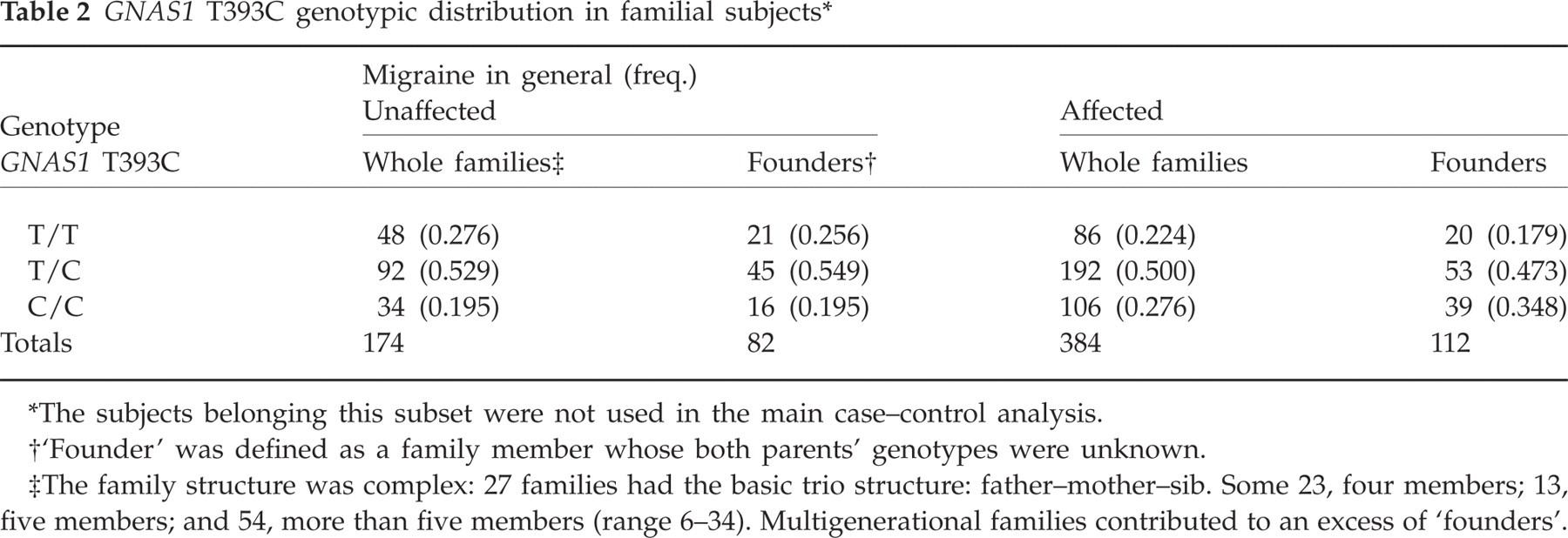
The subjects belonging this subset were not used in the main case–control analysis.
‘Founder’ was defined as a family member whose both parents' genotypes were unknown.
The family structure was complex: 27 families had the basic trio structure: father–mother–sib. Some 23, four members; 13, five members; and 54, more than five members (range 6–34). Multigenerational families contributed to an excess of ‘founders’.
Results of transmission disequilibtrium test for those alleles maternally transmitted

Alleles
maternally transmitted and
not transmitted.
NS, Not significant.
Discussion
Our study shows a genetic association of GNAS1 T393C with migraine in two independent subsets of subjects. We observed that the C393 allele was significantly more frequent in migraineurs than in controls in the case–control subset of patients. These results were also confirmed in the family study using the parents as a case–control sample. The C393C genotype conferred a twofold higher risk of suffering from migraine also in both subsets of patients. Even when the C393 allele was more frequent in MA patients, we found no significant differences between MA and MoA patients, nor was this polymorphism associated with migraine endophenotypes in the case–control subset. As GNAS1 is maternally imprinted, only maternal alleles are expressed. In this case, family studies permit us to recognize the precise origin of both alleles. In the family study we failed to demonstrate any association using the ETDT algorithm specific for maternally transmitted alleles, which may be due to loss of power after selecting only maternal transmissions.
The GNAS1 T393C polymorphism has been associated with different autonomic nervous system sensitivity in healthy Japanese subjects (24). In the Japanese study, those subjects with TT/TC genotypes had a lower parasympathetic index (the ratio between high-frequency components and total power of the spectrum of cardiac frequencies) than those sharing the CC genotype. The T allele has also been associated mainly with the development of hypertension, although the exact relation of the GNAS1 T393C polymorphism with hypertension is unclear (25–28). The contribution of the GNAS1 T393C to β-blocker response has also been well documented in hypertensive patients (25).
The sympathetic nervousd system (SNS) seems to play a role in migraine pathophysiology. Both the relative deficit in SNS function during the interictal phase and the hypersensitivity shown in migraine patients to norepinephrine (NE), dopamine (DA) and 5-hydroxytryptamine (5-HT) suggest sympathetic hypoactivity (29–33). Contingent negative variation, an event-related slow, negative cerebral potential recorded over the scalp in simple reaction-time tasks, used as a measure of central catecholaminergic activity (34), has also disclosed higher amplitudes in those more disabled patients and those more likely to respond to β-blockers (35). These observations could be summarized by saying that central catecholaminergic hyperactivity coexists with a peripheral sympathetic overflow. In addition to the possible role of GNAS1 in SNS activity, this complex locus is also expressed in platelets, where its activation results in increased cAMP levels and inhibition of platelet responses to activation (36), and is up-regulated in chronic migraine (37).
The GNAS1 T393C is a synonymous polymorphism at codon 131 (ATT to ATC) leaving the amino acid isoleucine unchanged. The effect caused by the T393C polymorphism may be related to some other genetic variation in linkage disequilibrium with T393C which leads to disfunctional Gαs protein.
In conclusion, we found that the GNAS1 C393C genotype almost doubles the risk of suffering from migraine compared with TT/TC genotypes. Our family-based study partly corroborates the role of this gene in migraine. Our observations need to be replicated in other population-based studies.
Acknowledgements
This work was supported by Grants API/22/05 of ‘Fundación Marqués de Valdecilla-IFIMAV’, Pfizer Fdn, FISS Project (PI05388) of the Instituto Carlos III, and by the ‘Centro de Investigación de Enfermedades Neurológicas’, Nodo HUMV/UC, ISCIII, Spain.
