Abstract
Introduction: As migraine attacks pose insult to cerebral circulation and ion homeostasis, migraine has the potential to interfere with the development of different brain structures, producing functional deficits. It is known that visual contour integration (CI) is a function with a protracted development. Therefore, we sought to establish whether migraine interferes with its development.
Methods: Forty-eight migraineurs (without aura) and 48 age- and sex-matched controls participated in the study, divided into three cohorts by age. Stimuli were presented on cards with a contour consisting of Gabor patches embedded in random noise. Difficulty was varied by the manipulation of relative noise density. The task was to identify and show the contour.
Results: A significant difference was found between the performance of migraineurs and controls in the 10–14-year-old and 15–18-year-old cohorts (p < 0.05). Development between all three cohorts was significant in the control group (p < 0.017), while it was not significant in migraineurs between 6 and 14 years. Correlation between age and CI threshold was stronger in controls than in migraineurs.
Conclusion: Children with paediatric migraine exhibited a less marked development in the Gabor patch-based CI task.
Introduction
Basic visual functions, such as flicker sensitivity, colour and depth discrimination and visual acuity have traditionally been considered as reaching adult levels in the first few years of postnatal life, making the general impression that the postnatal development of human vision is finished early in life (1–4). However, a vast body of evidence has emerged to suggest that certain aspects of human vision take a long time to reach their functional maximum, as in functioning as observed in healthy adults. Aspects developing beyond the second year of life include visual segmentation and form identification based on texture (5,6), motion (7,8), colour (9) and flicker contrast (10). Visual spatial integration has been reported to show significant development until adolescence (11). It is easy to see that such a wide developmental window also means a prolonged period of vulnerability to various pathological processes.
A migraine attack is obviously an insult to the cerebral circulation and ion homeostasis, due to processes related to cortical spreading depression (CSD) and local inflammation/pain. Although it is often mentioned in connection with the aura phase, it is not unequivocally proven that the characteristic CSD (12) is a trait of migraine with aura only (13,14). This disturbance of cerebral cortical function is associated with considerable metabolic and haemodynamic changes. Electrophysiologically, the process begins with depolarization associated with an increase in extracellular potassium, followed by a short-term depression of electrical activity. The “depolarization front” starts out from the occipital cortex and spreads in a rostral direction, over all major visual cortical areas. At the biochemical level, glutamate seems to play a key role through NMDA (N-methyl-
It is assumed, based mainly on animal studies, that the headache phase is related to the large cerebral vessels, pial vessels, large venous sinuses and dura mater as pain sensitive structures and the trigemino-cervical complex (TCC) innervating them in the form of a plexus of largely unmyelinated fibres. This plexus originates in the ophthalmic division of the trigeminal ganglion and in the upper dorsal cervical roots of the spinal cord (18). A central eventis a neurogenic vasodilation of the intracranial vessels, initiated by calcitonin generelated peptide (CGRP) from the trigeminal ganglion. This brings about a kind of “sterile inflammation”, coming with plasma protein extravasation and sensitization of the sensory nerve endings. Simultaneously, mast cell degranulation (serotonin release) (19) and platelet aggregation (20) also occur. Serotonin release is a direct link between the neurogenic and vascular theories, the latter postulating that the starting point of migraine is in fact pathological serotonin release, followed by vasoconstriction and compensatory vasodilation causing the actual painful sensation. It has recently been pointed out that the neurotransmitter systems involved in this general process are largely modulated by sex hormones (21), giving rise to a sex-influenced trait model (22). It is to be noted here, though, that evidence does not support a major role of such vascular events in human migraine, as pointed out by Schwedt and Dodick in a recent review (23). Migraine has also been proposed to be a channelopathy, most likely a disorder of the Ca2+P/Q channel, however, this has been proven only in the case of the relatively rare familial hemiplegic migraine (24). An alternative ocular theory of migraine has been proposed by Gupta (25), but that theory is possibly too specificto account for the general picture.
Interictally, the most obvious findings are lowered ionized magnesium levels in the serum (26) and lack of habituation at the cortical level (27), both pointing to a hyperexcitability of the cortex.
Based on these, we propose that repeated attacks have the potential to interfere with the development of the visual brain, which, in turn, should be observable as deficits in any of the long-developing aspects of vision, including contour integration. However, the question is largely unanswered, and we shouldmention that any functional alteration observed in migraineurs’ vision might also be a genetic trait, part of an endophenotype, either having nothing to do with the actual attacks, or, on the contrary, making carriers more vulnerable to the adverse effects of the attacks.
Contour integration
Gestalt psychology, which flourished in the 1930s, suggested that contour perception is basically carried out via the integration of local contour elements (28). Evidently, the task applied in this study, requiring the subject to identify a circular shape made up of Gabor patches placed at a distance from one another, necessitates a shape perception system capable of just such an integration, with the extra requirements that it has to be able to solve the task of bridging the gaps between the individual elements and that it has to be able to overcome contextual noise.
The first reasonable physiological cues concerning the nature of contour/shape perception came in the 1960s, from the pioneering experiments of Hubel and Wiesel (29,30), supporting the gestaltist insight by the demonstration of cellular level orientation preference in the primary visual cortex of both cats and monkeys. For our approach the group of cells later named “simple cells” in the 4Cβ layer of the primary visual cortex is important. These cells, acting as local filters, fire the most vigorously when their receptive field is matched by a line stimulus of their preferred orientation. It is easy to see that such simple cells are fit to process fine detail, and indeed, V1 4Cβ is the primary cortical receptive layer of parvocellular geniculostriatal input (31). Linking local filters and bridging gaps (even in noise) is possibly carried out via an intrinsic plexus of horizontal connections in V1 4Cβ (32–35). That is, perception of a shape, a contour might be interpreted as cells with receptive fields of the proper orientations responding simultaneously, linked by their horizontal connections.The very method of testing contour integration with Gabor patches emerged as a result of looking for the most ideal stimuli for the simple cells of the primary visual cortex (36). As a general hypothesis, therefore, we assumed that the migrainous process might interfere with the optimal performance of the local element integration system described above.
In a recent study (37), we pointed out that visual contrast sensitivity at low spatial frequencies is impaired in migrainous children of 6 to 14 years of age, as compared to age- and sex-matched controls. We also learnt from previousstudies (11,38,39) that in healthy children of the same age cohort, development of task performance in visual contrast sensitivity and visual contour integration tasks exhibit parallel development, especially in the scotopic and dynamic condition and at low spatial frequencies of the Venuscontrast sensitivity test (NeuroScientific Corporation, Farmingdale, NY, USA). The parallel development of performance in those two test types led us to assume shared underlying mechanisms/structures, and to test this hypothesis we sought to test the performance of migrainous children in a visual contour integration task based on Gabor patches. To our knowledge, we are the first to report on such data in connection with paediatric migraine.
Methods
Characteristics of migraineurs by cohort
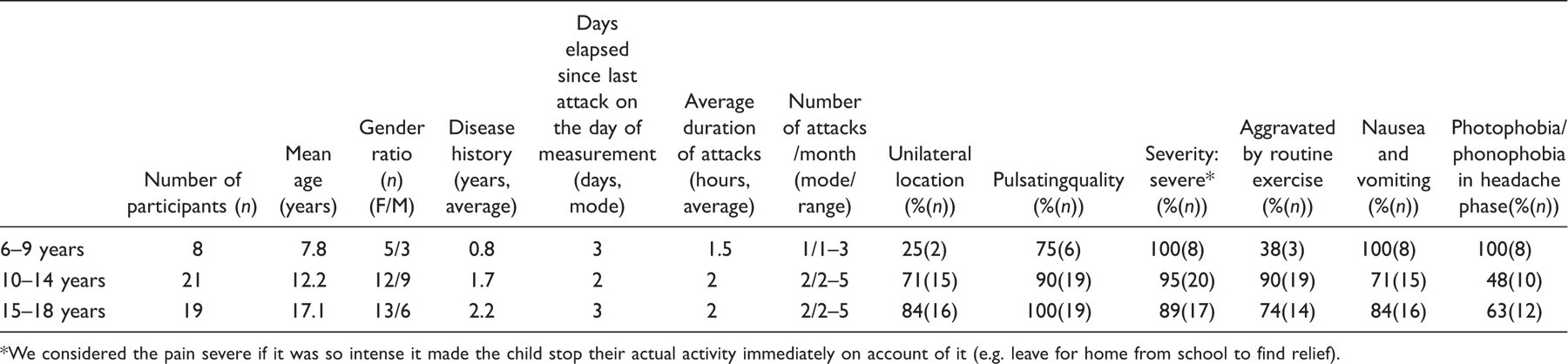
We considered the pain severe if it was so intense it made the child stop their actual activity immediately on account of it (e.g. leave for home from school to find relief).
Contour detection stimuli were presented on cards, at a distance of 0.5 m (size: 18 × 24.5 cm, subtending approximately 21° × 28°). On each card, a circular contour consisting of 12 Gabor patches was embedded in a backgroundof randomly placed patches (noise). The task was to locate and show the contour. The cards were presented in an increasing order of difficulty. Contour visibility (difficulty) was varied by manipulating relative noise density (D). The D-value was defined as the ratio of average noise spacing over contour spacing. We used a set of 10 cards in which D ranged between 1.1 and 0.65 and was varied with a step size of 0.05. At D > 1, the contour elements are closer to each other than the noise elements. However, at D < 1, this cue is not available, and it is impossible to detect the contour without orientation-specific long-range interactions.
Data analysis
Just like in our previous study (37), participants were assigned to age cohorts reflecting the normal (healthy) developmental steps of this function, as found by Benedek et al. (38), i.e. 6–9 years, 10–14 years, and 15–18 years. The dependent variable was the minimum value of D (D min), signifying the subject’s absolute detection threshold.
As our data did not follow a normal distribution, and given that splitting the total number of participants into three cohorts left us with a relatively small number of elements for each particular comparison, we applied non-parametric analyses. For the between-group comparisons (as in migraineurs vs. controls by age cohort) we applied the Wilcoxon matched pairs test at a significance level of p < 0.05, while for the within-group comparisons (comparison of age cohorts within the migraineur and control groups) the Mann-Whitney U-test (MWU) was applied at p < 0.017 (after the Bonferroni correction). MWU (at the same level of significance) was also performed for all cohorts to see if gender (in both groups) mightcause any significant variance in D min. To better characterize developmental tendencies, Spearman’s correlation coefficient (r) was computed for cohort-wise comparisons in both groups, and to find out whether there was significant correlation between attack frequency and D min.
Results
Exact significance values for the within-group and between-group comparisons.

The numbers signify the cohorts, as in 1: 6–9 years; 2: 10–14 years; 3: 15–18 years. That is, 1 vs. 2 means a comparison between the 6–9 and 10–14-year-olds. Asterisk (*) marks significant values.
Within-group (cohort-wise) comparisons for the migraine group, in order of significance: 6–9 years vs. 15–18 years (MWU 7, n 1 = 8, n 2 = 19, p < 0.001, two-tailed); 10–14 years vs. 15–18 years (MWU 16, n 1 = 20, n 2 = 19, p = 0.044, two-tailed); 6–9 years vs. 10–14 years (MWU 55.5, n 1 = 8, n 2 = 20, p = 0.53, two-tailed). Spearman’s r for the total development between 6 and 18 years was 0.51 (p < 0.05).
The same comparisons were also performed for the cohorts of age- and sex-matched controls, yielding the following results: 6–9 years vs. 10–14 years (MWU 8, n 1 = 8, n 2 = 20, p = 0.043, two-tailed); 10–14 years vs. 15–18 years (MWU 4.5, n 1 = 20, n 2 = 19, p < 0.001, two-tailed); 6–9 years vs. 14–18 years (MWU 6, n 1 = 8, n 2 = 19, p < 0.001, two-tailed). Spearman’s r for the total development between 6 and 18 years was 0.65 (p < 0.05).
As for the possible role of gender in migraineurs: 6–9 years (MWU 7, n = 8, p = 1.0, two-tailed); 10–14 years (MWU 51, n = 21, p = 0.86, two-tailed); 15–18 years (MWU 37.5, n = 19, p = 0.9, two-tailed); and in controls: 6–9 years (MWU 6.5, n = 8, p = 0.79, two-tailed); 10–14 years (MWU 40, n = 21, p = 0.35, two-tailed); 15–18 years (MWU 37, n = 19, p = 0.9, two-tailed).
Spearman’s r for attack frequency vs. D min by cohort: 6–9 years: 0.52 (NS); 10–14 years: 0.33 (NS), 15–18 years: 0.17 (NS); Spearman’s r for attack frequency for all cohorts taken together: 0.31 (p < 0.05). All results are summarized in Figure 1.
Discussion
Between-group comparisons revealed that children with migraine in all three studied cohorts exhibited a poorer performance in the contour integration task than age- and sex-matched controls, the difference reaching the level of statistical significance in the cohort of the 10–14-year-olds.Performance was characterized by D
min, i.e. the absolute detection threshold of the individual, expressed as the lowest value of relative noise density between 1.1 and 0.65, at which the individual was still able to identify the target contour (see Methods).
A. A group- and cohort-wise summary of performance. Small solid squares indicate the median value of D
min in the given group. Boxes indicate 25–75 percentiles, whiskers mark the non-outlier range. B: A plot of individual performance. D
min values of each migraineur–control pair are given. Light grey bars indicate controls, dark grey bars indicate migraineurs. Data have been ordered according to migraineurs’ performance in each cohort. Numbers on the lower horizontal axis refer to pairs, not individuals.
As for the within-group (cohort-wise) comparisons, the largest step in the migraine group was observed when comparing the youngest and the eldest cohorts. Somewhat less pronounced, but still significant, development was seen when comparing 10–14-year-olds with the eldest cohort. However, the remarkable development one would expect based on the study conducted with a large number of healthy subjects between 6–9 and 10–14 years (4) did not appear.Controls exhibited significant development in all of these comparisons.
Gender does not seem to have a significant effect on performance in this type of contour integration test. Correlation between attack frequency and D min was significant only when it was computed for all 48 subjects.
Finally, when the performance of all 48 subjects was considered, age and D min were more strongly correlated in controls than in migraineurs, reflecting a more pronounced development.
Our present study has therefore corroborated our hypothesis built up on the basis of our previous findings, namely that if children with migraine show contrast sensitivity deficits (37), they will exhibit contour integration deficits as well. What does this tell us about migraine and the developing human visual brain?
Possibly the most important finding of this study is the remarkable development between 6 and 14 years of age – described in healthy subjects, and appearing in the actual controls – is missing in migraineurs. This came as no real surprise, as in a previous study (34) we found the same pattern for contrast sensitivity at low spatial frequencies. As mentioned in the Introduction, the plexus of horizontal connections between the direction-sensitive cells of the primary visual cortex is considered to be of key importance in a Gabor-based contour integration task. Could it be that migraine (and especially the pain-related pathophysiological phenomena) interferes with the development of these connections? Simple and elegant as it may sound, such an interpretation alone fails to explain a number of observations.
First of all, Burkhalter and colleagues (32) established in their post mortem study that horizontal connections within the 4Cβ and 2/3 layers of the primary visual cortex are structurally adult-like by the age of 15 months. This early development makes complete sense, given the hierarchical nature of neocortical development (41), progressing from the primary sensory areas toward the association areas, which Flechsig was among the first to point out, as early as 1920 (42). Accordingly, functionality in terms of contour detection appears quite early: at 3 months of age it is yet to be seen (43), but 6-month-old babies perform well over chance level, even if their noise tolerance is quite limited (44). Taken together, this rather makes the impression of mature primary cortical circuits without fully effective filtering mechanisms than primary cortical circuits at the beginning of a decade-long developmental path. We propose, therefore, that the observed deficit of contour integration in paediatric migraine cannot be approached exclusively from the primary visual cortex, even though the primary visual cortex is of central importance in such tasks. To come up with a better explanation, one has to find structures whose development is well established and fits the findings. Literature on cortical development unequivocally considers the frontal/prefrontal cortical areas to be among the last to mature (41,45–48). In terms of our findings, two particular developmental studies deserve special attention. Kanemura et al. (49) examined the development of the prefrontal cortex in children in a three-dimensional volumetric MRI study, and found that this cortical area reaches its final, adult-like size by the age of 18, with a period of rapid growth between the ages of 8 and 14. Fornari (50) examined the relationship between the total amount of white matter and performance on a spatial integration task, and found that there is a linear relationship between age and white matter volume between the ages 7 and 13, also reflected by improving performance on psychophysical tasks. It can be seen that the age ranges strikingly resemble the range in which the most significant differences in our study were found. Therefore, we propose that taking the frontal/prefrontal areas and their connectivity into account might offer an insight into why contour integration develops rapidly in healthy subjects between 6 and 14 years of age, and why it fails to do so in migraineurs. A possible scenario is that the bottom-up visual connectivity of the frontal/prefrontal cortical areas fails to develop at a normal rate due to repeated attacks, which brings about a slightly delayed development of the related cortical parts, and this, in turn, results in deficient top-down inhibition. We also point out that our results fit well with the theory of Bar et al. (51), who consider the orbitofrontal cortex as a structure of key importance in object recognition, as the conclusion to a number of MEG studies. That is, based on our findings and the literature, we find it reasonable to assume that it is not contour/shape perception per se that is deficient in paediatric migraine but rather visual noise suppression. This assumption is supported by a number of imaging studies. Schmitz and colleagues showed that impaired executive function in migraine can be linked to decreased frontal and parietal gray matter density in adult migraineurs (52). Rocca et al. (53) and Ruscheweyh et al. (54) similarly come to the conclusion that the frontal areas are especially vulnerable to both migraine and chronic pain in general, respectively. What exactly causes this loss of density is yet unclear.
Is it really the attacks? Although we did not find significant correlation between the monthly frequency of attacks and D min when computed cohort-wise, it must be seen that the correlation was significant – if weak – when all subjects were included. This we consider a statistical issue due to the relatively small number of participants (especially if divided into subsamples) and the fact that the frequency of attacks turned out to be quite uniform in the sample (median: 2 days, range: 1–5 days). It seems to be clear enough that with a larger and more variable sample, this issue would disappear.In other words, we assume that the attacks do have a role in bringing about the pattern that we found, even though this particular sample is not the most optimal to demonstrate it.
As migraine is known to be a familial disease, it is possible that the explanation for the early appearance of the described difference between migraineurs and controls is that it reflects an endophenotype. Di Clemente et al. (55) make a similar argument in connection with the interictal habituation deficit of the nociceptive blink reflex in migraineurs. However, even if we consider the deficit described here as reflecting an endophenotype, it remains an open question whether increased vulnerability to insults or a relatively stable (fixed) degree of functional deficit (affecting a range of functions) constitutes that endophenotype. The former scenario, of course, predicts that the longer the migraine history is, the more pronounced the contrast sensitivity deficit, which, in spite of the relative paucity of research in this direction, seems to be the case (56). It is still an interesting question whether that vulnerability is part of the migrainous endophenotype only. Recent evidence (57) suggests that theincreased developmental sensitivity of the magnocellular visual pathways is part of the normal endophenotype. In our opinion, the role of the magnocellular pathways indeed deservesconsideration, as our previous contrast sensitivity study (34) conducted with child migraineurs also yielded results suggesting a magnocellular deficit.
Finally, why in the cohort of 6–9-year-olds we found no. significant difference might have a number of possible reasons. First, this is a really young cohort, with quite a brief disease history (see Table1). We find it unlikely that such a short exposure to migraine attacks should result in significant deterioration. Second, although migraine does exist at such a young age (58–60), the diagnosis poses challenging dilemmas (61), even following the ICHD-II as closely as possible. Thus, it cannot be ruled out that’false positives’ were included in the sample, and given that the number of subjects in this cohort was small, we admit that it is difficult to draw firm conclusions from this part of the data.
In conclusion, our study has shown that contour integration performance in paediatric migraine without aura is poorer than in age- and sex-matched controls, revealing a less marked development of performance in migraine. Considering relevant literature and our previous study on contrast sensitivity, we find it reasonable to hypothesize that the effect is mediated by the developmental vulnerability of the magnocellular visual pathways/dorsal stream (62), possibly leading to a deficit of frontal top-down noise suppression. Talking about migraine without aura, it appears reasonable to assume that it is rather the pain-related processesthat are possibly responsible for the effect than CSD. However, as said before, it cannot be ruled out that CSD might still play a role, especially given that it comes with a prolonged decrease in posterior blood flow (63), thus having the potential to cause developmental disturbances. Certainly, further studies are necessary to clarify the exact background of the described phenomenon.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
